Advances in Chemical Engineering and Science
Vol. 2 No. 3 (2012) , Article ID: 20825 , 7 pages DOI:10.4236/aces.2012.23039
Application of Polymeric Membrane in CO2 Capture from Post Combustion
University of Maryland, College Park, Maryland, USA
Email: *wangshuangzhen@gmail.com
Received April 27, 2012; revised May 28, 2012; accepted June 8, 2012
Keywords: Polymeric Membrane; CO2 Capture; Hollow Fiber Membrane; Selectivity; Membrane Wetting
ABSTRACT
CO2 capture from post combustion does not need significant alteration of the current power generation facilities and is therefore of more interests to the research and industrial circles. Polymeric membrane separations, which are based mainly on physical phenomena, are easy for operation and to scale up. The details and future research trends are covered in this most updated review, which serve as an excellent technique reference for the research circle and technology evaluation for the related industrial circle.
1. Introduction
Since the industrial revolution took place in the middle 18th century, 321 billion tons of CO2 has been released to the atmosphere and half of the CO2 emission has taken place in the recent 30 years (1971 to 2002) [1,2]. Therefore, the increasingly intensive energy requirement, which comes mainly from the fossil fuel combustion, accounts for the significant atmospheric CO2 increase and thus potentially the global climate change concerns.
While renewable energy will reduce the CO2 emission in the long term, by switching the energy resource from the current fossil fuel based toward to emerging solar, biomass, wind and tides, CO2 capture and sequestration from the large stationary coal/natural fired power plants, cement plants, oil refineries and steel plants, will reduce CO2 emission in the intermediate time span. Therefore, CO2 capture from the post combustion facilities, which does not need significant modification of the current industrial infrastructures, has aroused more research interests and industrial attentions.
The easy operation, reliable performance (based mainly on physical phenomena) and easy scale up has made polymer membrane as favorite media to capture CO2 from post combustion [3]. Historically, polymer membrane has been applied successfully in ammonia synthesis/purge, petrochemical/refinery and CO2 separation on industrial scale in the recent 30 years. The latter mainly includes natural sweetening, CO2 recovery from land fill gas (biogas) and Enhanced Oil Recovery (EOR/natural gas processing) [4]. Therefore, the 30-year’s commercial success strongly suggests polymer membrane’s potential for CO2 capture from flue gas [5-7].
2. Systematic Optimizations
Polymer membrane applied in gas separation falls mainly into three categories: 1) physically selective membrane; 2) hollow fiber membrane; and 3) facilitated transport. They have some common advantages and disadvantages. Compared to packed/tray columns, membrane operations are more flexible, economic, linear scale up, predictable, compacted and higher mass transfer rate per volume [3,8]. However, compared to the liquid adsorbent scrubbing (amine for example), membrane separation consumes more energy, has the low driving force due to the low CO2 concentration in flue gas, smaller flow rate (laminar) and higher transport resistance; in addition, the high temperature and the potential fouling membrane of flue gas are also major concerns, which need to be addressed adequately for CO2 capture [3,9,10].
To improve membrane’s performance, pressure, vacuum and dual/multi stages of membrane are exerted or simulated to reach the competitive performance and minimum cost of CO2 capture [7,9-17].
2.1. Pressure and Vacuum
Pressure and vacuum have been introduced to membrane for CO2 capture improvement. In a modeling followed with validated lab tests, the CO2 capture efficiency of 90% was achieved with gas pressure increase [17]. However, pressure increase has some potential drawbacks. Pressure increase could cause significant decrease of permeability and selectivity [18] and significant power loss (for example, as much as 20% of the power plant output for 5 bar compression) due to the dilute concentration of CO2 in the flue gas [7]. In comparison, under lab vacuum permeate conditions, a cost of $54/ton CO2 captured was avoided, which is equivalent to 2/3 capture cost of a pressurized bed [12]. Although popular lab scale vacuum for CO2 capture with membrane is cost effective, practically, vacuum as low as 0.2 baris only available for large scale CO2 capture [7]. Therefore, an adequate combination of pressure and vacuum seems to improve the membrane’s performance for CO2 capture and hold a potentially competitive cost benefit. The lab experimental results of pressure (1.5 bar) and vacuum (0.05 bar) combination [13] and modeling [10] are in favor of this prediction.
