Food and Nutrition Sciences
Vol.5 No.3(2014), Article ID:42252,6 pages DOI:10.4236/fns.2014.53032
Comparison of the Effectiveness of Different Traditional Soaking Processes on the in Vitro Digestibility of Taro (Colocasia esculenta L. SCHOTT) Flour
1Institut Universitaire des Sciences et Techniques, Abéché, Tchad; 2Biotechnopôle, Institut de Recherche en Élevage pour le Développement, N’Djamena, Tchad; 3Laboratoires TBC TransCell-Lab, Faculté de Médecine Xavier Bichat, Université ParisDiderot Paris 7, Paris, France; 4Laboratoire de Nutrition et Alimentation, VetAgro Sup-Campus Vétérinaire, Université de Lyon, Lyon, France.
Email: *soudyidji@yahoo.fr
Received October 18th, 2013; revised November 18th, 2013; accepted November 25th, 2013
ABSTRACT
A traditional process used by farmers in Chad consists in soaking slices of taro (Colocasia esculenta L. SCHOTT) in tamarind infusion, or in corn solution or in water over a 24-hour period to reduce the acridity of taro and facilitate cooking. The aim of this study was to assess the effect of traditional soaking on the in vitro digestibility of taro flour using or not using an α-amylase enzyme. The digestion without the enzyme has shown that the soaking processes improve the digestibility of taro flour (from 39.30% for the control sample to 75.11% (after tamarind infusion) and 78.67% (treatment with water) after 24 hours of soaking). Soaking over a 6-hour period and preferentially in tamarind infusion or in corn solution obtains highly digestible flour (around 95% of digestibility rate after 3 hours of enzymatic digestion).
Keywords:Colocasia esculenta L. SCHOTT; Soaking; Corn; Zea mays L.; Tamarind; Tamarindus indica L.; In Vitro Digestibility; Amylase
1. Introduction
Taro is a carbohydrate rich tuber, between 73% and 80% of dry matter (DM) basis [1], mainly starch (77%) and 1.4% crude fibre of DM basis [2]. Because of its high carbohydrate content, it represents one of the principal sources of energy in many parts of tropical and subtropical countries providing about a third of the food intake for more than 400 million people in these countries [3]. Africa represents 75% of the total world production [4].
The traditional process of treatment of taro used in the Mayo Kebbi area (Chad) is reputed to decrease the acridity of this tuber and to facilitate cooking [5]. It has been shown that these processes also modify the chemical composition of taro flour and its nutritional value [6]. This traditional process reduces the cooking time from 2 (two) hours to 30 (thirty) minutes. This method is based on the soaking of fresh taro chips in a tamarind infusion or in a solution of corn or in water during 24 hours. After drying in the sun, the dried chips are ground into flour. This flour is then used to prepare mixtures of porridge, soups and cakes. Moreover, according to the FAO report [3], taro corn has good nutritional properties and is supposed to contain anti-allergic factors.
Tamarind (Tamarindus indica L.) used in this process, has many uses, but it is best known as a souring agent in food flavoring. The fruit pulp of this tree is particularly rich in carbohydrates (60% - 70%) and contains an appreciable amount of linoleic acid and oleic acid. Among vitamins, the fruit of tamarind is particularly rich in ascorbic acid and beta carotene [7]. In the west and the central Africa, drinks of tamarind infusion are popular. It also has medicinal properties and it is mainly used against constipation.
Generally, the apparent digestibility is a global concept involving all the phenomena that concern the bowels in the small intestine and the large intestine [8]. It is also represented by the coefficient of apparent digestibility that is to say the measurement of the disappearance of nutrients and the difference between dietary intake and the faecal output [9]. This quantitative notion is quite often associated with the notion of kinetics in time. It suggests that the food is digested more quickly and more easily.
Njintang [10] mentioned that the cooking of unpeeled taro improved the digestibility of “Achu”, a taro-based food widely consumed in Cameroon.
