Open Journal of Anesthesiology
Vol.3 No.6(2013), Article ID:35236,3 pages DOI:10.4236/ojanes.2013.36066
Case Series of Methylene Blue Injections for the Treatment of Zygapophysial and Sacroiliac Joint Pain: Results of 5 Cases
![]()
University of Texas Health Science Center at San Antonio, San Antonio, USA.
Email: jparekh81@gmail.com
Copyright © 2013 Jay Parekh et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received May 17th, 2013; revised June 17th, 2013; accepted July 10th, 2013
Keywords: Radio Frequency Ablation; Facet; Zygapophysial; Sacroiliac; Methylene Blue; Pain; Interventional
ABSTRACT
Introduction: Shealy is credited with the use of fluoroscopy guided radiofrequency ablation (RFA) for denervation of the lumbar zygapophysial (L-Z) joint in the mid-1970’s. Peng and colleagues described the use of methylene blue (MB) injection for discogenic pain; MB may work through direct neurotoxicity or inhibition of nitric oxide synthesis. We present a retrospective case series of five patients with L-Z or SI joint pain where MB used as an alternative to RFA. Materials and Methods: This is a retrospective case series of five patients who received MB for the chemical neurolysis of either medial or/and lateral branch’s for zygapophysial or SI joint pain. All of the MB blocks were done under fluoroscopic guidance and performed in the same manner as diagnostic medial and lateral branch blocks but with care to consider the medication as neurotoxic in relation to spinal nerves. On average 1cc of 0.05% final concentration MB was injected per nerve or sacral foraminal level. Results: Four of the five patients had significant lasting pain relief. Discussion: Chemical neurolysis has potential to achieve technical success in these refractory cases of spine pain possibly due to spread over a broader area; for this same reason the procedure should be performed carefully under fluoroscopic guidance.
1. Introduction
Today lumbar, thoracic, cervical zygapophysial (L-Z) and sacroiliac (SI) joint pain are routinely treated with Radiofrequency Ablation (RFA) of the medial branch and lateral branch nerves, respectively [1-3]. Success rates for L-Z RFA are reported anywhere from 27% to 87% [1]. Unsuccessful lumbosacral RFA despite positive response to controlled diagnostic blocks, and diminishing efficacy of repeat RFA treatments, are possibly due to aberrant or additional innervation of the joints [1].
Peng and colleagues describe the use of intradiscal methylene blue (MB) for the treatment of chronic discogenic pain [4]. While MB inhibits nitric oxide synthesis, it is also neurotoxic and possibly destroys nerve fibers such as those innervating annual fissures or intradermal nerve endings [4-6]. Methylene Blue may be an option in the neurolysis of painful L-Z and SI joints refractory to RFA.
2. Materials and Methods
This is a retrospective case series of five [5] patients who received MB for the chemical neurolysis of medial and/ or lateral branch nerves for L-Z and/or SI joint pain as an alternative to RFA. All of the procedures were done in an outpatient academic pain clinic by multiple providers. All patients had two sessions of controlled diagnostic medial and/or lateral branch blocks prior to neurolysis. Diagnostic blocks were considered indicative of a strong L-Z or SI joint pain syndrome if diagnostic blocks resulted in greater than 50% pain relief on a Numeric Rating Scale (NRS, 0-10).
