Journal of Agricultural Chemistry and Environment
Vol.04 No.04(2015), Article ID:61166,7 pages
10.4236/jacen.2015.44011
Analysis of the trans-Cinnamic Acid Content in Cinnamomum spp. and Commercial Cinnamon Powder Using HPLC
Jaemin Lee1, Dong Gu Lee1, Jun Yeon Park2, Sungwook Chae3, Sanghyun Lee1*
1Department of Integrative Plant Science, Chung-Ang University, Anseong, South Korea
2College of Korean Medicine, Gachon University, Seongnam, South Korea
3KM Convergence Research Division, Korea Institute of Oriental Medicine, Daejeon, South Korea

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 24 September 2015; accepted 14 November 2015; published 17 November 2015
ABSTRACT
The present study aimed to quantify the content of trans-cinnamic acid (CA) in Cinnamomum japonicum, C. loureirii, and C. camphora and the commercial application of CA using high performance liquid chromatography (HPLC). A C18 column was used, with water/methanol as the mobile phase gradient. The highest content of CA was observed in the bark of C. loureirii (16.97 mg/g) and cinnamon powder A (47.60 mg/g). The lowest content of CA was observed in leaf and heartwood of C. japonicum (0.10 and 0.10 mg/g, respectively) and cinnamon powder C (22.87 mg/g). This result could be utilized as a guideline for the analysis of the commercial applications of Cinnamomum.
Keywords:
Cinnamomum spp., trans-Cinnamic Acid, HPLC

1. Introduction
Cinnamomum spp., which belongs to the Lauraceae family, is an evergreen dicotyledon that includes C. loureirii, C. camphora, and C. japonicum species. Lauraceae includes about 55 genera and more than 2000 species, which are generally grown in warm, tropical, and mild climates [1] [2] . Lauraceae has diverse commercial applications such as food supplements and pesticides. Previous research has demonstrated that the oils of the Himalayan Lauraceae species show anti-oxidant and anti-bacterial properties [3] . Moreover, the essential oils of Lauraceae tree leaves have been screened for cytotoxicity activity [4] .
The term “Cinnamomum”, meaning “sweet wood”, has been derived from the Greek word “kinnamomon” [5] . Previous research has revealed that the bark of C. loureirii branches contains chemical constituents such as (E)- cinnamaldehyde and α-copaene. Of the three aforementioned species, C. japonicum has the strongest anti-mi- crobial, anti-tyrosinase, and anti-oxidant properties. C. camphora also exhibits properties that enable it to be used as natural fumigants and insecticides [6] - [8] .
“Cinnamon”, the general term for the bark of several Cinnamomum spp., has long been used as a food ingredient, because of its sweet and spicy flavor [9] . This spice is used in countries such as India, Madagascar, Sri Lanka, and Vietnam for worldwide distribution [10] . Other research has revealed that cinnamon can prevent contamination by Salmonella typhimurium and its use has improved food safety, particularly in dairy applications [11] . Moreover, cinnamon has been shown to substantially improve liver glycogen synthase in case of reduced insulin levels, and the extract from the bark of C. cassia has been used to inhibit tumor, angiogenesis, and vascularization [12] [13] .
trans-Cinnamic acid (CA), an isomer of cinnamic acid, and its derivative cinnamaldehyde, is found in cinnamon and strawberry. CA has anti-fungal, nematicidal, and anti-microbial properties, and is known for its radio protective effect against skin damage [14] - [18] . Moreover, this substance has anti-oxidant, anti-inflammatory, anti-cancer, and anti-malarial properties [19] .
High-performance liquid chromatography (HPLC) has been used in a previous research to analyze cinnamon twigs and bark [20] . In addition, coumarin found in cinnamon has been analyzed by direct inlet probe-atmos- pheric pressure chemical ionization-mass spectrometry (DIP-APCI-MS) and liquid chromatography-mass spectrometry (LC-MS) [21] .
The objective of the present study is to analyze the CA content in C. loureirii, of C. camphora, and C. japonicum as well as in commercially produced cinnamon powder by HPLC.