Favier [11] showed that fermentation did not increase the sensitivity of cassava starch in relation to the action of the amylase enzyme. On the contrary, even brief and moderate heating facilitated the enzymatic activity of the animal to metabolize the starch. On the other hand, Elkhalifa et al. [12] showed that fermentation significantly improved the in vitro digestibility of sorghum starch grains. Admassu and Kumar [13] reported that the natural lactic fermentation of ground beans induced a higher protein digestibility. In addition, they observed that natural or controlled fermentation diminished the antinutrients and improved the nutritional value of the bean flour. Nour et al. [14] showed that sprouting changed the levels of nutrients, significantly reducing tannins and phytates and increasing protein digestibility in different types of Sudanese sorghum cultivars. All biological treatments improved the global digestibility of the food by modifying the chemical structure of the nutrients and/or reducing the antinutrients levels under different enzymatic actions on phytates or fibre [15].
Previous research has shown that the rate of enzymatic hydrolysis of starch depends on physical and chemical structure. The structure of starch varied with the botanical origin of the starch and with the applied technological treatments. The grains of starch of taro are very small (10 - 20 mm). This size is greater than rice (4 - 6 µm), close to corn (15 - 25 µm) but much lower than cassava (25 - 35 µm) or potato (100 - 200 µm) [3]. This reduces size of grains improving the digestibility of the starch and makes it more suitable for feeding infants and patients.
Literature concerning the effects of traditional soaking on digestibility does not exist. Therefore it is advisable to estimate and to compare the influence of these various treatments of soaking on the in vitro digestibility of taro flour.
2. Materials and Methods
2.1. Samples Source
Samples of taro were collected from Kolobo (village in the Mayo-Kebbi area (Chad), latitude 9˚93'N; longitude 15˚44'E) at the end of November, at the end of the rainy season, as usual, after 6 months of growth. Tubers harvested in November are all processed and eaten by local people. Among the tubers harvested in February, only the largest are processed for human consumption and the smallest are stored for the next crop.
2.2. Experimental Design
Fresh tubers for human consumption might be preserved in baskets for 3 weeks in fresh, dry and well ventilated places [5]. But, to avoid any fermentation and in agreement with the local practices they were immediately processed according to the traditional techniques used by the farmers of this region.
The freshly harvested tubers of taro were carefully washed in tap water and peeled (thickness of peelings: about 5 mm). The peeled tubers were then cut into 1 cm thick slices (or chips).
The chips were immediately dipped into three different solutions: water or corn infusion or tamarind infusion. Both corn and tamarind infusions were prepared with 45% (mass/volume) of product macerating respectively 3 days and 3 hours in water. They were then filtered through a cooking sieve.
The proportion of taro chips used in relation to the soaking solution was 40% (m/v) and the temperature of the soaking solutions was 22˚C. Five different soaking durations were applied: 0, 1 h, 3 h, 6 h, 12 h and 24 h. An untreated sample of dried chips (Time 0) served as a control. This is equivalent to a type of experimental design 1 × 3 × 5:1 variety of tuber, 3 soaking solutions and 5 soaking times and 2 replicate analyses were performed for each sample. Once soaked, the chips were dried in the sun for 24 hours at a temperature of about 46˚C. The dried chips were ground in the laboratory with an electric grinder to obtain flour with a particle size less than 500 µm (Figure 1).
We measured the enzymatic digestibility using an alpha amylase method. The method reported by Zhang et al. [16], slightly modified by Huijing et al. [17], was used. An 1.0 g quantity of dried taro flour was mixed with 30 ml phosphate buffer (0.2 M, pH 6.9) [Na2HPO4 (PROLABO-France) KH2PO4 (PROLABO-France)] in a 50 ml test tube. Once heated in a water bath (GYRO-

Figure 1. Traditional production of taro flour used in Chad.
TORY Water Bath Shaker, model G 76) at 95˚C for 30 min and cooled to 25˚C, 1ml enzymatic suspension of α-amylase (320 Units bacterial, Sigma Chemical Co., St. Louis, MO) was added. After incubation at 30˚C in a water bath shaker, the reaction was stopped by addition of 5 ml of a 1.0% (w/v) sulphuric acid solution. Samples were centrifuged at 5600 rpm, and the undigested flour residue was washed with 80% ethanol, centrifuged again and dried to constant weight. For each sample, a portion of taro flour without enzymatic hydrolysis was included to correct for initial concentration of soluble sugars. Starch digestibility was expressed as percentage weight loss after α-amylase digestion.