2.2. Dual/Multistage Membrane
Dual stage membrane has been introduced to enhance driving force and increase purity of captured CO2. Two staged pressure membrane (with 4 bar pressure at the first stage) was modeled and a cost of $57/ton CO2 captured was avoided with final CO2 concentration of 90% [18,19]. Two staged vacuum membrane was simulated to achieve 90% capture efficiency at a cost of $39/ton CO2 captured [7]. However, since multi staged membrane consumes more energy and its facility cost is more than those of a single stage one [15], a compromise between the stage number of membranes and the capture efficiency has to be taken into consideration.
2.3. Membrane Length and Module Number
Similar to vacuum, pressure and multi stage factors, optimization of membrane length and number of membrane module can also contribute to high selectivity. Like the scenario in distillation tower, making membrane longer can only improve CO2 capture when the membrane module is shorter than the maximum effective module length Leff [20]. Therefore, there is a balance between number of parallel modules (more of the membrane areas with higher driving force than the according length increase however with more cost of module manufacture) and the according cost [21].
2.4. Scale up and Capture Cost
Polymer membrane for CO2 capture is still in lab scale, since rare pilot tests have been reported and evaluated. Pilot scale of membrane operation for capturing SO2 from flue gas has been reported to run smoothly six months with no problems from CO2, NOx and dust [22]. A screening of suitable polymer materials for pilot scale set up and no further progress is followed yet [23]. Although energy cost for CO2 capture of polymer membrane is estimated to be $23/ton CO2 with 90% capture efficiency from modeling [7], the other researchers have insisted that membrane is much more expensive than chemical absorption, therefore, it still has a long way to go for commercialization, despite its promising future for CO2 capture [3,9,19].
3. Physically Selective Membranes
Ideally, a highly selective CO2/N2 and permeable membrane works well for post combustion CO2 capture; however, there is always a trade-off between selectivity and permeability [10,15]. The highly selective membrane is comparable to a filter and it only allows filtrate to go through, which can be enhanced by a sweeping media as a “vacuum”.
There are still some arguments about the appropriate selectivity of CO2/N2 in flue gas capture. An earlier argument stated that a CO2/N2 selectivity should be more than 200 [6] or higher than 100 with CO2% > 20% [24]; therefore, the current level of 50 is too low for the post CO2 capture from flue gas (about 13% CO2) [9,11,24,25]. Other researchers mentioned that with the CO2/N2 = 50, capture efficiency of 90% and cost of $23/ton CO2 can be achieved with combined membrane technologies [7]. Because the above statements are all based on modeling and parametric study, more experimental verification, including lab and pilot scales, will be crucial to evaluate CO2/N2 selectivity from flue gas for the most promising polymeric membranes.
4. Hollow Fiber Membranes
Hollow fiber membrane is highly gas permeable but not gas selective by itself. The gas mixtures on one side of membrane permeate into the lumen of the membrane, and the solvent selectively dissolve or reacts with and remove CO2 from the flue gas [9,11,26]. Therefore, the selectivity role in this membrane separation is realized through the solvent. The illustration and SEM picture of Hollow fibre membrane are illustrated below, as in Figures 1 and 2 [27,28].
4.1. Absorbents
Ideal absorbents for hollow fiber membrane should preferably have 1) high reactivity with CO2 (reduction of CO2 transport resistance in the solvents); 2) are highly hydrophobic and high surface tension (minimization of the liquid penetration into the fiber lumen significantly reduces the mass transfer in liquid solvent); 3) low vapor pressure (reduction of solvent evaporation into the flue
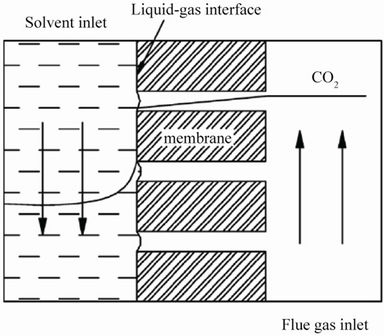
Figure 1. Illustration of Hollow fibre membrane (Yan et al. 2007).
gas mixtures and thus increase in the gas transport efficiency); 4) chemical compatibility with membrane; and 5) easy regeneration (low energy consumption) [3].
