Journal of Minerals and Materials Characterization and Engineering
Vol.2 No.1(2014), Article ID:41840,6 pages DOI:10.4236/jmmce.2014.21003
Electrical Properties of Nano-TiO2 Thin Film Using Spin Coating Method
1Department of Materials Science and Engineering, Obafemi Awolowo University, Ile-Ife, Nigeria
2Engineering Materials Development Institute, Akure, Nigeria
3Centre for Energy and Research Development, Obafemi Awolowo University, Ile-Ife, Nigeria
4Department of Metallurgical and Materials Engineering, Federal University of Technology, Akure, Nigeria
5Department of Mechanical and Manufacturing Engineering, University of Manitoba, Winnipeg, Canada
Email: *daniyanayodele@yahoo.com
Copyright © 2014 Ayodele Abeeb Daniyan et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Ayodele Abeeb Daniyan et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received September 17, 2013; revised November 9, 2013; accepted November 24, 2013
KEYWORDS
Titanium Oxide; ITO; Spin Coating; KFPPT; Sheet Resistance and Conductivity
ABSTRACT
This work investigated the electrical properties of TiO2 thin film on empty glass and ITO glass by spin coating method. Highly transparent titanium oxide thin films were prepared on empty glass and Indium Tin Oxide (ITO) from a titanium (III) chloride precursor. The sheet resistance of the film prepared from both the synthesised and the commercially available TiO2 was measured by the Keithley four-point probe tester (KFPPT). A comparable sheet resistance of 10.69E + 00 Ω/sq was obtained on the ITO. The electrical conductivity of the TiO2 film on ITO glass substrate was found to be very high (3.46E + 05 Ω−1·cm−1) and comparable to that of the bare ITO glass.
1. Introduction
Materials reduced to the nanoscale can display very different properties compared to what they exhibit on a micro scale, enabling unique applications. For instance, opaque substances become transparent (copper); inert materials attain catalytic properties (platinum); stable materials turn combustible (aluminium); solids turn into liquid at room temperature (gold); insulators become semi conductors (silicon). A wide range of these materials are now available and in use in many commercial, industrial and military applications [1]. Similarly, when TiO2 is reduced to nanoscale, it shows unique properties, of which the electrical aspect is highly important.
Some of the most current researches and development works are in the area of these nanomaterials. This describes applications in which material nanostructures are used to produce optical electronics or magnetic properties [2].
Titanium (IV) oxide is the naturally occurring oxide of titanium, chemical formula TiO2. It has a wide range of applications, from paint to sunscreen and then to food colouring. Titanium dioxide (TiO2) has attracted significant attention from researchers because of the many interesting physical and chemical properties that make it suitable for a variety of applications. For instance, TiO2 has high corrosion resistance and chemical stability and an excellent optical transparency in the visible and near infrared regions. It also has high refractive index that makes it useful for anti-reflection coatings in optical devices [3]. It has been used mostly as a pigment in paints, sunscreens, ointments toothpaste etc.
Recently, titanium oxide (TiO2) thin films have emerged as one of the most promising oxide materials owing to their optical, electrical and photo electrochemical properties.
Many studies on TiO2 thin films formed by conventional and advanced sol-gel processes have been reported [4,5].
Previous studies have shown that the properties of TiO2 films appear to strongly depend on the process conditions and starting materials used in the processes; many researchers have used sol-gel method to synthesize TiO2, for instance, in 2001, Tang et al. worked on synthesis of nano rutile TiO2 powder at low temperature by sol-gel method using Ti(OC4H9)4 and HNO3 and obtained mean particle size of about 50 nm after calcination at 600˚C in rutile phase [6]. Similarly, Yu et al. researched into the photocatalytic activity of nano-sized TiO2 powders by sol-gel method, using TTIP and EtOH/H2O solution. The results obtained were anatase (75.1%) and brookite (24.9%) phases at 400˚C. The particle sizes obtained were 7.9 nm and 7.4 nm respectively [7]. Likewise, Li et al., 2004 also studied the “Preparation and characterization of nano-TiO2 powder by sol-gel method” and yielded a mean size of about 10 nm after calcinations [8]. By similar method, we investigated the “Microstructure control of thermally stable TiO2 obtained by hydrothermal process”. The results obtained were: Hydrothermally treated TiO2 nanoparticles at pH 3 were 13 and 34 nm in average diameter after calcinations at 600˚C and 800˚C. Hydrothermally treated TiO2 nanoparticles at pH 2 were 11 and 26 nm in average diameter after calcinations at 600˚C and 800˚C [9]. Furthermore, by sol gel route, Qiu and Kalita, 2006 studied on the synthesis, characterization of nano-TiO2. The results obtained were: 5 to 10 nm in diameter after calcination at 400˚C, in pure anatase phase, anatase (53.4%) and rutile (46.6%) phase after calcinations at 600˚C and the particle sizes were 22.6 nm and 29.3 nm respectively, and 46.2 nm in diameter after calcinations at 800˚C, in pure rutile phase [10]. Therefore, this work seeks to investigate the electrical properties of the synthesized nano-TiO2 for opto-electronics application.
