Journal of Biosciences and Medicines
Vol.03 No.08(2015), Article ID:58698,11 pages
10.4236/jbm.2015.38003
The Impact of Some Widely Probiotic (Iraqi Probiotic) on Health and Performance
Hozan Jalil Hamasalim
Department of Animal Production, Faculty of Agricultural Sciences, University of Sulaimani, Sulaimani, Iraq
Email:hozan.hamasaleem@univsul.edu.iq
Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 June 2015; accepted 7 August 2015; published 10 August 2015
ABSTRACT
Probiotics (Greek: for life) are commonly defined as mono- or mixed cultures of live microbes. When applied to animals or humans, they exert a beneficial effect on health of the host. These beneficial effects include disease treatment and prevention as well as improvement of nutrients’ digestion and absorption. Probiotic microorganisms are generally, albeit not exclusively, lactic acid bacteria (LAB) including Lactobacillus acidophilus, L. bulgaricus, L. casei, L. plantarum, and L. rhamnosus. However, use of other bacterial species such as Bacillus and Bifidobacterium spp. as probiotic strains has also been described in several commercial products. This article intends to present an up-to-date version regarding Iraqi probiotics currently used and health benefit obtained from their consumption. However, the employing of microbes to promote a good health is very common, while the current review paper provides a comprehensive view of some common probiotics in Iraq as all strains are very important to be familiar with them before any applying process. Ideally, microbial probiotics should have a beneficial effect and not cause any harm to the host. Therefore, all strains must have been studied comprehensively prior to use in humans or animals and thus are given GRAS (Generally Regarded as Safe) status.
Keywords:
Iraqi Probiotic, Gastrointestinal Tract, Production and Health

1. Introduction
Mucous membranes are the unique location where different microbial species are able to live and to express their effects. About 1014 bacteria of 200 species, 40 - 50 genera live on these membrane surfaces. The majority of microbial population on mucous membranes occurs in the distal section of the small intestine and in the proximal part of the colon [1] . Micro-flora of the digestive tract plays a crucial role in the physiological, immunological and anatomical development of the host. It stimulates some systems to respond rapidly to infection with pathogens and through microbial antagonism it inhibits the colonisation of the gut by unsafe bacteria [2] .
Probiotics have been well defined as live microbes when administered in adequate amounts. Confer a wellbeing benefit on the host [3] . The most widely used probiotics come from the genera Lactobacillus and Bifidobacterium while strains of E. coli complete the demand of probiotics. Others include nonpathogenic strains of E. coli, Enterococcus, Streptococcus thermophilus, Bacillus, and yeasts such as Saccharomyces boulardii [4] . Iraqi probiotics are defined as “live microbial supplements or components of bacteria and yeast” which have been shown to have beneficial effects on animal efficiency and health [5] . The Iraqi probiotic are useful bacteria like Lactobacillus acidophilus, Bifidobacterium and Bacillus subtilis and yeast such as Saccharomyces cerevisiae [30] .
The probiotic is a combination of beneficial microbes mixed with animal feeds to make benefits and healthy microbial balance in the intestine [6] , which leads to improve animal efficiencies especially in stressed animal which face a heat stress, feed on toxic or improper diets [7] [8] . However, the Iraqi probiotic in animal diets seemed to improve performance [9] , increase live weight gain [10] [11] , increase digestibility and enhance feed conversion ratio [12] .
In this review article, the up-to-date knowledge regards the impacts of Iraqi probiotics and this review focuses on collection of most scientific evidence concerning aspects of Iraqi probiotic including its components. The review will cover its effect on animal growth, production and health including the immune system, digestive tract, metabolic, intestinal organ and blood. However, this review summarizes the current knowledge on Iraqi probiotics and discusses both limits and acquired evidence to support their use in prevention and benefit.
2. The Intestinal Environment
The intestinal microbiota is an ecosystem formed by a variety of ecological niches, made of some bacterial species and a very large quantity of strains. The physiological activities of the microbiota are manifold and are just being unraveled. Based on the observations of the multiple roles of played by the microbiota in health and disease, the notion of adapting it with appropriate formulations, for instance, probiotics is being tested in several settings [13] . The mammalian intestine is colonized by 100 trillions of microbes (called “microbiota”) that are essential for health [14] [15] .
The transition from plants and soil to the animal gut has three areas of genomic adaptation [16] . The three major sections of the gastrointestinal tract are the stomach, the small intestine, and the large intestine. Every section has its own distinct microbiota [17] - [19] . The quantity and composition of microbial species differs along the digestive tract. Families, genera and phyla of the microbiota enriched in each particular niche are listed. Main bacterial phyla are represented in the mammalian gut microbiota; Streptococcus, lactobscilus, Bacteroides, Clostridium, Streptococci, Lactobacilli, Eubacterium, Peptococcus, Streptococcus Fusobacterium and Bifidobacterium. Most of the bacterial species found in the gastrointestinal tract Figure 1.
3. Probiotic as Good Microorganism
The tradition those times back to Metchnikoff includes both the use of a diet matrix fermented by a “useful” bacterium and a “concentrated” bacterial supplementation of diet. Both scenarios provide consumers with live bacteria that are able to pass the ileal and gastric environments and, then, to reproduce themselves in the large intestine. This idea, ecological in nature, supports the use of live bacteria able to settle among other live bacteria, i.e. the microbiota and exercise functions involving metabolic activity [13] .The studies supporting the effectiveness of live bacteria are numerous; conversely. There are few and conflicting results on the effects of the same strain administered in either viable or non-viable forms [13] . Probiotic and prebiotic foods have been con- sumed for centuries, either as natural components of food, or as fermented foods. Interest in intestinal microbiology and the dietary use of prebiotics and probiotics blossomed in the late 1800s and early 1900. The growing enthusiasm was motivated by Escherichs isolation of Escherichia coli in the late 1800s, as well as, active investigation on the benefits of feeding lactic acid bacteria and lactose near the turn of the 20th century [20] . Several related organizations have defined probiotics as live microorganisms that when administered at appropriate doses confer a health benefit to the host by regulation of the gastrointestinal flora [21] . Probiotics are defined as “live microorganisms which when administered in adequate amounts confer a health benefit on the host’’. Most
Figure 1. Spatial distribution and composition of the microbiota along the gastrointestinal tract [9] .
probiotics fall into the group of organisms’ known as lactic acid-producing bacteria and are normally consumed in the form of yogurt, fermented milks, cereal or other fermented foods [22] .
