Agricultural Sciences
Vol.06 No.02(2015), Article ID:53862,6 pages
10.4236/as.2015.62019
Optimization of nitrogen fertilization input on Zea mays L. cultivation through the biological inhibition of nitrification
Purwanto, S. Minardi, Supriyadi
Soil Science Department, Agriculture Faculty, Sebelas Maret University, Surakarta, Indonesia
Email: purwahadi.ph@gmail.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 19 January 2015; accepted 2 February 2015; published 6 February 2015
ABSTRACT
Introduction: Nitrogen (N) fertilizer is the most widely used fertilizer (300 - 350 Kg/Ha) on the corn plant, mean while it has mobile character, so it becomes the lowest utilization (40% - 50%) compared to other nutrient elements. Aims: The aim of this research is to examine the effect of various qualities and dose of litter on the dynamics of N mineral soil and soil nitrification potential on maize cultivation system. Method: The treatment is set by Randomized Completely Block Design (RCBD). Four kinds of litter is chosen to represent the groups of low quality (Tectona grandis and Pterocarpus indicus) and the groups of high quality (Oryza sativa and Eupatorium inulifolium) are given on four doses: 4, 8, 12 and 16 mg/Ha. All treatments are fertilized with urea 150 kg/Ha. Variables measured include the soil concentration of 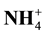 and
and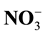 , potential nitrification conducted in 2, 4, 6, 8 and 10 weeks after planting the corn. Result: The research shows that the addition of low-quality litter is influenced very significantly by release of
, potential nitrification conducted in 2, 4, 6, 8 and 10 weeks after planting the corn. Result: The research shows that the addition of low-quality litter is influenced very significantly by release of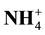 , formation of
, formation of 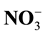 and potential nitrification of soil. Others results show that the content of litter (lignin, polyphenol and C/N ratio) has the most powerful influence as regulator of
and potential nitrification of soil. Others results show that the content of litter (lignin, polyphenol and C/N ratio) has the most powerful influence as regulator of 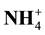 released (p < 0.01), the formation of
released (p < 0.01), the formation of 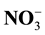 (p < 0.01) and potential nitrification of soil (p < 0.01) rather than the content of lignin, polyphenol and C/N ratio of litter separately. Conclusion: The conclusion of this research is that the management of litter quality input can be applied in the field to regulate the process of nitrification in soil and potential to increase N used efficiency of corn plant.
(p < 0.01) and potential nitrification of soil (p < 0.01) rather than the content of lignin, polyphenol and C/N ratio of litter separately. Conclusion: The conclusion of this research is that the management of litter quality input can be applied in the field to regulate the process of nitrification in soil and potential to increase N used efficiency of corn plant.
Keywords:
Litter Quality, 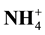 ,
, 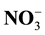 , Biological Nitrification Inhibition, Corn Plant
, Biological Nitrification Inhibition, Corn Plant

1. Introduction
Nitrogen fertilizer is the most widely used fertilizer (300 - 350 kg/Ha) on the corn plant, but because it has mobile characterization, it becomes the lowest utilization (only 40% - 50%) compared to other nutrient elements [1] . Approximately-estimated 50% of the fertilizer N in wet tropical areas will be lost [2] . Although corn plants can absorb the N in the form of  and
and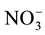 , but
, but 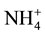 form is more efficient because it requires lower photosynthesis energy for reduced in NH3 i.e. 5ATP per
form is more efficient because it requires lower photosynthesis energy for reduced in NH3 i.e. 5ATP per 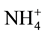 molecule, where as
molecule, where as 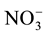 requires 20ATP per molecule [3] . Inhibition of nitrification can increase the efficiency usage of photosynthesis so it can increase the dry weight of the plant. Proven that N uptake in wheat would increase by 35%, if 25% of its fertilizer N was in the form of
requires 20ATP per molecule [3] . Inhibition of nitrification can increase the efficiency usage of photosynthesis so it can increase the dry weight of the plant. Proven that N uptake in wheat would increase by 35%, if 25% of its fertilizer N was in the form of  [4] . It is expected that the optimization of nitrification can be developed with a variety of efforts such as the selection of the right litter, so as to achieve the welfare of the corn harvest.
[4] . It is expected that the optimization of nitrification can be developed with a variety of efforts such as the selection of the right litter, so as to achieve the welfare of the corn harvest.
Corn is one of the most important food crops after rice and wheat. Corn is the main source of carbohydrates and as an alternative source of food in some developed countries. Indonesian peoples also use corn as a staple food. Corn’s cobs and leaves are also used as a source of carbohydrates for livestock. Corn’s plants can also be taken its oil, made of flour or cornstarch and used as industrial raw material. Corn cobs have rich pentose. Genetically engineered corn has also been widely cultivated [5] . Because of the importance of this corn cultivation, it must continue to increase in yields and quality, so that it can improve the quality of human resource as a whole.
Nitrification is the oxidation process of 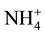 by successive chemoautotroph bacteria produce
by successive chemoautotroph bacteria produce 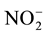 and
and







