Journal of Biomaterials and Nanobiotechnology
Vol.4 No.2A(2013), Article ID:29846,8 pages DOI:10.4236/jbnb.2013.42A003
Biological Synthesis of Colloidal Gold Nanoprisms Using Penicillium citrinum MTCC9999
![]()
1Department of Biochemistry, University of Calcutta, Kolkata, India; 2Department of Physiology, Krishnagar Government College, Krishnagar, India.
Email: *ghoshs71@hotmail.com, sgbioc@caluniv.ac.in
Copyright © 2013 Achintya Mohan Goswami, Sanjay Ghosh. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received January 16th, 2013; revised February 23rd, 2013; accepted April 5th, 2013
Keywords: Gold Nanoprism; Green Synthesis; Penicillium citrinum; Transmission Electron Microscopy (TEM); Zeta Potential; Fourier Transformed Infra Red Spectroscopy (FTIR)
ABSTRACT
This report provides for the first time rapid novel environment friendly cell surface based synthesis of stable gold nanoprisms at room temperature using Penicillium citrinum MTCC9999 biomass. The UV-Visible spectral scan of dispersed gold nanoparticles (GNPs) solution showed absorption maxima at 540 nm due to surface plasma resonance (SPR) of gold nanoparticles. Typical Transmission Electron Microscopic (TEM) images showed that most of them were prism (55%) shaped with a diameter ranging from 20 - 40 nm. These results obtained from TEM correlated well with the data obtained from Dynamic Light Scattering (DLS) experiment. Average zeta potential of GNPs was −20 mV suggesting some biomolecules capped the nanoparticles imparting a net negative charge over it. FTIR analysis also showed that protein molecules were involved in stabilization.
1. Introduction
In recent years the synthesis of stable gold nanoparticles is one of the most active areas of research in the field of nanotechnology because of their applications in a number of areas like bio-imaging, bio-medicine, bio-sensors, bio-labels, catalysis and others [1-5]. Metal nanoparticles are produced mainly by means of chemical processes involving chemical aggressive reducing agents (e.g. sodium borohydride, hydroxylamine, or tetrakishydroxymethylphosphonium chloride) [6,7], a capping agent (e.g. trioctyl phosphine oxide) [8,9], and organic solvent (e.g. toluene, chloroform) [10,11] for better results. Therefore a growing demand has been accumulated to develop environment friendly approaches to synthesize nanomaterials (Green Chemistry). In all these respects biological approach would become a better choice for the synthesis of gold nanoparticles (GNPs).
There are other reports which showed the intracellular synthesis of spherical GNPs in live Alfaalfa plants [12], truncated triangular and spherical GNPs from sun dried Cinamonum camphora leaf extract [13], triangular gold nanoprism from lemon grass extract [14], hexagonal, prism and spherical shaped GNPs using plant surfactants [15], triangular gold nano plates using algal solutions of Chlorella vulgaris [16] and Sargassum sp. [17] etc. Escherichia coli has been found to secrete proteins with specific polypeptide repeat sequences have been shown to synthesize flat, triangular gold nanocrystals with a relative yield of 4% to the total nanoparticle production [18]. It is advantageous to use fungi over bacteria for development of natural “nano-factories” because the handling of fungal biomass will be much simpler [19].
However, there is limited information available regarding the synthesis of GNPs using fungus. Synthesis of spherical gold nanoparticles (GNPs) have been reported using the fungus Fusarium oxysporum and Verticillium sp. [20,21]. By conventional chemical and physical methods nanocubes [22], nanorods [23], nanowires [24], nanoplates [25] can be synthesized but that require high environmental cost. The chemical synthesis of metal nanoprisms in large quantities using sphere-triangle shape transformation [26-28] or seeded growth [29] has been found to be more difficult. Synthesis of triangular gold nanoprism using lemon grass leaf extract showed large size of nanoprisms ranging from 500 - 1800 nm length and truncated vertices [14]. However, bio application of large nano particles (greater than 50 nm) are limited due to reduced endocytosis and poor renal clearance [30].
In this work, we report for the first time a cell surface based synthesis of prism shaped gold nanoparticles of smaller diameter 20 - 40 nm by reducing chloroauric acid (HAuCl4·3H2O) at room temperature using Penicillium citrinum MTCC9999 biomass and characterized the gold nanoparticles using UV-Visible Spectroscopy, Transmission Electron Micrograph (TEM), Dynamic Light Scattering (DLS) and Fourier Transformed Infra Red Spectroscopy (FTIR).
