Green and Sustainable Chemistry
Vol.4 No.1(2014), Article ID:42859,5 pages DOI:10.4236/gsc.2014.41003
A Green Approach towards the Synthesis of Enantio Pure Diols Using Horse Radish Peroxidase Enzyme Immobilized on Magnetic Nanoparticles
1Medicinal Chemistry and Pharmacology Division, CSIR-Indian Institute of Chemical Technology, Tarnaka, India
2Natural Products Chemistry Division, CSIR-Indian Institute of Chemical Technology, Tarnaka, India
3Inorganic and Physical Chemistry Division, CSIR-Indian Institute of Chemical Technology, Tarnaka, India
Email: *adarirao2002@yahoo.co.in
Copyright (c) 2014 Seelam Siva Deepthi et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights (c) 2014 are reserved for SCIRP and the owner of the intel-lectual property Seelam Siva Deepthi et al. All Copyright (c) 2014 are guarded by law and by SCIRP as a guardian.
Received January 3, 2014; revised February 3, 2014; accepted February 10, 2014
Keywords: Magnetic Nanoparticles; Horseradish Peroxidase; Immobilization; Vicinal-Diols
ABSTRACT
Enantiopure epoxides and their corresponding chiral vicinal diols serve as valuable intermediates in the synthe- sis of biologically active pharma and agro-compounds and also value added fine chemicals. Biocatalysts are well known for their selective hydrolysis of racemic epoxides to give optically pure chiral diols. This study highlights an efficient process of synthesis of chiral vicinal diols in good yields and enantioselectiviy using horse radish pe- roxidase enzyme immobilized on the amine functionalized magnetic nano particles (Fe3O4 nanoparticles) as en-zyme carriers. It also facilitates separation of MNP-immobilized enzymes by applying external magnetic field. The immobilization of magnetic nano particles was confirmed by transmission electron microscope (TEM) and scanning electron microscope (SEM). The MNP-immobilized peroxidase enzyme improved stability of the en- zyme and has shown broader substrate specificity in enantioselective hydrolysis of racemic epoxides, under mild and environmentally friendly conditions. The methodology MNP-immobilized enzyme developed in the synthesis of chiral diols has a potential for use in large-scale applications.
1. Introduction
Chiral compounds play an important role in both chemical and biotech industries. Among the optically active compounds, enantiopure epoxides and their corresponding vicinal diols are important intermediates for pharma, agro and fine chemical industries. Though there are several chemical methodologies for the resolution of epoxides, yet they have their own limitations in terms of efficiency and enantioselectivity. Enzymes/Biocatlysts have been used in the synthesis of chiral alcohols of biological importance. This strategy constitutes an attractive “green chemistry” alternative to the existing chemical methodologies [1-6]. Several epoxide hydrolases (EHs) and Horse radish peroxidase (HRP) have recently been recognized as versatile biocatalysts in synthesis of enantiopure epoxides and their diols. They exhibit high enantioselectivity with broad substrate specificity and these enzymes do not require any cofactors in aqueous/buffer solution [7-9]. However, the use of biocatalysts in the industrial application is limited due to the difficulties associated with the enzyme stability and reuse. Therefore, immobilization of the biocatalysts on certain supports tends to be an efficient solution for the problems associated with the use of free or soluble enzymes [10]. There are many organic and inorganic carriers available to immobilize the biocatalysts. Among them, magnetite (Fe3O4) nanoparticles (MNPs) exhibit superparamagnetic properties therefore surface modification of such magnetic nanoparticles can make improvements in their surface properties, which may be useful for enhancing the activity and stability of the enzyme [11-14]. The large surface-area-to-volume ratio of magnetic nanoparticles allows serving as an efficient carrier of biocatalyst and also helps in separation of enzymes from the reaction medium using external magnet, thus the magnetic nano particles were employed as supports for the immobilization of the enzymes [15-18].
Therefore, the aim of our present study is to immobilize the enzyme Horseradish peroxidase onto amine functionalized magnetic nanoparticles for the synthesis of chiral vicinal diols from the racemic epoxides. The immobilization of the enzyme on MNPs was characterized by TEM and SEM studies. The enantiomeric excess and yield of the products were confirmed by optical rotation and NMR spectral studies.
