Food and Nutrition Sciences
Vol. 2 No. 1 (2011) , Article ID: 3640 , 9 pages DOI:10.4236/fns.2011.21003
Quality Characteristics of Dark Muscle from Yellowfin Tuna Thunnus albacares to Its Potential Application in the Food Industry
![]()
1Agro-food Department–IPOA, Universidad Miguel Hernández, Ctra, Orihuela, Alicante, Spain; 2Biology Department, Science Faculty, Abdelmalek Essaâdi University, Tetouan, Morocco.
Email: j.fernandez@umh.es
Received September 30th, 2010; revised November 16th, 2010; accepted November 24th.
Keywords: Dark Muscle, Tuna Fish, Haemopigments, Myoglobin, Heme Iron, Physicochemical Properties
ABSTRACT
Dark muscle from yellowfin tuna is an important edible fish by-product. However, it has a low commercial value, and it is underutilized. The present study was conducted to establish the characteristic of this by-product. Myoglobin concentration in tuna dark muscle is high (9650.12 mg/kg). Total iron in tuna muscle was 32.11 mg/kg, higher than other animal foods like veal or pork, and heme iron concentration was 23.56 mg/kg (73.38% of the total iron), indicating a high bioavailability of heme iron in dark muscle from yellowfin tuna, which is a nutritional advantage. As for the technological properties, yellowfin tuna dark muscle had a water holding capacity of 8.37 g water/g and oil holding capacity of 8.11 g oil/g. This indicates that tuna dark muscle has possible applications to elaborate products, such as emulsionated foods or cooked products, so its industrialization is possible.
1. Introduction
Yellowfin tuna Thunnus albacares is an intensely exploited fish. Large quantities of yellowfin tuna are commercially used in canned and dry-salted products, like cured tuna loin (a typical product from Spanish Southeast, called “mojama”) [1] and as sashimi, a delicate-tasting raw fish product popular in Korea and Japan. The use of yellowfin tuna as sashimi is increasing in several other countries, with an annual worldwide production of 3,400,000 MT [2]. Yellowfin tuna industrialization is increasing fishery by-products. The amount of by-products produced during fish processing can be as high as 75% of the total weight of fish [3,4]. Some of the by-products are used to produce fish sauces and food products such as dry-salted roe, also could be used in animal feed, but much of it is discarded and is a source of environmental contamination. The amount of hazardous waste produced from fish processing has tended to increase annually [3].
Optimal utilisation of fishery by-products is becoming increasingly important [5]. Large quantities of proteinrich fish processing by-products are discarded as wastewithout any attempt to recover the essential nutrients. This would cause serious disposal and pollution problems, especially in developing countries. These wastes, which represent an environmental problem to the fishing industry, constitute an important source of proteins. Traditionally, this material is transformed into fish flour to animal feed. Nevertheless, novel processing methods are required to convert fish by-products into more profitable and marketable products [6,7]. In other cases the fish wastes have been use to produce collagen, chondroitin sulphate and gelatine which have indirect environmental benefits [5].
Fish muscle consists of light and dark muscle, the dark muscle is a band of dark tissue that lies beneath the skin throughout the body. In tuna, dark muscle is also located near the backbone. Although generally the relationship between light and dark muscle varies with the fish activeity, fatty fish contains higher proportion of dark muscle, reaching values up to 48% [8], like in yellowfin tuna. This is because the fatty fish are migratory species, so they need more fat, glycogen and myoglobin for their long journeys. There are many differences in the chemical composition of the two types of fish muscle, but the most important are the high fat and heamopigments content in dark muscle. Consequently, the higher lipid contents, less stable proteins, greater concentrations of heme proteins, lower ultimate pH values, higher concentrations of sarcoplasmic proteins of dark muscle have been suggested to contribute to the difficulties in its industrialization and making high-quality products from raw material containing high contents of dark muscle. Because of this it is considered as a by-product of the tuna industry [4-9].
Yellowfin tuna dark muscle (“sangacho” in Spanish) is not commonly used by the canned and dry-salted tuna fish industry [10], being a by-product for these industries. However, the characteristics of yellowfin tuna dark muscle (DM) that make it not acceptable for these industries (strong dark color and highly susceptible to lipid oxidation speeding up its deterioration) [11] could give it a high nutritive value, even higher than that of light muscle. Fish dark muscle contains many n-3 polyunsaturated fatty acids (n-3 PUFA) such as icosapentaenoic acid (EPA: C20:5n-3) and docosahexaenoic acid (DHA:C22:6n-3). These fatty acids have various bioactive functions, such as anti-cancer activity, recovery from heart failure, attenuation of cerebrovascular disease, and anti-arteriosclerosis action [12]. Dark muscle is also a high source of iron which is an essential mineral. When it is as heme iron is several times more absorbable than non heme iron present in other foods [13].
Several methods have been successfully applied to preserve fish and to extend the shelf life of fish products but none has been tested to use in DM [14]. The aim of this paper was to characterize (chemical, physicochemical and microbiologically) the yellowfin tuna dark muscle to determine their possible industrialization and thus exploit by-products from the tuna industry.
