Modern Research in Catalysis
Vol.03 No.03(2014), Article ID:47851,4 pages
10.4236/mrc.2014.33011
γ-Al2O3 Supported  /ZrO2 Solid Superacid Catalysts for n-Pentane Isomerization
/ZrO2 Solid Superacid Catalysts for n-Pentane Isomerization
Li Zhao, Xiaoshuang Cheng, Ye Hu, Shuqing Ma, Yingjun Wang
College of Chemistry & Chemical Engineering, Provincial Key Laboratory of Oil & Gas Chemical Technology, Northeast Petroleum University, Daqing, China
Email: zhaolikele9903@163.com, chengxiaoshuang@aliyun.com, 87581812@qq.com, msq6101@163.com, wangying-jun@163.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 20 April 2014; revised 20 May 2014; accepted 3 June 2014

ABSTRACT
A solid superacid catalyst Pt- /ZrO2-A12O3 for n-pentane isomerization, was prepared by incipient-wetness impregnation. Preparetion conditions, namely, calcination temperature, concentration of sulfuric acid solution used in impregnation and Al2O3 concentration, were varied to investigate the effects on catalytic performance of Pt-
/ZrO2-A12O3 for n-pentane isomerization, was prepared by incipient-wetness impregnation. Preparetion conditions, namely, calcination temperature, concentration of sulfuric acid solution used in impregnation and Al2O3 concentration, were varied to investigate the effects on catalytic performance of Pt-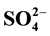 /ZrO2-A12O3. The results showed that the PtSZA catalyst exhibited excellent catalytic performance for n-pentane isomerization. Under optimized preparation conditions of calcination temperature of 650˚C, reaction time for 3 h, concentration of sulfuric acid solution for 0.5 mol/L, 30% of Al2O3 concentration and 0.3% of Pt concentration, the n-pentane conversion and isopentane selectivity of Pt-
/ZrO2-A12O3. The results showed that the PtSZA catalyst exhibited excellent catalytic performance for n-pentane isomerization. Under optimized preparation conditions of calcination temperature of 650˚C, reaction time for 3 h, concentration of sulfuric acid solution for 0.5 mol/L, 30% of Al2O3 concentration and 0.3% of Pt concentration, the n-pentane conversion and isopentane selectivity of Pt-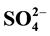 /ZrO2-A12O3 could reach up to 62.17% and 91.60%, respectively.
/ZrO2-A12O3 could reach up to 62.17% and 91.60%, respectively.
Keywords:
SO42−/ZrO2, n-pentane isomerization, γ-Al2O3, Supported superacidcatalysts 
1. Introduction
The isomerization of light paraffins to branched isomers is an important process in refining industry, which has a wide range of applications in increasing gasoline octane number, reducing diesel oil solidifying point and improving low temperature property of lubricant base oil. Although the early light hydrocarbon isomerization catalyst (such as H2SO4 and HF) has high acid strength, it is strong corrosive and toxic. Therefore, it has been abandoned with the growing awareness of environmental protection and safety. At present, Chloride treatment of Pt-A12O3 type low-temperature isomerization catalysts are widely used in the industry and usually have high activity and selectivity. The isomerization catalyst in the use process generally requires the addition of a small amount of chlorine to maintain catalytic activity. However, there are corrosion and pollution problems over the catalyst loading and handling process. It is sensitive to water, sulfur and thus raw materials are not allowed to contain water and sulfur, which limits the application of the process. In 1979, Hino and co-workers impregnated amorphous zirconium hydroxide with sul-furic acid to give upon calcination a solid superacid (Hammett acidity < −16) capable of catalyzing the isomerization of n-butane [1] [2] . The study on the 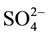 /ZrO2 isomerization reaction for for n-butane at room temperature was performed by Hino, and
/ZrO2 isomerization reaction for for n-butane at room temperature was performed by Hino, and 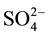 /ZrO2 showed very high catalytic ctivity. Since then,
/ZrO2 showed very high catalytic ctivity. Since then, 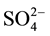 /ZrO2 solid superacid catalysts caused extensive concern of the researchers. Compared with liquid acid catalysts, solid acid catalyst has the advantages of high catalytic efficiency, simple preparation method, easy separation of products, being non-corrosive, environmentally friendly and reusable. Solid super acid not only overcomes many disadvantages of liquid acid catalyst, but also presents high catalytic activity in many reactions of isomerization, alkylation, dehydration and esterification. However, from the view point of practice, the activity of SZ needs to be improved further, at the same time the use of isomerization catalysts can reduce costs. The task of decreasing its cost remains important and challenging. Some researchers [3] suggested that supporting sulfated zirconia on a cheaper porous material may be a good way to enhance the amount of superacid site by increasing the surface area and lowering the cost of catalysts.