Live probiotic cultures are available in fermented dairy products and probiotic-fortified foods. Also, tablets, capsules, powders and sachets containing the bacteria in freeze-dried form are also available [23] . Probiotics beneficially affect the host animal by improving its intestinal balance and create gut conditions that suppress harmful microorganisms and favor beneficial ones [24] [25] . They have been shown to maintain health by reducing risk diseases, possibly through a reduction in proliferation of pathogenic species, maintaining microbiota balance in the gut enhancing immune system and increasing resistance to infection [25] [26] . Although, there are several dozen products imported to the market that claim to have probiotic activity representatives of only a handful of species dominate the market or have been used in multiple scientific trials but few of them local product. The Iraqi probiotic were purchased from the faculty of agriculture at Bagdad University. According to the manufacture information, each one gram of Iraqi probiotic contain of Lactobacillus acidophilus, Bacillus subtilis, Bifidobacterium and Saccharomyces cervisia. Iraqi probiotic produced by Prof. Dr. Saad Abd Al-Hus- sien Naji, this probiotic use in the animal feed and scientific application. However, Iraqi probiotic contain three useful bacteria and yeast with quantity, see Table 1.
4. Probiotic―Mechanism of Action
There are various proposed mechanisms that describe how probiotics work, and they vary depending on the strain of probiotic used. The effects of probiotics also depend on the dosage and route of administration. Thus the mechanisms of action cannot be extrapolated to all the probiotics. Proposed mechanisms include:
- Compete against the pathogenic microbes and to bind to the intestinal epithelial cells [27] . However, Iraqi probiotic change bacterial in intestine microflora [28] - [30] .
- Down-regulate pro-inflammatory cytokine production [31] , prevent apoptosis [32] and suppress T cell proliferation [33] , thus preventing various inflammatory conditions.
- Enhance phagocytosis [34] , increase activity of natural killer cells [35] , promote cell-mediated immunity [36] [37] , and stimulate various other nonspecific immune responses against pathogens. Though the Iraqi probiotic promote mediated immunity [28] .
- Probiotic Improve performance, increase live weight gain [5] , promote of Live weight gain and feed conversion ratio [38] , improving the qualities of productivity and the quality of the egg [39] [40] , stimulate
Table 1. Commercial Iraqi probiotic products use in market and scientific application.
- growth and improve performance [29] , increase in body weight gain [41] , improvement in averages of body weight, weight gain and feed conversion efficiency [42] , stimulate of total and daily growth [43] , Enhance growth [9] , improvement of carcass characteristics [44] and increase of Production Performance [45] .
- Enhance intestinal epithelial barrier function by increasing the production of mucin [46] , preventing injury of the epithelium from pathogens [47] and reducing cell permeability [48] . They may also enhance the mucosal barrier function by inducing expression of antimicrobial peptides like defensins [49] . However, Iraqi probiotic increase villi height, crypt depth, Percentage of villi height to crypt depth in duodenum, jejunum and Ileum [28] .
- Iraqi probiotic improve blood components, change physiological and biochemical blood parameters [38] [41] , improve the hormonal status and enhance the productivity [40] , and enhance the hematological blood [42] and changes in the hematological and biochemical blood parameters [50] - [52] .
- Enhance the production of serum IgA as well secretory IgA, which plays a crucial role in intestinal humoral immunity [53] [54] .
- Inhibit the growth of the pathogens by secreting another class of antimicrobial peptides like bacteriocins [55] and reuterin [56] . Some of the probiotics, particularly lactic acid bacteria, inhibit the growth of the pathogens by creating an acidic environment through the production of organic acids [57] . However, various mechanisms of action of probiotics can be seen in Diagram 1.
5. Probiotic and Performance
Probiotics have been used as growth promoters to replace the widely used antibiotic and synthetic chemical feed supplements. However, there are few published reports of good controlled field experiments and the comprehensive assessment of their value has not been attempted in the form of a large-scale co-ordinated field trial. The results of probiotic supplementation of diets have been variable but there have been reports of statistical effects on growth [58] . Protein supplementation, natural feed additives such as probiotic are very important materials that can improve, growth rate, daily weight gain, feed efficiency utilization and poductive performance [59] - [62] . The probiotic in animal diets seemed to improve performance [9] [63] - [65] and increased live weight gain [37] [66] and digestibility [12] and enhanced feed conversion ratio [67] . Hassan and Hassan, 2008 [9] [68] [69] reported significantly improvement in live weight gain and feed conversion ratio was associated with lamb fed diet supplemented with local Iraqi probiotic as compared with control diet. A locally prepared probiotic were actually tested and compared with foreign imported probiotic products in Iraq [70] [71] . Results its superior activity in improve poultry performance. Although Zubaidi [43] , found Iraqi probiotic increase of milk production of ewes, body weight, total and daily growth of lambs until weaning, this results give indicator of important of this treatment in increase of production total milk with ratio 26.65% also increasing body weight for lambs with ratio 17.49% finally increase of economic gain. Fuller [72] noticed that after using Lactobacillus probiotics in animals leads to competition for nutrients, improved gut-wall function and production of enzymes that support digestion and increase growth. El-Shaer [12] reported that probiotic microorganisms improve nutrients digestibility and increased the availability and absorbability of all nutrients in elementary tract such as rumen undegradable protein [9] and rumen degradable protein [68] . Similar observation was reported by Smirnov et al. [73] . The increased body weight gain in non-ruminant fed probiotic may be due to improvement in digestibility and availability of many nutrients such as proteins, fats and carbohydrates, as well as, some mineral elements and vitamins [74] . That is due to probiotics which improve digestion, absorption and availability of nutrition accompanying with beneficial change in gut micro flora with reduction in population of E. coli increasing lactate production
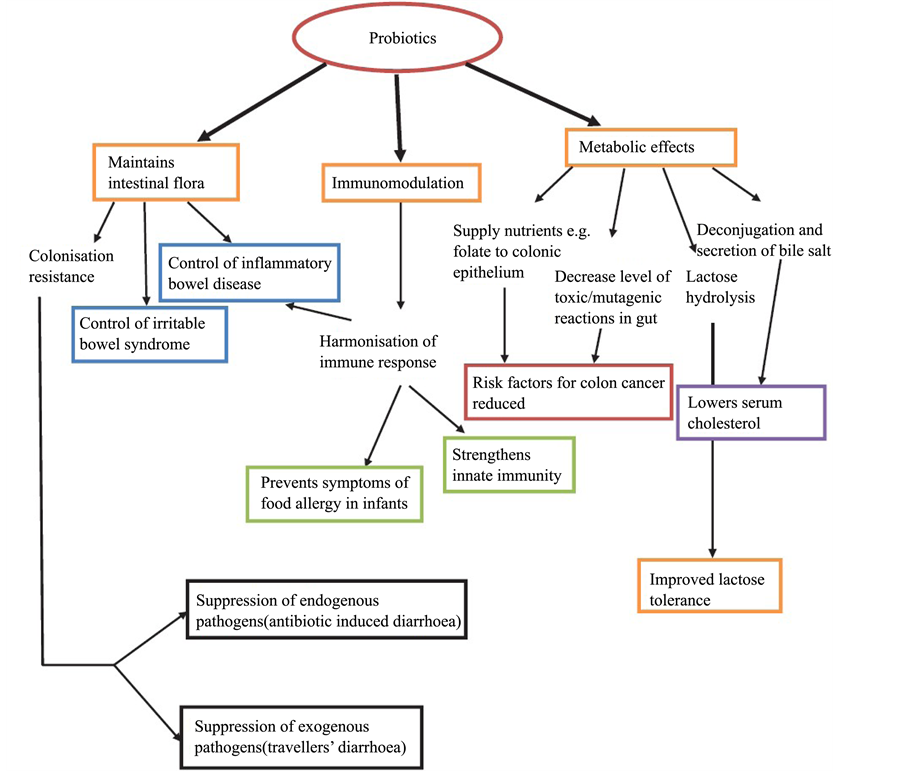
Diagram 1. The mechanism action of probiotic [75] .