2. Materials and Methods
2.1. Raw Material
The raw material (frozen tuna dark muscle) was obtained from a local fish manufacturer. It had been rapidly frozen at –40℃ and then stored at –18℃ for 1 month. It was transported to the pilot plant facilities of the IPOA Research Group, at the Universidad Miguel Hernández (Orihuela, Alicante, Spain) in refrigerated conditions (4℃). Tuna dark muscle was thawed before each analysis in chill storage (4℃) over night.
2.2. Proximate Composition
For all chemical analysis, samples were grounded in an electric mini food chopper Moulinex 390 (Moulinex, Paris, France). Moisture, ash, protein and fat content were determined by AOAC methods 24.003, 24.009, 24.027 and 24.005, respectively [15].
2.3. Technological Properties
2.3.1. Physicochemical Analysis
The pH was measured using a pH-meter (Mod. pH/Ion 510, Eutech Instruments Pte Ltd., Singapore) by AOAC method 14.022 [15]. Water activity (Aw) was determined in a Novasina Thermoconstanter Sprint TH-500 (Pfäffikon, Switzerland) at 24.9℃. The CIELAB color space was selected for color determination. Color measures were made using a spectrophotometer Minolta CM-2600d (Minolta Camera Co. Osaka, Japan), with D65 as illuminant and an observer angle of 10˚. Low reflectance glass (Minolta CR-A51/1829-752) was placed between the samples and the equipment. The CIELAB coordinates studied were: Lightness (L*), co-ordinate red/green (a*) and co-ordinate yellow/blue (b*) [16].
2.3.2. Functional Properties
Water-holding capacity (WHC) and oil-holding capacity (OHC) were determined according to El Khalifa et al. [17]. For the water holding capacity (WHC), 1 g of sample was placed in previously weighted centrifuge tubes, and 14 mL of water was added. For the oil holding capacity (OHC), 14 mL of sunflower oil was added. Both samples were stirred in a tube stirrer and kept at rest for 30 min at room temperature before being centrifuged at 5,000 × g for 25 min. The excess of water or oil was removed by tube inversion over tissue paper. The difference between the sample’s weight before and after water or oil absorption was taken as the amount of water or oil absorbed. WHC was expressed as g of water held per g of sample, and OHC was expressed as g of oil held per g of sample. Water adsorption capacity (WAdC) and water absorption capacity (WAbC) were determined according to Vázquez-Ovando et al. [18]. Water absorption capacity (WAbC) is indicative of a structure’s aptitude to spontaneously absorb water when placed in contact with a constantly moist surface or when immersed in water, while the water adsorption capacity (WAdC) is the ability of a structure to spontaneously adsorb water when exposed to an atmosphere of constant relative humidity. It is initially a surface phenomenon but, at higher hydration levels, absorption can occur inside the structure, leading to swelling and eventual solubilization [18]. WAdC was expressed as g of water adsorbed per g of sample, and WAbC was expressed as g of water absorbed per g of sample. Emulsifying activity (EA) and emulsion stability (ES) were evaluated according to Chau et al., [19] with slight modifications. 100 mL of 2% (w/v) sample suspension in water was homogenized at 11000 rpm for 30 s at room temperature, using a homogenizer IKA T-25. 100 mL of sunflower oil was then added and homogenized for another 1 min. The emulsions were centrifuged in 10 mL graduated centrifuged tubes at 1200 g for 5 min, and the volume of the emulsion left was measured. Temperature was controlled during the emulsion formation. EA was calculated as volume of emulsified layer/volume of whole layer in centrifuge tube × 100. To determine the thermal emulsion stability (ES), emulsions prepared by the above procedures were heated at 80℃ for 30 min, cooled to room temperature, and centrifuged at 1200 g for 5 min. ES was calculated as volume of remaining emulsified layer/original emulsion volume × 100.
2.4. Oxidation Parameters
2.4.1. Lipid Oxidation
Lipid oxidation was assessed by the 2-thiobarbituric acid (TBA) method following the recommendations of Buege and Aust [20]. TBARS values were calculated from a standard curve of malonaldehyde (MA) and expressed as mg MA/kg sample.
2.4.2. Haemopigments Oxidation
2.4.2.1. Myoglobin(Mb) and Metmyoglobin (MMb)
Myoglobin and metmyoglobin wer determinate according to Krywicki [21]. Five g of minced meat was used to determine MMb concentration in each sample. Myoglobin was extracted with cold 0.04 M phosphate buffer, pH 6.8, with a sample to buffer ratio of 1:10. Samples were homogenized for 15 s with an Ultraturrax homogenizer T20 Standard (IKA, Staufen, Alemania) at speed 13500 rpm. The homogenates were then centrifuged for 30 min at 5000 rpm. The absorbance of the filtered supernatant was read at 525, 572 and 730 nm. Mb was determining using the formula of Krywicki [21]:
Mb (mg/kg) = (A525 – A730) × 2,303 × 5 (dilution factor)
× 1000;
and percentage MMb was determining using the formula of Krywicki [21]:
MMb (%) = 1.395 – ((A572 – A730)/(A525 – A730)) × 100
Samples were kept on ice at all points during the assay.
2.4.2.2. Total Iron
Total iron concentration was determined in wet-ashed samples using the ferrozine assay [22].