2. Experimental Procedure
2.1. Materials
The Titanium (III) Chloride (MW = 154.23 g) used for the research work was obtained from the Engineering Materials Development (EMDI), Akure. Chitosan powder was obtained from the Department of Chemistry, Federal University of Technology, Akure. Acetic acid (60.06 g/mol), acetone (M = 58.08 g/mol), ethanol (MW = 46.0 g/mol), ultra pure deionized water and other reagents which are analytical grade were obtained from Pascal Chemical Nigeria Limited, Akure.
2.2. Synthesis of TiO2 (Rutile)
5 grams of chitosan powder was poured into a 250 ml vessel containing 100 ml of deionized water and 5 ml of acetic acid. The mixture was stirred for 6 h at 90˚C, and finally cooled naturally to room temperature. 10 grams of this as-synthesized chitosan solution was added dropwise into a 250 ml vessel containing 40 ml of acetone and 4 ml of TiCl3 solution, and then the vessel was covered with parafilm and left at room temperature for 2 weeks. The resulting white deposit was immersed in water to dissolve the chitosan, and then the suspension was centrifuged and washed several times with deionized water and ethanol. Finally, the powder was dried at 60˚C in an oven of 250˚C capacity [11].
2.3. Preparation of TiO2 Thin Film
A commercially available TiO2 powder was obtained to comparatively study the synthesised TiO2 powder sample.
