Open Journal of Forestry
Vol.07 No.03(2017), Article ID:78014,20 pages
10.4236/ojf.2017.73021
Structure and Natural Regeneration Status of Woody Plants of Berbere Afromontane Moist Forest, Bale Zone, South East Ethiopia; Implication to Biodiversity Conservation
Tesfaye Bogale1*, Demeke Datiko2, Shiferew Belachew3
1Mettu Biodiversity Center, Ethiopian Biodiversity Institute (EBI), Mettu, Ethiopia.
2Hawassa Biodiversity Center, Ethiopian Biodiversity Institute (EBI), Hawassa, Ethiopia.
3Biology Department, Madda Walabu University, Bale-Robe, Ethiopia

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 7, 2017; Accepted: July 25, 2017; Published: July 28, 2017
ABSTRACT
The study was conducted on Berbere Forest in Bale Zone of Oromia Regional State, south east of Ethiopia with the objective of determining the structural analysis and natural regeneration status of the forest. Systematic sampling method was used to collect vegetation data. Seventy two nest quadrat sizes of 400 m2 (20 m × 20 m) for trees and shrubs were used. Within the main quadrat, two opposite side of each sub-quadrat of 25 m2 (5 m × 5 m) for sapling, 4 m2 (2 m × 2 m) for seedling of woody plants. The diameter and height were measured for all individual trees and shrubs having DBH (Diameter at Breast Height) ≥ 10 cm thick and ≥2 m height by using a diameter tape or caliper and clinometer respectively. For description and analysis of vegetation structure Diameter at Breast Height (DBH), basal area, tree density, height, frequency and important value index were used. Structural analysis of some selected tree species was revealed four different population patterns (bell shaped, inverted J-shaped, irregular and U-shaped). The total basal area of Berbere forest was 87.49 m2/ha, but most of the basal area was contributed by few large sized Moraceae family (Ficus vasta, Ficus ovate and Ficus thonningii) plant species. Analysis of regeneration status of woody plants in the forest showed 37.09% trees/shrubs species exhibited “good”, 19.35% showed “fair”, 6.45% showed “poor” and 25.81% trees/shrubs species were “not regenerating” at all and 11.29% trees/shrubs species were available only in sapling or seedling stage. Studies on the structure and regeneration of the forest indicated that there are species that require urgent conservation measures. Therefore, based on the results of this study, we recommended detail regenerating studies of seed bank in relation to various environmental factors such as soil type and properties.
Keywords:
Bale Zone, Berbere Forest, Structural Analysis, Regeneration Status

1. Introduction
Ethiopia is an important regional center for biological diversity due to its wide ranges of altitude, its great geographical diversity with high and rugged mountains, flat-topped plateaus and deep gorges, incised river valleys and rolling plains ( Kelbessa et al., 1992 ; Woldu, 2008 ). These make Ethiopia have about 6500 - 7000 species of higher plants that are estimated to exist in the country of which about 12% plants species are estimated to be endemic ( Gebre-Egziabher, 1991 ; Kelbessa et al., 1992 ; WCMC, 1992 ; Bekele et al., 1999 ; Vivero et al., 2006 ; Hedberg et al., 2009 ). However, deforestation and habitat fragmentation pose a serious threat to the conservation of biodiversity in general and forest genetic resources in particular. The ultimate cause that has to be addressed for the forest destruction in Ethiopia is poverty, population growth, agricultural land expansion, extensive deforestation and encroachment ( Kelbessa et al, 1992 ; Woldemariam et al., 2002 ).
Population structure is the distribution of individuals of each species in arbitrarily to provide the overall regeneration profile of the study based on tree density, height, frequency, diameter at breast height, species importance value and basal area ( Peters, 1996 ; Tesfaye et al., 2002 ; Shibru & Balcha, 2004 ). Information on population structure of a tree species indicates the history of the past disturbance to that species and the environment and hence, used to forecast the future trend of the population of that particular species ( Peters, 1996 ). From the population dynamics point of view, examination of patterns of species population structure could provide valuable information about their regeneration and/or recruitment status as well as viability status of the population that could further be employed for devising evidence-based conservation and management strategies ( Teketay, 2005 ; Tilahun et al., 2011 ).