Thus, the in vitro digestibility of the starch of various taro flours was determined by the study of the kinetics of its hydrolysis by the bacterial amylase over 5 periods of digestion. It corresponded to an experimental design of factorial type 1 × 3 × 5 × 5:1 variety of tuber (taro), 3 soaking solutions (water, infusion of tamarind, solution of corn); 5 soaking times (0, 1 hour, 3 hours, 6 hours, 12 hours and 24 hours) and 5 digestion times (0, 3, 6, 9 and 12 hours). The soaking time “T0” represents the control or sample not treated whereas the time zero digestion (0) represents the sample without enzyme.
2.3. Statistical Analysis
The effects of soaking for each type of sample were evaluated by the analysis of variance and every sample was repeated according to the soaking solution and digestion time. Calculations of data were carried out by SPSS software (SPSS Inc, Chicago, Il, USA) and by Excel: Mac 2004, version 11.1.1 on the Apple-iBook G4 computer. The threshold significance of all the tests was fixed at P < 0.05.
3. Result and Discussion
The evaluation of the digestibility of taro flour “T0” is equivalent to the incubation without enzymes. The results presented in Figure 2 show that compared with the untreated sample, the traditional soaking processes induce an improvement of taro digestibility. This digestibility was estimated at 39.30% for the control sample and a maximum rate of 78.67% has been reached for the samples of taro that have been soaked in water. Thus, we considered that the fermentation process initiated during soaking and followed by cooking has led to a hydrolysis of taro flour starch. There might also be a degradation of the chain of taro flour starch during the cooking process. According to the FAO [3], taro flour presented as its main advantage a high digestibility factor. Due to this significant property, this food is used as ingredient in the preparation of food for infants and sick patients.
This first digestion test showed that the digestibility of taro starch varied according to the nature of the soaking solution (Figure 2). After one hour of soaking, all the samples were seen to be differently digestible and the results of each different soaking solution were significantly different (p < 0.05).
After three hours of soaking, the taro flour starch treated with the corn solution was seen to be significantly more digestible (77.12%) than the taro starch resulting from the two other treatments, respectively 60.86% after tamarind soaking and 60.45% after dipping in water.
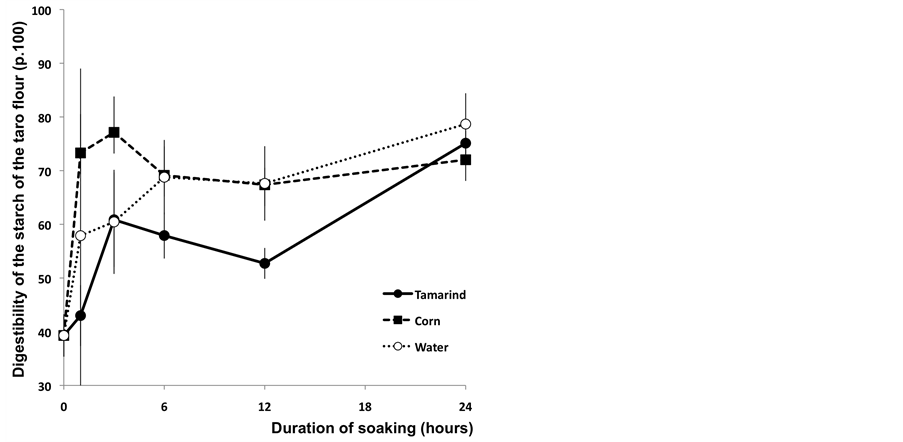
Figure 2. In vitro digestibility of starch (p.100) of Taro flour after a 30 minutes cooking at 95˚C without enzymatic digestion according to the duration of soaking.
After six hours of soaking, the taro samples dipped into the corn solution and those dipped in water presented almost the same level of digestibility.