2. Results and Discussion
The hydrolysis of the racemic epoxide was carried out with both free and immobilized enzyme and their respective yields are presented in Table 1. The structures of the products were confirmed by NMR, LC-MS and IR spectroscopy. The recovered MNPs bound HRP enzyme had shown 5% decrease in enzyme activity. Though the recycled enzyme showed good enantioselectivity, the yields were, gradually decreased to 5% - 10% in each cycle. Mono-substituted racemic aryl epoxides were resolved using MNP-bound peroxidase to obtain the (R)-diol, thus the process of enzymatic hydrolysis of the racemic epoxide show (R)-selectivity.
Thus the results show that, the peroxidase boundMNPs can be applied to large-scale reactions with acceptable yields and enantio-purity in ecofriendly environment. The above enzyme-immobilized MNPs methodology may be applicable in the kinetic resolution of a broad range of racemic epoxides in obtaining chiral diols and enantiopure epoxides.
3. Conclusion
Enzymes have long been used in the industry as catalysts in process for production of chiral alcohols of biological interest. MNP-enzyme conjugates (MNP-Es) represent a successful application of immobilized enzyme in the synthesis of value added chiral compounds of biological interest. In this study it is conclude that the enzyme horseradish peroxidase enzyme was immobilized on amine functionalized magnetic nanoparticles in synthesis of enantio pure vicinal diols from corresponding racemic epoxides in environmentally friendly conditions. Thus the MNP-Es methodology developed in synthesis of chiral alcohols has the potential for industrial application.
4. Experimental
4.1. General
All chemicals (FeSO4·7H2O, Fe2(SO4)3, ammonium hydroxide, tetraethylorthosilicate, (3-aminopropyl)-triethoxysilane, glutaraldehyde, Horse radish peroxidase; EC 1.11.1.7) were purchased from Sigma Aldrich. The racemic epoxides used in the study were synthesized in our laboratory. Melting points were measured on MettlerTemp apparatus; uncorrected. IR Spectra were recorded with a Perkin-Elmer-1600 FT-IR spectrometer in KBr; υ in cm−1.1H-NMR spectra were recorded on Gemini-200 spectrometer; in CDCl3. Mass spectra were recorded on ESI-MS: Agilent 6510 Q-TOF LC/MS instrument, ESIMS: 7070H spectrometer with a direct inlet system.
4.2. Preparation of Fe3O4 (Magnetite) Nanoparticles
Magnetic nanoparticles were prepared by co-precipitation method [19-21]. FeSO4·7H2O (2.78 g) and Fe2(SO4)3 (4.0 g) were dissolved in water and ammonium hydroxide (25%) was added slowly to adjust the alkaline pH of the solution. The reaction mixture was then continually stirred for 1 h at 50˚C under nitrogen atmosphere till black magnetite (Fe3O4) particles were formed and the mixture was cooled down to room temperature.
4.3. Synthesis of Silica Coated Fe3O4 Nanoparticles
To prepare silica coated Fe3O4 nanoparticles, tetraethyl orthosilicate (TEOS) was readily added to the above solution and vigorously stirred for 18 h under nitrogen atmosphere. Then the silica coated Fe3O4 nanoparticles were separated and washed with water until the solution pH reached to neutral and finally the particles were vacuum dried.
4.4. (3-Aminopropyl) Triethoxysilane Functionalization on (-Fe2O3 @ SiO2
To prepare (3-aminopropyl)triethoxysilane functionalized g-Fe2O3@SiO2, 3 g of g-Fe2O3@SiO2 was dispersed in dry toluene under N2 atmosphere, to which 2 mL of (3-aminopropyl)triethoxysilane was added and stirred at reflux temperature for 24 h and the material was collected with external magnet, washed with isopropanol and dried under vacuum.
4.5. Immobilization of HRP Enzyme onto Magnetic Nanoparticles and Synthesis of Chiral Diols
For immobilization of the enzyme: MNPs (50 mg) were washed with phosphate buffer of pH 6.0 and 1% (v/v) of glutaraldehyde solution was added under stirring for 1 h.