/ZrO2 solid superacid catalysts caused extensive concern of the researchers. Compared with liquid acid catalysts, solid acid catalyst has the advantages of high catalytic efficiency, simple preparation method, easy separation of products, being non-corrosive, environmentally friendly and reusable. Solid super acid not only overcomes many disadvantages of liquid acid catalyst, but also presents high catalytic activity in many reactions of isomerization, alkylation, dehydration and esterification. However, from the view point of practice, the activity of SZ needs to be improved further, at the same time the use of isomerization catalysts can reduce costs. The task of decreasing its cost remains important and challenging. Some researchers [3] suggested that supporting sulfated zirconia on a cheaper porous material may be a good way to enhance the amount of superacid site by increasing the surface area and lowering the cost of catalysts.
By using solid superacid catalyst in light hydrocarbon isomerization process, the isomerization products are environmental gasoline harmonic components that do not contain aromatics and olefins. Moreover, the process itself is environmental as well, which overcomes the corrosion problem in the traditional low-temperature isomerization process. Hence, it has attracted increasingly attention from word-wide researchers [4] .
2. Experiment
In this paper, the Pt- /ZrO2-A12O3 catalyst was prepared by two step method. The n-pentane selected as reaction mediums for catalyst evaluation, and n-pentane isomerization performance of catalysts was evaluated in a 10ml fixed bed microreactor-chromatography unit with high pressure continuous flow. The catalyst was placed in the middle of the reactor while the rest empty volume was filled with quartz sand. The effect of different preparetion conditions on n-pentane isomerization performance of Pt-
/ZrO2-A12O3 catalyst was prepared by two step method. The n-pentane selected as reaction mediums for catalyst evaluation, and n-pentane isomerization performance of catalysts was evaluated in a 10ml fixed bed microreactor-chromatography unit with high pressure continuous flow. The catalyst was placed in the middle of the reactor while the rest empty volume was filled with quartz sand. The effect of different preparetion conditions on n-pentane isomerization performance of Pt-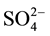 /ZrO2-A12O3 was mainly investigated.
/ZrO2-A12O3 was mainly investigated.
Chemical reagents: ZrOCl2∙8H2O (analytical pure, Shanghai richjoint Chemical Reagent Co. Ltd.); single water of aluminum hydroxide (Fushun No.3 oil factory); n-pentane (analysis purity, Shenyang Huadong Reagent Factory); H2PtC16∙6H2O (analytical pure, Shenyang Jinke Reagent Factory); sulfuric acid (analysis purity, Kunshan Jincheng Reagent Co. company); nitric acid (analysis purity, Kunshan Jincheng Reagent Co. Ltd.); concentrated ammonia solution (concentration 28%); silver nitrate solution (0.1 mol/L−1); hydrogen (purity over 99%).
Preparation of catalyst: The catalyst was prepared as follows: The required amount of ZrOCl2・8H2O was dissolved in deionized water. Aqueous ammonia was added dropwise to a solution of ZrOCl2・8H2O until pH = 9 - 10, and then the precipitation was obtained. After being stored at room temperature for 24 h, the precipitation was washed repeatly with deionized water until the washing liquid is no longer able to check out the Cl−1 ion (0.1 mol/L−1 AgNO3 solution). And then dried in oven (383 K, 24 h), the obtained powder was denoted as Zr(OH)4 and grinded into less than 100 mesh. Zr(OH)4 was mixed with proper amount of alumina. The obtained mixture immersed in sulfuric acid solution, dried at 383 K overnight then dipped H2PtC16∙6H2O solution. After drying at 383 K overnight, suitable amount of sesbania powders and 18% dilute nitric acid solution were added. Followed by extruding forming, drying and calcining in muffle oven at given temperature is to obtain the Pt-SO42−/ZrO2-A12O3 catalysts, denoted as PtSZA.
