American Journal of Analytical Chemistry
Vol.5 No.1(2014), Article ID:41861,5 pages DOI:10.4236/ajac.2014.51003
Emulsion Polymerization of Vinyl Acetate in the Presence of a New Cationic Surfactant
1Department of Chemistry, Yıldız Technical University, Istanbul, Turkey
2Department of Chemistry, Istanbul Technical University, Istanbul, Turkey
Email: *ayfersarac2002@yahoo.com
Received November 23, 2013; revised December 23, 2013; accepted January 2, 2014
ABSTRACT
In this study, the emulsion homopolymerization system containing vinyl acetate, potassium persulfate, a new cationic polymeric surfactant and water was studied by the applying semi-continuous emulsion polymerization process. The effects of new polymeric emulsifier on the physicochemical properties of obtained vinyl acetate latexes were investigated depending on vinyl acetate percentage in the polymerization recipe, and two thermal initiators in homopolymerization.
Keywords:Emulsion Polymerization; Lattices; Cationic Surfactants; Critic Micelle Concentration; Viscosity
1. Introduction
The stability of polymeric dispersions is a property of practical importance and hence, the search for an optimized strategy to equip polymer dispersions with sufficient stability is a matter of continuous research during the past years. The kernel is to reach a sufficient stability, as it is required during polymerization, conditioning, or storage and to allow coagulation or coalescence of particles when it is needed, such as during separation of polymer from latex or during film formation. Stability is an important topic in the field of polymer colloid research such as under the conditions of the polymerization process [1].
Conventional surfactants are typically characterized by a chemical structure that combines a hydrophilic group with one or two hydrophobic flexible alkyl chains of moderated length. In aqueous phase, small amounts of surfactant are enough to self-assemble into micellar microaggregates [2].
In general, particle stabilization is achieved by the use of various surfactants which are employed in emulsion polymerization formulations. The anionic and nonionic surfactants are the most widely utilized because of enhanced compatibility with negatively charged latex particles (usually as a result of persulfate initiator fragments) as compared to other emulsifiers [3-5]. Recent years, particle stabilization can be achieved via novel surfactants such as oligomeric, polymeric, AB type, ABA type etc. The hydrophilic-hydrophobic nature of polymeric surfactants has a significant influence on the properties of micelles. In contrast to small surfactants, polymeric surfactant molecules exhibit rather restricted mobility in the heterogeneous polymerization system and this characteristic feature may have an impact on the polymerization mechanisms and kinetics. In general, latex products stabilized by polymeric surfactants show better water resistance than those stabilized by conventional surfactants. Among various polymeric stabilizers used in industry, amphiphilic blocks or graft copolymers are most effective for stabilizing emulsion polymerization [5,6].
Many studies have been carried out to analyze the effect of the type and amount of surfactant in an emulsion polymerization, but almost all cases anionic or anionic and nonionic mixed emulsifier systems are used [7-14]. However, the effect of the type and amount of cationic emulsifier in a cationic emulsion polymerization is reported in a few cases [15-19].
Cationic surfactants are only infrequently used in vinyl acetate (VAc) emulsion polymerization, because they are not compatible with anionic emulsifiers or negatively charged latex particles [5]. They are frequently (generally) based on amine-containing polar head-groups. Due to their charged nature, the properties of cationic surfactants, e.g., surface activity or structure formation, are generally strongly dependent on the salt concentration, and on the valence of anions present. Generally cationic surfactants are used as antibacterial and/or limited foam producing agent. However, they are irritant and sometimes even toxic [14].
In the present work, a new polymeric cationic surfacetant was synthesized and used in the emulsion homopolymerization of vinyl acetate (VAc) in different concentrations for the latex stabilization. The aim of this work was to study the effects of the cationic surfactant and two thermal initiators on the colloidal characteristics of a semicontinuous cationic emulsion polymerization of VAc.
2. Experimental
2.1. Materials
Vinyl acetate (VAc) was used Fluka purification. Stability of the latexes was achieved by synthesized new cationic surfactant. This substance was acted like a protective colloid because of given viscose solution even very low concentration, ammonium persulfate (APS) (Merck) and potassium persulfate were used as thermal initiators. Sodium bicarbonate (Merck) was used for adjusting the pH value of the polymerization media. All substances were used as received. Deionized water was used in all studies.
2.2. Preparation of the Polymeric Surfactant
5 mL of TEMED was added to 20 mL of diethylether and 5.2 mL of Dibromohexane was added to this solution. The reaction was preceded at room temperature for 48h. Precipitated white solid product was filtered and was washed with excess of diethylether to remove soluble fractions. The white polymeric product was dried under vacuum at room temperature for 24 h. The yield was 10.33 g (86%). The polymeric surfactant was characterized with FT-IR, critical micelle concentration and viscosity analysis.