Neurolysis: We injected 1ml of a 1:1:1 mixture of 0.5% bupivicaine, contrast, and 1% MB over the lumbar medial branch nerves (Figure 1) and/or sacral lateral branch nerves under fluoroscopic guidance with care to consider the medication neurotoxic in relation to spinal nerves. MB was chosen for a variety of reasons in these
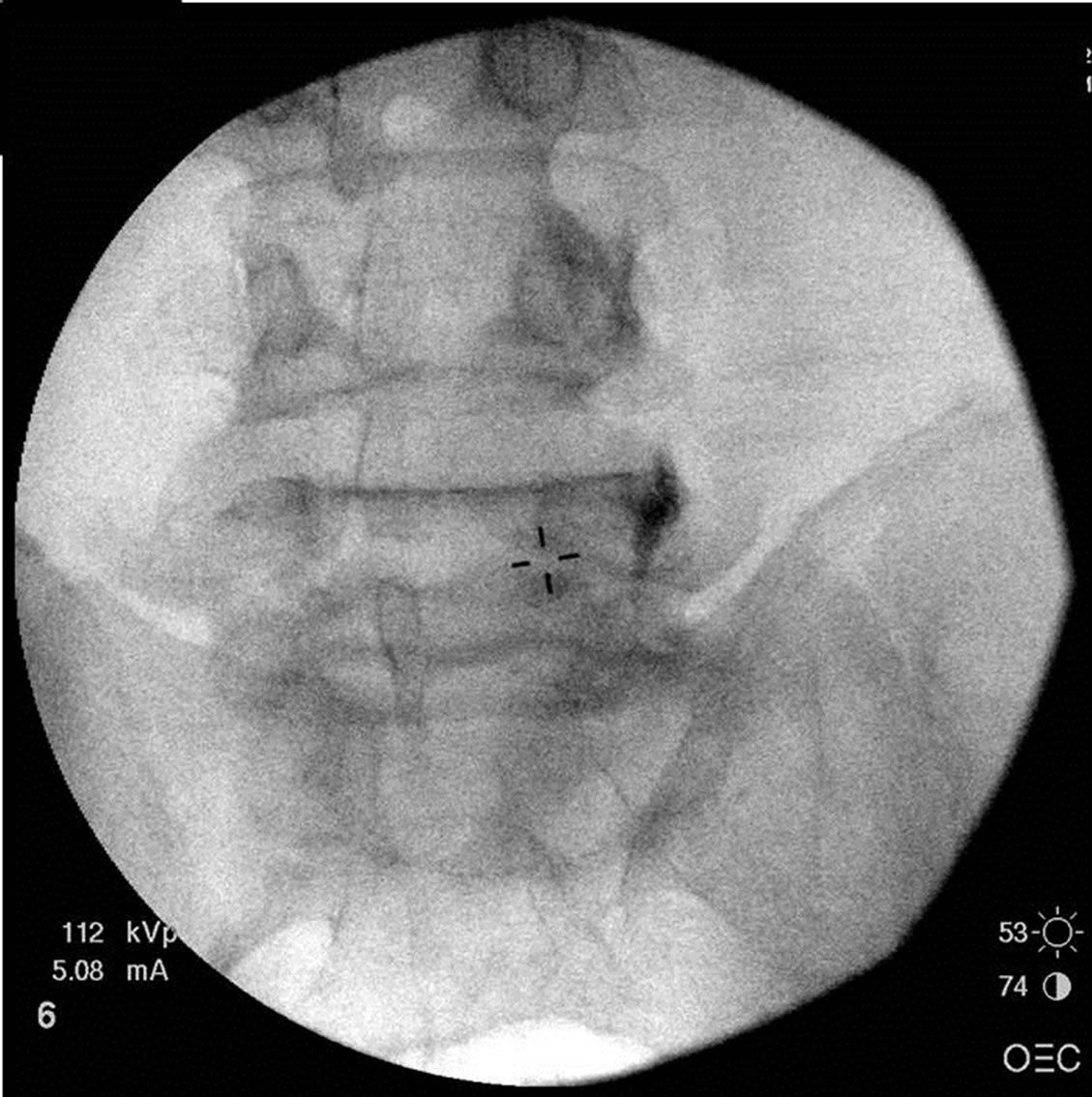
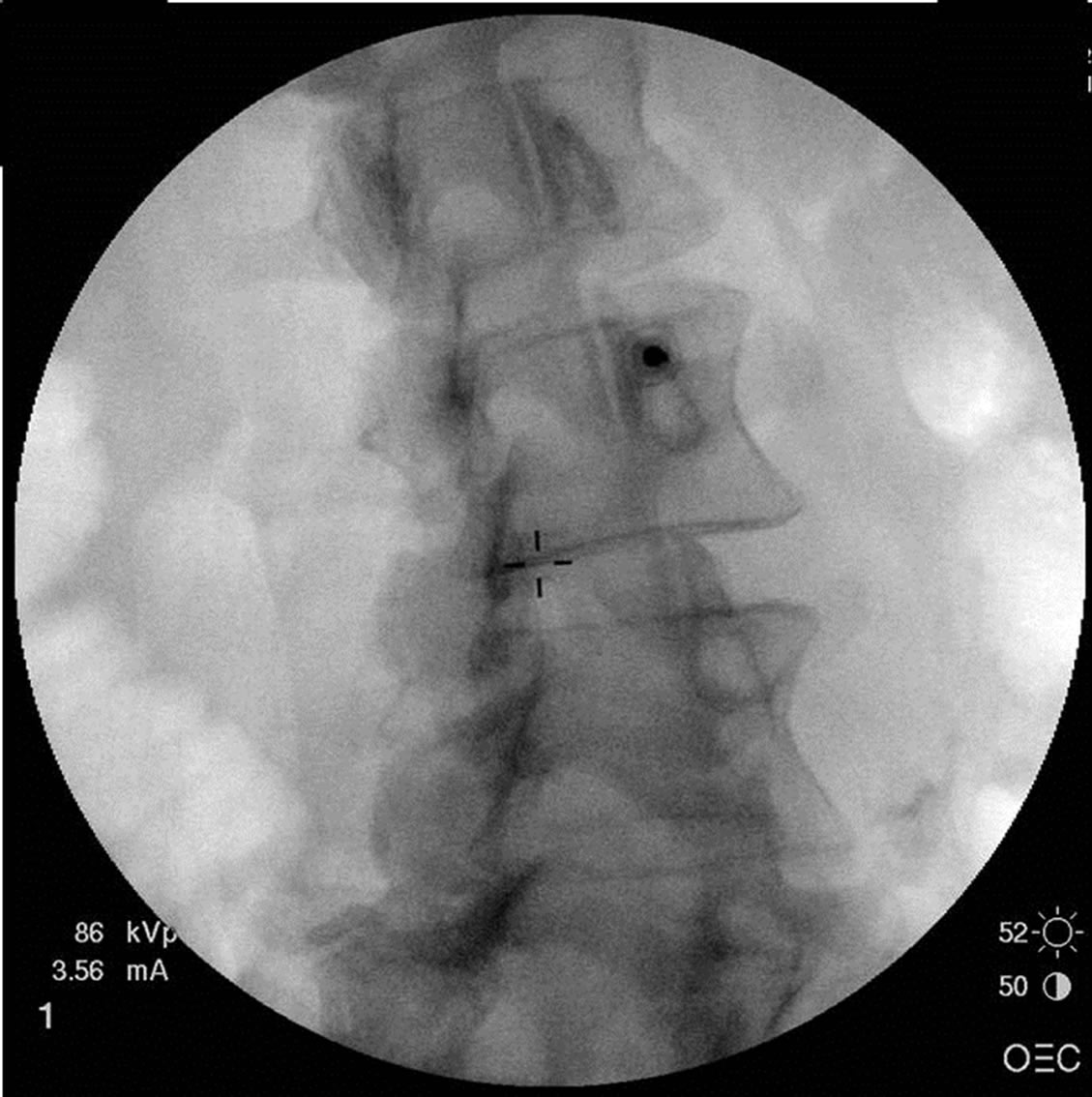
Figure 1. Placement of needle (left), and injection (right) containing equal parts methylene blue (1%), bupivacaine (0.5%), and radiopaque contrast, over the medial branch nerves.
patients.
3. Results
Results are summarized in Table 1. Patient A had RFA for pain related to the left SI joint with four months of relief; patient A returned to clinic for another RFA after successful diagnostic blocks but only received 10 days of pain relief. We proceeded to MB in this patient.Patient B had RFA for pain related to the L4/L5 and L5/S1 facets bilaterally with six months of relief; patient B returned for repeat RFA, but MB was utilized alternatively because of technical malfunction of the RFA machine. Patient C had RFA for pain related to the left L4/5 and L5/S1 facets with two weeks of pain relief; we then proceeded with MB treatment. Patient D had multiple RFA’s for pain related to the right L4/5 and L5/S1 facets, with initally good results but eventual loss of duration. Patient D then underwent prolotherapy with dextrose on two occasions with three to four months of relief and then underwent treatment with MB. Patient E presented with left L4/5 and L5/S1 facet pain, and SI joint pain. The patient inquired about alternative options to RFA and MB was a decision made by the treating physician and patient.