Modeling result has indicated that aqueous diethanolamine (DEA) is faster than water to dissolve CO2 since the mass transfer of CO2 in the aqueous phase is dominating the transfer resistance, is thus the overall control step [20]. The application of amino salts as solvents of hollow fiber membrane for CO2 capture proved to be successful in both ways: 1) enhancing the hydrophobic property and thus resulting in high mass transfer efficiency; and 2) reduction of amine corrosion concerns [29-31].
Furthermore, the interaction hydrophobic property, the non wetting of the membrane material with the aqueous solution, can minimize the transport resistance of CO2 in the lumen of hollow fiber [3,10].
4.2. Wettability
Membrane wetting has significantly increased the mass transfer resistance, as can be seen in Equation (1). Modeling and experimental results indicate that wetting of

Figure 2. SEM picture of Hollow fibre membrane (Mansourizadeh and Ismail 2010).
membrane pores significantly affects the mass transfer coefficients of the membrane module [32,33]. Simulation indicates that CO2 absorption rateis six time slower when membrane operated in wetting than unwetting mode; even 5% wetting of the inner membrane leads to 20% reduction of the overall mass transfer coefficient [17]. This was validated by the following experiments that overall mass transfer is reduced to 20% of the control (no wetting) because of the wetting [17].
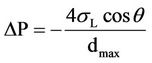 (1)
(1)
ΔP minimum permeance pressure kPa;
σL surface tension mN/m;
θ contact angel between liquid surface and membrane;
dmax the maximum porosity diameter m.
Membrane wetting can result from the membrane materials, the liquid absorbents and possibly its interaction between materials and liquid absorbents. The membrane materials should be hydrophobic, have small pores and possibly thick membrane wall difficult for absorbents to penetrate through [3]. A queous water (maybe with inorganic salts) easily penetrates into many materials, thus leading to the dominant organic adsorbents to react with CO2 as first priority. The optimization of polymer concentration (polyetherimide) can lead to smaller pore size and effective porosity increased the CO2 adsorption and less wetting [8]. Low molecules additives can also lead to small pore size, high surface porosity and thus high wetting resistance [34].
However, the interaction between the absorbents and membrane becomes complicated, which needs to be carefully addressed so as to minimize the membrane wetting. Three months’ immersion of PP in ionized water, MEA and MDA showed that diffusion of the absorbents molecules cause PP membrane swelling and thus a strong hydrophobicity of the inner membrane wall is recommended [35]. Other researchers have also attributed membrane wetting either to the chemical reaction between the absorbents or membrane materials [17]. A degradation of low density polyethylene with 8 - 45 days of immersion in MEA solution has been identified, which was assumed to be the result of oxidative degradation [36]. In parallel, plasticizing effect of CO2’s interaction with the polymer materials was also suggested for the degradation [23]. Screening of four polymer materials (PP, PVDF, PTFE and Nylon based) for pilot scale membrane operation has been based on the criteria of chemical aging with MEA [37].
Hydrodynamics and flow conditions could also possibly result in membrane wetting. While high flow pressure could be one reason for membrane wetting from theoretical calculations of Laplace equation [3], modeling and experiments results from complete, partial and non wetting of PP with MEA, water and NaOH solution illustrated that high flow rate could easily lead to membrane wetting [38].
5. Cross Linking (Facilitated Transport) Membrane
From the comparison that DEA can speed up CO2 capture through chemical absorption than purified water in hollow fiber membrane [20], it is expected that coupling of CO2 affinity coatings/cross linked materials (for example, organo amines) at the inner wall of membrane, which is compared to the catalyst in the chemical reaction, will improve the CO2 selectivity in membrane and thus speed the subsequent CO2 mass transport.