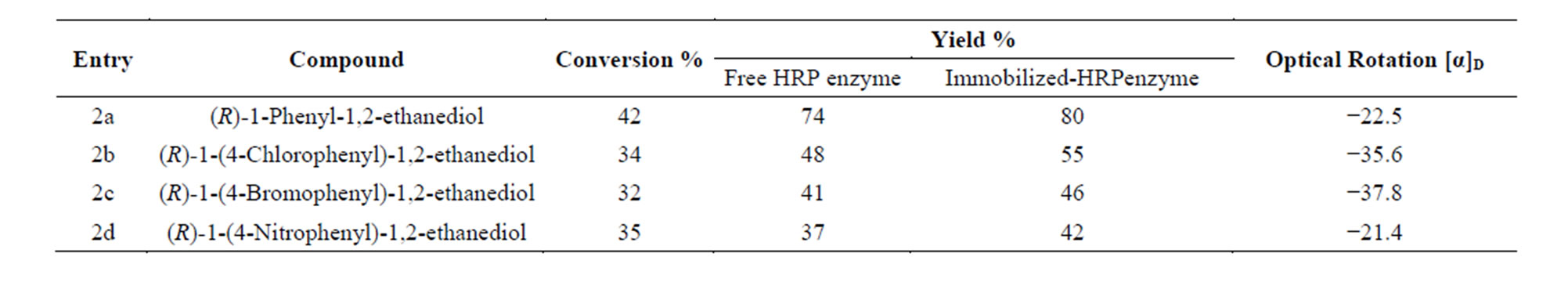
Table 1. Synthesis of chiral vicinal diols using immobilized enzyme.
The cross-linker treated MNPs were washed thoroughly with phosphate buffer to remove the un-reacted aldehyde. 4 mL (0.5 mg/mL) of HRP enzyme solution was then added to glutaraldehyde treated MNPs in 6 mL of phosphate buffer and stirred gently for 2 - 3 h to immobilize the enzyme [22-24]. The immobilized enzyme solution was stored in buffer solution at 4˚C until use. The size of nanoparticles and immobilization of the HRP onto magnetic nanoparticles was characterized using TEM and SEM studies (Figure 1). The activity of the HRP enzyme before and after immobilization was determined by pyrogallol method [25].
To the enzyme-immobilized onto MNPs taken in buffer, was added 1 mol of racemic epoxide and incubated for optimum conversion period. The product (diol) formation was monitored using TLC at regular intervals of time (Scheme 1). Upon completion of the reaction, the enzyme (HRP) bound to the magnetic nanoparticles was separated by using external magnet and thus retained MNP-bound enzyme was re-suspended in the buffer solution containing styrene epoxide for further kinetic resolution. The reaction mixture was separated and extracted with organic solvent and the product was purified using column chromatography.
4.6. (R)-1-Phenyl-1,2-Ethanediol (2a)
White solid., m. p. = 58˚C - 60˚C. [a]D25= −22.5 (c, 1.0, Ethanol). 1H NMR (200 MHz, CDCl3): d = 2.63 (brs, 1H), 3.05 (brs, 1H), 3.55 - 3.70 (m, 2H), 4.73 - 4.77 (dd, 1H, J = 3.0, 8.3 Hz), 7.24 - 7.31 (m, 5H. IR (KBr): n = 3315, 2925, 1600, 1454, 752 cm-1; MS: m/z = (M+, 138).
4.7. (R)-1-(4-Chlorophenyl)-1,2-Ethanediol (2b)
White solid, m.p. = 73˚C - 75˚C. [a]D25 = −35.6 (c, 1.0, CHCl3). 1H NMR (CDCl3, 200 MHz): d = 2.0 (brs, 1H), 2.62 (brs, 1H), 3.65 (dd, 1H, J = 8.1, 11.1 HZ), 3.71 (dd, 1H, J = 3.7, 11.1 Hz), 4.77 (dd, 1H, J = 3.7, 8.1 Hz) 7.28 - 7.30 (m, 4 H). IR (KBr): n 3369, 2923, 1595, 1511, 459, 829, 892 cm−1. MS: m/z = (M+, 172).