2. Material and Methods
All chemical reagents were purchased from Sigma (St. Louis, MO, USA) and were of analytical grade. All components for growth media were purchased from Becton-Dickinson (Rutherford, NJ, USA).
2.1. Microorganism
Penicillium citrinum (strain number MTCC9999) was isolated from soil, collected from Dhapa situated near Kolkata, West Bengal, and was identified by the Institute of Microbial Technology (IMTECH), Chandigarh, India. The strain was subcultured on potato dextrose agar (PDA). Each experiment was repeated thrice using freshly grown culture of P. citrinum in PDA.
2.2. Biomass Production
The fungus was grown aerobically in liquid media containing (g/l) KH2PO4: 7.0, K2HPO4: 2.0, MgSO4·7H2O: 0.1, (NH4)2SO4: 1.0, Yeast Extract: 0.6, Glucose 10.0. The conical containing media was inoculated within fungal spores and incubated at orbital shaker at 29˚C for 84 hours at 140 rpm. Then the biomass was harvested by sieving through a plastic filter and washed several times with double distilled water to remove any traces of media components. Typically 2 g biomass (fresh weight) was dispersed in 20 ml of double distilled water (pH 5.1 ± 0.1).
2.3. Synthesis of Gold Nanoparticles by Fungal Mycellia
Chloroauric acid (HAuCl4·3H2O) at a final concentration of 500 mg/L was added to 20 ml of resuspended biomass. The mycelial suspension was incubated at 110 rpm at 25˚C. Control set (only biomass) without HAuCl4·3H2O was also run side by side. Another negative control containing only HAuCl4·3H2O was maintained under the same conditions. Gold nanoparticles (GNPs) synthesized on cell surface were isolated by placing the mycelial suspension on a bath sonicator and sonicated for 15 minutes. Most of the GNPs came to the solution after sonication, it was then passed through Whatman Filter paper No. 1 and the filtrate containing GNPs was collected which was used for further studies.
2.4. Characterization of Gold Nanoparticles
Gold nanoparticles were characterized by following techniques.
2.4.1. Visual Inspection
Formation of gold nanoparticles resulted in a sharp colour change from yellow to dark pinkish. Thus a visual inspection was routinely performed.
2.4.2. UV-Vis Spectra Analysis
UV-Visible spectra of gold nanoparticles by bio-reduction of Chloroauric acid in aqueous solution was recorded in Jasco UV-Visible spectrophotometer (V-530) operated at a resolution of 1 nm in absorption mode.
2.4.3. Dynamic Light Scattering (DLS) Analysis
DLS was performed in MALVERAN Nano Series to measure the hydrodynamic diameter and zeta potential of nanoparticles.
2.4.4. Transmission Electron Microscopic (TEM) Measurement
The samples for Transmission Electron Microscopy (TEM) analysis were prepared by drop-casting the GNPs solution on a carbon-coated copper TEM grid. Before casting to the grid the GNPs solution was centrifuged at 10,000 rpm for 5 minutes and the isolated GNPs were dispersed in 100 µl double distilled water and sonicated in a bath sonicator for 15 minutes. The TEM images were recorded on a high resolution electron microscope (HRTEM: JEOL JEM 2010) operating at an accelerating voltage of 200 kV.
2.4.5. Atomic Force Microscopic (AFM) Study
AFM was conducted to record the morphological changes of fungal mycelia following GNP synthesis on their surface. The images were recorded by a multimode atomic force microscope (Veeco Metrology, Autoprobe CPII, Model No. AP0100) at ambient conditions (20˚C ± 2˚C) using silicon probes (RTESPA-M, Veeco, Santa Barbara, CA) in tapping mode for minimization of sample damage by scanning tip. Sample images were processed using ProScan image processing software.
2.5. Fourier Transformed Infra Red Spectroscopy (FTIR)
For FTIR spectrum analysis the GNPs were centrifuged at 10,000 rpm for 10 minutes to remove free proteins or other compounds present in the solution. The GNPs then resuspended in double distilled water and again centrifuged. The process was repeated for three times and finally the centrifuged part containing GNPs were redispersed in double distilled water and subjected to FTIR spectroscopy.