Heme iron
Heme iron was determined using the method of Hornsey [23]. Total pigments, as acid hematin, were calculated using the formula:
Total pigment (ppm) = A640 × 680;
and heme iron was calculated as follows [24]:
Heme iron (ppm) = total pigment (ppm) × 8.82/100
2.5. Microbial Analysis
Serial dilutions of samples were prepared in sterile peptone water for most microbial determinations and in the De Man, Rogosa, Sharpe (MRS) broth for lactic acid bacteria counts. Total viable counts (TVC) were determined by plating the diluted samples on TVC 3 M PetrifilmÔ plates followed by incubation at 35℃ for 48 h; lactic acid bacteria (MRS broth) and anaerobic bacteria on TVC 3M PetrifilmÔ plates incubated at 37℃ for 48 h in anaerobic conditions; psychotrophic bacteria on TVC 3 M PetrifilmÔ plates incubated at 7℃ for 10 days; enterobacteria on 3 M PetrifilmÔ plates for enterobacteria incubated at 37℃ for 24 h; Staphylococcus aureus on 3 M PetrifilmÔ Staph express plates incubated at 37℃ for 24 h; Clostridium perfringens on SPS agar at 37℃ for 24 h in anaerobic conditions; and moulds and yeasts on Rose Bengal Agar with Chloramphenicol incubated at 28℃ for 5 days.
2.6. Statistic
All tests were carried out in triplicate (4 × 3 = 12 samples). Results are expressed as means ± standard deviations (SD). All statistics were performed using a SPSS statistics packaged software (SPSS 16.0 for Windows, SPSS Inc., Chicago, Illinois).
3. Results and Discussion
3.1. Proximate Composition
The results for proximate composition of yellowfin tuna dark muscle are shown in Table 1. Tuna dark muscle showed similar fat and protein content than values reported by Saito et al. [25] (4.3 g fat/100 g and 26.4 g protein/100 g), but lower moisture content (71.9 g water/100 g). Proximate composition of the yellowfin tuna dark muscle was similar to those of other tuna fish, like bonito fish Sarda sarda [26] and wild Pacific bluefin tuna Thunnus orientalis [12]. However, it is difficult to compare composition data from different fish muscles be cause there are a lot of factors that can modify it. Furthermore, those of identical fish species are also affected by season, sea area, age and physiological stage [9,11].
3.2. Technological Properties
3.2.1. Physicochemical Properties
Table 2 shows the physicochemical properties of DM.

Table 1. Proximate composition of yellowfin tuna dark muscle (DM) (g/100 g).

Table 2. Physicochemical properties of tuna dark muscle (DM) (mean ± sd).
The water activity of this muscle was 0.990. As regards the pH, the value was 5.36. In fish, the decrease in pH after slaughter is lower than in meat, contributing to the high perishable condition of this food. In many fish species, the postmortem pH is higher than 6 because the low carbohydrate content in muscle leads to low production of lactic acid postmortem [27]. However, in this experiment, the pH was not near to neutrality, it could be due to microbial growth, as lactic acid bacteria (Table 5). Microorganisms originally located on the fish skin, gills and intestinal tract can contaminate the tissues during filleting [4]. The high water activity of DM may also reflect greater water availability for microbial growth. A high pH and water activity together with the storage under refrigerated conditions, which are closed to the temperatures of the natural habitat of tuna [9], provides favourable conditions for the growth of present microbiota and determines an extremely short self-life.
Colour is one of the most important quality parameters in food. In the CIE L*, a*, b* system, L* denotes lightness on a scale from 0 to 100 from black to white; a* denotes (+) red or (-) green; and b* denotes (+) yellow or (-) blue [16]. The values of L*, a* and b* coordinates for tuna dark muscle were lower than those reported by Sanchez-Zapata and Pérez-Álvarez [8] for tuna muscle although in this case no separation between light and dark muscle was made before color measurement.
Onyango et al. [28] studied the colorimetric coordinates of loin meat from different animals (cow, zebra, oryx and kongoni) and determined the myoglobin content of these meats. The results of this study indicated that there was an inverse correlation between lightness (L*) and myoglobin content (y = –2.24x + 51.2; where y = lightness (L*) and x = myoglobin content (mg/g)). Applying this relationship to the lightness of tuna dark muscle, it would be expected a myoglobin content of 10.78 mg/g. This myoglobin concentration would be approximately three times the concentration of myoglobin determined by Onyango et al. [28] in cow, which would agree with the work of Kanoh et al. [29], which determined that the concentration of myoglobin in the tuna muscle was 3-14 times higher than regular content in mammal muscle. The myoglobin content determinates in this experiment (9.65 mg/g) was near to the myoblobin content obtained by Onyango et al. [28] formula.
3.2.2. Functional Properties
The water-holding capacity (WHC) is one of the most important quality parameters of meat and fish products, because of reduced weight loss during cutting and storage, and improved ability of meat to retain water during processing. The WHC of DM (Table 3) was high (8.37 g water/g sample), compared to the WHC of horse mackerel fillet Trachurus mediterraneus (0.99 g water/g sample) [30], and other fish by-products like protein fractions generated from hydrolysed cod Gadus morhua (0.70 g water/g sample) [5] and enzymatic hydrolysates from Bluewing searobin Prionotus punctatus (2.37) [31]. This indicates that DM has possible applications to elaborate products such as emulsifyed foods or cooked products because binding capacity of emulsion results from denaturation and decreased solubility proteins. Once fat is coated, the emulsion is stabilized. Heating the emulsion coagulates the protein and stabilizes the emulsion so that the protein holds the fat in suspension. Emulsion stability is influenced by the WHC of the product, and the mechanical and heat treatment. Formation of a protein matrix is one of the main factors contributing to creating of a successful emulsion [32].