4.8. (R)-1-(4-Bromophenyl)-1,2-Ethanediol (2c)
White solid, m.p = 94˚C - 96˚C. [a]D25 = −37.8 (c, 1.0, CHCl3). 1H NMR (CDCl3, 200 MHz): d = 1.88 (brs, 1H), 2.42 (brs, 1H), 3.55 (dd, 1H, J = 8.0, 11.0 Hz), 3.70 (dd, 1H,

Figure 1. TEM and SEM images of magnetic nanoparticles. TEM (A & B) and SEM (C & D): (A & C) amine functionalized silica coated magnetic nanoparticles; (B & D) HRP enzyme immobilized MNPs.
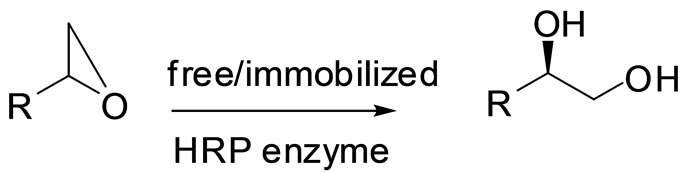
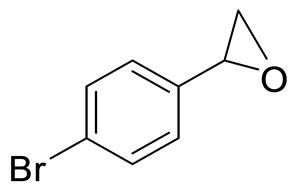
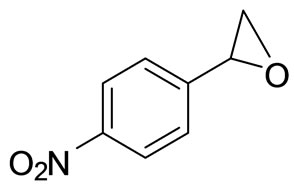
Scheme 1. Synthesis of vicinal diols.
J = 3.6, 11.0 Hz), 4.74 (dd, 1H, J = 3.6, 8.0 Hz), 7.22 (d, 2H, J = 8.8 Hz), 7.45 (d, 2H, J = 8.8 Hz). IR (KBr): n 3368, 2918, 1588, 1487, 894, 830 cm−1. MS: m/z = (M+, 216).
4.9. (R)-1-(4-Nitrophenyl)-1,2-Ethanediol (2d)
Light yellow solid. m.p = 70˚C - 72˚C. [a]D25 = −21.4 (c = 1.0, MeOH). 1H NMR (CDCl3, 200 MHz): d = 3.48 - 3.59 (m, 2H, CH2), 4.33 (m, OH), 4.77 (m, OH), 5.07 - 5.08 (m, 1H, CH), 7.58 (d, 2H, J = 9.0 HZ), 7.58 (d, 2H, J = 9.0 HZ), 8.14 (d, 2H, J = 8.2 HZ). IR (KBr): n 3304, 2932, 1603, 1514, 854 cm−1. MS: m/z = (M+, 183).
Acknowledgements
S. S. D thanks to CSIR, New Delhi, India for financial support for this project. B.V.S and A.B.R thanks CSIR, New Delhi for financial support as a part of XII five year plan program under title ORIGIN (CSC-0108) and Biocatalysis… industrial applications (CSC-0106).