2. Method
This research used a Randomized Complete of Block Design (RCBD). We used four types of litter materials selected based on their quality, i.e. lignin, polyphenol, C/N content. Corn seeds use BISI 2 and soils use Alfisol.
Experimental plots were divided into subplots in 3 × 1 m2 dimensions. The basis fertilizers are given simultaneously with the application of litter as a substrate of nitrification. Basis fertilizer dose calculated based on the results of preliminary analysis of soil and nutrient needs of corn crops. Given fertilizer was urea (213.32 kg/Ha), SP 36 (437.81 kg/Ha), and KCl (237.68 kg/Ha). Planting corn spacing was 75 × 25 cm2.
Soil samples for measurement of 



Soil samples for potential nitrification measurements taken by aseptic at a depth of 0 - 20 cm. Nitrification potential was measured from the total of 
The measurement variables of

3. Results and Discussion
3.1. Doses and Quality of Litter
Litter used in this research content of lignin, polyphenols, and C/N ratio varies (shown in table 1).
Litter decomposition rate is determined by the quality of litter or the content of soluble carbohydrates, amino acids, active polyphenols, lignin, as well as the C/N ratio [15] . High quality litter categorized by ratios C/N less than 25%, lignin <15%, and polyphenols <3% contents, thus they were rapidly decomposes. Based on the lignin content and C/N ratio content, Rice (Oryza sativa) straw and Teak (Tectona grandis) were classified as low quality, where as Angsana (Pterocarpus indicus) litter, and Kirinyu (Eupatorium inulifolium) were classified as high-quality litter. Litter quality differences will result in the release patterns of different 
3.2. Dynamic of N
The length of stay 

High-quality litter (Pterocarpus indicus and Eupatorium inulifolium) increase the concentration of 





Concentration of 


Table 1. The analysis results of the litter quality.
Table 2. Rate of 
Figure 1. 
in soil with the pattern varies between species. High concentration of 


The concentration of 









Indicated that the process has not been held fast immobilizes 







3.3. Potential Nitrification of Soil
Potential nitrification were measured by the number of 
Analysis of variance results showed a difference of species/quality and time of incubation effect quite significant (p < 0.01), whereas the dosage litter affect significant (p < 0.05) against potential nitrification of soils. The increasing of potential nitrification in soils is closely connected and significantly (r = 0.802) increased concentrations of 