4. Discussion
Lumbar L-Z and SI joint pain are among the most common sources of low back pain. Ideally the treatment should entail a multimodal approach of non-interventional and interventional treatment [1]. Currently accepted practice guidelines recommend two diagnostic
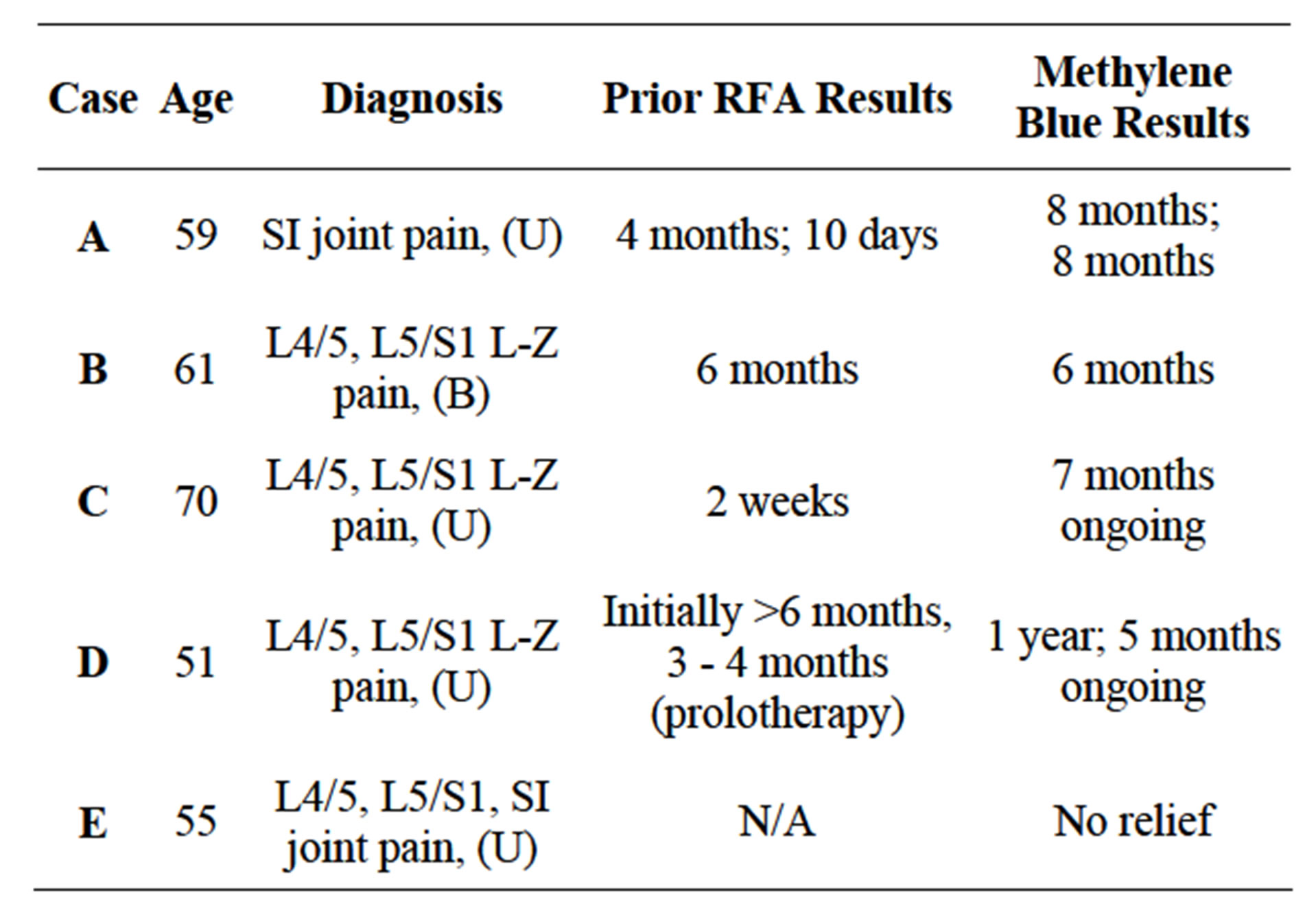
Table 1. Pain relief duration after different methods of neurolysis.
blocks to confirm the source of back pain before proceeding to RFA, with expected relief of 6 to 12 months. RFA has become the standard evidence-based interventional treatment of L-Z and SI joint pain, however challenges to efficient treatment remain in some cases.
Where the diagnosis is correct, chemical neurolysis has potential to achieve technical success in refractory cases of spine pain possibly due to spread over a broader area than the extents of typical RFA lesions. Utilization of chemical neurolysis in the treatment of nonmalignant pain is less frequently preferred due to its potential to cause neuritis, long term neurologic deficits, damage to nonneural tissue, and less ability to control where the substance may track [7].