REFERENCES
- W. J. Choi, “Biotechnological Production of Enantiopure Epoxides by Enzymatic Kinetic Resolution,” Applied Microbiology and Biotechnology, Vol. 84, No. 2, 2009, pp. 239-247. http://dx.doi.org/10.1007/s00253-009-2110-9
- D. Sareen and R. Kumar, “Prospecting for Efficient Enantioselective Epoxide Hydrolases,” Indian Journal of Biotechnology, Vol. 10, 2011, pp. 161-177. http://nopr.niscair.res.in/bitstream/123456789/11459/1/IJBT%2010(2)%20161-177.pdf
- C. Mateo, A. Archelas, R. Fernandez-Lafuente, J. M. Guisanb and R. Furstoss, “Enzymatic Transformations. Immobilized A. niger Epoxide Hydrolase as a Novel Biocatalytic Tool for Repeated-Batch Hydrolytic Kinetic Resolution of Epoxides,” Organic & Biomolecular Chemistry, Vol. 1, No. 15, 2003, pp. 2739-2743. http://pubs.rsc.org/en/content/articlepdf/2003/OB/B303307D
- W. Adam, M. Lazarus, C. R. Saha-Möller, O. Weichold, U. Hoch, D. Häring and P. Schreier, “Biotransformations with Peroxidases,” Advances in Biochemical Engineering/ Biotechnology, Vol. 63, 1999, pp. 73-108. http://dx.doi.org/10.1007/3-540-69791-8_4
- H. Lin, J. Y. Liu, H. B. Wang, A. A. Q. Ahmed and Z. L. Wu, “Biocatalysis as an Alternative for the Production of Chiral Epoxides: A Comparative Review,” Journal of Molecular Catalysis B: Enzymatic, Vol. 72, No. 3-4, 2011, pp. 77-89. http://dx.doi.org/10.1016/j.molcatb.2011.07.012
- Y. Genzel, A. Archelas, Q. B. Broxterman, B. Schulze and R. Furtoss, “Microbiological Transformations. 47. A Step toward a Green Chemistry Preparation of Enantiopure (S)-2-,3-Pyridyloxirane via an Epoxide Hydrolase Catalyzed Kinetic Resolution,” The Journal of Organic Chemistry, Vol. 66, No. 2, 2001, pp. 538-543. http://dx.doi.org/10.1021/jo001406x
- S. Colonna, N. Gaggero, C. Richelmi and P. Pasta, “Recent Biotechnological Developments in the Use of Peroxidases,” Trends in Biotechnology, Vol. 17, No. 4, 1999, 163-168. http://dx.doi.org/10.1016/S0167-7799(98)01288-8
- A. J. Carlsson, P. Bauer, H. Ma and M. Widersten, “Obtaining Optical Purity for Product Diols in Enzyme Catalyzed Epoxide Hydrolysis: Contributions from Changes in Both Enantioand Regioselectivity,” Biochemistry, Vol. 51, No. 38, 2012, pp. 7627-7637. http://dx.doi.org/10.1021/bi3007725
- N. S. Finney, “Enantioselective Epoxide Hydrolysis: Catalysis Involving Microbes, Mammals and Metals,” Chemistry and Biology, Vol. 5, No. 4, 1998, pp. 73-79. http://dx.doi.org/10.1016/S1074-5521(98)90630-5
- H. C. Zhou, W. Li, Q. H. Shou, H. S. Gao, P. Xu, F. L. Deng and H. Z. Liu, “Immobilization of Penicillin G Acylase on Magnetic Nanoparticles Modified by Ionic Liquids,” Chinese Journal of Chemical Engineering, Vol. 20, No. 1, 2012, pp. 146-151. http://dx.doi.org/10.1016/S1004-9541(12)60374-7
- P. A. Johnson, H. J. Park and A. J. Driscoll, “Enzyme Nanoparticle Fabrication: Magnetic Nanoparticle Synthesis and Enzyme Immobilization,” Methods in Molecular Biology, Vol. 679, 2011, pp. 183-191. http://www.ncbi.nlm.nih.gov/pubmed/20865397 http://dx.doi.org/10.1007/978-1-60761-895-9_15
- Y. Ren, J. G. Rivera, L. He, H. Kulkarni, D. K. Lee and P. B. Messersmith, “Facile, High Efficiency Immobilization of Lipase Enzyme on Magnetic Iron Oxide Nanoparticles via Biomimetic Coating,” BMC Biotechnology, Vol. 11, 2011, pp. 63-70. http://dx.doi.org/10.1186/1472-6750-11-63
- A. Dyal, K. Loos, M. Noto, S. W. Chang, C. Spagnoli, K. V. P. M. Shafi, A. Ulman, M. Cowman and R. A. Gross, “Activity of Candida rugosa Lipase Immobilized on g-Fe2O3 Magnetic Nanoparticles,” Journal of the American Chemical Society, Vol. 125, No. 7, 2003, pp. 1684- 1685.