The commercially available TiO2 powder and the synthesized TiO2 and were used to prepare TiO2 thin film on both empty glass substrate and Indium Tin Oxide (ITO) coated glass substrate using Spin coater. Prior to the preparation of TiO2 thin film, the glass substrate and the Indium Tin Oxide (ITO) coated glass substrate used were cut and ultrasonically cleaned by degreasing with acetone, methanol, rinsed in isopropanol, kept in staining jar and allow to dry in vacuum oven.
5 mg of commercially available powder of TiO2 and synthesized sample were thoroughly mixed with 5 ml of Polyethylene Oxide (PEO) solution. The resulting colloidal solution was spin coated on glass substrate and ITO at 1500 rpm. The thickness of the film was measured using the weight differential method [12].
3. Results and Discussion
The results are shown in Figures 1-4 and Tables 1 and 2.
X-ray diffraction (XRD) pattern (radiation used: CuKα) of both the synthesized and commercially available powder samples are shown in Figures 1 and 2 (from the diffraction pattern). There are appreciable similarities in their structures, which suggest that the two samples are of similar material. The d-values of synthesized sample obtained in this work are in agreement with the standard d-values of rutile structure with error of about ±1. The

Table 1. Diffraction angles of the synthesized (S 2θ˚) commercially available TiO2 powder (R 2θ) and the standard (STD2θ˚) with the lattice planes.
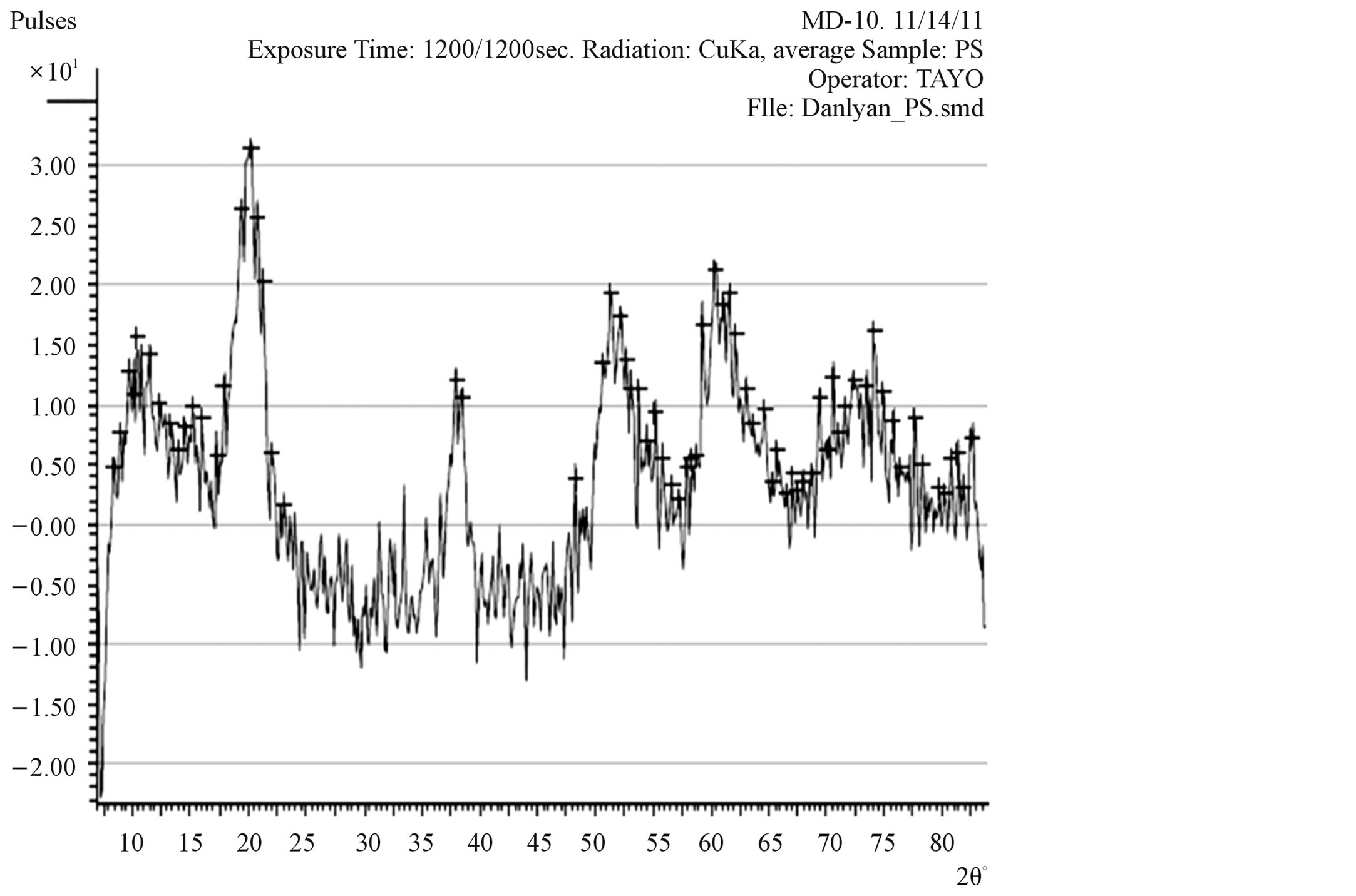
Figure 1. XRD pattern of synthesised TiO2 powder.
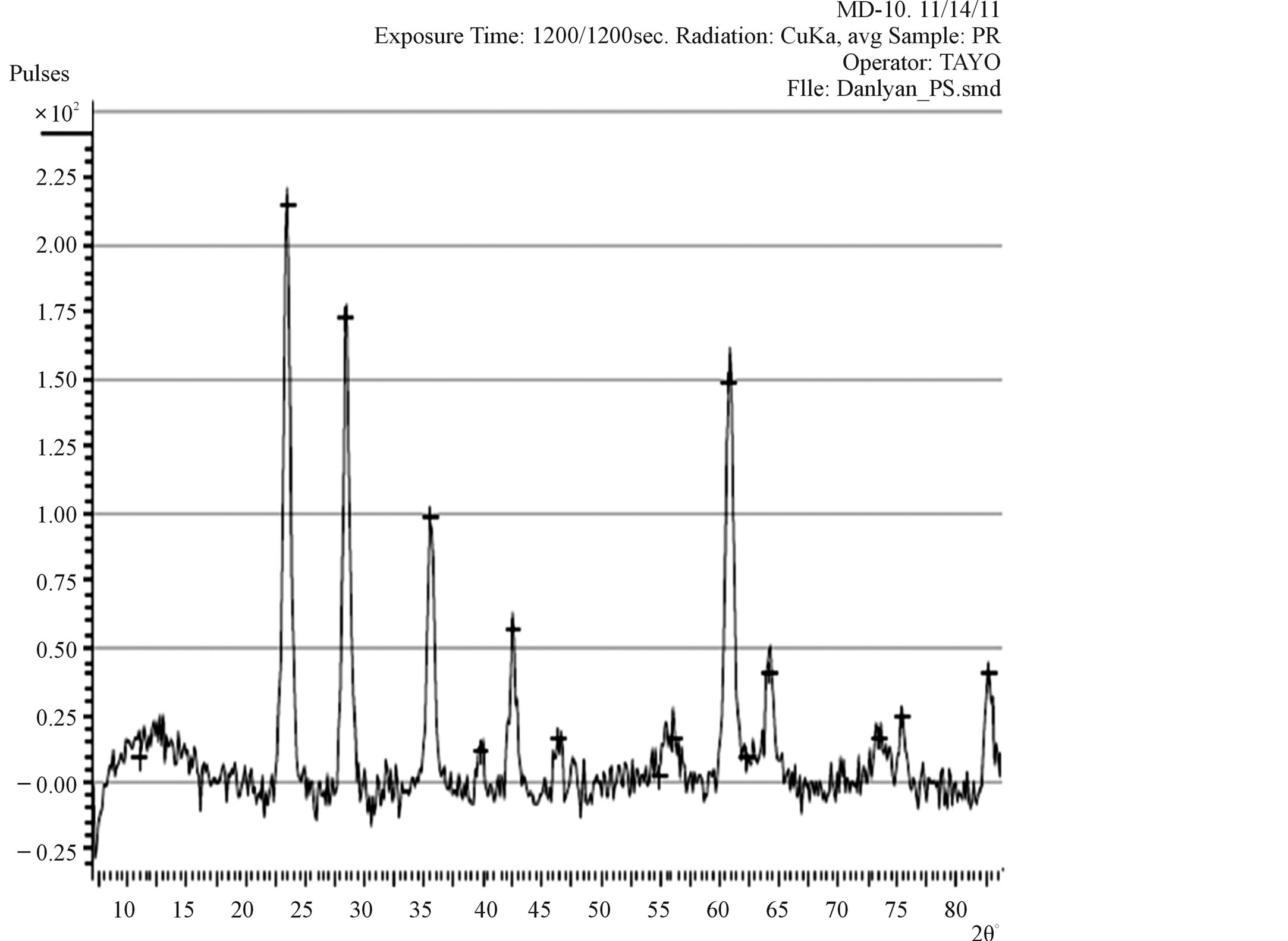
Figure 2. XRD Pattern of the commercially available TiO2 powder.