Methylene Blue has been used to treat methemoglobinemia, malaraia [8], psoriasis [9], AIDS-related Kaposi’s sarcoma [10], west nile virus [11], and to localize tissues or identify urinary leaks; it’s neurolytic properties may lend utility in the treatment of discogenic pain, pruritus ani, and fracture pain. In 1970 Poppers and colleagues confirmed that multiple doses of 1.5 ml of 1% MB into the epidural space of cats caused varying degrees of permanent paraplegia in all 5 cases [5]. According to Poppers and colleagues, epidural administration of MB is contraindicated [5]. This contraindication was challenged by Peng and colleagues when they choose to administer intradiscal MB, given the close proximity of the intrathecal and epidural space [4]. With no major adverse outcomes reported in the Peng study [4], with caution, we believed we could safely administer MB for neurolysis in the treatment of L-Z and SI joint pain.
5. Conclusion
With meticulous fluoroscopic technique we administered MB to five patients on the spine without major adverse outcome. Successful results (4/5) in our case series appear to last as long as our expected relief from RFA, often salvaging previously diminishing results. Chemical neurolysis has potential to achieve technical success in refractory cases of L-Z or SI joint pain possibly due to spread over a broader area.Further prospective studies should be performed to evaluate the efficacy of MB for pain of the L-Z and SI joints.
REFERENCES
- S. P. Cohen and S. N. Raja, “Pathogenesis, Diagnosis, and Treatment of Lumbar Zygapophysial Joint Pain,” Anesthesiology, Vol. 106, No. 3, 2007, pp. 591-614. doi:10.1097/00000542-200703000-00024
- P. Dreyfuss, B. Halbrook, K. Pauza, A. Joshi, J. McLarty and N. Bogduk, “Efficacy and Validity of Radiofrequency Neurotomy for Chronic Lumbar Zygapophysial Joint Pain,” Spine, Vol. 25, No. 10, 2000, pp. 1270-1277. doi:10.1097/00007632-200005150-00012
- S. M. Lord, L. Barnsley, B. J. Wallis, G. J. McDonald and N. Bogduk, “Percutaneous Radio-Frequency Neurotomy for Chronic Cervical Zygapophyseal-Joint Pain,” The New England Journal of Medicine, Vol. 335, 1996, pp. 1721- 1726. doi:10.1056/NEJM199612053352302
- B. Peng, Y. Zhang, S. Hou, W. Wu and X. Fu, “Intradiscal Methylene Blue Injection for the Treatment of Chronic Discogenic Low Back Pain,” European Spine Journal, Vol. 16, No. 1, 2007, pp. 33-38. doi:10.1007/s00586-006-0076-1
- P. J. Poppers, A. R. Mastri, M. Lebeaux and B. G. Covino, “The Effect of Methylene Blue on Neural Tissue,” Anesthesiology, Vol. 33, No. 3, 1970, pp. 335-340. doi:10.1097/00000542-197009000-00014
- B. B. Mentes, M. Akin, S. Leventoglu, F. A. Gultekin and M. Oguz, “Intradermal Methylene Blue Injection for the Treatment of Intractable Idiopathic Pruritus Ani: Results of 30 Cases,” Techniques in Coloproctology, Vol. 8, No. 1, 2004, pp. 11-14. doi:10.1007/s10151-004-0043-y
- P. J. Christo and D. Mazloomdoost, “Interventional Pain Treatments for Cancer Pain,” Annals of the New York Academy of Sciences, Vol. 1138, 2008, pp. 299-328. doi:10.1196/annals.1414.034
- R. H. Schirmer, B. Coulibaly, A. Stich, M. Scheiwein, H. Merkle, J. Eubel, K. Becker, H. Becher, O. Müller, T. Zich, W. Schiek and B. Kouyaté, “Methylene Blue as an Antimalarial Agent,” Redox Report, Vol. 8, No. 5, 2003, pp. 272-276. doi:10.1179/135100003225002899
- M. Salah, N. Samy and M. Fadel, “Methylene Blue Mediated Photodynamic Therapy for Resistant Plaque Psoriasis,” Journal of Drugs in Dermatology, Vol. 8, No. 1, 2009, pp. 42-49.
- J. P. Tardivo, A. Del Giglio, L. H. Paschoal and M. S. Baptista, “New Photodynamic Therapy Protocol to Treat AIDSRelated Kaposi’s Sarcoma,” Photomedicine and Laser Surgery, Vol. 24, No. 4, 2006, pp. 528-531. doi:10.1089/pho.2006.24.528
- J. F. Papin, R. A. Floyd and D. P. Dittmer, “Methylene Blue Photoinactivation Abolishes West Nile Virus Infectivity in Vivo,” Antiviral Research, Vol. 68, No. 2, 2005, pp. 84-87. doi:10.1016/j.antiviral.2005.07.001