DM had a higher OHC that the cod fish by-products [5] and the bluewing searobin by-products [31]. The high OHC in DM, compared to the values of OHC found in the literature [5,31], is attributed mostly to physical entrapment of the oil, and thus the higher bulk density of the protein [33]. Fat-binding capacity of DM correlates with surface hydrophobicity and by the fat content [5,33]. In cod, Šlžiytė et al. [5] reported that higher amounts of lipids causes a higher fat absorption ability and a positive relationship (r2 = 0.90) between fat absorption and the amount of phospholipids was observed in this fish samples. OHC is important to flavour retention and product yield, especially for cooked products, which normally lose fat during cooking [33]. Due to its high OHC, DM is a potential fish ingredient to elaborate cooked products, but not for fried products since it would provide a greasy sensation.
The water absorption and adsorption capacities of DM are presented in Table 3. The DM had a water absorption capacity of 0.33 g water/g product. DM water adsorption capacity was 0.14 g water/g product. The water absorption capacity and water adsorption capacity from tuna dark muscle could be due to the amount and type of protein present in tuna dark muscle. Each type of food protein has a unique molecular structure that determines its functional properties, in addition to a range of environmental conditions over which it exhibits such properties. The functional properties of proteins in a food system depend in part on the water-protein interaction, and the final outcome greatly depends on how well the protein binds and holds water in a food system [33].
Emulsifying capacity is a molecule’s ability to act as an agent that facilitates solubilisation or dispersion of two immiscible liquids, and emulsion stability is the ability to maintain an emulsion and its resistance to rupture under heating [18]. Table 3 shows the emulsifying activity (EA) and emulsion stability (ES) of DM. Emulsifying activity of DM was 16.84 mL/100 mL and its emulsifying stability was 100 mL/100 mL. The amount of proteins and amino acids in fish products seems to be important for emulsification capacity. It was observed that the EA values increased with increasing protein content in cod byproducts; however, a relationship between amount of preins and stability of emulsions was not found in this proct [5]. EA and ES of DM are higher than cod by-products [5].
The proteins of fish muscle tissue are usually grouped into three categories: sarcoplasmic proteins (25-30%), myofibrilar proteins (70-80%) and stromal proteins from connective tissue (3%) [9]. The current theory of gel formation from fish muscle proteins holds that a high concentration of salt (NaCl) is required to solubilize the myofribillar proteins, particularly miogobin and actomyin, which can then gel on heating as the proteins denature, interact, and aggregate. The dark muscle has been repted to contain higher concentrations of sarcoplasmic proins than the light muscle [4]. The high protein content of DM (26.92 g/100 g w. b.) would explain its high EA and ES, since most proteins are strong emulsifying agents [5]. It is noted that DM may be used to elaborate emulsifyed products with heat treatment because DM has an elevated stability to the heat treatment. The mechanism of binding between chunks of fish is a heat-initiated reactions. The mechanism is similar to the mechanism acting in heatnitiated emulsion-stabilization in emulsions products. In the heat-nitiated reaction, a concentrated emulsion forms between fish chunks and acts to bind adjacent pieces. A protein matrix develops and upon heating acts as a stable binder between fish pieces [32].
3.3. Pigment and Lipid Oxidation
Table 4 shows the mean and standard deviation of the determinations of lipid and pigment oxidative parameters (total Fe, Fe heme, myoglobin, % metmyoglobin and content in 2-thiobarbituric acid (TBA) in tuna dark muscle Thunnus albacares. TBA value was 1.23 mg malonaldehide/kg sample; total iron content was 32.11 mg Fe /kg sample; heme iron content was 23.56 mg Fe/kg sample; myoglobin content was 9650.12 mg/kg sample and metmyoglobin was 91.44% of myoglobin content.
Pigment and lipid oxidation are major deteriorative reactions in meat and meat products during storage. They are responsible for a significant loss in quality charactertics such as colour, flavour, texture and nutritive value [34]. The relationship between oxidation of lipids and hemigments has been well documented in fresh meat during storage [35]. However, the cause-effect relationip between the two processes is not completely clarified and it is not known when or whether lipid oxidation causes pigment oxidation or vice versa.
The tuna dark muscle is rich in iron, according to the values obtained in this work (Table 4), because the values are higher than those determined by Lombadi-Boccia et al. [13] in beef (20.9 mg/kg), in chicken (5.9 mg/kg) or in pork (4.2 mg/kg), and even to those obtained by Fernández-Lopez et al. [36], in ostrich liver (25.10 mg/ kg). The iron content of tuna dark muscle is also higher than in fish other species such as mackerel (30 mg/kg), herring (11 mg/kg), cod (9 mg/kg) or trout (4 mg/kg) [37]. This would be due to the increased concentration of myoglobin [13] in tuna, as the work of Kanoh et al. [29], which stated that the concentration of myoglobin in fresh tuna is 3-14 times higher than that normally found in meat of slaughter animals.
Foods from animal origin are, in diet, the main source of highly bioavailable iron [13]. The bioavai-lability of ir-

Table 3. Functional properties of tuna dark muscle (DM) (mean ± sd).