2. Experimental
2.1. General Experimental Procedure
C. loureirii bark was obtained from Omniherb Co., Ltd. (Yeongcheon, Republic of Korea) and was extracted with methanol (MeOH) under reflux. The MeOH extracts of the leaf of C. loureirii and leaf, heartwood, and bark of C. japonicum and C. camphora were purchased from the Korea Research Institute of Bioscience and Biotechnology (KRIBB; Daejeon, Republic of Korea). Cinnamon powders designated A, B, and C were purchased from a local retailer in Anseong, Republic of Korea.
2.2. Apparatus and Chemicals
Evaporation was conducted with an evaporator system (EYELA rotary vacuum evaporator N1100, Tokyo Radadidai Co. Ltd., Tokyo, Japan) under reflux in vacuo. The HPLC analysis was performed by using a Waters Breeze system (Waters Co., Milford, MA, USA) equipped with a Waters 1525 binary HPLC pump and a 2489 system ultraviolet-visible (UV/VIS) detector. HPLC-grade reagents were water containing 0.2% acetic acid and MeOH, and all other reagents were of analytical grade. CA (Figure 1) was purchased from Sigma-Aldrich (St. Louis, MO, USA).
2.3. Preparation of Standards and Samples
The CA was weighed and dissolved in 100% MeOH to obtain a stock standard (1.0 mg/mL). CA aqueous solutions were prepared in concentrations of 100, 10, 1, 0.1, and 0.01 mg/mL for the calibration curve. Then 10.1 g of C. loureirii bark was extracted with 250 mL MeOH, and 10 g of each commercial cinnamon powder sample
Figure 1. Structure of trans-cinnamic acid (CA).
was extracted with 200 mL MeOH under reflux and evaporated in vacuo. Cinnamomum spp. extracts were dissolved in 1 mL of MeOH and were filtered with a 0.45-μm syringe filter. The dissolved solutions were used for HPLC analysis.
2.4. HPLC Conditions
The quantitative analysis of CA separated by HPLC was performed by using a reverse phase system. Discovery C18 column (250 mm × 4.6 mm, particle size 5 μm) was used, and a mobile phase was a gradient of water and MeOH (20% to 100% over 30 min). An aliquot volume of 10 μl was eluted with a gradient solvent system at a flow rate of 1 mL/min. UV detection was conducted at an absorbance of 280 nm. The flow rate was 1 mL/min.
2.5. Calibration Curve
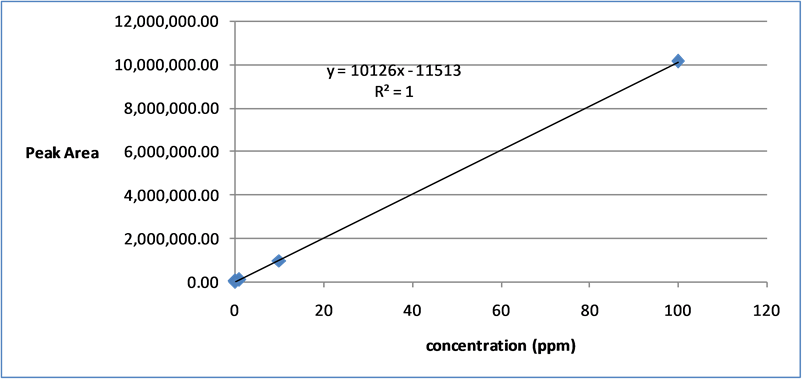
CA stock solutions (100 - 0.01 μg/mL) were prepared in MeOH. The CA contents of the samples were determined from the corresponding calibration curves. The calibration functions of CA were calculated by using the peak area (Y), concentration (X, μg/mL), and mean values (n = 5) ± standard deviation (SD; Figure 2).
