Open Journal of Urology
Vol.05 No.08(2015), Article ID:58862,4 pages
10.4236/oju.2015.58019
Cavernous Nerve Graft Reconstruction with a Novel Artificial Conduit during Robot-Assisted Laparoscopic Radical Prostatectomy
Yoshiyuki Matsui*, Toshinari Yamasaki, Takahiro Inoue, Tomomi Kamba, Osamu Ogawa
Department of Urology, Graduate School of Medicine, Kyoto University, Kyoto, Japan
Email: *ym1108@kuhp.kyoto-u.ac.jp
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 21 June 2015; accepted 15 August 2015; published 18 August 2015
ABSTRACT
The interposition sural nerve graft has been attempted occasionally during radical prostatectomy for the recovery of continence and erectile function; however, nerve autograft may result in adverse events for the patient. Here, we present our initial experiences using NerbridgeTM, a novel conduit for peripheral nerve regeneration, rather than utilizing sural nerve grafting, in robot-as- sisted laparoscopic radical prostatectomy to overcome autograft problems such as prolongation of operation time and postoperative abnormal sensation. This novel artificial conduit interposition can be technically feasible when combined with robotic surgery, and prospective randomized con- trolled trials with high patients-numbers and long follow-up periods are warranted.
Keywords:
Robot-Assisted Laparoscopic Radical Prostatectomy (RALP), Nerbridge, Cavernous Nerve, Graft Reconstruction, Erectile Function

1. Introduction
Radical prostatectomy (RP) is considered a safe and effective treatment for localized prostate cancer; however, the risk of urinary incontinence and erectile dysfunction are significant disadvantages compared to other modalities. Preservation of the cavernous nerves during RP is one of the most important points for the recovery of continence and erectile function, yet is not always appropriate in men with high grade tumors or relatively advanced disease extending toward the neurovascular bundles (NVBs).
The use of interposition sural nerve graft (SNG) instead of nerve sparing has been attempted in such patients, and some investigators have reported its effectiveness in the recovery of continence and erectile function [1] -[3] . In particular, the introduction of robot-assisted laparoscopic radical prostatectomy (RALP) has provided technical advantages for nerve grafting due to its ability to allow easier intracorporeal thin suturing and knot tying, and its improved 3-dimentional visualization with 10-fold magnification. Presently, SNG is now easily performed by only an urologist with robotic assistance [4] ; yet nerve autograft, which requires the extraction of a peripheral nerve from a healthy portion of the patient’s own body, carries the risk of adverse event such as prolongation of operation time and postoperative abnormal sensation. In the present report, NerbridgeTM was utilized as a conduit for peripheral nerve regeneration, rather than SNG in robotic surgery, to attempt to overcome the disadvantages associated with autografting.
2. Case Presentation
NerbridgeTM grafting was performed in three patients with clinically organ-confined prostate cancer who received RALP with or without lymphadenectomy. No patients had a history of diabetes mellitus. Cavernous nerve preservation was determined based on the results of the biopsy data. All patients received ipsilateral wide resection of the NVB on the side where greater than 33% of the biopsy cores were positive, and the contralateral NVB were preserved according to our surgical criteria [5] . Among the patients who had high sexual activity preoperatively based on IIEF-scores, three patients agreed with the use of NerbridgeTM for cavernous nerve reconstruction, which was approved by Japanese National Health Insurance.
3. Surgical Technique
RALP was performed as described previously [6] . Before resection of the NVB, electrostimulation of the bundle was performed. A monopolar stimulating needle electrode (Nippon Kohden) was placed at the proximal end of the NVB. Electrical stimulation was maintained for 15 to 20 seconds at 50 mA/10Hz with a monophasic rectangular pulse and pulse duration of 1.0 ms. The response to the electrical stimulation was observed as the change in cavernous pressure measured by a specific pressure-monitoring urethral balloon inserted into the middle portion of the penile urethra. An increase in pressure of >5 cm H2O was considered a ‘positive’ response [7] . The proximal end of the NVB was marked by 3-0 polyglactin suture and en-bloc dissection of the NVB was subsequently performed. The distal end of the NVB was also detected at the time of urethral dissection, and marked by a metal clip. Electrical stimulation was performed before the dissection of the bladder neck in the first case, and afterwards in the latter two cases, because the NVB was more easily observed when the prostate was pulled up to the opposite side.