Table 4. TBA, total Fe, heme Fe, % metmyoglobin and myoglobin content in tuna dark muscle (DM) (mean ± sd).
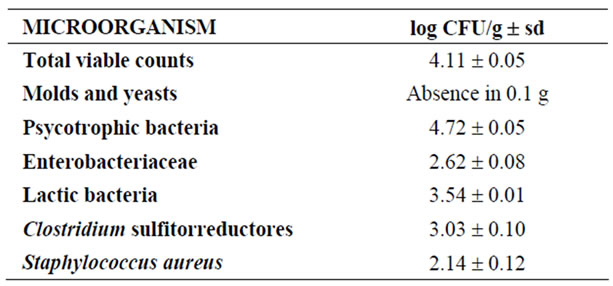
Table 5. Microbial counts in yellowfin tuna dark muscle (DM) (log colony forming units (CFU)/g ± sd).
on is considered to be the proportion of iron from food, or ingested as a supplement, which is absorbed and utilized by the human body. Iron deficiency is currently the most important nutritional deficiency in the world and it is particularly severe in developing countries [13]. This stems partly from a poor diet or a diet in which the bioavailability of iron is low. The total iron in the diet can be divided into heme and non-heme fraction, meat (including fish) is the sole source of heme iron. Heme iron has much greater bioavailability than non-heme. It is very important to have accurate knowledge of total iron levels and its chemical forms (heme, non-heme) in food, since there is a big difference in the availability of the heme and non-heme iron. The estimate amount of bioavailable iron in the diet depends largely on the accuracy of the determinations in heme iron content. Furthermore, an accurate knowledge of the concentration of heme iron in food, is extremely important because it allows to determine the storage stability of a food product, since the non-heme iron (released from the pigment) is a major lipid oxidation catalysts [13]. Therefore, the study of the ratios of heme and non-heme iron in the tuna muscle is of primary importance that the form of non-heme iron has greater ability to promote oxidation reactions than heme iron [38]. Heme iron concentration in tuna muscle was 23.56 mg/kg of sample and total iron was 32.11 mg/kg of sample, therefore, the amount of non-heme iron present in tuna would be 8.55 mg/kg of sample, which represents a 26.62% total iron, as against 73.38% constituting the he-me iron, indicating a high bioavailability of heme iron in raw tuna. However, the treatments that tuna suffers during processing (heating fundamentally) can affect the bioavailability of iron [13,39].
The concentration of myoglobin determined in DM tuna samples (Table 4) is high when compared with that present in the dark muscle of other fish species such as mackerel (0.39 mg/g) or black marlin (0.51 mg/g) [40] and even certain animals for slaughter as veal or pork, and is close to the estimated value (10.78 mg/g) by the Onyango et al. [28] formula. This high myoglobin concentration is also responsible for the high content of heme iron in the samples, as has been discussed previously. In addition, the metmyoglobin content of the sample (represented as % of total myoglobin) was high (91.44%). Myoglobin of tuna muscle is highly susceptible to lipid oxidation [41], and this reaction is also favored by the process of freezing and thawing to which our samples were subjected, making the muscle to oxidize rapidly, thus favouring the formation of metmyoglobin.
The lipid oxidation is a major cause of deterioration in the quality of animal products, both meat and fish. In this study, the TBA value of DM was 1.23 mg MA/Kg sample. This would indicate that there have been oxidation reactions in the lipid fraction of tuna, which would cause increased levels of oxidation as well as by exposure to oxygen and muscle lipase activity. This could also be due to greater concentration of fat in the tuna muscle. These oxidation reactions would be further promoted by the presence of iron in muscle and its prooxidant role [41].
3.4. Microbiological Quality
DM microbial counts are showed in Table 5. Total viable counts were 4.11 log CFU/g; molds and yeast absence in 0.1 g; psycotrophic bacteria counts were 4.72 log CFU/g; enterobacteriaceae counts were 2.62 log CFU/g; lactic bacteria counts were 3.54 log CFU/g; Clostridium sulfite reductor counts were 3.03 log CFU/g and Staphylococcus aureus counts were 2.14 log CFU/g.
The postmortem degradation of marine species is very fast, first caused by endogenous enzymes and, secondly, by endogenous microorganisms or contaminants from the raw materials or incorporated during processing [42]. Total viable counts (TVC; log CFU/g) in tuna dark muscle were similar to those of fresh yellowfin tuna [43]. Guizani et al. [44], reported that aerobic mesophilic bacteria and aerobic psychrotrophic bacteria dominate the microbiota on yellowfin tuna, as has been observed in DM. Because of the high water and protein content and neutral pH, dark muscle is an ideal medium for psycrotrophic bacterial growth. These seafood bacteria have an optimum growth of 20-25℃ but have the ability to grow at refrigeration temperature, although at lower rate [4]. However, reported counts did not exceed upper Spanish legal limits for fresh fish [45] which are 106 CFU/g of TVC, and are far below the limit of 107 CFU/g that indicates the spoilage of a food International Commission on Microbiological Specifications for Foods (ICMSF).