with subsequent change in intestinal and increasing digestive enzymes [76] . Finally, Lactobacillus bacteria can increase the protein digestibility and availability of minerals for its host like Cu, Mn, Ca, Fe, P, etc. [77] .
6. Probiotic and Immunity Stimulation
The relationships established between microbiota and host organism can be either symbiotic or commensal. The bacteria of the microbiota, as mentioned are essential in enabling the absorption of nutrients for example, by allowing the hydrolysis of some otherwise non-digestible carbohydrates for the body and in preventing the intestinal colonization by and, thus, the entering the body of pathogenic microbes [13] . Therefore, it is vital that the immune system recognizes the components of the microbiota such establish a state of acceptance towards them. External microorganisms can penetrate the gut wall by translocation through the epithelial layer or through Peyer’s patches. Indigenous intestinal bacteria including lactobacilli are able to cross the intestinal mucous layer and they can live in the spleen or in other body part for many days where they stimulate phagocytic activity [78] . The thickness and physical state of the intestinal mucus layer [79] [80] and its response to orally consumed lactobacilli [81] [82] are essential in the immune response.
Iraqi probiotic improve Humeral and Cellular immunity and enhance immune protein [5] and improvement in the weight of immune organ [29] . Improve the bacterial ecology of the gastrointestinal tract and reduced the level of Enterobacteriaceae in the different parts of gastrointestinal tract and improved immunity parameters in broiler chicks [30] . the explanation of the effect probiotic on immune system could be founds in Al-Khafaji [83] , probiotic has a huge effect on immunity, as in human, under the intestinal epithelia as it is the first line of defense against pathogens) and the probiotic affect this layer to produced more immuno proteins. However, Cao et al. [84] presented that feeding diet supplemented with probiotic was significantly increased the immunity against Escherichia coli. On the other hand, the live microbes in the fermented feed may also act as probiotic to enhance the humoral immune response [85] . Probiotics can increase the immunogenicity of orally administered vaccines such as rotavirus [86] , polio [87] , cholera [88] and influenza [89] .
7. Probiotic and Metabolic Effects
Ischemic heart disease is a major reason of illness and death that is often associated with raised cholesterol levels and primary prevention with lipid lowering drugs or dietary modification can reduce the incidence and mortality of ischemic heart disease in healthy individuals [90] . A wide variety of probiotic products have been used in clinical trials of serum lipid modulation [91] - [94] . Some of the studies report positive effects on improving cardiovascular risk factors and there seems to be a trend towards decreasing risk factors. But more long-term evaluation would be required before firm conclusions can be drawn. So far, there is no enough evidence to support the use of probiotics to modify serum lipids and to prevent atherosclerosis. Al-Samarrai et al. [50] , observed that Iraqi probiotic Significantly decrease in the concentration of serum cholesterol in host. However, Saed [95] found high decrease in uric acid and cholesterol concentration blood plasma of birds fed on diet with Iraqi probiotic. Though Lambs fed on diets supplemented with Iraqi probiotic had significantly reduced total cholesterol (TCL), low-density lipoprotein (LDL) and Serum triglyceride (TG) as compared with those fed on diets without probiotic. High-density lipoprotein (HDL) concentrations were not affected by probiotics supplementation [51] . Probiotic causes a significant decrease in serum triglyceride levels [40] . The description of the effect probiotic on lipid profile could be founds in Santose et al. [96] , that found some of the probiotic Micro-floras as Bacillus subtilis decrease the activity of the acetyl-CoA carboxylase which is the limiting enzyme in fatty acid synthesis the building units for triglycerides. Although Desmet et al. [97] reported that the lactobacilli and bifidobacteria (the most common used probiotic microorganisms) had the ability to conjugate with bile acid enzymatically increasing their rate of excretion and lead to the reduction of serum cholesterol. Taranto et al. [98] who attributed the low levels of cholesterol in probiotic-treated animals to the inhibition of cholesterol synthesis by direct assimilation. Major functions of the gut microbiota include metabolic activities that result in salvage of energy and absorbable nutrients, trophic effects on the intestinal epithelium and protection of the host against invasion by harmful microbes [99] [100] .
8. Probiotic and Intestinal Microorganism
The gut microflora is an essential constituent in the intestine's defense barrier [101] . The initial compositional development of the gut microflora is considered a key determinant in the development of normal gut barrier functions [102] . Specific aberrancies in the intestinal microbiota may predispose the host to disease. Intestinal mucosal defense mechanisms acting in lumen and mucosa restrict colonization by pathogenic bacteria by interfering with the adherence of microorganisms to the mucosal surface. The normal gut microbiota can prevent the overgrowth of potential pathogens in the gastrointestinal tract [90] . Probiotics introduce new microbes to the gastrointestinal tract to enhance microbiota maintenance and modification, while most prebiotic components have been shown to enhance the growth of Bifidobacterium biota. Probiotics have been shown to amplify the gut mucosal barrier functions. Iraqi probiotic have great potential to beneficially affect the gut micro-flora and hence improve gut and reducing mortality ratio by inhibiting pathogenic microorganisms such as E.coli, Clostridium sp which are sensitive to the earlier culture of beneficial bacteria like lactobacillus [29] . However, Iraqi probiotic effected on total count bacteria, proteolytic bacteria and lipolytic bacteria of common carp [103] . Though, Iraqi probiotic reduction of logarithmic numbers of total aerobic bacteria and Coliformis bacteria and increasing logarithmic numbers of Lactobacillus bacteria in the internal environment of the contents of duodenum [28] . Probiotics beneficially affect the host animal by improving its intestinal balance and create gut conditions that suppress harmful microorganisms and favor beneficial ones [24] - [26] . They have been shown to maintain health by reducing risk diseases, possibly through a reduction in proliferation of pathogenic species, maintaining micro biota balance in the gut enhancing immune system and increasing resistance to infection [25] [26] . Finally, the mechanisms implied in the positive effects of Iraqi probiotic on the animals' growth and health, see Diagram 2.