The regeneration status/potential of species in a community can be accessed from the total population dynamics of seedlings and saplings in the forest community ( Tesfaye et al., 2002 ; Duchok et al., 2005 ). The overall pattern of population dynamics of seedlings, saplings and adults of a plants species can exhibit the regeneration profile, which is used to determine their regeneration status ( Khan et al., 1987 ; Bekele, 1994 ). A population with sufficient number of seedlings and saplings depicts satisfactory regeneration behavior, while inadequate number of seedlings and saplings of the species in a forest indicates poor regeneration ( Khan et al., 1987 ; Tripathi & Khan, 2007 ).
2. Materials and Methods
2.1. Study Site
Berbere forest is administratively located in Berbere district, Bale zone, Oromia National Regional State, Ethiopia, and 530 km from Addis Ababa and 100 km from capital of Bale zone, Robe. The study area is located at longitudes between UTM 0742254 to 0751912 E and latitude 37N 0619214 to 37N 0629989 and altitude between 1100 m and 1880 m (Figure 1).
Climate: The mean annual temperature is about 20.0˚C and the mean annual minimum and maximum temperatures are 8.9˚C and 30.44˚C respectively. There is a slight difference in the temperature throughout the year. The hottest month is February with maximum temperature record 30.44˚C and the coldest month is December with minimum temperature of 8.91˚C. The mean annual rainfall of the study area is 771 mm. It is characterized by bimodal rainfall with the main rainy season occurring early March through May and the short rain late August through November.
2.2. Sampling Design
Following a reconnaissance survey, a systematic sampling technique was used to collect vegetation data in the Forest. Seventy two nest quadrat size of 400 m2 (20 m × 20 m) were used. Within the main quadrat two opposite side of each sub- quadrat of 25 m2 (5 m × 5 m) for sapling of woody plants, 4 m2 (2 m × 2 m)
Figure 1. The study area map.
for seedling of woody plants. Plots were laid systematically at every 300 m along transect lines, which were 200 m apart from each other ( Chauhan et al., 2008 ; Yineger et al., 2008 ; Adamu et al., 2012 ; Tiwari et al., 2010 ; Gebrehiwot & Hundera, 2014 ; Sharma & Ahmad, 2014 ).
2.3. Data Collection
Data collection was conducted from October 05 to November 25, 2015. The diameter and height were measured and recorded for all individual trees and shrubs having DBH (Diameter at Breast Height) ≥ 10 cm thick and ≥2 m height by using a diameter tape or caliper and clinometer respectively. The woody plants <10 cm DBH and <2 m height were also recorded. If the tree branched at breast height or below, the diameter were measured separately for the branches and averaged ( Bharali et al., 2012 ; Dibaba et al., 2014 ; Gebrehiwot & Hundera, 2014 ).
Regeneration status (seedling and sapling) density of woody plants was carried out with two opposite sub-quadrat with in the main quadrat. For sapling data, the sub-plot of 25 m2 (5 m × 5 m) for individual’s woody plant <10 cm DBH and >0.5 m <2 m height were recorded. For seedling data, the sub-plot of 4 m2 (2 m × 2 m) with individual’s woody plant species ≤0.5 m height was recorded ( Chauhan et al., 2008 ; Bharali et al., 2012 ; Dibaba et al., 2014 ).
2.4. Data Analysis
2.4.1. Structural Data Analysis
・ Diameter at Breast Height (DBH): The structural data of DBH was analyzed based on eight DBH classes (i.e., 10 - 20 cm, 20.1 - 30 cm, 30.1 - 40 cm, 40.1 - 50 cm, 50.1 - 60 cm, 60.1 - 70 cm, 70.1 - 80 cm, >80 cm) ( Hundera et al., 2007 ).
・ Basal area is the area outline of a plant near ground surface. It is expressed in square m/hectare ( Mueller-Dombois & Ellenberge, 1974 ).
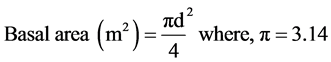 (1)
(1)
d = DBH by m
・ Density is a count of the numbers of individuals of a species within the quadrat ( Kent & Coker, 1992 ). Afterwards, the sum of individuals per species were calculated and analyzed in terms of species density per hectare ( Mueller- Dombois & Ellenberge, 1974 ).