3. Results
Treatment of Penicillium citrinum MTCC9999 biomass with chloroauric acid (HAuCl4·3H20) resulted in a sharp color change of fungal mycelia from light yellow to purple within 24 hours (Figure 1(A)) indicating formation of gold nanoparticles on the mycelial surface as showed by others [21]. Figure 1(B) represents GNPs solution collected from mycelial surfaces following sequential sonication and filtration. The UV-Visible spectral scan of dispersed GNPs solution showed absorption maxima at 540 nm due to surface plasma resonance (SPR) of gold nanoparticles (Figure 1(C)). To observe mycelial surface containing GNPs Atomic Force Microscopy was performed (Figure 1(D)).
Dynamic Light Scattering (DLS) study was used to measure the hydrodynamic diameter of GNPs present in the solution. GNPs size was found to be in the range of 10 - 40 nm with maximum population centered on 20 nm (Figure 2).
To investigate the physical nature of gold nanoparticles transmission electron microscopic analysis was done. A typical TEM image of GNPs (Figures 3(A)-(C)) showed that various types of GNPs were present in a field but most of them were prism (55%) shaped with a diameter ranging from 20 - 40 nm. Although some rod shaped, triangular, cubical, very small circular (3 - 4 nm diameter) and rectangular GNPs were present in smaller amounts (Figures 3(A) and (B)). The HRTEM image (Figure 4(A)) of GNPs showed that the fringe spacing of gold nanoprism was 0.24 nm which corresponded well to the spacing between (111) plane of face centered cubic (fcc) gold (0.235 nm) (JCPDS card No. 04-0784). The selected area electron diffraction pattern (SAED) of gold nanoprism (Figure 4(B)) indicated that the entire nanoparticle was nanocrystalline in nature [31].
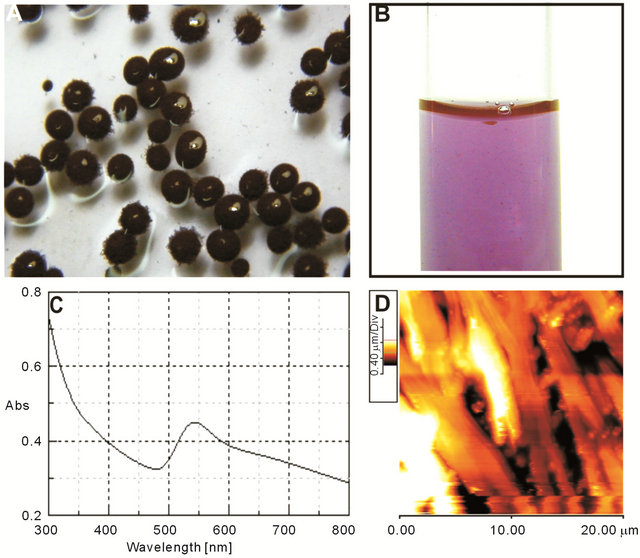
Figure 1. (A) Formation of GNPs on mycelial biomass resulting purple color. (B) Color of GNPs solution. (C) UV-Vis spectrum of GNPs presenting an absorption peak around 540 nm due to surface plasma resonance. (D) AFM image showing morphology of fungal mycelia following GNP synthesis on their surface.
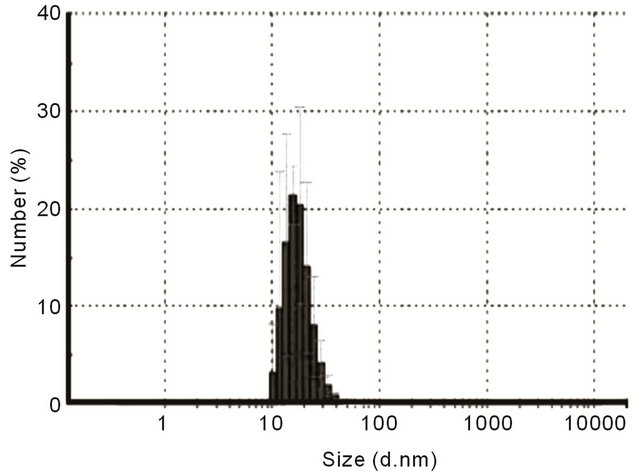
Figure 2. Dynamic Light Scattering (DLS) study was used to measure the hydrodynamic diameter of GNPs present in the solution.