Diagram 2. The mechanisms implied in the positive effects of Iraqi probiotic on the animals’ performance and health.
9. Conclusion
Probiotics have shown beneficial effects in a large number of health conditions and performances. Iraqi probiotics are live microbial feed supplements or components of bacteria and yeast. In animal diet, probiotics used as feed additives belong to different groups: lactic acid bacteria, yeasts and bacillus spores. They differ from one another in their properties, mode of action and source. The main activity of probiotics is the maintenance and re-formation of the equilibrium of the intestinal microflora which is achieved by many modes of action. The prerequisite for their probiotic action is reaching the gastrointestinal tract alive. The probiotics support the intestinal microflora by means of specific metabolic activities and/or stimulation of the host‘s immune system. There currently exists good evidence for the therapeutic use of Iraqi probiotic in performance productively, digestion and absorption, humeral and cellular immunity, metabolic effects, changing intestinal flora and preventing the emission of unpleasant odors. The influence on the microbiota composition, through probiotic consumption, may contribute significantly to host health and well-being. Iraqi probiotic associated with an overall improvement in functional intestinal disorders. Different mechanisms could influence the composition of the micro-or- ganisms that colonized the digestive tract. Further research should be done in order to specify the safety of various strains used for probiotic preparations.
Acknowledgements
The author gratefully acknowledges Prof. Dr. Saad Abd Al-Hussien Naji, Asst. prof. Dr. Saman A. Rashid and Dr. Nasreen M. Abdulrahman for providing sufficient researches.
Cite this paper
Hozan JalilHamasalim, (2015) The Impact of Some Widely Probiotic (Iraqi Probiotic) on Health and Performance. Journal of Biosciences and Medicines,03,25-36. doi: 10.4236/jbm.2015.38003
References
- 1. Savage, D.C., Ogra, P.L., Mestecky, J., Lamm, M.E., Strober, W. and McGhee, J.R. (1998) Mucosal Microbiota. In: Bienestock, J., Ed., Mucosal Immunology, Academic Press, San Diego, 216-238.
- 2. Cebra, J.J., Jiang, H.Q., Sterzl, J. and Tlaskalova-Hogenova, H. (1999) The Role of Mucosal Microbiota in the Development and Maintenance of the Mucosal Immune System. In: Ogra, P.L., et al., Eds., Mucosal Immunology, Academic Press, New York, 267-280.
- 3. FAO/WHO (2002) Guidelines for the Evaluation of Probiotics in Food. Food and Agriculture Organization of the United Nations/World Health Organization, London, Ontario.
www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf - 4. Dunne, C., O’Mahony, L. and Murphy, L. (2001) In Vitro Selection Criteria for Probiotic Bacteria of Human Origin: Correlation with in Vivo Findings. The American Journal of Clinical Nutrition, 73, 386S-392S.
- 5. Hamasalim, H.J. (2009) The Effect of Different Levels of Feeding on Karadi Lambs Response to Local Iraqi Probiotics. Master Thesis, College of Agriculture and Animal Production, Sulaimani University, Sulaimani.
- 6. Nunes, C.S. (1994) Microbial Probiotics and Their Utilization in Husbandry. Revista portuguesade Ciencias veterinarias, 89, 166-174.
- 7. Yoruk, M.A., Gul, M., Hayirli, A. and Macit, M. (2004) The Effects of Supplementation of Humate and Probiotic on Egg Production and Quality Parameters during the Late Laying Period in Hens. Poultry Science, 83, 84-88.
http://dx.doi.org/10.1093/ps/83.1.84 - 8. Zinedine, A., Faid, M. and Benlemlith M. (2005) In Vitro Reduction of Aflatoxin B1 by Strains of Lactic Acid Bacteria Isolated from Sourdough Bread. International Journal of Agriculture and Biology, 7, 67-70.
- 9. Hassan, S.A. and Hassan, K.M. (2009) The Effect of Supplementation of Medicinal Plants and Probiotic on Growth Rate and Some Blood Parameters of Karadi Lambs. Egyptian Journal of Nutrition and Feeds, 12, 53-63.
- 10. Hassan, S.A., Tawffek, J. A. and El-Saady, M.A. (2009) Effect Substitution Gradually Percentages of Reed Silage with Alfalfa Hay Fed with Probiotic to Awassi Lamb.2-On Carcass Characteristics. The Iraqi Journal of Agricultural Sciences, 40, 138-147.
- 11. Hassan, S.A., Tawffek, J.A. and El-Saady, M.A. (2009) Effect Substitution Gradually Percentages of Reed Silage with Alfalfa Hay Fed with Probiotic to Awassi Lamb. 3-On Some Blood Parameters. The Iraqi Journal of Agricultural Science, 40, 158-173.
- 12. El-Shaer, E.K.H. (2003) Nutritional Studies in Ruminants (Effect of Yeast Culture Supplementation and Concentrate: Roughage Ratio on Performance of Growing Lambs). Ph.D. Thesis, Faculty of Agriculture, Mansoura University, Mansoura.
- 13. Aureli, P., Capurso, L., Castellazzi, A.M., Clerici, M., Giovannini, M., Morelli, L., Poli, A., Pregliasco, F., Salvini, F. and Zuccotti, G.V. (2011) Probiotics and Health: An Evidence-Based Review. Pharmacological Research, 63, 366- 376.
- 14. Hooper, L.V. and Macpherson, A.J. (2010) Immune Adaptations That Maintain Homeostasis with the Intestinal Microbiota. Nature Reviews Immunology, 10, 159-169.
http://dx.doi.org/10.1038/nri2710 - 15. Kamada, N., Seo, S.-U., Chen, G.Y. and Núñez, G. (2013) Role of the Gut Microbiota in Immunity and Inflammatory Disease. Nature Reviews Immunology, 13, 321-335.
http://dx.doi.org/10.1038/nri3430 - 16. Lebeer, S., Vanderleyden, J. and De Keersmaecker, S.C. (2008) Genes and Molecules of Lactobacilli Supporting Probiotic Action. Microbiology and Molecular Biology Reviews, 72, 728-764.
http://dx.doi.org/10.1128/MMBR.00017-08 - 17. Dethlefsen, L., Eckburg, P.B., Bik, E.M. and Relman, D.A. (2006) Assembly of the Human Intestinal Microbiota. Trends in Ecology & Evolution, 21, 517-523.
http://dx.doi.org/10.1016/j.tree.2006.06.013 - 18. Savage, D. (1977) Microbiology of the Gastrointestinal Tract. Annual Review of Microbiology, 31, 107-133.
http://dx.doi.org/10.1146/annurev.mi.31.100177.000543 - 19. Simon, G.L. and Gorbach, S.L. (1984) Intestinal Flora in Health and Disease. Gastroenterology, 86, 174-193.