3. Results & Discussion
The CA contents of Cinnamomum spp. and commercial cinnamon powder are shown in Table 1, Table 2, and Figure 3. The CA concentration in the bark of C. loureirii was determined to be 16.97 mg/g of the extract (Omniherb), and that in the leaf of C. loureirii was 2.50 mg/g of extract. The CA concentrations in the bark, leaf, and heartwood of C. camphora, and C. japonicum (KRIBB) were detected to be 0.31, 0.26, and 0.69 mg/g of extract and 0.23, 0.10, and 0.10 mg/g of extract, respectively (Table 1). Thus, the CA content of C. loureirii was higher than that of the other Cinnamomum spp. CA also can be found in cinnamon, virgin olive oil, Viola betonicifolia, and strawberries. Previous research has demonstrated that strawberries contain 2.91 to 4.97 μg/g of CA in accordance with maturation stages. Immature green strawberries contain the highest content of 4.97 μg/g [14] [22] . Propolis, also known as bee glue, contains 1% - 1.5% CA [23] .
Among the three samples of commercially available cinnamon powder, the content of CA was the highest in cinnamon powder A (47.60 mg/g; Table 2). The CA content in all of the powders was higher than that in Cinnamomum spp. Generally, cinnamon refers to several Cinnamomum spp., and thus percentage values can vary. For examples, a previous study determined that the CA content of the oils of four Cinnamomum spp. compositions differed [24] . In addition, commercially produced cinnamon powder includes two types of Cinnamomum trees: true cinnamon (C. verum) and cassia (C. aromaticum). Therefore, the results of CA content analysis can differ [5] .
Various studies have been conducted on cinnamon derivatives in commercial products. Ultra performance liquid chromatography (UPLC) has been used to analyze the coumarin content in cinnamon contained in food
Figure 2. Calibration curve of trans-cinnamic acid (CA).
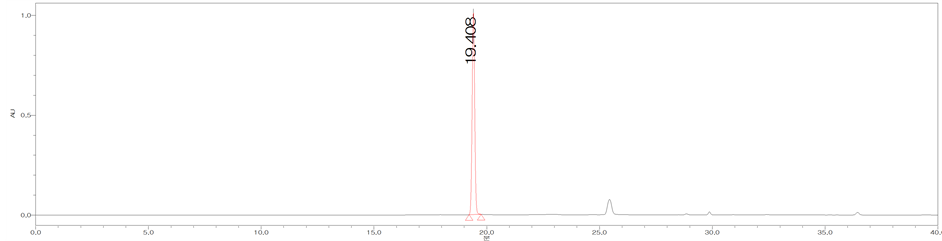
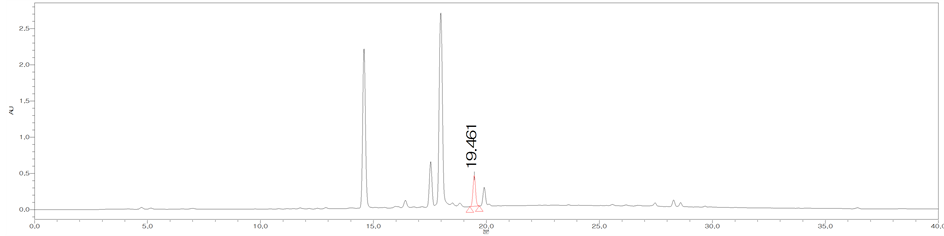
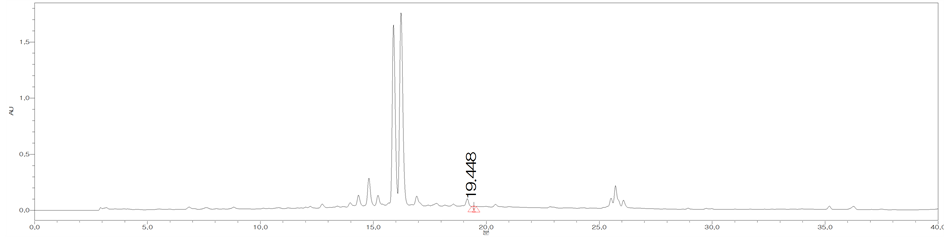
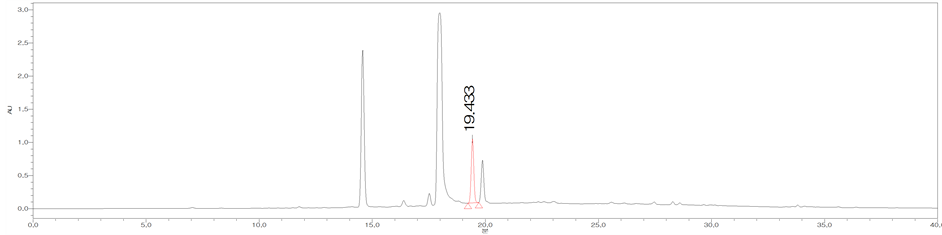
Figure 3. HPLC chromatograms of trans-cinnamic acid (CA) (a); The MeOH extract of the bark of C. loureirii (b); The leaf of C. loureirii (c); and Cinnamon powder A (d).