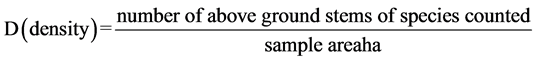 (2)
(2)
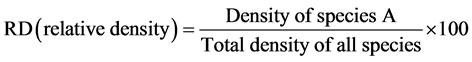 (3)
(3)
・ Frequency is defined as the probability or chance of finding a species in a given sample area or quadrat. It is dependent on quadrat size, plant size and patterning in the vegetation ( Kent & Coker, 1992 ). It was calculate with this formula:
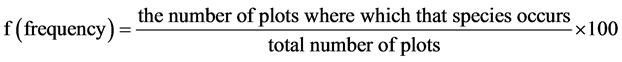 (4)
(4)
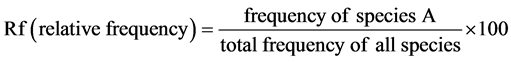 (5)
(5)
・ Importance Value Index (IVI): It combines data for three parameters (relative frequency, relative density and relative abundance) or it often reflects the extent of the dominance, occurrence and abundance of a given species in relation to other associated species in an area ( Kent & Coker, 1992 ).
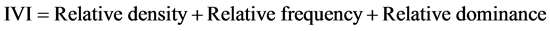
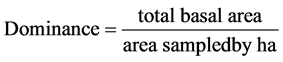 (6)
(6)
Relative Dominance (RDO) was calculated as:
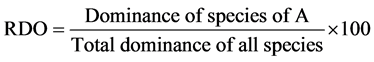 (7)
(7)
Height: Individual trees and shrubs having height greater than or equal to 2 m within sampling quadrats were collected and analyzed by classifying into ten classes (2 - 6 m, 6 - 9 m, 9 - 12 m, 12 - 15 m, 15 - 18 m, 18 - 21 m, 21 - 24 m, 24 - 27 m, 27 - 30 m, >30 m) ( Hundera et al., 2007 ).
2.4.2. Regeneration Data Analysis
The regeneration status of sample species in the forest was analyzed by comparing seedling with sapling and sapling with matured trees data ( Shankar, 2001 ; Dhaulkhandi et al., 2008 ; Tiwari et al., 2010 ; Gebrehiwot & Hundera, 2014 ) in the following categories: 1) “good” regeneration, if present in seedling > sapling > mature tree; 2) “fair” regeneration, if present in seedling > sapling < mature tree; 3) “poor” regeneration, if a species survives only in the sapling stage, but not as seedlings (even though saplings may be less than, more than, or equal to mature); 4) “none”, if a species is absent both in sapling and seedling stages, but present as mature; and 5) “new”, if a species has no mature, but only sapling and/or seedling stages.
3. Result and Discussions
3.1. Vegetation Structure
3.1.1. Tree and Shrub Density
Tree and shrub density, expressed as the number of individuals with DBH greater than 10 cm was 355.2/ha and those individuals with DBH between 10 and 20 cm and with DBH greater than 20 cm were 216.58/ha and 138.62/ha respectively. The ratio described as a/b, is taken as the measure of size class distribution ( Grubb et al., 1963 ). Accordingly, the ratio of individuals with DBH between 10 and 20 cm (a) to DBH > 20 cm (b) was 1.56 for Berbere Forest. This
Table 1. Comparisons of tree densities with Diameter at Breast Height (DBH) between 10 and 20 (a) and tree density with DBH > 20 cm (b) from Berbere forest with 10 other forests in Ethiopia.
indicates that the proportion of medium-sized individuals (DBH between 10 and 20 cm) is larger than the large sized individuals (DBH > 20 cm). When compare ratio (a/b DBH) of Berbere forest to the other forests of Ethiopia is relatively larger than that of Dodola, Jibat, Komto and Kimphe Lafa forests but Alata-Bolale, Jima, Bibita, Menna Angetu, Belete and Masha Anderacha forests have more a/b ratio values than Berbere forest indicating that there is more predominance of trees in the lower DBH class in these forests than in Berbere forest (Table 1).