On the other hand, the digestibility of taro dipped in the tamarind infusion was relatively low compared with the two other treatments. This difference could be linked with the slightly higher fibre content reported by Soudy et al. [6] in the sample of taro treated with the infusion of tamarind (3.24% on the dry matter (DM) basis) compared with the water treatment (2.63% DM) and the corn solution (2.75% DM). The digestibility of taro starch soaked in tamarind infusion and samples resulting from the other two treatments (water and corn) was significantly different at the threshold of 5%. The same observation has been made after 12 hours of soaking. The taro starch resulting from water treatment and the corn solution was significantly more digestible than samples treated with the tamarind infusion.
At the end of the soaking process (24 hours), we observed that the samples soaked with the tamarind infusion presented a statistically similar digestibility compared with the two other treatments (Figure 2). These results could be explained by the decline of the pH value of the solution of tamarind (pH 2) that would have facilitated the digestibility of taro starch. This test allowed the assessment of the influence of the soaking process on the in vitro digestibility of different taro flour during three hours of incubation under the action of bacterial enzyme. In general, it was noticed that digestibility was greatly improved when the samples were dipped into the tamarind infusion or into the corn infusion (respectively 93.77% and 90.80% after 3 hours of soaking time).
On the contrary, the digestibility level remains low for samples dipped in water (77.52%) and the digestibility level is significantly higher at the end of three hours of soaking in the case of corn and tamarind treatments as opposed to the samples treated with water (P < 0.05). After 24 hours of soaking time, the same results were obtained (Figure 3). Based on these results, we suggested that the tamarind infusion and the corn solution have increased the sensitivity of the starch to the bacterial enzyme. We could suspect the process of fermentation which would have prematurely begun in the soaking solutions, or the endogenous enzymes of these solutions to have initiated the digestion of the taro chip starch during the soaking process. This initiation of digestion allows increasing in vitro digestibility of starch [17,18].
Steinkraus [19] reported that most fermentation processes are able to improve the digestibility of food products and at the same time destroy the toxic factors.
From the beginning of the soaking process up to a 6-hour soaking time, the digestibility of taro treated with the tamarind infusion is significantly higher (95.23%) than with the other two treatments (corn solution: 93.16% and water : 88.49%) (Figure 4).
Thus, the tamarind infusion (pH = 2) could have led to the elimination of antinutritional factors existing in these tubers such as the inhibitors of enzymes or phytates during the soaking process. This would partially explain the improvement of the enzymatic digestibility of taro starch with this soaking solution. Madhuri and Pratima [20] showed that the germination of cereal and the fermentation of sweet potatoes during the soaking process in water significantly increased the digestibility of carbohydrates. Moreover, the soaking of millet in water at 30˚C
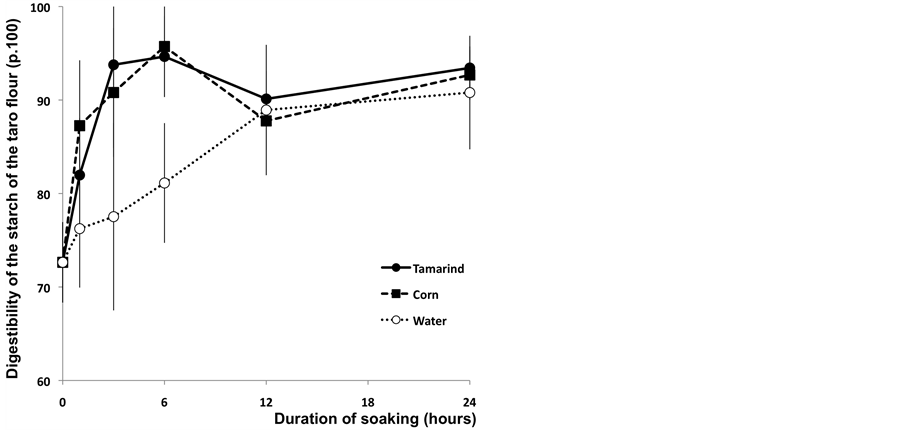
Figure 3. In vitro digestibility of starch (p.100) of Taro flour after a 30 minutes cooking at 95˚C after 3 hours of enzymatic incubation according to the duration of soaking.