2.3. Determination of the Critical Micelle Concentration (CMC)
For this purpose 0.5 g of polymeric surfactant sample was dissolved in 30 mL water. This solution was placed in thermostat bath at 25˚C. 1 mL of water at a time was added to the surfactant solution until the volume reaches up to 50 mL and the conductivity of the solution was measured after every addition continuously.
2.4. Emulsion Polymerization of Vinyl Acetate
Semicontinuous emulsion polymerizations of VAc with polymeric surfactant were carried out in a 250 mL five neck glass reactor equipped with a condenser, a thermometer, two dropping funnels, and a mechanical stirrer having a constant speed of 400 rpm and a reflux condenser in a total batch period of 2 h. Only 10% of the total monomer was introduced at the beginning of the reaction at 60˚C and the remaining monomer was added dropwise at 71˚C ± 2˚C from dropping funnels during the remaining time. Initiator was dissolved in a small amount of water and these freshly prepared initiator portions were added at 30 minute intervals throughout the reaction. Polymerization reactions were performed by using different surfactant concentration.
2.5. Cationic Surfactant and Latex Characterizations
New synthesized polymeric surfactant was characterized by FT-IR and its critical micelle concentration was determined by conductometric methods. Latexes were characterized by determination of conversion, and measuring Brookfield viscosity, viscosity average molecular weight (Mv), glass transition temperature (Tg), and surface tension of latexes to air. Conversions were monitored gravimetrically. The original viscosities of the homopolymer latexes were determined by Brookfield Programmable DV-II model viscometer with spindle number 4 at 20˚C.
Viscosity average molecular weights of polymers were determined by capillary intrinsic viscometry at 30˚C. Mv values of the polymers were determined using Ubbelohde-type viscometer in an acetone solvent for poly (vinyl acetate). For this purpose stable latexes were precipitated by adding salt and obtained polymers were filtered and was washed excess of hot water and were dried under vacuum at room temperature for 48 h. Dried polymers were dissolved in different concentration by using acetone for PVAc. Molecular weights of the polymers were determined viscosimetrically by using Mark-Houwink-Sakurada equation:
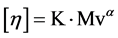
Mark-Houwink constant values of VAc were used as α = 0.72 and K = 1.01 × 10−4 (dL/g) in the calculations.
The surface tension measurements were done with a Sigma 701 model tensiometer (KSV instruments, Helsinki, Finland) using ring-detachment method and a platinum ring at 23.7˚C.
A Perkin Elmer Pyris 6 model differential scanning calorimeter was used to determine Tg of the homopolymers. The scan was performed at a heating rate of 10˚C/min under N2 atmosphere.
3. Results and Discussion
3.1. Synthesizing of Polymeric Surfactant
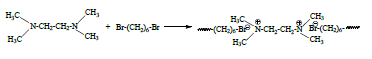
Polymeric surfactant was prepared by starting from TEMED and 1,6-dibromohexane. The reaction was carried out in ether at room temperature for 3 days. White precipitated solid product was filtrated and was washed with excess of ether. Chemical structure of synthesized new cationic surfactant was given in Scheme 1.
The polymeric product was characterized by FT-IR, viscosity measurements and critical micelle concentration. The FT-IR spectrum of cationic surfactant (Figure 1) was as expected, with bands for the alkyl group at 2900 - 2800 cm−1. The new bands were observed at 1133 cm−1 and 3010 cm−1 because of quaternization.
3.2. Determination of CMC of the Polymeric Surfactant
Critical micelle concentration of the surfactant was determined by conductometric method by measuring conductance. Critical micelle concentration of the new cationic surfactant was calculated by Figure 2 as 1.67 × 10−2 g/mL.
3.3. Inherent Viscosity of Polymeric Surfactant
The inherent viscosities of the polymeric surfactant in various solutions calculated from the data taken from the Ubbelohde type viscometer. Inherent viscosity of the polymeric surfactant was found in different solvent (Table 1).
3.4. Preparation of Emulsion Polymers
Stable latexes were precipitated by adding NaCl and obtained polymers were filtered and washed excess of hot water and were dried under vacuum at room temperature for 48 h. Dried polymers were dissolved in different concentration by using acetone for PVAc. All obtained results were given in Table2
The surface tensions and Brookfield viscosity of PVAc latexes changed regularly with increasing monomer concentration.