3.1.2. Diameter at Breast Height (DBH)
The patterns of diameter class distribution indicate the general trends of population dynamics and recruitment processes of a given species ( Steininger, 2000 ). The distribution of trees in different DBH classes was analyzed. The DBH was classified into eight classes: 1) 10.0 - 20.0 cm; 2) 20.1 - 30.0 cm; 3) 30.1 - 40.0 cm; 4) 40.1 - 50.0 cm; 5) 50.1 - 60.0 cm; 6) 60.1 - 70.0 cm; 7) 70.1 - 80.0 cm; and 8) >80.0 cm. The majority of the tree individuals are distributed in the first DBH class (10 - 20 cm) with 216.58 ha−1 (60.02%). The distribution of trees in DBH classes of 2, 3, 4, 5, 6, 7, 8 was 73.61 individuals ha−1 (20%), 43.05 (12.12%), 14.93 (4.20%), 3.82 (1.07%), 1.74 (0.49%), 0.69 (0.19%) and 2.78 (0.78%) individuals ha−1 respectively. As the DBH class size increases, the number of individuals gradually decreases toward the higher DBH classes. Similar results were reported by Lulekal et al. (2008) from Mana Angetu, Burju et al. (2013) from Jibat and Gebrehiwot & Hundera, 2014 from Belete forest.
The general pattern of DBH class distribution of Berbere woody plant species was showed an inverted J-shaped distribution (Figure 2). However, this pattern does not describe the general trends of population dynamics and recruitment
Figure 2. Diameter at Breast Height classes versus the number of individuals/ha of woody plant species in Berbere forest.
processes of a given individual species. Analysis of population structures for each individual tree and shrub species could provide more realistic and distinctive information for forest conservation and management activities ( Kelbessa & Soromessa, 2008 ; Yineger et al., 2008 ; Didita et al., 2010 ; Dibaba et al., 2014 ). The population structure of selected species of Berbere forest followed four general diameter class distribution patterns, which indicated different population dynamics among species.
The first pattern was an inverted J-shaped distribution showed a pattern where species frequently had the highest frequency in low diameter classes and a gradual decrease towards the higher class. Species such as Combretum molle, Filicium decipiens, Mimusops kummel, Psydrax schimperiana and Maesa lanceolata were characterized by this distribution pattern. As Ayalew et al. (2006) , Kelbessa & Soromessa (2008) , Yineger et al. (2008) and Dibaba et al. (2014) indicated in different forest such pattern is normal population structure and shows the existence of species in healthier condition.
The second pattern was Bell-shape, which showed that the number of individuals were high in the middle classes and decreased towards the lower and higher diameter classes. Example Podocarpus falcatus, Warburgia ugandensis, Diospyros abyssinica, Celtis africana, Tamarindus indica, and Buddleja polystachya were characterized by such. This pattern indicates a poor reproduction ( Senbata, 2006 ) and recruitment of species, which may be associated with the over harvesting of seed bearing individual ( Senbata, 2006 ).
The third pattern was formed by species having irregular distribution over diameter classes. Some DBH classes had small number of individuals while other DBH classes had large number of individuals and even some were missed. This irregular pattern distribution was might be due to selective cutting by the local people for construction and firewood. Overgrazing which affects the seedlings under the mother tree could be another reason for such irregularities. This pattern is exemplified by Oleacapensis subsp. hochstetteri and Pouteria adolfifriederici.
The fourth pattern was with no individual in DBH class one and two and five relatively equal numbers in DBH class 3 and 4 and DBH class 6 and 8, example Ficus ovate. This pattern represents abnormal population dynamics. The underlying reason for such pattern is may be due to the nature of seeds germination and recruitments of its seedling. The seeds of this plant germinate on the stems of other plants and the seedlings also grow on stems of other plants as parasite until they become larger and replace the host plant by killing it ( Putz & Holbrook, 1989 ; Kelbessa & Soromessa, 2008 ).
Information on population structure of a tree species indicates the history of the past disturbance to that species and the environment and hence, used to forecast the future trend of the population of that particular species ( Bekele, 1994 ; Teketay, 1997 ).