Barros-Velázquez et al. [42] obtained lower counts in whole fish kept under refrigeration on board than the observed in DM. Probably this is because to obtain DM, tuna has undergone a process of gutting, freezing and thawing. Enterobacteria, E. coli, lactic acid bacteria (LAB) and Clostridium provide information on fish contamination and spoilage. Reported counts are moderate, as Enterobacteria counts are under the Spanish legal limit of 103 CFU/g of fish for Enterobacteria. No other microorganisms are regulated by Spanish laws in fresh raw fish. E. coli and St. aureus are over the legal limits for ready to eat fishery products (10 CFU/g and 102 CFU/g, respectively) but both groups would be eliminated by heat treatment and would pose no risk for consumers. Counts of E. coli and Clostridium sp. evidence fecal contamination, probably from gutting process and handling. LAB counts, together with Enterobacteria counts would indicate early stages of microbial spoilage. Molds and yeasts were not detected in DM. Overall microbial quality of this by-product is acceptable as it may not be directly consumed but used for food formulation and processing including heat treatment.
4. Conclusion
Yellowfin tuna dark muscle has a high nutritive value refereed to its total iron content and that it is mainly in the form of heme iron (73.45%) which has a high bioavailability. It sow high values for important technological properties as water holding capacity (8.37 g water/g), oil holding capacity (8.11 goil/g), emulsifying activity (16.84 mL/100 mL) and emulsifying stability (100 mL/100 mL), that make it appropriate to elaborate products requiring hydration and viscosity development, such as emulsionated foods or cooked products. Because its technological properties show that heat-induced gelation and changes in viscoelastic properties of the protein matrix are related to development of characteristic textures in fish product and to binding between pieces and emulsion stabilization. Myosin forms an irreversible gel, which is initiated by heat. The gel has a high WHC and strong elastic properties. After gel is formed no syneresis takes place, probably as a result of interaction stability. The mechanism of myosin gelation seems to involve formation of bonds that are not rupture by heat [32]. However, TBA value of dark muscle is high (1.23 mg MA/Kg) which means that there have been oxidation reactions in the lipid fraction of tuna.
These nutritive and technological characteristics of yellowfin tuna dark muscle indicate that is suitable as raw material for the food industry, despite having a high tendency to lipid oxidation. This could be avoided by preserving the product under conditions that prevent the lipids oxidation (such as modified atmosphere packaging) or by adding antioxidants during food processing.
5. Acknowledgements
We thank Albaladejo Hnos, S. A. for kindly providing the tuna dark muscle samples for use in the study.
REFERENCES
- E. Sánchez-Zapata, E. Fuentes-Zaragoza and J. A. PérezAlvarez, “Processes of Salted Fish,” In: J. A. PérezAlvarez, Ed., Industrialización de Productos de Origen Animal, Universidad Miguel Hernández, Elche, Spain, 2007, pp. 49-73.
- Lee and S. B. Kim, “Extraction Optimization and Properties of Collagen from Yellowfin Tuna (Thunnus albacares) Dorsal Skin,” Food Hydrocolloids, Vol. 22, No. 5, 2008, pp. 879-887.
- F. Shahidi, “Seafood Processing By-Products,” In: F. Shahidi, Ed., Seafoods Chemistry, Processing, Technology and Quality, Blackie Academic & Professional, Glasgow (UK), 1994, pp. 320-334.
- H. O. Hulting and S. D. Kelleher, “Surimi Procedding from Dark Muscle Fish,” In: J. W. Park, Ed., Surimi and surimi seafood. Marcel Dekker, Inc, New York (USA), 2000, pp. 59-77.
- R. Šlžiytė, E. Daukšas, E. Falch, I. Storrø and T. Rustad, “Characteristics of Protein Fractions Generated from Hydrolysed Cod (Gadus morhua) By-products,” Process Biochemistry, Vol. 40, No. 6, 2005, pp. 2021-2033. doi:10.1016/j.procbio.2004.07.016
- A. Bougatef, N. Nedjar-Arooume, L. Manni, R. Ravallec, A. Barkia, D. Guillochon and M. Nasri, “Purification and Identification of Novel Antioxidant Peptides from Enzymatic Hydrolysates of Sardinelle (Sardinella aurita) Byproducts Proteins,” Food Chemistry, Vol. 118, No. 3, 2010, pp. 559-565. doi:10.1016/j.foodchem.2009.05.021
- M. Chalamaiah, G. Narsing Rao and T. Jyothirmayi, “Protein Hydrolysates from Meriga (Cirrhinus mrigala) Egg and Evaluation of Their Functional Properties,” Food Chemistry, Vol. 120, No. 3, 2010, pp. 652-657. doi:10.1016/j.foodchem.2009.10.057
- E. Sánchez-Zapata and J. A. Pérez-Alvarez, “The Color in Different Fish Species,” Alimentación, Equipos y Tecnología, Vol. 219, 2007, pp.39-43.
- H. H. Huss, “Fresh Fish: Its Quality and Quality Changes,” Food and Agricultural Organization, Rome, 1998.
- E. Sánchez-Zapata, J. Fernández-López, E. Sendra, E. Sayas-Barberá, C. Navarro, M. Viuda-Martos and J. A. Pérez-Alvarez, “Application of Orange Fiber for the Control of Oxidation in Paté from Yellowfin (Thunnus albacares) Dark Muscle (sangacho),” Alimentaria, Vol. 400, 2009, p. 95.