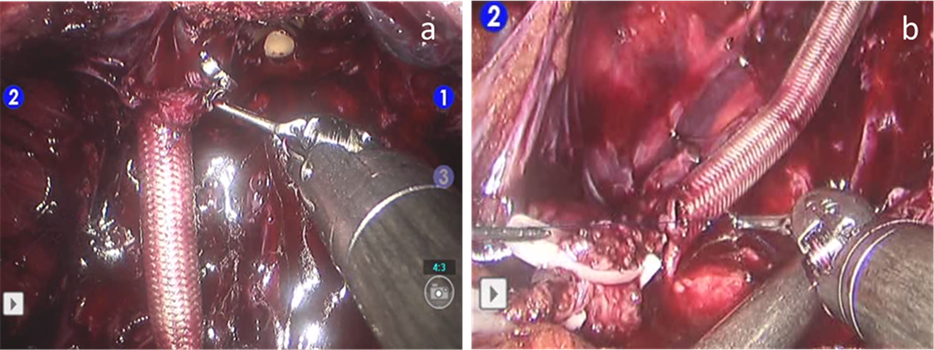
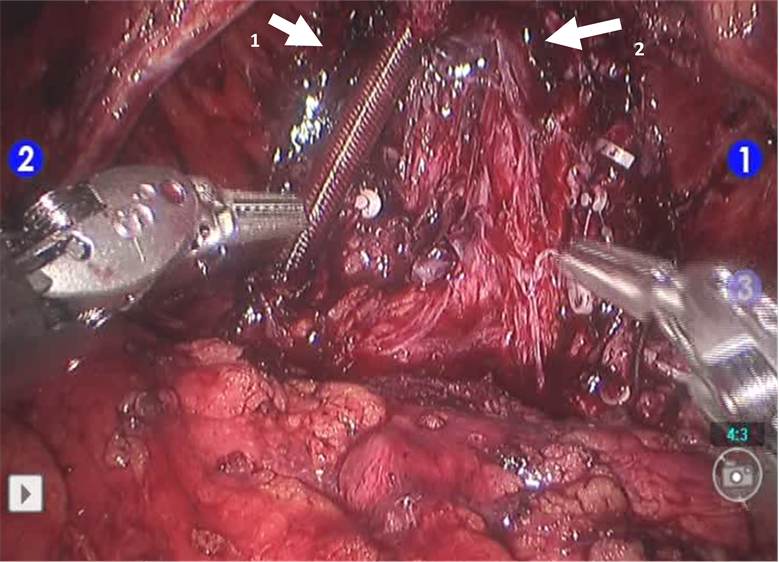
After resection of the prostate, the surgical field was inspected, and both the proximal and distal ends of one of the cavernous nerves in the NVB was identified. The length of the 4 mm-diameter NerbridgeTM was adjusted to the length of the dissected nerve. Using fine needle drivers and the ultrafine mode of the robotic surgical system, anastomosis was completed using two 6-0 polypropylene mattress sutures on each end. Both ends of the nerve were pulled into the NerbridgeTM conduit for a length of 2 to 3 mm (Figure 1 and Figure 2). After the completion of grafting, Rocco’s stitch and vesicourethral anastomosis were performed.