products such as breakfast cereal, bakery products, desserts, crisp bread, and tea. In addition, gas chromatography-flame ionization detection (GC-FID) was used to quantitatively determine the concentrations of cinnamaldehyde, cinnamylalcohol, and salicylaldehyde in cinnamon contained in biopesticides [25] [26] . Moreover, the therapeutic potential of CA has been reported. For example, CA and its derivatives have been used for α-gluco- sidase inhibition, and CA showed that anti-auxin effects in plants can be utilized as pesticides [15] [27] [28] . The CA contents in the Cinnamomum spp. extracts and in the cinnamon powder were quantified by using a
Table 1. trans-Cinnamic acid (CA) contents of the MeOH extracts of Cinnamomum spp.
Data is represented as the mean ± SD (n = 3) in mg/g of the extract. 1The bark of C. loureirii was purchased from Omniherb. 2The MeOH extracts were purchased from KRIBB.
Table 2. trans-Cinnamic acid (CA) contents of the MeOH extracts in commercially produced cinnamon powder.
Data is represented as the mean ± SD (n = 3) in mg/g of the extract.
calibration curve. Its data for the extracts confirmed a good linear relationship, and the resulting equations were operational in the concentration range. The linear calibration equation is Y = 10,126 X − 11,513, where Y is the peak area and X is the content of CA. The correlation coefficient (r2) was 1 (Figure 2).
The contents of CA in Cinnamomum spp. and in commercially available cinnamon powder were determined by using HPLC. Our results demonstrated that this simple method gives rapid and accurate results. And our results can be utilized as a guideline for analysis of CA in Cinnamomum spp. and its powders.
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (No. 2006-2005173), and grants (K14101) from the Korea Institute of Oriental Medicine.
Cite this paper
Jaemin Lee,Dong Gu Lee,Jun Yeon Park,Sungwook Chae,Sanghyun Lee, (2015) Analysis of the trans-Cinnamic Acid Content in Cinnamomum spp. and Commercial Cinnamon Powder Using HPLC. Journal of Agricultural Chemistry and Environment,04,102-108. doi: 10.4236/jacen.2015.44011
References
- 1. Lee, S.C., Lee, S.C., Lin, M.Y. and Ho, K.Y. (2010) Genetic Identification of Cinnamomum Species Based on Partial Internal Transcribed Spacer 2 of Ribosomal DNA. Journal of Food and Drug Analysis, 18, 225-231.
- 2. Al-Mamun, R., Hamid, A., Islam, M.K., Chowdhury, J.A. and Zafrul Azam, A.T.M. (2011) Lipid Lowering Activity and Free Radical Scavenging Effect of Cinnamomum tamala (Fam: Lauraceae). International Journal of Natural Sciences, 1, 93-96.
- 3. Joshi, S.C., Verma, A.R. and Mathela, C.S. (2010) Antioxidant and Antibacterial Activities of the Leaf Essential Oils of Himalayan Lauraceae Species. Food and Chemical Toxicology, 48, 37-40. http://dx.doi.org/10.1016/j.fct.2009.09.011
- 4. Palazzo, M.C., Agius, B.R., Wright, B.S., Haber, W.A., Moriarity, D.M. and Setzer, W.N. (2009) Chemical Compositions and Cytotoxicity Activities of Leaf Essential Oils of Four Lauraceae Tree Species from Monteverde, Costa Rica. Records of Natural Products, 3, 32-37.