3.1.3. Height Class Distributions
The woody species individuals obtained in the study were classified into 10 height classes: 1) 2.0 - 6.0 m; 2) 6.1 - 9.0 m; 3) 9.1 - 12.0 m; 4) 12.1 - 15.0 m; 5) 15.1 - 18.0 m; 6) 18.1 - 21.0 m; 7) 21.1 - 24.0 m; 8) 24.1 - 27.0 m; 9) 27.1 - 30.0 m; 10) >30.0 m and these were described. There is higher number of trees/shrubs individuals in the height class 2, 3, 4, 5 and 6 which accounts 75.85% of the total height classes. The rest lower and upper class intervals are accounts 24.15%. The woody species height class distribution of all individuals in different size class was more or less showed an inverted J-shaped like DBH distribution pattern (Figure 3). As Tesfaye et al. (2002) indicated the decrease in number of each height class towards the highest classes showed that the dominance of small- sized individuals in the forest, which was the characteristic of normal rate.
3.1.4. Vertical Structure
The vertical structure of the woody species occurring in the Berbere forest was analyzed using the IUFRO classification scheme ( Lamprecht, 1989 ). The scheme classifies the storey into upper, where the tree height is greater than 2/3 of the top height; middle, where the tree height is in between 1/3 and 2/3 of the top height and the lower storey where the tree height is less than 1/3 of the top height. The top height for trees in Berbere forest was 40 m (Podocarpus falcatus). Accordingly, the emergent tree species that occupied the upper storey in-
Figure 3. Height classes versus density of individual species per hectar in Berbere forest.
Table 2. Density, species number and individuals to species ratios by Storey.
clude Podocarpus falcatus, Warburgia ugandensis, Pouteria adolfi-friederici, Diospyros abyssinica and Buddleja polystachya etc. In addition, only few individuals attained the upper storey as the ratio of individuals to species is lower. From this it is important to note that the highest proportion of species was concentrated in the lower storey (49.58%) followed by the middle (43.94%) and upper storey (6.48%) of the vertical structure of the forest given in Table 2.
3.1.5. Basal Area and Density
The total basal area of Berbere forest was 87.49 m2/ha. About 49.67 m2/ha (56.77%) of the total basal area was contributed by eleven tree species. The basal area of family Moraceae (Ficus vasta, Ficus ovate and Ficus thonningii) had contribute 27.76 (31.73%) of the total basal area of the study area. This is might be due to the Ficus species is not currently used for wood, charcoal and construction materials. In addition the society of the area has respected the Moraceae family plant species culturally.
The basal area of Berbere Forest is much greater than that of Chilimo forest ( Bekele, 1993 ), Donkoro forest ( Ayalew et al., 2006 ), Alata-Bolale forest ( Enkossa, 2008 ), Komto forest ( Gurmessa et al., 2012 ) and Jibat forest ( Burju et al., 2013 ). But it was much less basal area than that of Dodolla forest ( Hundera et al., 2007 ), Menna Angetu forest ( Lulekal et al., 2008 ), Belete forest ( Gebrehiwot & Hundera, 2014 ) and Kimphe lafa forest ( Aliyi et al., 2015 ) (Figure 4).
Basal area provides a better measure of the relative importance of the species than simple stem count ( Cain & Castro, 1959 ), cited in Bekele (1994) . Thus, species with the largest basal area can be considered the most important woody species in the study area. Accordingly, Ficus vasta, Ficus ovate, Ficus thonningii, Pappea capensis and Podocarpus falcatus species were the most important species in the study area. Species like, Diospyros abyssinica, Warburgia ugandensis, Celtis africana and Tamarindus indica although they have high density; their basal area is not as high as their density (Table 3). This is due to the nature of the plants not to grow to higher basal area and the level of exposure to human activities. It also indicates that species with the highest basal area do not necessarily have the highest density, indicating size difference between species ( Bekele, 1994 ; Shibru & Balcha, 2004 ; Denu, 2006 ).
3.1.6. Important Value Index (IVI)
Important value index is the degree of dominancy and abundance of a given species in relation to the other species in the area ( Kent & Coker, 1992 ). The re-
Figure 4. Basal area (in m2/ha) of different forests in Ethiopia.
Table 3. Basal area, density and their percent contribution of trees/shrubs species with greater than two BA m2 ha−1 in the Berbere forest.
sult of IVI which is calculated from relative density, relative basal area (relative dominance) and relative frequency, of woody species is shown in Table 4. It is useful to compare the ecological significance of species and for setting conservation priority ( Lamprecht, 1989 ).