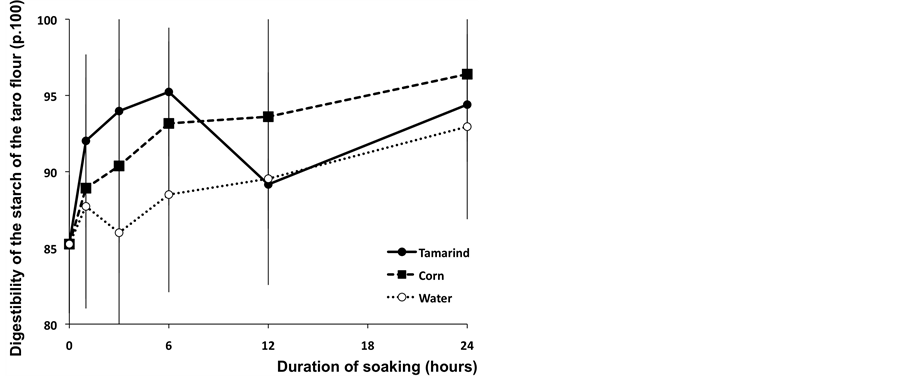
Figure 4. In vitro digestibility of starch (p.100) of Taro flour after a 30 minutes cooking at 95˚C after 6 hours of enzymatic incubation according to the duration of soaking.
for 48 hours significantly reduced phytates, inhibitors of amylases and phenols and improved the digestibility of this cereal.
From the start of soaking, the samples of taro treated with the tamarind infusion as well as the corn solution were significantly more digestible than the samples soaked in water (P < 0.05). From six hours of soaking, all treatments showed a statistically similar and high level of digestibility (more than 90) (Figure 5).
Although taro starch is known for its digestive qualities [3], the process of soaking used by the farmers of the Mayo-Kebbi area improves the digestibility of taro starch especially after it has been soaked in a tamarind infusion or a corn solution.
The in vitro digestibility of starch of different taro flours after 12 hours of enzymatic incubation quickly reached its maximum level (Figure 6). After one hour of soaking, the taro chips treated with the corn infusion were significantly more digestible than the other two treatments (P < 0.05). Nevertheless, for all treatments, the in vitro digestibility was more than 90% from the first hour of the soaking process (Figure 6). The highest level of digestion of the taro starch has been measured with the corn solution (97.46%) after 24 hours of soaking (Figure 6). The differences observed on the digestibility of taro flours could be linked to the physical or chemical properties of the starch.
4. Conclusions
All the processes of soaking used in Mayo-Kebbi area (Chad) significantly improved the in vitro digestibility of taro flour. Simply cooking taro (30 minutes at 95˚C
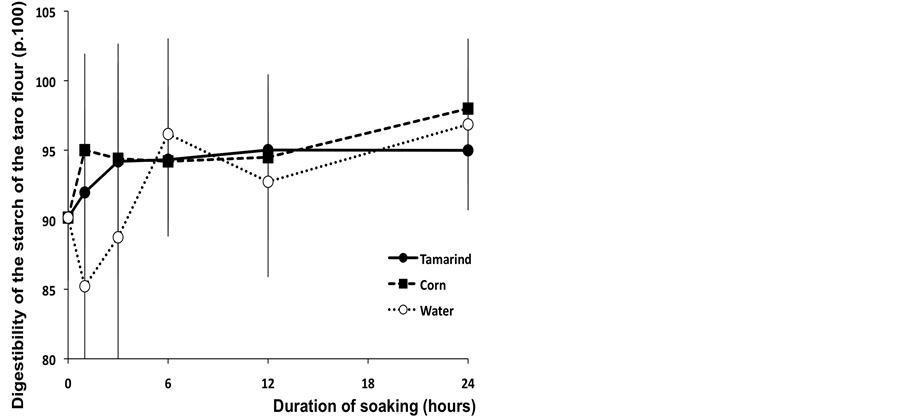
Figure 5. In vitro digestibility of starch (p.100) of Taro flour after a 30 minutes cooking at 95˚C after 9 hours of enzymatic incubation according to the duration of soaking.