- 5. Sangal, A. (2011) Role of Cinnamon as a Beneficial Antidiabetic Food Adjunct: A Review. Advances in Applied Science Research, 2, 440-450.
- 6. Li, R., Wang, Y. and Jiang, Z.T. (2010) Chemical Constituents of the Essential Oils of Cinnamomum loureirii Nees. from China Obtained by Hydrodistillation and Microwave-Assisted Hydrodistillation. Journal of Essential Oil Research, 22, 129-131. http://dx.doi.org/10.1080/10412905.2010.9700281
- 7. Lin, C.C., Yang, C.H., Wu, P.S., Kwan, C.C. and Chen, Y.S. (2011) Antimicrobial, Anti-Tyrosinase and Antioxidant Activities of Aqueous Aromatic Extracts from Forty-Eight Selected Herbs. Journal of Medicinal Plants Research, 5, 6203-6209.
- 8. Chen, H.P., Yang, K., You, C.H., Lei, N., Sun, R.Q., Geng, Z.F., Ma, P., Cai, Q., Du, S.S. and Deng, Z.W. (2014) Chemical Constituents and Insecticidal Activities of the Essential Oil of Cinnamonmum camphora Leaves against Lasioderma serricorne. Journal of Chemistry, 2014, 1-5.
- 9. Shareef, A.A. (2011) Evaluation of Antibacterial of Essential Oils of Cinnamomum sp. and Boswellia sp. Journal of Basrah Researches, 37, 60-71.
- 10. Li, Y.Q., Kong, D.X. and Wu, H. (2013) Analysis and Evaluation of Essential Oil Components of Cinnamon Barks Using GC-MS and FTIR Spectroscopy. Industrial Crops and Products, 41, 269-278. http://dx.doi.org/10.1016/j.indcrop.2012.04.056
- 11. Pina-Pérez, M.C., Martínez-López, A. and Rodrigo, D. (2012) Cinnamon Antimicrobial Effect against Salmonella typhimurium Cells Treated by Pulsed Electric Fields (PEF) in Pasteurized Skim Milk Beverage. Food Research International, 48, 777-783. http://dx.doi.org/10.1016/j.foodres.2012.06.027
- 12. Couturier, K., Qin, B., Batandier, C., Awada, M., Hininger-Favier, I., Canini, F., Leverve, X., Roussel, A.M. and Anderson, R.A. (2011) Cinnamon Increase Liver Glycogen in an Animal Model of Insulin Resistance. Metabolism, 11, 1590-1597. http://dx.doi.org/10.1016/j.metabol.2011.03.016
- 13. Kwon, H.K., Jeon, W.K., Hwang, J.S., Lee, C.G., So, J.S., Park, J.A., Ko, B.S. and Im, S.H. (2009) Cinnamon Extract Suppresses Tumor Progression by Modulating Angiogenesis and the Effector Function of CD8+ T Cells. Cancer Letters, 278, 174-182. http://dx.doi.org/10.1016/j.canlet.2009.01.015
- 14. Kim, D.S., Na, H., Song, J.H., Kwack, Y., Kim, S.K. and Chun, C.H. (2012) Antimicrobial Activity of Thinned Strawberry Fruits at Different Maturation Stages. Korean Journal of Horticultural Science and Technology, 30, 769-775. http://dx.doi.org/10.7235/hort.2012.12199
- 15. Bock, C.H., Shapiro-Ilan, D.I., Wedge, D.E. and Cantrell, C.L. (2014) Identification of the Antifungal Compound, Trans-Cinnamic Acid, Produced by Photorhabdus luminescens, a Potential Biopesticide against Pecan Scab. Journal of Pest Science, 87, 155-162. http://dx.doi.org/10.1007/s10340-013-0519-5
- 16. Mahajan, R., Singh, P. and Bajaj, K.L. (1992) Nematicidal Activity of Some Phenolic Compounds against Meloidogyne incognita. Nematologia Mediterranea, 20, 217-219.