3. Results and discussion
3.1. Effect of Calcination Temperatures on n-pentane isomerizationrate and Selectivity of Pt- /ZrO2-Al2O3 Catalyst
/ZrO2-Al2O3 Catalyst
The effect of calcination temperatures on conversion of n-pentane isomerization reaction rate and selectivity is shown in Figure 1. As can be seen, calcination temperature is varied from 550˚C to 650˚C, the conversion of n-pentane and isopentane yield increases and maintains a high selectivity. Increasing the calcined temperature up to 700˚C, conversion of n-pentane and isopentane yield are obviously decreased. Although there is the decomposition and loss of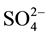 , the interaction of
, the interaction of 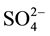 with the catalyst surface is strong when the calcination temperature is below 650˚C. The most ZrO2 exist mainly as tetragonal, monoclinic accounts for only a small proportion, thus the catalyst has high activity. After high temperature calcination rises up to 700˚C, most of
with the catalyst surface is strong when the calcination temperature is below 650˚C. The most ZrO2 exist mainly as tetragonal, monoclinic accounts for only a small proportion, thus the catalyst has high activity. After high temperature calcination rises up to 700˚C, most of 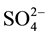 loss, sulfur content is very small and the average pore size of the catalyst becomes large, which resulting in very small specific surface area. And the thermally treated reduces the proportion of the ZrO2 tetragonal and increases monoclinic crystal ratio, so leading to the decreasing activity of the catalyst for n-pentane isomerization.
loss, sulfur content is very small and the average pore size of the catalyst becomes large, which resulting in very small specific surface area. And the thermally treated reduces the proportion of the ZrO2 tetragonal and increases monoclinic crystal ratio, so leading to the decreasing activity of the catalyst for n-pentane isomerization.
3.2. Units Effect of sulfuric acidconcentrations on the Catalyst performance
Figure 2 presents n-pentane isomerization performance for the Pt-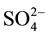 /ZrO2-A12O3 catalysts with different sulfuric acid concentrations. The experimental results show that solid superacid catalysts with superior performance must ensure to have the appropriate concentration of
/ZrO2-A12O3 catalysts with different sulfuric acid concentrations. The experimental results show that solid superacid catalysts with superior performance must ensure to have the appropriate concentration of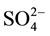 . The relationship between
. The relationship between 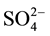 and ZrO2 must achieve the best match with each other, so that we can get highly active and selective solid super acid catalysts.
and ZrO2 must achieve the best match with each other, so that we can get highly active and selective solid super acid catalysts.
This is because high concentration of H2SO4 forms the sulfate and then covers the part of the acid site. So that the problems caused by uneven distribution of the acid site will affect the activity and the selectivity of the reaction. When the H2SO4 concentration is 0.25 mol/L, under the same reaction conditions, the conversion of n-pentane and selectivity of the Pt-

3.3. Effect of Al2O3 Contents on the properties of solid Superacid Catalyst
To clarify the influence of Al2O3 and ZrO2 over properties of solid superacid catalysts, the Pt-
Figure 1. Effect of calcined temperature on n-pentane conversion or selecti- vity.
Figure 2. Effect of 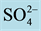
Figure 3. Effect of Al2O3 content on n-pentane conversion and selectivity.
declines. It indicates that forming strong acid centers should reduce the ZrO2 content requiring. However, the number of the active center is reduced, leading to decreasing activity. Therefore, introducing the appropriate amount of Al2O3 can increase acid strength of the catalyst, so as to improve catalytic activity and stability of the Pt-

4. Conclusion
The best conditions for preparation of Pt-

Acknowledgements
The authors gratefully acknowledge the support from the Provincial Key Laboratory of Oil & Gas Chemical Technology of Daqing. This study was supported by scientific research fund of Heilongjiang provincial education department (12521063).
References
- Hino, M., Kobaya, S. and Arata, K. (1979) Reaction of Butane and Isobutane Catalyzed by Zirconium Oxide Treated with Sulfate Ion. Journal of the American Chemical Society, 101, 6439-6445. http://dx.doi.org/10.1021/ja00515a051
- Tian, G., Xu, Y.P. and Xu, Z.S. (2008) Effect of Aluminum on the Mechanical Stress Stability of WOx/ZrO2 Superacid. Chinese Journal of Catalysis, 29, 415-417. http://dx.doi.org/10.1016/S1872-2067(08)60041-8
- Laosiripojana, N., Kiatkittipong, W., Sutthisripok, W. and Assabumrungrat, S. (2010) Synthesis of Methyl Esters from Relevant Palm Products in Near-Critical Methanol with Modified-Zirconia Catalysts. Bioresource Technology, 101, 8416-8423. http://dx.doi.org/10.1016/j.biortech.2010.05.076
- Shu, Q., Zhang, Q., Xu, G.H., Nawaz, Z., Wang, D.Z. and Wang, J.F. (2009) Synthesis of Biodiesel from Cottonseed Oil and Methanol Using a Carbonbasedsolid Acid Catalyst. Fuel Processing Technology, 90, 1002-1008. http://dx.doi.org/10.1016/j.fuproc.2009.03.007