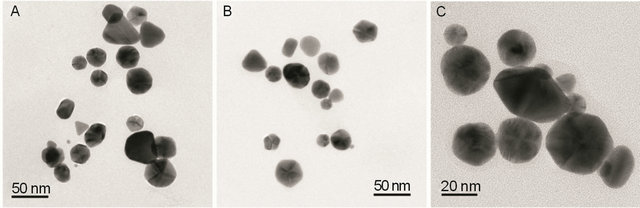
Figure 3. TEM image of GNPs. Some rod shaped, triangular, cubical, very small circular (3 - 4 nm diameter) and rectangular GNPs were present in smaller amounts (Figures A and B). A higher magnification image of gold nanoprism is shown in Figure C.

Figure 4. HRTEM image, SAED and FFT image of gold nanoparticles. (A) HRTEM image of gold nanoprism showing a fringe spacing of 0.24 nm. (B) A selected area electron diffraction pattern of GNPs. The Scherrer ring patterns indicate the fcc gold which is nanocrystalline in nature. (C) HRTEM image of a part of triangular gold nanoparticle and its corresponding FFT image (D). (E) HRTEM image of a gold nanoprism and its corresponding FFT image (F). The arrow in (F) indicates the splitting of spots in the FFT image of gold nanoprism.
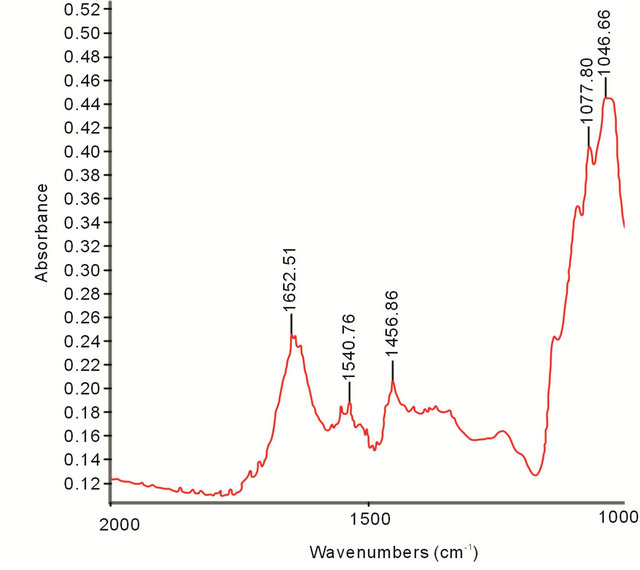
Figure 5. FTIR spectra of gold nanoparticles.
The HRTEM image (Figure 4(A)) of GNPs showed that the fringe spacing of gold nanoprism was 0.24 nm which corresponded well to the spacing between (111) plane of face centered cubic (fcc) gold (0.235 nm) (JCPDS card No. 04-0784). The selected area electron diffraction pattern (SAED) of gold nanoprism (Figure 4(B)) indicated that the entire nanoparticle was nanocrystalline in nature (31). The ring structure of SAED pattern is a characteristic of polycrystalline gold (14). The patterns of SAED were indexed according to (111), (200), (220), and (311) reflections of fcc gold on the basis of their d-spacings of 2.40 Å, 2.05 Å, 1.467 Å, and 1.22 Å. Figure 4(C) shows a HRTEM image of an area of traingular GNP in (111) zone axis. The fringe spacing was 2.38 Å, closely related to fcc gold structure. The d-spacing of the corresponding FFT image (Figure 4(D)) was calculated to be 2.3 Å. Figure 4(E) shows a HRTEM image of a gold nanoprism in (111) zone axis. d-spacings were calculated from the corresponding FFT image (Figure 4(F)) which varied from 1.86 to 2.2 Å.
Average zeta potential of GNPs measured to be −20 mV. FTIR spectroscopy of GNPs was performed to know about the bio-molecules present in the surface. Figure 5 showed the FTIR spectrum obtained from GNPs showing several absorption peaks at 1652 cm−1, 1540 cm−1, 1456 cm−1, 1077 cm−1, and 1046 cm−1.