Figure 2. 
Figure 3. Soil nitrification potential after addition of various types of quality and dosage of litter.
On the field condition, N which is not absorbed by the corn plant rooting potentially lost leached to the lower layers. 
4. Conclusion
The conclusion of this research is that the management of the quality of the litter input can be applied in the field to hinder the nitrification process of soil N from leaching and improving the efficiency of the utilization of N on corn plants.
Acknowledgements
We thank Higher Education Competitive Research Project Ministry of Education and Culture Republic of Indonesia for Grand Featured Research Mail Agreement No. 2342/UN 27.16/PN/2012 State Budget Funds and No. 159a/UN 27.11/PN/2013 and especially thank Prof. Dr. Rafik Karsidi, MSc as a Rector of Sebelas Maret University Surakarta Indonesia; Prof. Dr. Ir. Darsono, M.Si as a Chairman of the Institute for Research and Community Service; Prof. Dr. Ir. Bambang Pujiasmanto, M.Si as a Dean of Agriculture Faculty of Sebelas Maret University Surakarta Indonesia and Dr. Adi Prayitno, DDS, PhD for all kindness.
References
- Verchot, L.V., Hutabarat, L., Hairiah, K. and Van Noordwijk, M. (2007) Nitrogen Availability and soil N2O Emissions Following Conversion of Forests to Coffee in Southern Sumatra. Global Biochemical Cycles, 20, (In press).
- Van Noordwijk, M. and de Willigen, P. (1987) Root as Sinks and Sources of Carbon and Nutrient in Agricultural Sys- tems. In: Brussaard, L. and Ferrera-Cerrato, R., Eds., Soil Biology in Sustainable Agricultural Systems, CRC Lewis Pub- lication, Boca Raton, 71-89.
- Taiz, L. and Zeiger, E. (2002) Plant Physiology (Third Edition). Sinauer Associates, Inc., Publishers, Sunderland, 67- 86.
- Raun, W.R and Johnson, G.V. (1999) Improving Nitrogen Use Efficiency for Cereal Production (Review & Interpretation). Agronomy Journal, 91, 357-363. http://dx.doi.org/10.2134/agronj1999.00021962009100030001x
- James, M.G., Robertson, D.S. and Myers, A.M. (1995) Characterization of the Maize Gene Sugary 1, a Determinant of Starch Composition in Kernels. The Plant Cell, 7, 417-429. http://dx.doi.org/10.1105/tpc.7.4.417
- Myrold, D.D. (1999) Transformation of Nitrogen. In: Sylvia, D.M., Jeffry, J.F., Peter, G.H. and David, A.Z., Eds., Principles and Application of Soil Microbiology, Prentice Hall, New Jersey, 259-294.
- Bardgett, R.D. (2002) The Biology of Soil. A Community and Ecosystem Approach. Oxford University Press Inc., New York, 242.
- Aarnio, T. and Martikainen, P.J. (1995) Mineralization of C and Nitrification in Scot Pine Forest Soil treated with Nitrogen Fertilizers Containing Different Proportions of Urea and Its Slow-Releasing Derivative, Ureaformaldehyde. Soil Biology & Biochemistry, 27, 1325-1331.
- Erickson, A.J., Ramsewak, R.S., Smucker, A.J. and Nair, M.G. (2000) Nitrification Inhibitors from the Roots of Leucaena leucocephala. Journal of Agricultural and Food Chemistry, 48, 6174-6177. http://dx.doi.org/10.1105/tpc.7.4.417
- Paul, E.A. and Clarck, F.E. (1989) Soil Microbiology and Biochemistry. Academic Press, Inc., Waltham.
- Filali, A., Fayolle, Y., Peu, P., Philippe, L., Nauleau, F. and Gillot, S. (2013) Aeration Control in a Full-Scale Activated Sludge Wastewater Treatment Plant: Impact on Performances, Energy Consumption and N2O Emission. Proceedings of the 11ème Conférence IWA sur l’instrumentation, le contrôle et l’automatisation, Narbonne, 18-20 September 2013, 4.
- Anderson, J. and Ingram, J. (1993) Tropical Soil Biology and Fertility: A Handbook of Methods. Second Edition, CABI, Wallington, 221.
- Alves, J.R., Boodey, R.M. and Urquiga, S.S. (1993) A Rapid and Sensitive Flow Injection Technique for the Analysis in Soil Extract. Communication Soil Science Plant Analysis, 24, 277-284. http://dx.doi.org/10.1080/00103629309368798
- Kandeler, E. (1995) Potential Nitrification. In: Schinner, F., Kandeler, E., Ohlinger, R. and dan Margesin, R., Eds., Methods in Soil Biology, Spinger-Verlag Berlin Heidelberg, Berlin, 146-149.
- Handayanto, E. (1994) Nitrogen Mineralization from Legume Tree Prunings of Different Quality. Thesis for Doctor of Philosophy, Department of Biological Sciences, Wye College, University of London, London, 176 p.
- Palm, C.A. and Sanchez, P.A. (1991) Nitrogen Release from Some Tropical Legumes as Affected by Lignin and Polyphenol Contents. Soil Biology Biochemistry, 23, 83-88. http://dx.doi.org/10.1016/0038-0717(91)90166-H
- Brady, N.C. and Weil, R.R. (2002) The Nature and Properties of Soils. Thirteenth Edition, Pearson Education, Inc., Upper Saddle River, 960.
- Tate, R.L. (1995) Soil Microbiology. Wiley, New York.
- Madigan, M.T., Martinko, J.M. and Parker, J. (2000) Biology of Microorganisms. Ninth Edition, Pearson Prentice Hall, Upper Saddle River, 991.
- McColl, J.G. (1995) Forest Clear-Cutting, Soil Response. In: Lederberg, J., Ed., Encyclopedia of Microbiology, Academic Press, Inc., Waltham, 959-1103.
- Wolf, B. and Snyder, G.H. (2003) Sustainable Soils the Place of Organic Matter in Sustaining Soils and Their Productivity. Food Product Press, Haworth Press, Inc., New York.