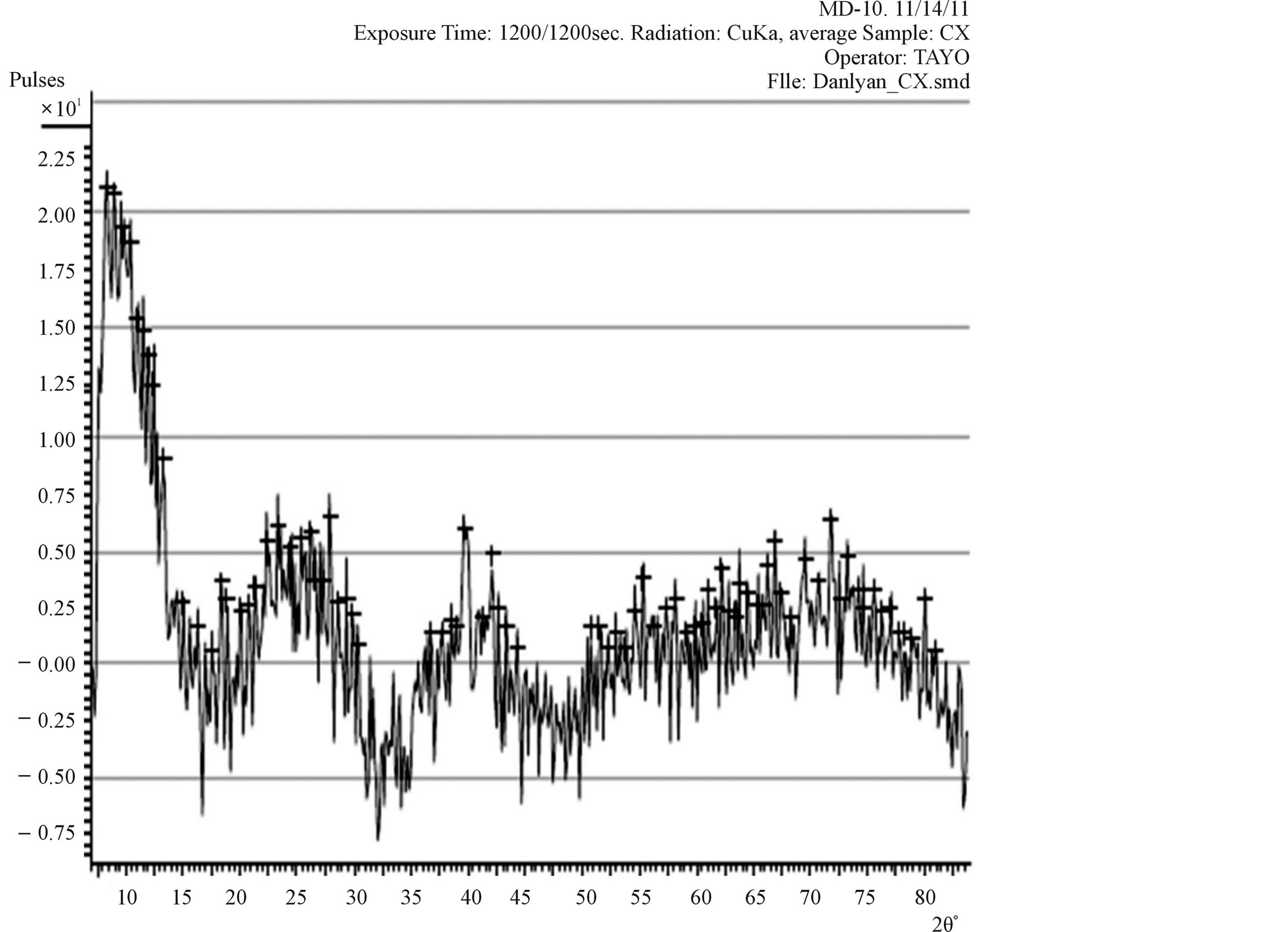
Figure 3. The XRD pattern of the TiO2 film prepared from synthesised TiO2 powder.