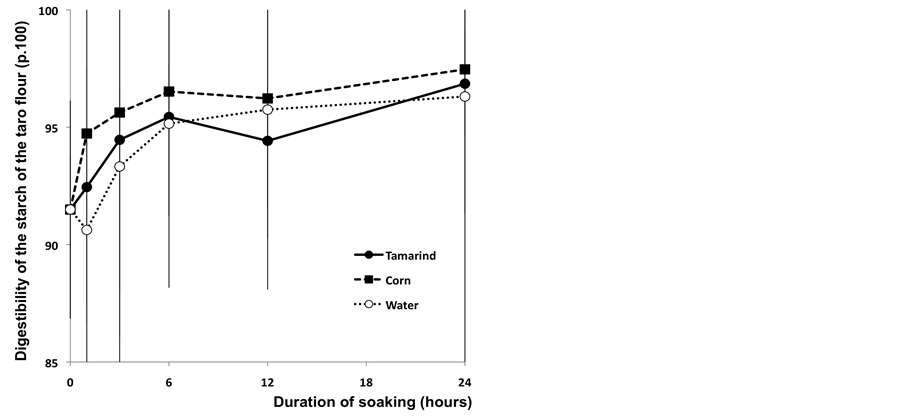
Figure 6. In vitro digestibility of taro flour (p.100) after 12 hours of enzymatic incubation.
without enzymatic action) after soaking improves the digestibility of taro starch from 39% (without any soaking) to 72% (after 24 hours of soaking time). This maximal digestibility was reached after just one single hour of soaking with the corn solution. A similar level of digestibility was only obtained after 24 hours of soaking with the other two treatments of soaking (tamarind and water).
Whatever the enzymatic digestion time fixed, the taro samples treated with tamarind infusion or corn solution obtained a better in vitro digestibility than the taro treated in the water. The 6-hour or more soaking process allowed a highly digestible flour to be obtained (around 95% of digestibility).
After a 3-hour soaking time and 3 hours of enzymatic incubation, the digestibility of taro flour reached 93% and 90% respectively with the tamarind infusion and the corn solution. Therefore soaking in water should be of a much longer duration (6 hours) and requires a longer enzymatic incubation (9 hours) to reach a digestibility of 90%.
It requires only 3 hours of incubation time with the α-amylase action to obtain an almost complete digestion of starch.
In conclusion, these traditional practices are particularly beneficial for the humans or animals with an amylasic incapacity, such as infants being weaned from maternal milk or people with lower pancreatic capacity.
Acknowledgements
The authors are grateful to Chad French Ambassy for the financing of this project.
REFERENCES
- Jane, L. Shen, J. Chen, S. Lim, T. Kasemsuwan and W. K. Nip, “Physical and Chemical Studies of Taro Starches and Flours,” Cereal Chemistry, Vol. 69, No. 5, 1992, pp. 528-535.
- I. C. Onwueme, “Tropical Root and Tuber Crops: Production, Perspectives and Future Prospects,” FAO, Rome, 1994, 228p.
- FAO, “Racines, Tubercules, Plantains et Bananes dans la Nutrition humaine,” Alimentation et Nutrition, FAO, Rome, 1991, 200p.
- A. Bell, O. Mueck and B. Schuler, “Les richesses du sol: les Plantes à Racines et Tubercules en Afrique: Une Contribution au Développement des Technologies de Récolte et d’après—Récolte,” Ed. Deutsche Stiftung fuer Internationale Entwicklung, Feldafing, 2000, 237p.
- I. D. Soudy, D. Grancher, P. Delatour and B. Facho, “Mode de Culture et Techniques Traditionnelles de Transformation et de Conservation Post Récolte du Taro (Colocasia esculenta L. SCHOTT) au Tchad,” Les Annales de l’Université de N’Djaména, Série C, Vol. 3, 2008, pp. 71-85.