The facilitated transport membrane has been successfully patented for its promising application in CO2 capture [39]. Based on this process, the membrane has a support coated and cross linked polyvinilamine, which serves as a fixed carrier of CO2 and a “catalyst” (in the form of  with combination of moisture), helps quickly remove and transport CO2 through the membrane lumen. It is reported through the improvement of this process, CO2/N2 selectivity has approached 174 and 200 by the same research group [40,41].
with combination of moisture), helps quickly remove and transport CO2 through the membrane lumen. It is reported through the improvement of this process, CO2/N2 selectivity has approached 174 and 200 by the same research group [40,41].
Other researchers have also come up with the similar approaches of cross linking. Examples of the chemical structure of carriers and the related reactions are as shown in Figure 3 and Equations (2)-(4). Amines in cross linked poly (vinyl alcohol) of polymeric membrane reached CO2/N2 selectivity of 450 and the membrane has a descent performance even at the temperature range of 100˚C - 170˚C, much higher than the current prevalent working temperature of polymer membrane [18]. Highly hydrophilic compounds containing quaternary ammonium moieties attached to the reactive trimethoxysilane have CO2/N2 selectivity up to 1500 and the permeability
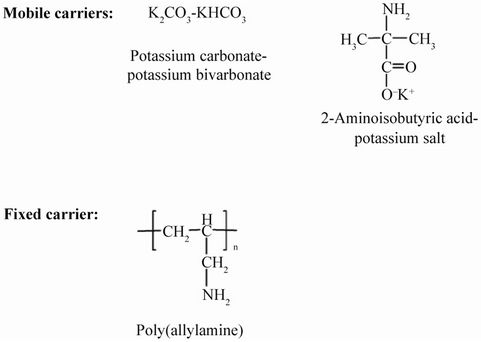
Figure 3. Chemical structure of crossing linking media in facilitated transported membrane for CO2 capture [18].
increased by 35 fold in moisture than that of dry mode without CO2/N2 loss [42].
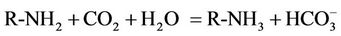 (2)
(2)
 (3)
(3)
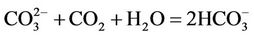 (4)
(4)
Modeling results indicate that facilitated transport membranes could capture CO2 efficiently even in the concentration of as low as 10% in flue gas, with 90% efficiency, 90% CO2 purity with competitive cost to that of aqueous amine scrubbing [41].
6. Conclusions
Polymer membrane separation process is simple, easy to scale up; therefore, it has a huge potential in the postcombustion capture of carbon dioxide applications. Tree typical polymer membrane, physical-selective membrane, hollow fiber membrane and facilitated transport membrane, as well as the operation optimization of pressure, vacuum and multi stag, have been comprehensively discussed in depth in this paper. The major conclusions and suggestions can be summarized below:
1) Pressure and vacuum combination will accelerate the mass transfer rate, reasonable cost, and thus more suitable for the polymer membrane separation of carbon dioxide;
2) Increase in membrane length and parallel membrane devices will increase the separation efficiency, but to balance thus increasing the cost of equipment;
3) Physical selective membrane due to the low selectivity and separation efficiency to reach the burning need for separation of carbon dioxide;
4) The selectivity of the hollow fiber membrane greatly increased, but its chemical adsorbent will increase the secondary pollution and increase the separation costs, a careful analysis of the factors to be studied later;
5) Auxiliary coating/cross-connecting material film should be in the future vigorously research, high selectivity and high mass transfer rates and may withstand 170˚C high temperature, can adapt to the needs of the flue gas of large-scale separation of carbon dioxide.
REFERENCES
- IEA, “CO2 Emissions from Fuel Combustion 1997- 2001,” IEA/OECD, Paris, 2003.