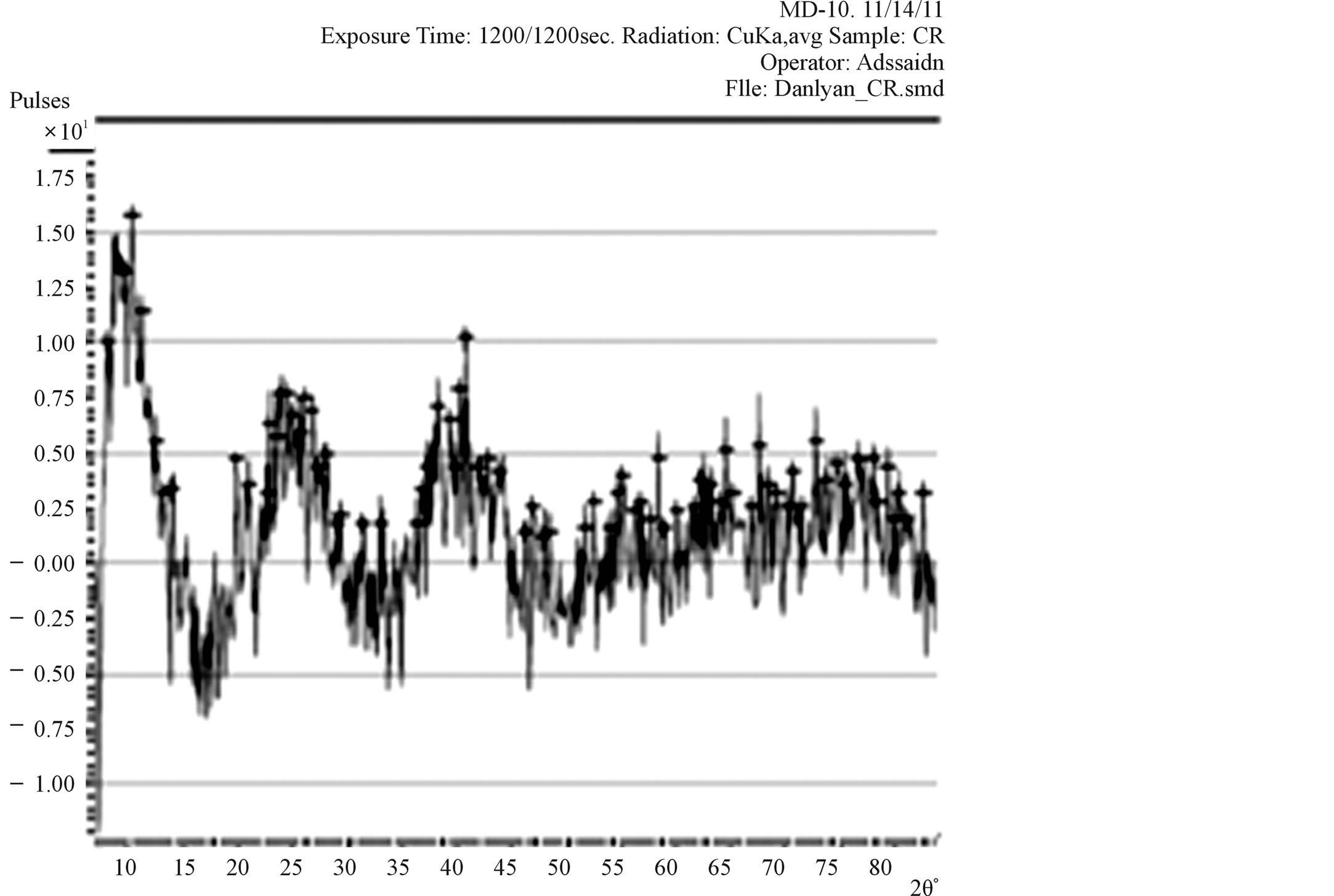
Figure 4. XRD Pattern of TiO2 film prepared from commercially available TiO2 powder.