Cationic surfactants are used in high limited in industrial emulsion polymerization, because of economic aspects. Studies concerning dimeric surfactants are focused on the relationship between surfactant structure and the critical micelle concentration by using the alkyl chainlength and the chemical nature of the spacer as rele-

Figure 1. The FT-IR spectrum of the polymeric surfactant.
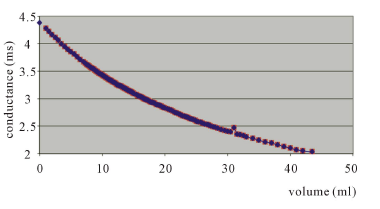
Figure 2. The CMC graph of the polymeric surfactant.
Table 1. Inherent viscosity values in different solutions.
vant parameters, on the effect of alkyl chain and spacer length on critical micelle concentration and micelle ionization degree [20]. The evolution of the Brookfield viscosity and glass transition temperature of VAc homopolymer as a function of the monomer concentration is shown in Figures 3 and 4.
PVAc produces higher viscosity values than expected and this is clearly reflected in the stabilization of the already mentioned number of polymer particles, Tg and particle size correlation. Because using amounts of new cationic surfactant are indeed very low in the VAc homopolymerization. The surface tensions of polymer changed regularly with the increasing surfactant percentage in the VAc polymerization in Figure 5.

Figure 3. Brookfield viscosity of VAc homopolymers as a function of monomer concentration and initiators.
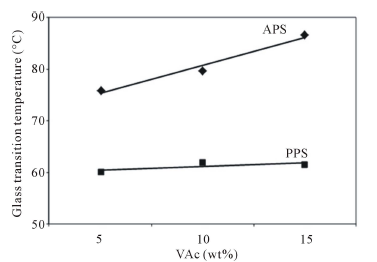
Figure 4. Tg of VAc homopolymers as a function of monomer concentration and initiators.
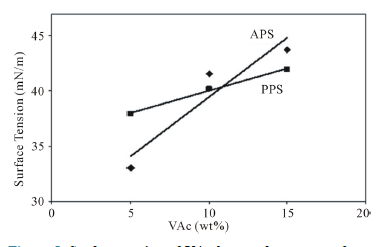
Figure 5. Surface tension of VAc homopolymers as a function of monomer concentration and initiators.
Surface tension depends on free emulsifier amounts in the latex, and the raising of this amount leads to the tendency of surface tension to increase. But emulsifier adsorption onto polymer particles cause to decrease free emulsifier concentration in latex, the increasing adsorption cause to increase surface tension of latexes.
4. Conclusion
In this work, a new cationic polymeric surfactant was synthesized and was used in emulsion polymerization of vinyl acetate (VAc). The features of the cationic polymerizations of vinyl acetate were analyzed by the consideration of the percentage of the monomer and the final viscosity of different latexes obtained from the polymerizations.
REFERENCES
- T. Aslamazova and K. Tauer, “On the Colloidal Stability of Polystyrene Particles Prepared with Surface-Active Initiators,” Advances in Colloid and Interface Science, Vol. 104, No. 1, 2003, pp. 273-283. http://dx.doi.org/10.1016/S0001-8686(03)00046-0
- E. A. Zaragoza-Contreras and D. M. Navarro-Rodriguez, “On the Role of an Unconventional Rigid Rodlike Cationic Surfactant on the Styrene Emulsion Polymerization. Kinetics, Particle Size and Particle Size Distribution,” Polymer Vol. 44, No. 19, 2003, pp. 5541-5546. http://dx.doi.org/10.1016/S0032-3861(03)00621-9
- M. S. El-Aasser and J. W. Van der Hoff, “Emulsion Polymerization of Vinyl Acetate,” Applied Science Publication, London and New Jersey, 1981. http://dx.doi.org/10.1007/978-94-009-8114-0
- P. A. Lovell and M. S. El-Aasser, “Emulsion Polymerization and Emulsion Polymers,” John Wiley and Sons, Chichester, 1997.