- I. D. Soudy, P. Delatour and D. Grancher, “Effects of Traditional Soaking on the Nutritional Profile of Taro Flour (Colocasia esculenta L. Schott) Produced in Chad,” Revue de Médecine Vétérinaire, Vol. 161, No. 1, 2010, pp. 37-42.
- D. Martinez Bou, “Tamarind Characteristics,” 2011. http://www.botanical-online.com/english/tamarind.htm
- J. Adrian, J. Potus and R. Frangne, “La Science Alimentaire de A à Z,” 2nd Editipn, Ed Lavoisier: Technique et Documentation, Paris, 1995, 477p.
- INRA, “Alimentation des Bovins, Ovins et Caprins,” Ed. INRA, Paris, 1978, pp. 23-45.
- Y. N. Njintang, “Studies on the Production of Taro (Colocasia esculenta L. Schott) Flour for Use in the Preparation of Achu,” Ph.D. Thesis, University of Ngaoundéré— Cameroon, Department of Food Sciences and Nutrition, National School of Agro-Industrial Sciences (ENSAI), 2003.
- J. C. Favier, “Etude de la Digestibilité ‘in Vitro’ de l’amidon de Diverses Plantes Alimentaires du Sud-Cameroun, Influence des Transformations Technologiques sur l’Amidon de Manioc,” Centre ORSTOM de Yaoundé, Cameroon, 1969, pp. 9-14.
- A. E. Elkhalifa, B. Schiffer and R. Bernhard, “Effect of Fermentation on the Starch Digestibility, Resistant Starch and Some Physicochemical Properties of Sorghum Flour,” Food/Nahrung, Vol. 48, No. 2, 2004, pp. 91-94. http://dx.doi.org/10.1002/food.200300322
- E. S. Admassu and S. R. Kumar, “Influence of Natural and Controlled Fermentations on α-Galactosides, Antinutrients and Protein Digestibility of Beans (Phaseolus vulgaris L.),” International Journal of Food Science and Technology, Vol. 43, No. 4, 2008, pp. 658-665. http://dx.doi.org/10.1111/j.1365-2621.2006.01506.x
- A. A. M. Nour, M. A. I. Ahmad, E. E. Babiker and A. E. A. Yagoub, “Investigations on Winter Season Sudanese Sorghum Cultivars: Effect of Sprouting on the Nutritional Value,” International Journal of Food Science & Technology, Vol. 45, No. 5, 2010, pp. 884-890.
- Y. Luo, W. Xie and Q. Cui, “Effects of Phytase, Cellulase and Dehulling Treatments on Zinc in Vitro Solubility in Faba Bean (Vicia faba L.) Flour and Seed Fractions,” International Journal of Food Science & Technology, Vol. 45, No. 2, 2010, pp. 358-364. http://dx.doi.org/10.1111/j.1365-2621.2009.02152.x
- D. Zhang, W. W. Collins and M. Andrade, “Estimation of Genetic Variance of Starch Digestibility in Sweetpotato,” Horticultural Science, Vol. 30, No. 2, 1995, pp. 348-349.
- L. Huijun, R. Lawrence and C. Harold, “Physical Properties and Enzymatic Digestibility of Acetylated ae, wx, and Normal Maize Starch,” Carbohydrate Polymers, Vol. 34, No. 4, 1997, pp. 283-289.
- L. N. Panlasigui, L. U. Thompson, B. O. Juliano, C. M. Perez, S. H. Yiu and G. R. Greenberg, “Rice Varieties with Similar Amylose Content Differ in Starch Digestibility and Glycemic Response in Humans,” The American Journal of Clinical Nutrition, Vol. 54, No. 5, 1991, pp. 871-877.
- K. H. Steinkraus, “Handbook of Indigenous Fermented Foods,” 2nd Edition, Marcel Dekker Inc., New York, 1996, 40p.
- K. Madhuri and S. Pratima, “Effect of Processing on in Vitro Carbohydrates Digestibility of Cereals and Legums,” Journal of Food Science and Technology-Mysore, Vol. 33, No. 6, 1996, pp. 493-497.
NOTES
*Corresponding author.