- R. Thiruvenkatachari, S. Su, H. An and X. X. Yu, “Post Combustion CO2 Capture by Carbon Fibre Monolithic Adsorbents,” Progress in Energy and Combustion Science, Vol. 35, No. 5, 2009, pp. 438-455. doi:10.1016/j.pecs.2009.05.003
- J.-L. Li and B.-H. Chen, “Review of CO2 Absorption Using Chemical Solvents in Hollow Fiber Membrane Contactors,” Separation and Purification Technology, Vol. 41, No. 2, 2005, pp. 109-122. doi:10.1016/j.seppur.2004.09.008
- D. R. Paul and Y. P. Yampol’skii, “Polymeric Gas Separation Membranes”, CRC Press, Boca Raton, 1994.
- C. A. Hendricks, K. Blok and W. C. Turkenburg, “The Recovery of Carbon Dioxide from Power Plants,” In: P. A. Okken, R. J. Swart and S. Zwerver, Eds., Climate and Energy, Kluwer Academic Publishers, Dordrecht, 1989.
- J. P. Van Der Sluijs, C. A. Hendricks and K. Blok, “Feasibility of Polymer Membranes for Carbon Dioxide Recovery from Flue Gases,” Energy Conversion and Management, Vol. 33, No. 5-8, 1992, pp. 429-436. doi:10.1016/0196-8904(92)90040-4
- T. C. Merkel, H. Q. Lin, X. T. Wei and R. Baker, “Power Plant Post-Combustion Carbon Dioxide Capture: An Opportunity for Membranes,” Journal of Membrane Science, Vol. 359, No. 1-2, 2010, pp. 126-139. doi:10.1016/j.memsci.2009.10.041
- Gh. Bakeri, A. F. Ismail, M. Shariaty-Niassar and T. Matsuura, “Effect of Polymer Concentration on the Structure and Performance of Polyetherimide Hollow Fiber Membranes,” Journal of Membrane Science, Vol. 363, No. 1-2, 2010, pp. 103-111. doi:10.1016/j.memsci.2010.07.018
- E. Favre, “Membrane Processes and Post Combustion Carbon Dioxide Capture: Challenges and Prospects,” Chemical Engineering Journal, Vol. 171, No. 3, 2011, pp. 782-793. doi:10.1016/j.cej.2011.01.010
- R. Khalilpour, A. Abbas, Z. P. Lai and I. Pinnau, “Modelling and Parametric Analysis of Hollow Fibre Membrane System for Carbon Capture from Multicomponent Flue Gas,” Processing System Engineering, Vol. 58, No. 5, 2011, pp. 1550-1561. doi:10.1002/aic.12699
- A. Brunetti, F. Scura, G. Barbieri and E. Drioli, “Membrane Technologies for CO2 Separation,” Journal of Membrane Science, Vol. 359, No. 1-2, 2010, pp. 115-125. doi:10.1016/j.memsci.2009.11.040
- M. T. Ho, G. W. Allison and D. E. Wiley, “Reducing the Cost of CO2 from Flue Gas Using Pressure Swing Adsorption,” Industrial Engineering Chemical Research, Vol. 47, No. 5, 2008, pp. 1562-1568.
- M. T. Ho, G. W. Allison and D. E. Wiley, “Reducing the Cost of CO2 from Flue Gas Using Pressure Swing Adsorption,” Industrial Engineering Chemical Research, Vol. 47, No. 5, 2008, pp. 4883-4890.
- J. Kotowicz, T. Chmielniak and K. Janusz-Szymanska, “The Influence of Membrane CO2 Separation on the Efficiency of a Coal-Fired Power Plant,” Energy, Vol. 35, No. 2, 2010, pp. 841-850.
- L. Zhao, E. Riensche, R. Menzer, L. Blum and D. Stolten, “A Parametric Study of CO2/N2 Gas Separation Membrane Process for Post-Combustion Capture,” Journal of Membrane Science, Vol. 325, No. 1, 2008, pp. 284-294.