4. Results
All procedures were successfully completed using the robotic system in all three patients. For the interposition of the resected cavernous nerve, unilateral NerbridgeTM grafting was performed in all patients. The results of the surgeries are shown in Table 1. After changing the timing of nerve stimulation (case 2 and case 3), the required time for nerve stimulation was shortened. The final pathologic stage was pT2c in cases 1 and 3, pT3a in case 2. The excised segment of the NVB was histologically identified on the grafting side. Foley catheters were removed 5 days postoperatively in cases 1 - 3, but was removed after 20 days in case 2 due to urinary leakage from the vesicourethral anastomosis. There were no complications associated with the use of NerbridgeTM grafting. Postoperatively, PSA declined to less than 0.008 ng/ml in all patients.
No patients have yet had the need for PDE5 inhibitor. Case 2 patient recovered his potency just one month postoperatively, which was considered not because of the effect of Nerbridge but because of contralateral spared cavernous nerve from the shortness of recovery period.
Figure 1. The anastomosis of NerbridgeTM was completed using two 6-0 polypropylene mattress sutures on distal (a) and proximal end (b). Both ends of the nerve were pulled into NerbridgeTM conduit for the length of 2 to 3 mm.
Figure 2. The final appearance of left-sided nerve interposition by NerbridgeTM (arrow 1). Right-sided NVB was preserved (arrow 2).

Table 1. Patients’ characteristics and operative records.
*Corresponding author.
SNG for the interposition of bilateral or unilateral cavernous nerves has been used to improve the recovery of erectile function after RP, although the published data on cavernous nerve graft reconstruction has been controversial [1] -[3] [8] . Several non-randomized prospective and retrospective studies or case reports with small numbers of subjects reported the benefits of SNG; however, Davis et al. showed in a randomized phase II trial that addition of unilateral SNG to a unilateral nerve-sparing RP did not result in a 50% relative improvement in potency at 2 years [8] . On the other hand, as suggested by Saito et al., because interposition nerve grafting may contribute to the recovery of urinary function as well as sexual function, nerve grafting may be beneficial for some patients to promote nerve regeneration.
Disadvantages of SNG include prolongation of operation time for nerve grafting, and risk of postoperative complications such as numbness, hypesthesia, and anesthesia of the lower limbs, resulting in a decrease in quality of life (QOL). To avoid these disadvantages, NerbridgeTM, a conduit for peripheral nerve regeneration, was utilized in the present study. This novel conduit utilizes a newly developed medical-grade collagen (NMP Collagen PS, produced by Nippon Meat Packers, Inc. (NMP)), which is applied to the inside of a polyglycolic acid conduit, resulting in an increase in angiogenesis within the conduit and thus providing the necessary nourishment for the regeneration and growth of peripheral nerves. Furthermore, NerbridgeTM dissolves and is absorbed by the patient’s body in approximately 3 months. A clinical trial for the treatment of peripheral nerve injury using NerbridgeTM conduits showed that it was equally effective or more effective (as defined by the recovery of sensation and other attributes) than previous nerve autograft and nerve suturing techniques.
However, this is just the preliminary results of our initial experiences with NerbridgeTM , and there are some limitations to discuss the usefulness of NerbridgeTM. All follow-up were still less than one year, and longer-follow-up period and high patients-numbers would be required to see the effect. The neurovascular bundle consists of a visceral nerve plexus with different autonomous nerve fibers, which first have to build Schwann cell canals and find their way into the graft. The growth under these circumstances is not comparable to peripheral nerve system. In addition we don’t know what the influence of newly developed collagen material with its angiogenesis stimulus and absorption after 3 months on visceral nerve fibers could be.
Further, Takenaka et al. recently suggested that cavernous nerves run in a spray-like fashion forming the pelvic splanchnic nerves to the prostate [9] . We may need to optimize the shape and length of NerbridgeTM, which can cover a wider neural fibers and effectively assist to extension of cavernous nerves.
6. Conclusion
In conclusion, cavernous nerve graft reconstruction with this novel artificial conduit interposition can be technically feasible when combined with robotic surgery. It may contribute to the improvement in QOL of patients treated by RP, and prospective randomized controlled trials with high patients-numbers and long follow-up periods are warranted.