4. Discussions
Biological systems are capable of synthesizing a remarkably diverse range of inorganic materials. These systems are resourceful and assemble a vast assortment of materials using precursors that are readily available and abundant in the biosphere. Biomaterials have inspired a growing research effort in which biomolecules are used to synthesize and assemble materials in the laboratory. The synthesis of nanoparticles using biological approaches offers several advantages than chemical synthesis like environmentally acceptable solvent, eco friendly reducing and capping agents. We emphasized on bio-inspired synthesis of gold nanoparticles by Penicillium citrinum MTCC9999 in room temperature. The purple colour of Penicillium citrinum biomass is due to the interaction of GNPs with light [21]. The surface plasma resonance of GNPs at 540 nm was due to collective coherent electronic oscillation of metal nanopartticles in presence of electromagnetic field of light. AFM picture revealed that GNPs are present on the surface of the mycelia. It was quite exciting for us to find this bioprocess where mycelial biomass suspended in double distilled water in presence of chloroauric acid could produce GNPs on their surfaces. This microbial synthesis of GNPs could be explained by the fact that microorganisms often face metal ions or metals in their environment and thereby developed mechanism to detoxify metals by changing the redox states of metal ions and (or) intracellular precipitation of metals forming a basis for synthesis of nanoparticles [32].
In our study, GNPs formed a single population in the size range of 10 - 40 nm. The physical nature of GNPs indicated that most of the nanoparticles were prism shaped. Due to symmetric face centered cubic (fcc) lattice of gold nanoparticles, the formation of anisotropic structures like cube, prism, star, branched nanoparticles require a selective capping agent during growth (31). Ha et al reported that I− promoted the growth of gold nanoprism formation [33] by preferential binding to (111) facet of gold nanocrystal with binding energies that scale with polarizability (I− > Br− > Cl−) and crystal facet ((111) > (110) > (100)) [34]. Recently cetyltrimethylammonium bromide (CTAB) has been shown to induce the formation of gold nanorods, triangular prism and spheres through seed mediated approaches depending on iodide contamination present in CTAB which act as shape directing agent [35]. Thus this is quite exciting for us to synthesize gold nanoprism for first time using biological approach. Development of strain in crystals of gold nanoprism was observed from the analysis of crystals. The d-spacings calculated from FFT images of a triangular gold nanoparticle and a gold nanoprism were 2.3 Å and 1.86 to 2.2 Å respectively. Thus d-spacing of (111) plane of gold nanoprism revealed a deviation from the standard value of fcc gold crystal (2.36 Å). FFT image also showed an elongation of the diffraction reflexes. So it can be assumed that a micro stress was developed due to the formation of anisotropic nanoprism.
The zeta potential of GNPs was measured to know their colloidal stability and the nature of the charge carried in their surface. Average zeta potential of GNPs measured to be −20 mV suggesting that some biomolecules capped the nanoparticles imparting a net negative charge over it. FTIR spectroscopy data also supported the fact that biomolecules were present in the surface. The absorption peaks at 1652 cm−1 and 1540 cm−1 are characteristic of amide I and amide II bands [36]. The two absorption peaks located at around 1077 cm−1, and 1046 cm−1 can be assigned as the absorption peaks of -C-Oand -C-O-C- [13]. Free amine, carboxyl or phosphate groups present in the protein can interact with gold nanoparticles [32,37] and possibly involved in stabilization of GNPs.
5. Conclusion
The biomolecules present in the cell surface of Penicillum citrinum may perform three important functions: 1) acts as reducing agent involved in the synthesis, 2) acts as shape directing agent involved primarily in the shaping anisotropic structure of gold nanoparticles to gold nanoprism and 3) acts as capping agent involved in stabilization of gold nanoprisms. Moreover, the synthesis of other anisotropic nanostrctures of gold like nano rod, cube, triangle (which are present in smaller quantities as per TEM images) may be achieved by further tuning the synthesis protocol using the fungus. In addition gold nanoprism offers several advantages in biomedical applications. In conclusion, in situ synthesis of gold nanoprisms by reduction of Au3+ on mycelial surface and stabilization of the synthesized GNPs, provides an environment friendly synthetic route.