- K. Okabe, H. Mano and Y. Fujioka, “Separation and Recovery of Carbon Dioxide by a Membrane Flash Process,” International Journal of Greenhouse Gas Control, Vol. 1, No. 1, 2009, pp. 1281-1288. doi:10.1016/j.ijggc.2008.06.004
- R. Wang, H. Y. Zhang, P. H. M. Feron and D. T. Liang, “Influence of Membrane Wetting on CO2 Capture in Microporous Hollow Fiber Membrane Contactors,” Separation and Purification Technology, Vol. 46, No. 1-2, 2005, pp. 33-40. doi:10.1016/j.seppur.2005.04.007
- J. Zou and W. S. W. Ho, “CO2-Selective Polymeric Membranes Containing Amines in Crosslinked Poly(Vinyl Alcohol),” Journal of Membrane Science, Vol. 286, No. 1-2, 2006, pp. 310-321. doi:10.1016/j.memsci.2006.10.013
- X. Z. He and M.-B. Hägg, “Hollow Fiber Carbon Membranes: Investigations for CO2 Capture,” Journal of Membrane Science, Vol. 378, No. 1-2, 2010, pp. 1-9. doi:10.1016/j.memsci.2010.10.070
- H.-Y. Zhang, R. Wang, D. T. Liang and J. H. Tay, “Modeling and Experimental Study of CO2 Absorption in A Hollow Fiber Membrane Contactor,” Journal of Membrane Science, Vol. 279, No. 1-2, 2006, pp. 301-310.
- A. Gabelman and S.-T. Hwang, “Hollow Fiber Membrane Contactors,” Journal of Membrane Science, Vol. 159, No. 1-2, 1999, pp. 61-106. doi:10.1016/S0376-7388(99)00040-X
- M. Mavroudi, S. P. Kaldis and G. P. Sakellaropoulos, “Reduction of CO2 Emissions by a Membrane Contacting Process,” Fuel, Vol. 82, No. 15-17, 2003, pp. 2153-2159.
- D. Shekhawat, D. R. Luebke and H. W. Pennline, “A Review of Carbon Dioxide Selective Membranes—A Topical Report,” National Energy Technology Laboratory, United States Department of Energy, 2003.
- R. Bounaceur, N. Lape, D. Roizard, C. Vallieres and E. Favre, “Membrane Processes for Post-Combustion Carbon Dioxidecapture: A Parametric Study,” Energy, Vol. 31, No. 14, 2006, pp. 2556-2570. doi:10.1016/j.energy.2005.10.038
- E. Favre, “Carbon Dioxide Recovery from Post-Combustion Processes: Can Gas Permeation Membranes Compete with Absorption?” Journal of Membrane Science, Vol. 294, No. 1-2, 2007, pp. 50-59. doi:10.1016/j.memsci.2007.02.007
- C. E. Powell and G. G. Qiao, “Polymeric CO2/N2 Gas Separation Membrane for the Capture of Carbon Dioxide from Power Plant Flue Gases,” Journal of Membrane Science, Vol. 279, No. 1-2, 2006, pp. 1-49. doi:10.1016/j.memsci.2005.12.062
- S.-P. Yan, M.-X. Fang, W.-F. Zhang, S.-Y. Wang, Z.-K. Xu, Z.-Y. Luo and K.-F. Cen, “Experimental Study on the Separation of CO2 from Flue Gas Using Hollow Fiber Membrane Contactors without Wetting,” Fuel Processing Technology, Vol. 88, No. 5, 2007, pp. 501-511.
- A. Mansourizadeh and A. F. Ismail, “A Developed Asymmetric PVDF Hollow Fiber Membrane Structure for CO2 Absorption,” International Journal of Greenhouse Gas Control, Vol. 5, No. 2, 2010, pp. 374-380. doi:10.1016/j.ijggc.2010.09.007
- U. E. Aronu, H. F. Svendsen and K. AndersHoff, “Investigation of Amine Amino Acid Salts for Carbon Dioxide Absorption,” International Journal of Greenhouse Gas Control, Vol. 4, No. 5, 2010, pp. 771-775.