- M. Nishioka, Y. Tanioka, E. Miyamoto, T. Enomoto and F. Watanabe, “TLC Analysis of a Corrinoid Compound from Dark Muscle of the Yellowfin Tuna (Thunnus albacares),” Journal of Liquid Chromatography & Related Technologies, Vol. 30, No. 13-16, 2007, pp. 2245-2252. doi:10.1080/10826070701451605
- Y. N. Nakamura, M. Ando, M. Seoka, K. Kawasaki and Y. Tsukamasa, “Changes of Proximate and Fatty Acid Compositions of the Dorsal and Ventral Ordinary Muscles of the Full-cycle Cultured Pacific Bluefin Tuna Thunnus Orientalis with the Growth,” Food Chemistry, Vol. 103, No. 1, 2007, pp. 234-241. doi:10.1016/j.foodchem.2006.07.064
- G. Lombardi-Boccia, B. Martínez-Domínguez and A. Aguzzi, “Total Heme and Non Heme Iron in Raw and Cooked Meats,” Journal of Food Science, Vol. 67, 2002, p. 5. doi:10.1111/j.1365-2621.2002.tb08715.x
- E. Sánchez-Zapata, J. A. Pérez-Alvarez, J. FernándezLópez, E. Sendra, E. Sayas-Barberá, C. Navarro and M. Viuda-Martos, “Characterization of Dark Muscle (‘Sangacho’) Yellowfin Tuna (Thunnus albacares) as Raw Material for the Production of ‘Pate’,” Procceding of Congress CESIA-CIBSA, Barcelona, Spain, 2008.
- AOAC, “Official Methods of Analysis of AOAC International,” 16th Edition, Association of Official Analytical Chemistry, Washington, 1997.
- J. A. Pérez-Alvarez and J. Fernández-López, “Aspectos Físicos, Químicos, Psicológicos, Fisiológicos y ultraestructurales Del Color De La Carne,” CD-Rom, Universidad Miguel Hernández, Elche, Spain, 2000.
- A. E. O. El Khalifa, B. Schiffler and R. Bernhard, “Effect of Fermentation on the Functional Properties of Sorghum Flour,” Food Chemistry, Vol. 92, No. 1, 2005, pp. 1-5. doi:10.1016/j.foodchem.2004.05.058
- A. Vázquez-Ovando, G. Rosado-Rubio, L. Chel-Guerrero and D. Betancur-Ancona, “Physicochemical Properties of a Fibrous Fraction from Chia (Salvia hispanica L.),” Food Science & Technology, Vol. 42, No. 1, 2009, pp. 168-173.
- C. Chau, K. Cheung and Y. Wong, “Functional Properties of Protein Concentrate from Three Chinese Indigenous Legume Seeds,” Journal of Agriculture and Food Chemistry, Vol. 45, No. 17, 1997, pp. 2500-2503. doi:10.1021/jf970047c
- J. A. Buege and S. D. Aust, “Microsomal Lipid Peroxidetion,” In: M. S. Flesicher and L. Packer, Eds., Methods in Enzymology, Academic Press, New York, 1978, pp. 302- 310.
- K. Krzywicki, “The Determination of Haem Pigments in Meat,” Meat Science, Vol. 7, No. 1, 1982, pp. 29-36. doi:10.1016/0309-1740(82)90095-X
- L. L. Stookey, “Ferrozine—A New Spectrophotometric Reagent for Iron,” Analytical Chemistry, Vol. 42, No. 7, 1970, pp. 779-781.
- H. C. Hornsey, “The Color of Cooked Cured Pork. I. Estimation of Nitric Oxide-haem Pigments,” Journal of Agriculture and Food Chemistry, Vol. 7, No. 8, 1956, pp. 534 -540.
- E. M. Clark, A. W. Mahoney and C. E. Carpenter, “Heme and Total Iron in Ready-to-Eat Chicken,” Journal of Agriculture and Food Chemistry, Vol. 45, No. 1, 1997, pp. 124-126.
- H. Saito, K. Ishihara and T. Murase, ” Effect of Prey Fish Lipids on the Docohexaenoic Acid Contento of Total Fatty Acids in the Lipid of Thunnus Albaceres Yellowfin Tuna,” Bioscience, Biotechnology, and Biochemistry, Vol. 60, No. 6, 1996, pp. 962-965. doi:10.1271/bbb.60.962
- M. Emin-Erdem, F. Kakayci, H. Avni-Duyar and S. Samsun, “Shelf Life and Biochemical Composition of Bonito Fish (Sarda Sarda Bloch 1758) Stored at 4˚C and Fishing with Different Fishing Tools,” Journal of Muscle Foods, Vol. 20, No. 2, 2009, pp. 242-253. doi:10.1111/j.1745-4573.2009.00148.x
- L. Gram and H. H. Huss, “Microbial Spoliage of Fish and Fish Products,” International Journal of Food Microbiology, Vol. 33, No. 1, 1996, pp. 121-137. doi:10.1016/0168-1605(96)01134-8
- C. A. Onyango, M. Izumimoto and P. P. Kutima, “Comparison of Some Physical and Chemical Properties of Selected Game Meats,” Meat Science, Vol. 49, No. 1, 1998, pp. 117-125. doi:10.1016/S0309-1740(97)00116-2
- S. Kanoh, T. Suzuki, K. Maeyama, T. Takewa, S. Watabe and K. Hashimoto, “Comparative Studies on Ordinary and Dark Muscle of Tuna Fish,” Bulletin of the Japanese Society of Fisheries Oceanography, Vol. 52, No. 10, 1986, pp. 1807-1816.