- 20. Rettger, L.F. and Cheplin, H.A. (1921) A Treatise on the Transformation of the Intestinal Flora, with Special Reference to the Implantation of Bacillus acidophlus. Yale University Press, New Haven.
- 21. Rask, C., Adlerberth, I., Berggren, A., Ahrén, I.L. and Wold, A.E. (2013) Differential Effect on Cell-Mediated Immunity in Human Volunteers after Intake of Different Lactobacilli. Clinical & Experimental Immunology, 172, 321-332.
http://dx.doi.org/10.1111/cei.12055 - 22. Naidu, K.S.B., Adam, J.K. and Govender, P. (2012) The Use of Probiotics and Safety Concerns: A Review. African Journal of Microbiology Research, 6, 6871-6877.
- 23. Raoult, D. (2009) No Link between Probiotics and Obesity? Author Reply. Nature Reviews Microbiology, 7, 901.
http://dx.doi.org/10.1038/nrmicro2209-c3 - 24. Line, E.J., Bailey, S.J., Cox, N.A., Stern, N.J. and Tompkins, T. (1998) Effect of Yeast-Supplemented Feed on Salmonella and Campylobacter Populations in Broilers. Poultry Science, 77, 405-410.
http://dx.doi.org/10.1093/ps/77.3.405 - 25. Mead, G.C. (2000) Prospects for Competitive Exclusion Treatment to Control Salmonellas and Other Food Borne Pathogens in Poultry. Veterinary Journal, 159, 111-123.
http://dx.doi.org/10.1053/tvjl.1999.0423 - 26. Mountzouris, K.C., Tsirtisikos, P. and Kalamara, E. (2007) Evaluation of the Efficacy of a Probiotic Containing Lactobacillus, Bifidobacterium, Enterococcus and Pediococcus Strains in Promotion Broiler Performance and Modulation Cecal Micro Flora Composition and Metabolic Actives. Poultry Science, 86, 309-317.
http://dx.doi.org/10.1093/ps/86.2.309 - 27. Bernet, M.F., Brassart, D., Neeser, J.R. and Servin, A.L. (1994) Lactobacillus acidophilus LA 1 Binds to Cultured Human Intestinal Cell Lines and Inhibits Cell Attachment and Cell Invasion by Enterovirulent Bacteria. Gut, 35, 483- 489.
http://dx.doi.org/10.1136/gut.35.4.483 - 28. Bedy, I.F. (2014) The Effect of Diet Fermentation with Locally Probiotics on Some Productive, Microbiological, Immunological and Histological Properties of Broiler Chicks. Doctoral Thesis, College of Agriculture at the University of Baghdad, Baghdad.
- 29. Mahmmod, Z.A., Abdulrazaq, H.S., Salem, A.S. and Sideq, R.M. (2014) Effects of Supplementation Probiotic and Dried Yogurt Powder on Growth Performance, Carcass Characteristics, Intestinal Micro Flora and Immunity of Broiler Chickens. Zanco Journal of Pure and Applied Sciences, 26, 35-42.
- 30. Naji, S.A.H., Al-Zamil, I.F.B. and Al-Gharawi, J.K.M. (2015) The Effect of Feed Wetting and Fermentation on the Intestinal Flora, Humoral and Cellular Immunity of Broiler Chicks. College of Agriculture, Al-Qadisiya University-Iraq, Diwaniya.
- 31. Lammers, K.M., Vergopoulos, A. and Babel, N. (2005) Probiotic Therapy in the Prevention of Pouchitis Onset: Decreased Interleukin-1β, Interleukin-8, and Interferon-γ Gene Expression. Inflammatory Bowel Diseases, 11, 447-454.
http://dx.doi.org/10.1097/01.mpa.0000160302.40931.7b - 32. Lin, P.W., Nasr, T.R. and Berardinelli, A.J. (2008) The Probiotic Lactobacillus GG May Augment Intestinal Host Defense by Regulating Apoptosis and Promoting Cytoprotective Responses in the Developing Murine Gut. Pediatric Research, 64, 511-516.
http://dx.doi.org/10.1203/PDR.0b013e3181827c0f - 33. Pessi, T., Sutas, Y. and Saxelin, M. (1999) Antiproliferative Effects of Homogenates Derived from Five Strains of Candidate Probiotic Bacteria. Applied and Environmental Microbiology, 65, 4725-4728.
- 34. Schiffrin, E.J., Rochat, F., Link-Amster, H., Aeschlimann, J.M. and Donnet-Hughes, A. (1995) Immunomodulation of Human Blood Cells Following the Ingestion of Lactic Acid Bacteria. Journal of Dairy Science, 78, 491-497.
http://dx.doi.org/10.3168/jds.S0022-0302(95)76659-0 - 35. Gill, H.S., Rutherfurd, K.J. and Cross, M.L. (2001) Dietary Probiotic Supplementation Enhances Natural Killer Cell Activity in the Elderly: An Investigation of Age-Related Immunological Changes. Journal of Clinical Immunology, 21, 264-271.
http://dx.doi.org/10.1023/A:1010979225018 - 36. de Waard, R., Claassen, E. and Bokken, G.C. (2003) Enhanced Immunological Memory Responses to Listeria monocytogenes in Rodents, as Measured by Delayed-Type Hypersensitivity (DTH), Adoptive Transfer of DTH, and Protective Immunity, Following Lactobacillus casei Shirota Ingestion. Clinical and Diagnostic Laboratory Immunology, 10, 59-65.
- 37. Galyean, M.L., Nunnery, G.A., Defoor, P.J., Salyer, G.B. and Parson, C.H. (2000) Effect of Live Culture of Lactobacillus acidophilus (Strain 145 and 51) and Propionibacterium frendenreichii PF-24 on Performance and Carcass Characteristics of Finishing Beef Steers. Burnett Center Progress Report No. 8.
- 38. Hassan, K.M. (2009) Effect of Some Feed Additives on Performance and Some Blood Parameters of Karadi Lambs. Ph. D. Thesis, Department of Animal Production, Collage of Agriculture, University of Sulaimani, Sulaymaniyah.