Cite this paper
YoshiyukiMatsui,ToshinariYamasaki,TakahiroInoue,TomomiKamba,OsamuOgawa, (2015) Cavernous Nerve Graft Reconstruction with a Novel Artificial Conduit during Robot-Assisted Laparoscopic Radical Prostatectomy. Open Journal of Urology,05,118-122. doi: 10.4236/oju.2015.58019
References
- 1. Namiki, S., Saito, S., Nakagawa, H., Sanada, T., Yamada, A. and Arai, Y. (2007) Impact of Unilateral Sural Nerve Graft on Recovery of Potency and Continence Following Radical Prostatectomy: 3-Year Longitudinal Study. Journal of Urology, 78, 212-216; Discussion, 216.
http://dx.doi.org/10.1016/j.juro.2007.03.043 - 2. Saito, S., Namiki, S., Numahata, K., Satoh, M., Ishidoya, S., Ito, A., et al. (2007) Impact of Unilateral Interposition Sural Nerve Graft on the Recovery of Sexual Function after Radical Prostatectomy in Japanese Men: A Preliminary Study. International Journal of Urology, 14, 133-139.
http://dx.doi.org/10.1111/j.1442-2042.2007.01699.x - 3. Sim, H.G., Kliot, M., Lange, P.H., Ellis, W.J., Takayama, T.K. and Yang, C.C. (2006) Two-Year Outcome of Unilateral Sural Nerve Interposition Graft after Radical Prostatectomy. Urology, 68, 1290-1294.
http://dx.doi.org/10.1016/j.urology.2006.08.1064 - 4. Kaouk, J.H., Desai, M.M., Abreu, S.C., Papay, F. and Gill, I.S. (2003) Robotic Assisted Laparoscopic Sural Nerve Grafting during Radical Prostatectomy: Initial Experience. Journal of Urology, 170, 909-912. http://dx.doi.org/10.1097/01.ju.0000073208.18059.62
- 5. Mizuno, K., Inoue, T., Miyazaki, Y., Makino, Y., Terada, N., Kobayashi, T., et al. (2014) Development of a Preoperative Criterion to Select Candidates for Nerve-Sparing Radical Prostatectomy at Kyoto University Hospital. Hinyokika Kiyo, 60, 543-547.
- 6. Montorsi, F., Wilson, T.G., Rosen, R.C., Ahlering, T.E., Artibani, W., Carroll, P.R., et al. (2012) Best Practices in Robot-Assisted Radical Prostatectomy: Recommendations of the Pasadena Consensus Panel. European Urology, 62, 368-381. http://dx.doi.org/10.1016/j.eururo.2012.05.057
- 7. Terada, N., Arai, Y., Kurokawa, K., Ohara, H., Ichioka, K., Matui, Y., et al. (2003) Intraoperative Electrical Stimulation of Cavernous Nerves with Monitoring of Intracorporeal Pressure to Confirm Nerve Sparing during Radical Prostatectomy: Early Clinical Results. International Journal of Urology, 10, 251-256.http://dx.doi.org/10.1046/j.1442-2042.2003.00614.x
- 8. Davis, J.W., Chang, D.W., Chevray, P., Wang, R., Shen, Y., Wen, S., et al. (2009) Randomized Phase II Trial Evaluation of Erectile Function after Attempted Unilateral Cavernous Nerve-Sparing Retropubic Radical Prostatectomy with versus without Unilateral Sural Nerve Grafting for Clinically Localized Prostate Cancer. European Urology, 55, 1135-1143. http://dx.doi.org/10.1016/j.eururo.2008.08.051
- 9. Takenaka, A., Murakami, G., Soga, H., Han, S.H., Arai, Y. and Fujisawa, M. (2004) Anatomical Analysis of the Neurovascular Bundle Supplying Penile Cavernous Tissue to Ensure a Reliable Nerve Graft after Radical Prostatectomy. Journal of Urology, 172, 1032-1035.
http://dx.doi.org/10.1097/01.ju.0000135648.33110.df
NOTES

*Corresponding author.