6. Acknowledgement
This work is supported by Centre for Research in Nano Science and Nanotechnology, University of Calcutta. The authors would like to thank Shantiswarup Sinha and Prof. Anjan Dasgupta for assistance with the FTIR measurements. The authors would like to acknowledge DSTFIST, UGC-CAS programme, Govt. of India, Indian Association for Cultivation of Science (IACS), Jadavpur for equipment and infrastructural support.
REFERENCES
- C. A. Mirkin, R. L. Letsinger, R. C. Mucic and J. J. Storhoff, “A DNA-Based Method for Rationally Assembling Nanoparticles into Macroscopic Materials,” Nature, Vol. 382, No. 15, 1996, pp. 607-609. doi:10.1038/382607a0
- M. Han, X. Gao, J. Z. Su and S. Nie, “Quantum-DotTagged Microbeads for Multiplexed Optical Coding of Biomolecules,” Nature Biotechnology, Vol. 19, No. 7, 2001, pp. 631-635. doi:10.1038/90228
- A. K. Salem, P. C. Searson and K. W. Leong, “Multifunctional Nanorods for Gene Delivery,” Nature Materials, Vol. 2, No. 10, 2003, pp. 668-671. doi:10.1038/nmat974
- X. Huang, I. H. El-Sayed, W. Qian and M. A. El-Sayed, “Cancer Cell Imaging and Photothermal Therapy in the Near-Infrared Region by Using Gold Nanorods,” Journal of the American Chemical Society, Vol. 128, No. 6, 2006, pp. 2115-2120. doi:10.1021/ja057254a
- M. Moreno-Manas and R. Pleixats, “Formation of Carbon-Carbon Double Bonds under Catalysis by Transition Metal Nanoparticles,” Accounts of Chemical Research, Vol. 36, No. 8, 2003, pp. 638-643. doi:10.1021/ar020267y
- S. Meltzer, R. Resch, B. E. Koel, M. E. Thompson, A. Madhukar, A. A. G. Requicha and P. Will, “Fabrication of Nanostructures by Hydroxylamine Seeding of Gold Nanoparticle Templates,” Langmuir, Vol. 17, No. 5, 2001, pp. 1713-1718. doi:10.1021/la001170s
- S. L. Westcott, S. J. Oldenburg, T. R. Lee and N. J. Halas, “Formation and Adsorption of Gold Nanoparticle-Clusters on Functionalized Silica Nanoparticle Surfaces,” Langmuir, Vol. 14, No. 19, 1998, pp. 5396-5401. doi:10.1021/la980380q
- V. F. Puntes, K. M. Krishnan and A. P. Alivisatos, “Colloidal Nanocrystal Shape and Size Control: The Case of Cobalt,” Science, Vol. 291, No. 5511, 2001, pp. 2115- 2117. doi:10.1126/science.1057553
- Z. A. Peng and X. J. Peng, “Nearly Monodisperse and Shape-Controlled CDSE Nanocrystals via Alternative Routes: Nucleation and Growth,” Journal of the American Chemical Society, Vol. 124, No. 13, 2002, pp. 3343- 3353. doi:10.1021/ja0173167
- M. Brust, M. Walker, D. Bethell, D. J. Schiffrin and R. J. Whyman, “Synthesis of Thiol Derivatised Gold Nanoparticles in a Two-Phase Liquid/Liquid System,” Journal of the Chemical Society, Chemical Communications, No. 7, 1994, pp. 801-802. doi:10.1039/c39940000801
- D. I. Gittins, F. Caruso, “Biological and Physical Applications of Water-Based Metal Nanoparticles Synthesised in Organic Solution,” ChemPhysChem, Vol. 3, No. 1, 2002, pp. 110-113. doi:10.1002/1439-7641(20020118)3:1<110::AID-CPHC110>3.0.CO;2-Q
- J. L. Gardea-Torresdey, J. G. Parsons, E. Gomez, J. Peralta-Videa, H. E. Troiani, P. Santiago and M. Jose-Yacaman, “Formation and Growth of Au Nanoparticles inside Live Alfalfa Plants,” Nano Letters, Vol. 2, No. 4, 2002, pp. 397-401. doi:10.1021/nl015673+