- H. Y. Erbil, “Vinyl Acetate Emulsion Polymerization and Copolymerization with Acrylic Monomers,” CRC Press, Boca Raton, 2000. http://dx.doi.org/10.1201/9781420038804
- C. S. Chern, “Emulsion Polymerization Mechanisms and Kinetics,” Progress in Polymer Science, Vol. 31, No. 5, 2006, pp. 443-486. http://dx.doi.org/10.1016/j.progpolymsci.2006.02.001
- E. Unzueta and J. Forcada, “Semicontinuous Emulsion Copolymerization of Methyl Methacrylate and n-Butyl Acrylate: 1. Effect of Mixed Emulsifiers in Seeded Polymerization,” Polymer, Vol. 36, No. 5, 1995, pp. 1045- 1052. http://dx.doi.org/10.1016/0032-3861(95)93606-M
- B. Emelie, C. Pichot and J. Guillot, “Batch Emulsion Copolymerization of n-Butyl Acrylate and Methyl Methacrylate in the Presence of a Nonionic Surfactant,” Die Makromolekulare Chemie, Vol. 10, Suppl. 10-11, 1985, pp. 43-57. http://dx.doi.org/10.1002/macp.1985.020101985106
- B. Emelie, C. Pichot and J. Guillot, “Batch Emulsion Copolymerization of Butyl Acrylate and Methyl Methacrylate in the Presence of Sodium Dodecyl Sulfate,” Die Makromolekulare Chemie Vol. 192, No. 7, 1991, pp. 1629-1647. http://dx.doi.org/10.1002/macp.1991.021920716
- H. Chu and I. Piirma, “The Stabilization Effect of Mixed-Surfactants in the Emulsion Polymerization of Styrene,” Polymer Bulletin, Vol. 21, No. 3, 1989, pp. 301-308. http://dx.doi.org/10.1007/BF00955922
- H. Wang and H. Chu, “The Stabilization Effect of MixedSurfactants in the Emulsion Polymerization of Methyl Methacrylate,” Polymer Bulletin Vol. 24, No. 2, 1990, pp. 207-214. http://dx.doi.org/10.1007/BF00297319
- C. H. Lee and R. G. Mallinson, “Surfactant Effects in the Emulsion Polymerization of Vinyl Acetate,” Journal of Applied Polymer Science, Vol. 39, No. 11-12, 1990, pp. 2205-2218. http://dx.doi.org/10.1002/app.1990.070391102
- P. J. Feeney, D. H. Napper and R. G. Gilbert, “The Determinants of Latex Monodispersity in Emulsion Polymerizations,” Journal of Colloid and Interface Science, Vol. 118, No. 2, 1987, pp. 493-505. http://dx.doi.org/10.1016/0021-9797(87)90485-1
- J. Ramos and J. Forcada, “Semicontinuous Seeded Cationic Emulsion Polymerization of Styrene: The Effects of the Concentration and Type of Cationic Surfactant,” Journal of Polymer Science Part A: Polymer Chemistry, Vol. 41, No. 15, 2003, pp. 2322-2334. http://dx.doi.org/10.1002/pola.10775
- F. Miraballes-Martinez, A. Martin-Molina, F. GalisteoGonzalez and J. Forcada, “Synthesis of Amino-Functionalized Latex Particles by a Multistep Method,” Journal of Polymer Science Part A: Polymer Chemistry, Vol. 39, No. 17, 2001, pp. 2929-2936. http://dx.doi.org/10.1002/pola.1273
- H. H. Chu and H. Y. Hwang, “Stabilizing Effect of the Cationic Surfactant (CPB) in Emulsion Polymerization,” Polymer Bulletin, Vol. 38, No. 3, 1997, pp. 295-302. http://dx.doi.org/10.1007/s002890050051
- D. Cochin, A. Laschewaky and F. Nallet, “Emulsion Polymerization of Styrene Using Conventional, Polymerizable, and Polymeric Surfactants. A Comparative Study,” Macromolecules, Vol. 30, No. 8, 1997, pp. 2278-2287. http://dx.doi.org/10.1021/ma9614510
- J. Wieboldt, R. Zimehl, J. Ahrens and G. Lagaly, “Some Aspects of Polymer Colloids. III. Preparation and Properties of Different Types of Cationic Latex Particles,” In: F. Kremer and G. Lagaly, Eds., Progress in Colloid & Polymer Science, Dr. Dietrich Steinkopff Verlag GmbH & Co. KG, Darmstadt, 1998, Vol. 109, pp. 260-269.
- K. Landfester, N. Bechthold, F. Tiarks and M. Antonietti, “Miniemulsion Polymerization with Cationic and Nonionic Surfactants: A Very Efficient Use of Surfactants for Heterophase Polymerization,” Macromolecules Vol. 32, No. 8, 1999, pp. 2679-2683. http://dx.doi.org/10.1021/ma9819438
- R. Zana, M. Benrraou and R. Rueff, “Alkanediyl-Alpha Omega-Bis(Dimethylalkylammonium Bromide) Surfactants. 1. Effect of the Spacer Chain Length on the Critical Micelle Concentration and Micelle Ionization Degree,” Langmuir, Vol. 7, No. 6, 1991, pp. 1072-1075. http://dx.doi.org/10.1021/la00054a008
NOTES
*Corresponding authors.