- 17. Kuk, J.H., Ma, S.J. and Park, K.H. (1997) Isolation and Characterization of Cinnamic Acid with Antimicrobial Activity from Needle of Pinus densiflora. Korean Journal of Food Science and Technology, 29, 823-826.
- 18. Lukmanul Hakkim, F., Miura, M., Matsuda, N., Alharassi, A.S., Guillemin, G., Yamauchi, M., Arivazhagan, G. and Song, H. (2014) An in Vitro Evidence for Caffeic Acid, Rosmarinic Acid and Trans Cinnamic Acid as a Skin Protectant Agent against γ-Radiation. International Journal of Low Radiation, 9, 305-316. http://dx.doi.org/10.1504/IJLR.2014.063414
- 19. Rahman, A.U. (2014) Studies in Natural Products Chemistry. Vol. 42, Elsevier, Oxford, 276.
- 20. Ding, Y., Wu, E.Q., Liang, C., Chen, J., Tran, M.N., Hong, C.H., Jang, Y., Park, K.L., Bae, K.H., Kim, Y.H. and Kang, J.S. (2011) Discrimination of Cinnamon Bark and Cinnamon Twig Samples Sourced from Various Countries Using HPLC-Based Fingerprint Analysis. Food Chemistry, 127, 755-760. http://dx.doi.org/10.1016/j.foodchem.2011.01.011
- 21. Krieger, S., Hayen, H. and Schmitz, O.J. (2013) Quantification of Coumarin in Cinnamon and Woodruff Beverages Using DIP-APCI-MS and LC-MS. Analytical and Bioanalytical Chemistry, 405, 8337-8345. http://dx.doi.org/10.1007/s00216-013-7238-x
- 22. Carrasco Pancorbo, A., Cruces-Blanco, C., Segura Carrentero, A. and Fernández Gutiérrez, A. (2004) Sensitive Determination of Phenolic Acids in Extra-Virgin Olive Oil by Capillary Zone Electrophoresis. Journal of Agricultural and Food Chemistry, 52, 6687-6693. http://dx.doi.org/10.1021/jf0497399
- 23. Croci, A.N., Cioroiu, B., Lazar, D., Corciova, A., Ivanescu, B. and Lazar, M.I. (2009) HPLC Evaluation of Phenolic and Polyphenolic Acids from Propolis. Farmacia, 57, 52-57.
- 24. Chalchat, J.C. and Valade, I. (2000) Chemical Composition of Leaf Oils of Cinnamomum from Madagascar: C. zeylanicum. Blume, C. camphora L., C. frangas Baillon and C. angustifolium. Journal of Essential Oil Research, 12, 537- 540. http://dx.doi.org/10.1080/10412905.2000.9712153
- 25. Ballin, N.Z. and Sorensen, A.T. (2014) Coumarin Content in Cinnamon Containing Food Products on the Danish Market. Food Control, 38, 198-203. http://dx.doi.org/10.1016/j.foodcont.2013.10.014
- 26. Lim, S.J., Lee, J.H., Kim, H.J., Choi, G.H., Cho, N.J. and Park, B.J. (2014) Quantitative Analysis of Cinnamaldehyde, Cinnamylalcohol and Salicylaldehyde in Commercial Biopesticides Containing Cinnamon Extract Using Gas Chromatography—Flame Ionization Detector. Korean Journal of Environmental Agriculture, 33, 213-219. http://dx.doi.org/10.5338/KJEA.2014.33.3.213
- 27. Adisakwattana, S., Sookkongwaree, K., Roengsumran, S., Petsom, A., Ngamrojnavanich, N., Chavasiri, W., Deesamer, S. and Yibchok-anun, S. (2004) Structure-Activity Relationships of Trans-Cinnamic Acid Derivatives on Alpha-Glucosidase Inhibition. Bioorganic and Medicinal Chemistry Letters, 14, 2893-2896. http://dx.doi.org/10.1016/j.bmcl.2004.03.037
- 28. Van Overbeek, J., Blondeau, J. and Horne, V. (1951) Trans-Cinnamic Acid as an Anti-Auxtin. American Journal of Botany, 38, 589-595. http://dx.doi.org/10.2307/2437768
NOTES
*Corresponding author.