- J. Huang, Q. Li, D. Sun, Y. Lu, Y. Su, X. Yang, H. Wang, Y. Wang, W. Shao, N. He, J. Hong and C. Chen, “Biosynthesis of Silver and Gold Nanoparticles by Novel Sundried Cinnamomum camphora Leaf,” Nanotechnology, Vol. 18, No. 10, 2007, p. 105104. doi:10.1088/0957-4484/18/10/105104
- S. S. Shankar, A. Rai, B. Ankamwar, A. Singh, A. Ahmad and M. Sastry, “Biological Synthesis of Triangular Gold Nanoprisms,” Nature Materials, Vol. 3, No. 7, 2004, pp. 482-488. doi:10.1038/nmat1152
- M. N. Nadagouda, “Green Synthesis of Au Nanostructures at Room Temperature Using Biodegradable Plant Surfactants,” Crystal Growth and Design, Vol. 9, No. 11, 2009, pp. 4979-4983. doi:10.1021/cg9007685
- J. Xie, J. Y. Lee, D. I. Wang and Y. P. Ting, “Identification of Active Biomolecules in the High Yield Synthesis of Single-Crystalline Gold Nanoplates in Algal Solutions,” Small, Vol. 3, No. 4, 2007, pp. 672-682. doi:10.1002/smll.200600612
- B. Liu, J. Xie, J. Y. Lee, Y. P. Ting and J. P. Chen, “Optimization of High Yield Biological Synthesis of Single Crystalline Gold Nanoplates,” The Journal of Physical Chemistry B, Vol. 109, No. 32, 2005, pp. 15256-15263. doi:10.1021/jp051449n
- S. Brown, M. Sarikaya and E. Johnson, “A Genetic Analysis of Crystal Growth,” Journal of Molecular Biology, Vol. 299, No. 3, 2000, pp. 725-735. doi:10.1006/jmbi.2000.3682
- M. Sastry, A. Ahmad, M. I. Khan and R. Kumar, “Biosynthesis of Metal Nanoparticles Using Fungi and Actinomycete,” Current Science, Vol. 85, No. 2, 2003, pp. 162-170.
- P. Mukherjee, S. Senapati, D. Mandal, A. Ahmad, M. I. Khan, R. Kumar and M. Sastry, “Extracellular Synthesis of Gold Nanoparticles by the Fungus Fusarium oxysporum,” Chembiochem, Vol. 3, No. 5, 2002, pp. 461-463. doi:10.1002/1439-7633(20020503)3:5<461::AID-CBIC461>3.0.CO;2-X
- P. Mukherjee, A. Ahmad, D. Mandal, S. Senapati, S. R. Sainkar, M. I. Khan, R. Ramani, R. Parischa, P. V. Ajayakumar, M. Alam, M. Sastry and R. Kumar, “Bioreduction of
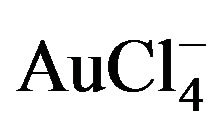 Ions by the Fungus, Verticillium sp. and Surface Trapping of the Gold Nanoparticles Formed,” Angewandte Chemie International Edition England, Vol. 40, No. 19, 2001, pp. 3585-3588. doi:10.1002/1521-3773(20011001)40:19<3585::AID-ANIE3585>3.0.CO;2-K
Ions by the Fungus, Verticillium sp. and Surface Trapping of the Gold Nanoparticles Formed,” Angewandte Chemie International Edition England, Vol. 40, No. 19, 2001, pp. 3585-3588. doi:10.1002/1521-3773(20011001)40:19<3585::AID-ANIE3585>3.0.CO;2-K - Y. Sun and Y. Xia, “Shape-Controlled Synthesis of Gold and Silver Nanoparticles,” Science, Vol. 298, No. 5601, 2002, pp. 2176-2179. doi:10.1126/science.1077229
- C. J. Murphy, A. M. Gole, S. E. Hunyadi and C. J. Orendorff, “One Dimensional Colloidal Gold and Silver Nanostructures,” Inorganic Chemistry, Vol. 45, No. 19, 2006, pp. 7544-7554. doi:10.1021/ic0519382
- L. Pei, K. Mori and M. Adachi, “Formation Process of Two Dimensional Networked Gold Nanowires by Citrate Reduction of