- 39. Al-Tememy, A.T.D. (2013) Effect of Adding Two Levels of Iraqi Soluble Probiotic on Performance and Egg Quality of Japanese Quail. Diyala Journal of Agricultural Sciences, 5, 81-91.
- 40. Sultan, K.H. and Abdul-Rahman, S.Y. (2011) Effect of Probiotic on Some Physiological Parameters in Broiler Breeders. International Journal of Poultry Science, 10, 626-628.
http://dx.doi.org/10.3923/ijps.2011.626.628 - 41. Abdullah, S.T. (2014) Effect of Probiotics Addition into Diet and Drinking Water in Collared Dove (Streptopelia decaocto) on Certain Physiological and Biochemical Parameters. Iraqi Journal of Veterinary Science, 28, 127-131.
- 42. Al-Bdeery, A.G.M. (2013) Effect of the Probiotics, Vitamins A and E on Growth Performance and Some Blood Parameters in Broiler Chickens. Kufa Journal for Veterinary Medical Sciences, 4, 34-42.
- 43. Zubaidi, K.H.A. (2010) Effect of Iraqi Probiotic to Awassi Ewes Diet on Milk Production & Growth until Weaning. Journal of the University of Karbala Scientific, 8, 277-282.
- 44. Al-Ruubii, A.M.S, Hassan, S.A. and Al-Qabani, A.A.M. (2008) Effect of Iraqi Probiotic as an Additives Feed on Carcass Characteristics and Composition of Awassi Lambs. Journal of Kerbala University, 6, 4.
- 45. Taha, M.W. and Omar, B.T. (2013) Effect of Adding Different Levels of Soluble Iraqi Probiotic on Production Performance of Broiler Chickens. Journal of Tikrit University of Agricultural Sciences, 13, 127-135.
- 46. Caballero-Franco, C., Keller, K., De Simone, C. and Chadee, K. (2007) The VSL#3 Probiotic Formula Induces Mucin Gene Expression and Secretion in Colonic Epithelial Cells. AJP: Gastrointestinal and Liver Physiology, 292, G315- G322.
http://dx.doi.org/10.1152/ajpgi.00265.2006 - 47. Johnson-Henry, K.C., Donato, K.A. and Shen-Tu, G. (2008) Lactobacillus rhamnosus Strain GG Prevents Enterohemorrhagic Escherichia coli O157:H7-Induced Changes in Epithelial Barrier Function. Infection and Immunity, 76, 1340-1348.
http://dx.doi.org/10.1128/IAI.00778-07 - 48. Ewaschuk, J.B., Diaz, H. and Meddings, L. (2008) Secreted Bioactive Factors from Bifidobacterium infantis Enhance Epithelial Cell Barrier Function. AJP: Gastrointestinal and Liver Physiology, 295, G1025-G1034.
http://dx.doi.org/10.1152/ajpgi.90227.2008 - 49. Wehkamp, J., Harder, J. and Wehkamp, K. (2004) NF-κB- and AP-1-Mediated Induction of Human Beta Defensin-2 in Intestinal Epithelial Cells by Escherichia coli Nissle 1917: A Novel Effect of a Probiotic Bacterium. Infection and Immunity, 72, 5750-5758.
http://dx.doi.org/10.1128/IAI.72.10.5750-5758.2004 - 50. Al-Samarrai, W.H., Ahmad, A.R.A.K., Al-Mashhadani, N.I., Abbas, S.M. and Fangan, K. (2014) Effect of Addition of Iraqi Probiotic on Blood Parameters in Awassi Lambs Feeding Barley Straw. Global Journal of Biochemistry and Biotechnology, 3, 84-90.
- 51. Hamasalim, H.J. and Abdulla, S.A. (2011) The Effect of Probiotic Supplementation with Levels of Feeding on Hematological and Biochemical Blood of Karadi Lambs. Proceedings of the 5th Scientific Conference of College of Agriculture, Tikrit, 26-27 April 2011, 200-210.
- 52. Hassan, S.A. and Hassan, K.M. (2009) Effects of Medicinal Plants and Probiotic Supplementation on Some Nutrients and Blood Parameters of Karadi Lambs. Euphrates Journal of Agriculture Science, 1, 1-13.
- 53. Link-Amster, H., Rochat, F. and Saudan, K.Y. (1994) Modulation of a Specific Humoral Immune Response and Changes in Intestinal Flora Mediated through Fermented Milk Intake. FEMS Immunology and Medical Microbiology, 10, 55-63.
http://dx.doi.org/10.1111/j.1574-695X.1994.tb00011.x - 54. Park, J.H., Um, J.I. and Lee, B.J. (2002) Encapsulated Bifidobacterium bifidum Potentiates Intestinal IgA Production. Cellular Immunology, 219, 22-27.
http://dx.doi.org/10.1016/S0008-8749(02)00579-8 - 55. De Vuyst, L. and Leroy, F. (2007) Bacteriocins from Lactic Acid Bacteria: Production, Purification, and Food Applications. Journal of Molecular Microbiology and Biotechnology, 13, 194-199.
http://dx.doi.org/10.1159/000104752 - 56. Spinler, J.K., Taweechotipatr, M. and Rognerud, C.L. (2008) Human-Derived Probiotic Lactobacillus reuteri Demonstrate Antimicrobial Activities Targeting Diverse Enteric Bacterial Pathogens. Anaerobe, 14, 166-171.
http://dx.doi.org/10.1016/j.anaerobe.2008.02.001 - 57. Ogawa, M., Shimizu, K. and Nomoto, K. (2001) Inhibition of in Vitro Growth of Shiga Toxin-Producing Escherichia coli O157:H7 by Probiotic Lactobacillus Strains Due to Production of Lactic Acid. International Journal of Food Microbiology, 68, 135-140.
http://dx.doi.org/10.1016/S0168-1605(01)00465-2 - 58. Dilworth, B.C. and Day, E.J. (1978) Lactobacillus Cultures in Brooder Diets. Poultry Science, 57, 1101.
- 59. AI-Jassim, R.A.M., AL-Ani, A.N., Hassan, S.A., Dana, T.K. and AL-Jerien, L.J. (1991) Effects of Dietary Supplementation with Rumen Undegradable Protein on Carcass Characteristics of Iraqi Awassi Lambs and Desert Goats. Small Ruminant Research, 4, 269-275.
http://dx.doi.org/10.1016/0921-4488(91)90150-O - 60. Hassan, S.A. (2005) Effect of Barley Straw Treated with Liquid Diet on Its Daily Intake, Digestion Coefficient and Live Weight Gain of Awassi Lambs. Iraqi Journal of Agricultural Science, 36, 133-138.