- C. G. C. M. Netto, H. E. Tomo and L. H. Andrade, “Superparamagnetic Nanoparticles as Versatile Carriers and Supporting Materials for Enzymes,” Journal of Molecular Catalysis B: Enzymatic, Vol. 85-86, 2013, pp. 71-92. http://dx.doi.org/10.1016/j.molcatb.2012.08.010
- L. Zhou, J. Yuan and Y. Wei, “Core-Shell Structural Iron Oxide Hybrid Nanoparticles: From Controlled Synthesis to Biomedical Applications,” Journal of Materials Chemistry, Vol. 21, 2011, pp. 2823-2840. http://dx.doi.org/10.1039/c0jm02172e
- A. K. Johnson, A. M. Zawadzka, L. A. Deobald, R. L. Crawford and A. J. Paszczynski, “Novel Method for Immobilization of Enzymes to Magnetic Nanoparticles,” Journal of Nanoparticle Research, Vol. 10, 2008, pp. 1009-1025. http://dx.doi.org/10.1007/s11051-007-9332-5
- H. M. R. Gardimalla, D. Mandal, P. D. Stevens, M. Yen and Y. Gao, “Superparamagnetic Nanoparticle-Supported Enzymatic Resolution of Racemic Carboxylates,” Chemical Communications, No. 35, 2005, pp. 4432-4434. http://dx.doi.org/10.1039/b504128g
- J. Wang, G. Meng, K. Tao, M. Feng, X. Zhao, Z. Li, H. Xu, D. Xia and J. R. Lu, “Immobilization of Lipases on Alkyl Silane Modified Magnetic Nanoparticles: Effect of Alkyl Chain Length on Enzyme Activity,” Plos One, Vol. 7, No. 8, 2012, Article ID: e43478. http://dx.doi.org/10.1371/journal.pone.0043478
- F. Mahdizadeh, A. Karimi and L. Ranjbarian, “Immobilization of Glucose Oxidase on Synthesized Superparamagnetic Fe3O4 Nanoparticles; Application for Water Deoxygenation,” International Journal of Scientific & Engineering Research, Vol. 3, No. 5, 2012, pp. 1-6. http://www.ijser.org/researchpaper
- J. Xu, C. Ju, J. Sheng, F. Wang, Q. Zhang, G. Sun and M. Sun, “Synthesis and Characterization of Magnetic Nanoparticles and Its Application in Lipase Immobilization,” Bulletin of the Korean Chemical Society, Vol. 34, No. 8, 2013, pp. 2408-2412. http://dx.doi.org/10.5012/bkcs.2013.34.8.2408
- X. Zheng, S. Luo, L. Zhang and J. P. Cheng, “Magnetic Nanoparticle Supported Ionic Liquid Catalysts for CO2 Cycloaddition Reactions,” Green Chemistry, Vol. 11, No. 4, 2009, pp. 455-458. http://dx.doi.org/10.1039/b823123k
- K. Khoshnevisana, A. K. Bordbarb, D. Zarec, D. Davoodic, M. Noruzic, M. Barkhic and M. Tabatabaei, “Immobilization of Cellulose Enzyme on Superparamagnetic Nanoparticles and Determination of Its Activity and Stability,” Chemical Engineering Journal, Vol. 171, No. 2, 2011, pp. 669-673. http://dx.doi.org/10.1016/j.cej.2011.04.039
- Y. Kim, I. Lee, S. Choi, O. Lee, J. Shim, J. Lee, J. Kim abd E. Y. Lee, “Enhanced Stability and Reusability of Marine Epoxide Hydrolase Using Ship-in-a-Bottle Approach with Magnetically-Separable Mesoporous Silica,” Journal of Molecular Catalysis B: Enzymatic, Vol. 89, 2013, pp. 48-51. http://dx.doi.org/10.1016/j.molcatb.2012.12.012
- K. Pospiskova and I. Safarik, “Low-Cost, Easy-to-Prepare Magnetic Chitosan Microparticles for Enzymes Immobilization,” Carbohydrate Polymers, Vol. 96, No. 2, 2013, pp. 545-548. http://www.ncbi.nlm.nih.gov/pubmed/23768599 http://dx.doi.org/10.1016/j.carbpol.2013.04.014
- B. Chance and A. C. Maehly, “Assay of Catalases and Peroxidases,” Methods in Enzymology, Vol. 2, 1955, pp. 773-775.
NOTES
*Corresponding author.