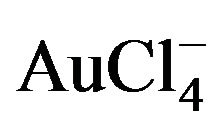 and the Shape Stabilization,” Langmuir, Vol. 20, No. 18, 2004, pp.7837-7843. doi:10.1021/la049262v
and the Shape Stabilization,” Langmuir, Vol. 20, No. 18, 2004, pp.7837-7843. doi:10.1021/la049262v - Z. Li, Z. Liu, J. Zhang, B. Han, J. Du, Y. Gao and T. Jiang, “Synthesis of Single-Crystal Gold Nanosheets of Large Size in Ionic Liquids,” The Journal of Physical Chemistry B, Vol. 109, No. 30, 2005, pp. 14445-14448. doi:10.1021/jp0520998
- R. Jin, Y. Cao, C. A. Mirkin, K. L. Kelly, G. C. Schatz and J. G. Zheng, “Photoinduced Conversion of Silver Nanospheres to Nanoprisms,” Science, Vol. 294, No. 5548, 2001, pp. 1901-1903. doi:10.1126/science.1066541
- Y. Sun, B. Mayers and Y. Xia, “Transformation of Silver Nanospheres into Nanobelts and Triangular Nanoplates through a Thermal Process,” Nano Letters, Vol. 3, No. 5, 2003, pp. 675-679. doi:10.1021/nl034140t
- R. Jin, Y. C. Cao, E. Hao, G. S. Metraux, G. C. Schatz and C. A. Mirkin, “Controlling Anisotropic Nanoparticle Growth through Plasmon Excitation,” Nature, Vol. 425, No. 6957, 2003, pp. 487-490. doi:10.1038/nature02020
- S. Chen and D. L. Carroll, “Synthesis and Characterization of Truncated Triangular Silver Nanoplates,” Nano Letters, Vol. 2, No. 9, 2002, pp. 1003-1007. doi:10.1021/nl025674h
- B. D. Chithrani, A. A. Ghazani and W. C. Chan, “Determining the Size and Shape Dependence of Gold Nanoparticle Uptake into Mammalian Cells,” Nano Letters, Vol. 6, No. 4, 2006, pp. 662-668. doi:10.1021/nl052396o
- A. Ahmad, S. Senapati, M. I. Khan, R. Kumar and M. Sastry, “Extracellular Biosynthesis of Monodisperse Gold Nanoparticles by a Novel Extremophilic Actinomycete, Thermomonospora sp.,” Langmuir, Vol. 19, No. 8, 2003, pp. 3550-3553. doi:10.1021/la026772l
- T. Klaus, R. Joerger, E. Olsson and C. G. Granqvist, “Silver-Based Crystalline Nanoparticles, Microbially Fabricated,” Proceedings of the National Academy of Sciences of the United States of America, Vol. 96, No. 24, 1999, pp. 13611-13614. doi:10.1073/pnas.96.24.13611
- T. H. Ha, H. J. Koo and B. H. Chung, “Shape-Controlled Syntheses of Gold Nanoprisms and Nanorods Influenced by Specific Adsorption of Halide Ions,” The Journal of Physical Chemistry C, Vol. 111, No. 3, 2007, pp. 1123- 1130. doi:10.1021/jp066454l
- O. M. Magnussen, “Ordered Anion Adlayers on Metal Electrode Surfaces,” Chemical Reviews, Vol. 102, No. 3, 2002, pp. 679-725. doi:10.1021/cr000069p
- J. E. Millstone, W. Wei, M. R. Jones, H. Yoo and C. A. Mirkin, “Iodide Ions Control Seed-Mediated Growth of Anisotropic Gold Nanoparticles,” Nano Letters, Vol. 8, No. 8, 2008, pp. 2526-2529. doi:10.1021/nl8016253
- F. Caruso, D. N. Furlong, K. Ariga, I. Ichinose and T. Kunitake, “Characterization of Polyelectrolyte-Protein Multilayer Films by Atomic Force Microscopy, Scanning Electron Microscopy, and Fourier Transform Infrared Reflection-Absorption Spectroscopy,” Langmuir, Vol. 14, No. 16, 1998, pp. 4559-4565. doi:10.1021/la971288h
- J. Xie, J. Y. Lee, D. I. Wang and Y. P. Ting, “Silver Nanoplates: From Biological to Biomimetic Synthesis,” ACS Nano, Vol. 1, No. 5, 2007, pp. 429-439. doi:10.1021/nn7000883
NOTES
*Corresponding author.