- 61. Hassan, S.A., Al-Jassim, R.A.M., AL-Ani, A.N. and Abdullah, N.S. (1991) Effects of Dietary Supplement of Rumen Undegradable Protein upon Carcass Composition of Fat-Tail Awassi Sheep. Small Ruminant Research, 5, 65-74.
http://dx.doi.org/10.1016/0921-4488(91)90031-K - 62. Hassan, S.A., Ahmed, A.A. and Alwan, M.F. (2008) Effect of Iraqi Probiotic Supplementation on Growth Rate, Blood Parameters and Carcass Characteristics of Awassi Lambs. Egyptian Journal of Nutrition and Feeds. (Accepted)
- 63. Abedo, A.A., El-Ashry, M.A., El-Babawi, A.Y., Helal, F.I.S. and Fadel, M. (2005) Effect of Feeding Biologically Treated Sugar Beet Pulp on Growth Performance of Sheep. Egyptian Journal of Nutrition and Feeds, 8, 579-590.
- 64. Ali, M.A. (2005) Effect of Probiotic Addition on Growth Performance of Growing Lambs Fed Different Roughages. Egyptian Journal of Nutrition and Feeds, 8, 567-578.
- 65. Hassan, S.A. (2008) Effect of Some Medicinal Plants Supplementation on Daily Intake, Live Weight Gain and Carcass Characteristics of Awassi Lambs. Egyptian Journal of Nutrition and Feeds. (Accepted)
- 66. Orr, C., Ware, D.R., Manfredi, E.T. and Hutheson, D.P. (1988) The Effect of Continuous Feeding of Lactobacillius acidophilus Strain BT1386 on Gain and Feed Efficiency of Feeder Calves. Journal of Animal Science, 66, 460-461.
- 67. Wysong, D.L. (2003) Retionale for Probiotic Supplementation. www.Wysong.net
- 68. Hassan, S.A. and Hassan, K.M. (2008) Response of Karadi Lambs to the Rosemary Officinal Supplementation Fed with Either Alkali Treated or Untreated Barley Straw Basal Diets. Egyptian Journal of Nutrition and Feeds. (In Press)
- 69. Hassan, S.A. and Hassan, K.M. (2008) Effect of Graded Levels of Rumen Degradable Nitrogen and Nigella Sativa on Daily Intake, Live Weight Gain, Feed Conversion Ratio and Some Blood Parameters of Karadi Lambs. Proceedings of the 7th Scientific Conference for Agriculture Research, Bagdad, 24-26 October 2008, 168-177.
- 70. Abbas, M.R. (2005) Effect of Adding Iraqi Probiotic to the Ration on the Productive Performance of Caged Laying Hens. The Iraqi Journal of Agricultural Sciences, 36, 97-104.
- 71. Al-Khalidi, R.A. (2005) Comparative Study of Imported Probiotic (Biomin) and Locally Probiotic (The Iraqi Probiotic) in Production, Performance and Intestinal Microbial Balance of Broiler. Master’s Thesis, College of Veterinary Medicine, University of Baghdad, Baghdad.
- 72. Fuller, R. (1989) Probiotics in Man and Animals. Journal of Applied Bacteriology, 66, 365-378.
http://dx.doi.org/10.1111/j.1365-2672.1989.tb05105.x - 73. Smirnov, A., Perez, R., Amit-Romach, E., Sklan, D. and Uni, Z. (2005) Mucin Dynamics and Microbial Populations in Chickens Small Intestine Are Changed by Dietary Prebiotic and Antibiotic Growth Promoter Supplementation. Journal of Nutrition, 135, 187-192.
- 74. Burkholder, K.M., Applegate, T.J. and Patterson, J.A. (2005) Performance and Intestinal Characteristics of Broilers Fedsalinomycin, Fructooligosaccharides, Probiotics and Synbiotics. Proceedings of the Southern Poultry Science Association Meeting, Atlanta, 20-21 January 2005.
- 75. Vandana, K.A. (2015) Probiotics: Nature’s Medicine. International Journal of Nutrition, Pharmacology, Neurological Diseases, 3, 219-228.
http://www.ijnpnd.com - 76. Leeson, S. and summers, J.D. (2005) Commercial Poultry Nutrition. Third Edition, Nottingham University Press, Nottingham.
- 77. Kalavathy, R., Abdullah, N., Jalaludin, S., Wong, C.M.V.L. and Ho, Y.W. (2008) Effect of Lactobacillus Cultures and Oxytetracycline on the Growth Performance and Serum Lipids of Chickens. International Journal of Poultry Science, 7, 385-389.
http://dx.doi.org/10.3923/ijps.2008.385.389 - 78. Deitch, E., Specian, E., Steffen, E. and Berg, R. (1990) Translocation of Lactobacillus murinus from the Gastrointestinal Tract. Current Microbiology, 20, 177-184.
http://dx.doi.org/10.1007/BF02091994 - 79. Atuma, C., Strugala, V., Allen, A. and Holm, L. (2001) The Adherent Gastrointestinal Mucus Gel Layer: Thickness and Physical State in Vivo. American Journal of Physiology Gastrointestinal Liver Physiology, 280, G922-G929.
- 80. Johansson, M.E., Phillipson, M., Petersson, J., Velcich, A., Holm, L. and Hansson, G.C. (2008) The Inner of the Two Muc2 Mucin-Dependent Mucus Layers in Colon Is Devoid of Bacteria. Proceedings of the National Academy of Sciences of the United States of America, 105, 15064-15069.
http://dx.doi.org/10.1073/pnas.0803124105 - 81. Troost, F.J., van Baarlen, P., Lindsey, P., Kodde, A., de Vos, W.M., Kleerebezem, M. and Brummer, R.J. (2008) Identification of the Transcriptional Response of Human Intestinal Mucosa to Lactobacillus plantarum WCFS1 in Vivo. BMC Genomics, 9, 374.
http://dx.doi.org/10.1186/1471-2164-9-374 - 82. Van Baarlen, P., Troost, F.J., van Hemert, S., van der Meer, C., de Vos, W.M., de Groot, P.J., Hooiveld, G.J., Brummer, R.J. and Kleerebezem, M. (2009) Differential NF-κB Pathways Induction by Lactobacillus plantarum in the Duodenum of Healthy Humans Correlating with Immune Tolerance. Proceedings of the National Academy of Sciences of the United States of America, 106, 2371-2376.
http://dx.doi.org/10.1073/pnas.0809919106 - 83. Al-Khafaji, Z.M. (2008) Probiotics (For Life). Published and Printed by House and Documentation Baghdad. Library of Control No. 84, 3-11.