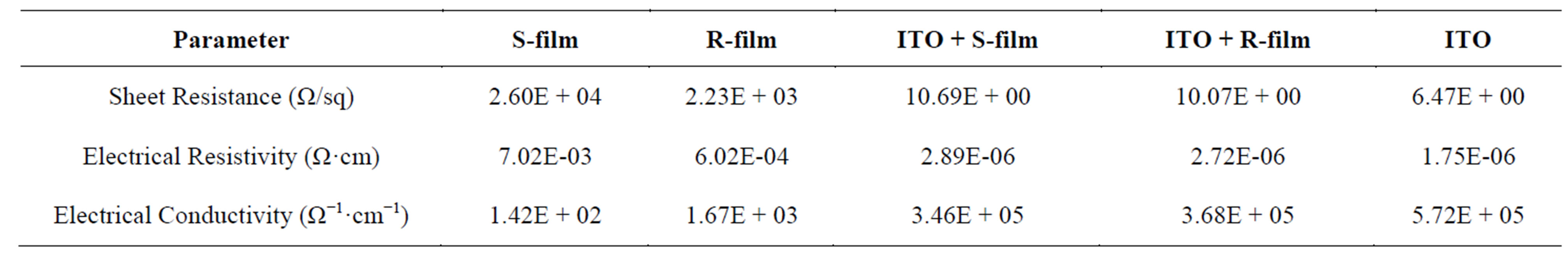
Table 2. Summary of the electrical characterization.
average particle sizes of Figures 1 and 2 were calculated using Sherrer’s equation. Sherrer’s equation is as follows:
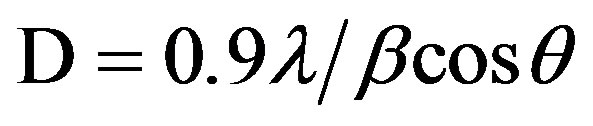 (1)
(1)
where λ is the wavelength of X-ray (0.1540 nm), β is the full-width at half-maximum of the peak (in radian), and the θ is the Bragg’s angle of the X-ray diffraction peaks.
The sharp diffraction peaks indicated the polycrystallinity of the TiO2 powder. The broadening of some peaks observed on the synthesised sample, according to [13], the broadening could be as a result of smaller crystals. Other causes of broadening are: instruments used and synthesis temperature of the powder [14]. The broadening could be improved by calcination [15]. The little shift observed in the diffraction peaks that correspond to the main peak of the synthesised samples could be caused by particle size variation [16]. Also, the correlation between the values of diffraction angles of the synthesized sample with the commercially available one with respect to the standard value of diffraction angles of rutile structure obtained by the XRD machine (Table 1) shows that the two powder samples are of the same material. Hence, the structure further confirmed the sample synthesised to be rutile.
The particle size, 15.9 nm obtained from the synthesised sample was smaller than the commercially available one (26.7 nm) and much smaller than 50 nm, the result obtained by [6] who used sol gel method from Ti (OC4H9)4 precursor.
The Electrical Characterisation
The electrical resistivity of the films were obtained using Four-point probe system coupled with Keithley 2400 Series Source meter, interfaced by a Lab view Tracer software. This was used to measure sheet Resistance Rs and by employing set of equations by [17] (Bautista, 2004), the value of the electrical conductivity can be calculated. Thus
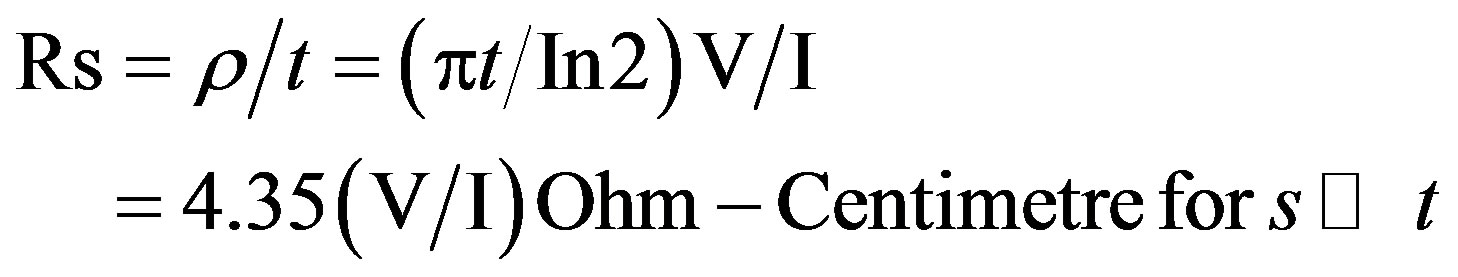 (2)
(2)
The electrical resistivity ρ was determined using the formula
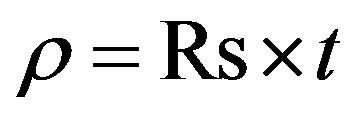 (3)
(3)
and henceThe electrical conductivity was obtained by using the equation
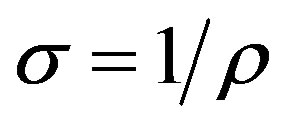 (4)
(4)
The results from the electrical characterization are given in the Table 1 below. S-film represent the TiO2 film prepared from synthesised TiO2 powder on bare glass, R-film represent TiO2 film prepared from commercially available TiO2 powder on empty glass, ITO + S represent the TiO2 film prepared from synthesised TiO2 powder on ITO glass substrate and ITO + R represent TiO2 film prepared from commercially available TiO2 powder on ITO glass substrate.