- Z. Tzikas, E. Papavergou, N. Soultos, I. Ambrosiadis and S. P. Georgakis, “Quality Loss of Mediterranean Horse Mackerel (Trachurus mediterraneus) Skinned Fillets Kept under Vacuum During Frozen Storage,” Journal of Aquatic Food Product Technology, Vol. 18, No. 3, 2009, pp. 266-283. doi:10.1080/10498850902762174
- S. D. dos Santos, V. G. Martins, M. Salas-Mellado and C. Prentice, “Evaluation of Functional Properties in Protein Hydrolysates from Bluewing Searobin Obtained with Different Microbial Enzymes,” Food and Bioprocess Technology, doi 10.1007/s11947-009-0301-0, 2010.
- Y. Pomeranz, “Proteins: Specific Foods,” In: S. L. Taylor, Ed., Functional Properties of Food Components, 2th Edition, Academic Press, Inc, New York (USA), 1991, pp. 210-247.
- H. G. Kristinsson and B. A. Rasco, “Fish Protein Hydrolysates: Production, Biochemical and Functional Properties,” Critical Reviews in Food Science and Nutrition, Vol. 40, No. 1, 2000, pp. 43-81. doi:10.1080/10408690091189266
- G. Akamittath, C. J. Brekke and E. G. Schanus, “Lipid Oxidation and Color Stability in Restructured Meat Systems during Frozen Storage,” Journal of Food Science, Vol. 55, No. 6, 1990, pp. 1513-1517. doi:10.1111/j.1365-2621.1990.tb03557.x
- Y. Mercier, P. Gatellier and M. Renerre, “Relationships between Lipid and Protein Oxidation in Different Beef Muscles,” In: Proceedings of the 41st ICoMST, San Antonio, USA, 1995, pp. 562-564.
- J. Fernández-López, A. Yelo, E. Sayas-Barberá, E. Sendra, C. Navarro and J. A. Pérez-Alvarez, “Shelf Life of Ostrich (Struthio camelus) Liver Stored under Different Packaging Conditions,” Journal of Food Protection, Vol. 69, No. 8, 2006, pp. 1920-1927.
- F. M. Teeny, E. J. Gauglitz, A. S. Hall and C. R. Houle, “Mineral Composition of de Edible Muscle Tissue of Seven Species of Fish from the Northeast Pacific,” Journal of Agriculture and Food Chemistry, Vol. 32, No. 4, 1984, pp. 852-855. doi:10.1021/jf00124a040
- J. Kanner, “Oxidative Processes in Meat Products: Quality Implications,” Meat Science, Vol. 36, No. 1-2, 1994, pp. 169-189. doi:10.1016/0309-1740(94)90040-X
- C. E. Carpenter and A. W. Clark, “Contributions of Heme and Non-Heme Iron to Human Nutrition,” Critical Reviews in Food Science and Nutrition, Vol. 31, No. 4, 1995, pp. 333-367. doi:10.1080/10408399209527576
- E. Sikorski, A. Kolakowska and B. Sun-Pan, “Nutrient Composition of the Major Groups of Marine Food Organisms,” In: E. Sikorski, Ed., Technology seafood: Resources, Nutritional Composition and Preservation, Acribia, Zaragoza, Spain, 1994, pp. 41-70.
- V. Papadopoulos, I. Chouliara, A. Badeka, I. N. Savvaidis and M. G. Kontominas, “Effect of Gutting on Microbiological, Chemical, and Sensory Properties of Aquacultured Sea Bass (Dicentrarchus labrax) Stored in Ice,” Food Microbiology, Vol. 20, No. 4, 2003, pp. 411-420. doi:10.1016/S0740-0020(02)00148-X
- J. Barros-Velázquez, J. M. Gallardo, P. Calo and S. P. Aubourg, (2008) “Enhanced Quality and Safety during on-Board Chilled Storage of Fish Species Captured in the Grand Sole North Atlantic Fishing Bank,” Food Chemistry, Vol. 2, No. 15, 2008, pp. 493-500. doi:10.1016/j.foodchem.2007.06.042
- H. G. Kristinsson, S. Crynen and Y. Yagiz, “Effect of a Filtered Wood Smoke Treatment Compared to Various Gas Treatments on Aerobic Bacteria in Yellowfin Tuna Steaks,” LWT-Food Science and Technology, Vol. 41, No. 4, 2008, pp. 746-750. doi:10.1016/j.lwt.2007.04.005
- N. Guizani, M. A. Al-Busaidy, I. M. Al-Belushi, A. Mothershaw and M. S. Rahman, “The Effect of Storage Temperature on Histamine Production and the Freshness of Yellowfin Tuna (Thunnus albacares),” Food Research International, Vol. 38, No. 2, 2005, pp. 215-222. doi:10.1016/j.foodres.2004.09.011
- B. O. E., Boletín Oficial del Estado, “Laying Down the Microbiological Standards of the Various Products of Fisheries and Aquaculture, as Well as the Limits of Heavy Metal Content and Analytical Methods for Their Determination,” Spain, 1991, p. 195.