- 84. Cao, G.T., Zing, X.F., Chen, A.G. and Yang C.M. (2013) Effects of a Probiotic, Enterococcus faecium, on Growth Performance, Intestinal Morphology, Immune Response, and Cecal Microflora in Broiler Chickens Challenged with Escherichia coli K88. Poultry Science, 92, 2949-2955.
http://dx.doi.org/10.3382/ps.2013-03366 - 85. Apata, D.F. (2011) Effect of Terminalia catappa Fruit Meal Fermented by Aspergillus niger as Replacement of Maize on Growth Performance, Nutrient Digestibility, and Serum Biochemical Profile of Broiler Chickens. Biotechnology Research International, 2011, 1-6.
http://dx.doi.org/10.4061/2011/907546 - 86. Isolauri, E., Joensuu, J., Suomalainen, H., Luomala, M. and Vesikari, T. (1995) Improved Immunogenicity of Oral D x RRV Reassortant Rotavirus Vaccine by Lactobacillus casei GG. Vaccine, 13, 310-312.
http://dx.doi.org/10.1016/0264-410X(95)93319-5 - 87. de Vrese, M., Rautenberg, P., Laue, C., Koopmans, M., Herremans, T. and Schrezenmeir, J. (2005) Probiotic Bacteria Stimulate Virus-Specific Neutralizing Antibodies Following a Booster Polio Vaccination. European Journal of Nutrition, 44, 406-413.
http://dx.doi.org/10.1007/s00394-004-0541-8 - 88. Paineau, D., Carcano, D., Leyer, G., Darquy, S., Alyanakian, M.A., Simoneau, G., Bergmann, J.F., Brassart, D., Bornet, F. and Ouwehand, A.C. (2008) Effects of Seven Potential Probiotic Strains on Specific Immune Responses in Healthy Adults: A Double-Blind, Randomized, Controlled Trial. FEMS Immunology and Medical Microbiology, 53, 107-113.
- 89. Kobayashi, N., Saito, T., Uematsu, T., Kishi, K., Toba, M., Kohda, N. and Suzuki, T. (2011) Oral Administration of Heat-Killed Lactobacillus pentosus Strain b240 Augments Protection against Influenza Virus Infection in Mice. International Immunopharmacology, 11, 199-203.
http://dx.doi.org/10.1016/j.intimp.2010.11.019 - 90. Harish, K. and Varghese, T. (2006) Probiotics in Humans—Evidence Based Review. Calicut Medical Journal, 4, e3.
- 91. Anderson, J.W. and Gilliland, S.E. (1999) Effect of Fermented Milk (Yogurt) Containing Lactobacillus acidophilus L1 on Serum Cholesterol in Hypercholesterolemic Humans. Journal of the American College of Nutrition, 18, 43-50.
http://dx.doi.org/10.1080/07315724.1999.10718826 - 92. Bukowska, H., Pieczul-Mroz, J., Jastrzebska, M., Chelstowski, K. and Naruszewicz, M. (1998) Decrease in Fibrinogen and LDL Cholesterol Levels upon Supplementation of Diet with Lactobacillus plantarum in Subjects with Moderately Elevated Cholesterol. Atherosclerosis, 137, 437-438.
- 93. Naruszewicz, M., Johansson, M.L., Zapolska-Downar, D. and Bukowska, H. (2002) Effect of Lactobacillus plantarum 299v on Cardiovascular Disease Risk Factors in Smokers. The American Journal of Clinical Nutrition, 76, 1249-1255.
- 94. Schaafsma, G., Meuling, W.J., van Dokkum, W. and Bouley, C. (1998) Effects of a Milk Product, Fermented by Lactobacillus acidophilus and with Fructo-Oligosaccharides Added, on Blood Lipids in Male Volunteers. The European Journal of Clinical Nutrition, 52, 436-440.
http://dx.doi.org/10.1038/sj.ejcn.1600583 - 95. Saed, S.H.M. (2005) Effect of Local Probiotic, Imported Yeast Baker and Multi Enzymes Supplementation in the Performance and Some Blood Biochemistry of the Broiler. Master’s Thesis, College of Agriculture, Sulaimani University, Sulaimani.
- 96. Santose, U., Tanaka, K. and Othani, S. (1995) Effect of Dried Bacillus subtilis Culture on Growth, Body Composition and Hepatic Lipogenic Enzyme Activity in Fimale Broiler Chicks. British Journal of Nutrition, 74, 523-529.
http://dx.doi.org/10.1079/BJN19950155 - 97. DeSmet, I., Van Hoorde, L., De Saeyer, N., Woestyne, M.V. and Verstraete, W. (1994) In Vitro Study of Bile Salt Hydrolase (BSH) Activity of BSH Isogenic Lactobacillus plantarum 80 Strains and Estimation of Cholesterol Lowering through Enhanced BSH Activity. Microbial Ecology in Health and Disease, 7, 315-329.
http://dx.doi.org/10.3109/08910609409141371 - 98. Taranto, M.P., Medici, M., Perdigon, G., Ruiz Holgado, A.P. and Valdez, G.F. (1998) Evidence for Hypocholesterolemic Effect of Lactobacillus reuteri in Hypercholesterolemic Mice. Journal of Dairy Science, 81, 2336-2340.
http://dx.doi.org/10.3168/jds.S0022-0302(98)70123-7 - 99. Backhed, F., Ding, H., Wang, T., Hooper, L.V., Koh, G.Y. and Nagy, A. (2004) The Gut Microbiota as an Environmental Factor That Regulates Fat Storage. Proceedings of the National Academy of Sciences of the United States of America, 101, 15718-15723.
http://dx.doi.org/10.1073/pnas.0407076101 - 100. Guarner, F. and Malagelada, J.R. (2003) Gut Flora in Health and Disease. The Lancet, 361, 512-519.
http://dx.doi.org/10.1016/S0140-6736(03)12489-0 - 101. Isolauri, E., Sutas, Y., Kankaanpaa, P., Arvilommi, H. and Salminen, S. (2001) Probiotics: Effects on Immunity. The American Journal of Clinical Nutrition, 73, 444S-450S.
- 102. Hooper, L.V., Wong, M.H., Thelin, A., Hansson, L., Falk, P.G. and Gordon J.I. (2001) Molecular Analysis of Commensal Host-Microbial Relationships in the Intestine. Science, 291, 881-884.
http://dx.doi.org/10.1126/science.291.5505.881 - 103. Abdulrahman, N.M. and Al shawi, S.A. (2014) Locally Produces Probiotic Their Effect on Total, Proteolytic Bacteria and Lipolytic Bacteria of Common Carp Fed Tow Level of Protein. Journal of Animal and Veterinary Advances, 13, 660-663.