The values obtained are shown in Table 1. The electrical conductivity, which may be influenced by carrier concentration, also increased from 1.42E + 02 Ω−1·cm−1 (on empty glass) to 3.46E + 05 Ω−1·cm−1 (on ITO substrate) for the synthesised sample. This was in the same trend as that of the commercially available one (1.67E + 03 to 3.68E + 05), which imply a little reduction in the value of conductivity of the bare ITO. The reason could be traced to composite effect of the ITO-TiO2 mixture. These results are in good agreement with those reported by [18,19]. The higher conductivity of the thin film TiO2 coated on ITO glass compared to the thin film coated on empty glass suggest that Indium Tin Oxide (ITO) affected the charge transport even when all the four electrical contacts are on the TiO2 layer. This indicated lower resistance across ITO/TiO2 interface. This lower resistance interface may be understood from the chemistry of growth for TiO2 shell. The value of sheet resistance obtained for the TiO2 coated on the empty glass substrate was reasonable and falls within the range of that of semiconductor, since it lies between the sheet resistance of TiO2 thin film (2.6 × 10−1 106 Ω/sq) [20]. To obtain material for the design of devices which requires very low sheet resistance similar to what is obtained from dielectric/metal/dielectric (DMD structure, TiO2 coated on ITO glass substrate is recommended. Example of such is TiO2/Ag/TiO2 which can have sheet resistance as low as 30 ohm/square. Since a pure Ag has lowest resistivity and relatively low absorption in the visible region [21] (Jia et al., 2003).
4. Conclusion
From the electrical characterization of the TiO2 film, it can be concluded that the film has exhibited semiconductor property. The crystallized TiO2 on ITO substrate decreases the electrical conductivity of ITO from 5.72E + 05 to 3.46E + 05 Ω−1·cm−1 and could be served as a raw material for the design of devices which requires very low sheet resistance.
Acknowledgements
The authors appreciate the support provided by the management of Engineering Materials Development Institute, Akure, Nigeria where the bench work was carried out.
REFERENCES
- A. A. Afonja, “Novel Materials for Energy Applications,” Nigerian Journal of Materials Science and Engineering, Vol. 1, No. 1, 2009, pp. 63-72.
- O. O. Adewoye, “Advances in Engineering Materials Development,” In Proceedings of Nigeria Materials Congress (NIMACON 2007), Centre for Energy, Research and Development (CERD), Obafemi Awolowo University, Ile-Ife, 2007, pp. 118-122.
- C. X. Shan, X. H. Hou and K.-L. Choy, “Corrosion Resistance of TiO2 Films Grown on Stainless Steel by Atomic Layer Deposition,” Surface and Coatings Technology, Vol. 202, No. 11, 2008, pp. 2399-2402. http://dx.doi.org/10.1016/j.surfcoat.2007.08.066
- T. Nishide, M. Sato and H. Hara, “Crystal Structure and Optical Property of TiO2 Gels and Films Prepared from Ti-Edta Complexes as Titania Precursors,” Journal of Materials Science, Vol. 35, No. 2, 2000, pp. 465-469. http://dx.doi.org/10.1023/A:1004731804075
- B. H. Kim, J. H. Ahn, J. H. Jeong, Y. S. Jeon, K. O. Jeon and K. S. Hwang, “Preparation of TiO2 Thin Film on SiO2 Glass by a Spin Coating—Pyrolysis Process,” Ceramics International, Vol. 32, 2006, pp. 223-225.