- J. Kittel, R. Idem, D. Gelowitz, P. Tontiwachwuthikul, G. Parrain and A. Bonneau, “Corrosion in MEA Units for CO2 Capture: Pilot Plant Studies,” Energy Procedia, Vol. 1, No. 1, 2009, pp. 791-797. doi:10.1016/j.egypro.2009.01.105
- A. F. Portugal, J. M. Sousa, F. D. Magalhães and A. Mendes, “Solubility of Carbon Dioxide in Aqueous Solutions of Amino Acid Salts,” Chemical Engineering Science, Vol. 64, No. 9, 2009, pp. 1993-2002. doi:10.1016/j.ces.2009.01.036
- H. Kreulen, C. A. Smolders, G. F. Versteeg and W. P. M. van Swaaij, “Determination of Mass Transfer Rates in Wetted and Non-Wetted Microporous Membranes,” Chemical Engineering Science, Vol. 48, No. 11, 1993, pp. 2093-2102. doi:10.1016/0009-2509(93)80084-4
- J. G. Lu, Y. F. Zheng and M. D. Cheng, “Wetting Mechanism in Mass Transfer Process of Hydrophobic Membrane Gas Absorption,” Journal of Membrane Science, Vol. 308, No. 1-2, 2008, pp. 180-190. doi:10.1016/j.memsci.2007.09.051
- A. Mansourizadeh and A. F. Ismail, “Effect of Additives on the Structure and Performance of Polysulfone Hollow Fiber Membranes for CO2 Absorption,” Journal of Membrane Science, Vol. 348, No. 1-2, 2010, pp. 260-267. doi:10.1016/j.memsci.2009.11.010
- Y. X. Lv, X. H.Yu, S.-T. Tu, J. Y. Yan and E. Dahlquist, “Wetting of Polypropylene Hollow Fiber Membrane Contactors,” Journal of Membrane Science, Vol. 362, No. 1-2, 2010, pp. 444-452. doi:10.1016/j.memsci.2010.06.067
- S. M. Sedeghi, J. Brisson, D. Rodrigue and M. C. Iliuta, “Chemical Alteration of LDPE Hollow Fibers Exposed to Monoethanolamine Solutions Used as Absorbent for CO2 Capture Process,” Separation and Purification Technology, Vol. 80, No. 2, 2011, pp. 338-344. doi:10.1016/j.seppur.2011.05.017
- F. Porcheron and S. Drozdz, “Hollow Fiber Membrane Contactor Transient Experiments for the Characterization of Gas/Liquid Thermodynamics and Mass Transferproperties,” Chemical Engineering Science, Vol. 64, No. 2, 2009, pp. 265-275. doi:10.1016/j.ces.2008.09.035
- M. H. El-Naas, M. Al-Marzouqi, S. A. Marzouk and N. Abdullatif, “Evaluation of the Removal of CO2 Using Membrane Contactors: Membrane Wettability,” Journal of Membrane Science, Vol. 350, No. 1-2, 2010, pp. 410- 416. doi:10.1016/j.memsci.2010.01.018
- M. B. Hägg, T.-J. Kim and B. Li, “Membrane for Separating CO2 and Process for the Production Thereof,” WO Patent No. 2005089907, 2005.
- L. Y. Deng, T.-J. Kim and M.-B. Hägg, “Facilitated Transport of CO2 in Novel PVAm/PVA Blend Membrane,” Journal of Membrane Science, Vol. 340, No. 1-2, pp. 154-163.
- A. Hussain and M.-B. Hägg, “A Feasibility Study of CO2 Capture from Flue Gas by a Facilitated Transport Membrane,” Journal of Membrane Science Volume, Vol. 359, No. 1-2, 2010, pp. 140-148.
- S. Shishatskiy, J. R. Pauls, S. P. Nunes and K.-V. Peinemann, “Quaternary Ammonium Membrane Materials for CO2 Separation,” Journal of Membrane Science, Vol. 359, No. 1-2, 2010, pp. 44-53. doi:10.1016/j.memsci.2009.09.006
NOTES
*Corresponding author.