- Z. L. Tang, J. Y. Zhang, Z. Cheng and Z. T. Zhang, “Synthesis of Rutile TiO2 at Low Temperature,” Materials Chemistry and Physics, Vol. 77, No. 2, 2001, pp. 314- 317. http://dx.doi.org/10.1016/S0254-0584(02)00003-2
- J. C. Yu, J. G. Yu, L. Z. Zhang and W. K. Ho, “Photocatalytic Activity of Nano-Sized TiO2 Powders by Sol-Gel Method, Using Titanium Tetraisopropoxide and EtOH/ H2O Solution,” Journal of Photochemistry and Photobiology A: Chemistry, Vol. 148, No. 1-3, 2002, pp. 263- 271. http://dx.doi.org/10.1016/S1010-6030(02)00052-7
- Y. Z. Li, N. H. Lee, E. G. Lee, J. S. Song and S. Kim, “Preparation and Characterization of Nano-TiO2 Powder by Sol-Gel Method,” Chemical Physics Letters, Vol. 389, No. 1-3, 2004, pp. 124-128. http://dx.doi.org/10.1016/j.cplett.2004.03.081
- A. M. Ruiz, G. Sakai, A. Cornet, K. Shimanoe, J. R. Morante and N. Yamazoe, “Microstructure Control of Thermally Stable TiO2 Obtained by Hydrothermal Process,” Sensors and Actuators B: Chemical, Vol. 103, No. 1-2, 2004, pp. 312-317. http://dx.doi.org/10.1016/j.snb.2004.04.061
- S. Qiu and S. Kalita, “Synthesis, Processing and Characterization of Nanocrystalline Titanium Dioxide,” Department of Mechanical, Materials and Aerospace Engineering College of Engineering and Computer Science, University of Central Florida, Orlando, Florida. http://www.google.com.ng/url?q=http://etd.fcla.edu/CF/CFE0001432/QIU_SHIPENG_200612_MS.pdf&sa=U&ei=4myxUpOtLaen0AXwxoAY&ved=0CB8QFjAA&usg=AFQjCNGnT3HeNSMtrE8rS0BDb4bbsDiXfw
- S. F. Chen, J. P. Li, K. Qian, W. P. Xu, Y. Lu, W. X. Huang and S. H. Yu, “Large Scale Photochemical Synthesis of M@TiO2 Nanocomposites (M = Ag, Pd, Au, Pt) and Their Optical Properties, CO Oxidation Performance, and Antibacterial Effect,” Nano Research, Vol. 3, No. 4, 2010, pp. 244-255. www.springerlink.com
- J. Y. Choi, K. Kim, J. Yoo and D. Kim, “Properties of Cadmium Sulphide Films Deposited by Chemical Bath Deposition with Ultrasonication,” Solar Energy, Vol. 64, No. 1-3, 1998, pp. 41-47. http://dx.doi.org/10.1016/S0038-092X(98)00047-4
- D. S. Reddy, D. R. Reddy, B. K. Reddy, A. M. Reddy, K. R. Gunasekhar and P. S. Reddy, “Annealing Effect on Physical Properties of Thermally Evaporated MnS Nanocrystalline Films,” Journal of Optoelectronics And Advanced Materials, Vol. 9, No. 7, 2007, pp. 2019-2022.
- R. K. Wahi, Y. Liu, J. C. Falkner and V. L. Colvin, “Solvothermal Synthesis and Characterization of Anatase TiO2 Nanocrystals with Ultrahigh Surface Area,” Journal of Colloid and Interface Science, Vol. 302, No. 2, 2006, pp. 530-536. http://dx.doi.org/10.1016/j.jcis.2006.07.003
- M. Addamo, V. Augugliaro, A. Paola, E. García-López, V. Loddo, G. Marcì and L. Palmisano, “Photocatalytic Thin Films of TiO2 Formed by a Sol-Gel Process Using Titanium Tetraisopropoxide as the Precursor,” Thin Solid Films, Vol. 516, No. 12, 2007, pp. 3802-3807. http://dx.doi.org/10.1016/j.tsf.2007.06.139
- B.-H. Kim, J.-H. Ahn, J.-H. Jeong, Y.-S. Jeon, K.-O. Jeon and K.-S. Hwang, “Preparation of TiO2 Thin Film on SiO2 Glass by a Spin Coating—Pyrolysis Process,” Ceramics International, Vol. 32, No. 2, 2006, pp. 223-225. http://dx.doi.org/10.1016/j.ceramint.2005.01.016
- K. Bautista, “Thin Film Deposition,” Four-Point Probe Operation, 2004, pp. 1-8. Accessed on 18th July, 2011.
- M. Stamate, G. Lazar and I. Lazar, “Anatase—Rutil TiO2 Thin Films Deposited in a D.C. Magnetron Sputtering System,” Romanian Journal of Physics, Vol. 53, No. 1-2, 2008, pp. 217-221.
- R. Li and L. Chen, “A Nano Silver, and Its Preparation Method,” Chinese Patent CN10027622, 2005.
- T. Miyata, S. Tsukada and T. Minami, “Preparation of Anatase TiO2 Thin Films by Vacuum Arc Plasma Evaporation,” Thin Solid Film, Vol. 496, No. 1, 2006, pp. 136- 140. http://dx.doi.org/10.1016/j.tsf.2005.08.294
- J. H. Jia, P. Zhou, H. Xie, H. Y. You, J. Li and L. Y. Chen, “Study of Optical and Electrical Properties of TiO2/ Ag/TiO2 Multilayers,” Journal of the Korean Physical Society, Vol. 44, No. 3, 2003, pp. 717-721.
NOTES
*Corresponding author.