The output of IVI analysis showed that Warburgia ugandensis 33.91 (11.32%), Diospyros abyssinica 29.85 (9.96%), Podocarpus falcatus 18.21 (6.08%), Combretum molle 17.92 (5.98%), Filicium decipiens 16.27 (5.43%) and Ficus vasta 13.59 (4.53%) were the first six most dominant species (Table 4). These species constituted 43.30% of the total importance value index; while the majority of the species (87.27%) had important value indices of less than 10 (Figure 5). Distribution of species among different IVI classes indicated that 7.27% less than 1, 60% 1.1 - 5.0, 20% 5.1 - 10.0, 3.64% 10.1 - 15.0, 5.45% 15.1 - 20.0 and 3.64% greater than 20.0 had importance value indices in the study forest.
Table 4. Importance Value Index (IVI) of the dominant tree species of Berbere forest (RD = Relative Density, RF = Relative Frequency and RDO = Relative Dominance IVI = Important Value Index).
Figure 5. IVI classes and individual species of woody plants in the Berbere forest.
Table 5. Frequency distribution of dominant woody species in Berbere forest.
3.1.7. Frequency
Frequency is the number of quadrats in which a given species occurred in the study area. The four most frequently observed species were Warburgia ugandensis which occurred 57 times out of 72 quadrats which having 15.20 relative frequency, Filicium decipiens 9.07 relative frequency, Diospyros abyssinica 7.20 relative frequency and Combretum molle 6.93 relative frequency. The nine most frequently occurred species together contributed 57.59% of total relative frequency of the forest (Table 5). The least occurred species were Trema orientalis, Cordia africana, Olea welwitschii, Erythrina brucei, Berchemia discolor, Acaccia abyssinica, Acacia seyal, Acacia bussei, Cassipourea malosana, Ficus thonningii, Ziziphus spinachristi, Rhus natalensis, Rhus ruspolii, Dichrostachys cinerea, Pappea capensis and Combretum collinum subsp. Binderianum each having 0.27 relative frequencies and a total of 4.32 relative frequencies.
3.2. Regeneration of Woody Plants in Berbere Forest
The future composition of the forests depends on the potential regenerative status of individual species within a forest stand in space and time ( Henle et al., 2004 ). The population structure, characterized by the presence of sufficient population of seedlings, saplings and adults, indicates successful regeneration of forest species ( Saxena & Singh, 1984 ), and the presence of saplings under the canopies of adult trees also indicates the future composition of a community. Climatic factors and biotic interference influence the regeneration of different species in the vegetation ( Henle et al., 2004 ; Dhaulkhandi et al., 2008 ).
In this study it has been observed that 37.09% tree/shrubs species exhibited “good” regeneration status, 19.35% showed “fair” regeneration condition and 6.45% showed “poor” regeneration status. A total of 25.81% tree species were “not regenerating” at all and 11.29 % tree species, which were available only in sapling or seedling stage, were considered as “new” in Berbere forest (Appendix 1). The “poor” regenerating tree species were Schrebera alata, Pappea capensis, Olea europaea subsp. cuspidata, and Acacia bussei. Species which were found in “not regenerating” category were Pouteria adolfifriederici, Lannea rivae, Ficus vasta, Ficus thonningii, Diospyros abyssinica, Tamarindus indica, Warburgia ugandensis, Erythrina brucei, Combretum aculeatum, Cordia africana, Buddleja polystachya, Fagaropsis angolensis, Caesalpinia volkensii, Nuxia congesta, Olea welwitschii and syzygium guineense. “New” regeneration status included Rosa abyssinica, Myrsine africana, Maytenus gracilipes, Acalypha volkensii, Asparagus aridicola, Carissa spinarum and Croton dichogamus. These species may have reached or colonized to the study site by different mechanisms like dispersal of seeds through drooping of birds and animals.
The “poor” and “none” regenerating categories which constitute around 32.26% of the woody plant in the study area have many important and useful tree species namely Pouteria adolfifriederici, Diospyros abyssinica, Warburgia ugandensis, Erythrina brucei, Combretum aculeatum, Cordia africana, Buddleja polystachya, Schrebera alata, Pappea capensis, Olea welwitschii and Acacia bussei etc. which have certain economic, medicinal and ecological values.
The overall regeneration status of the tree species of the study site is satisfactory at community level showing “good” regeneration status (Figure 6), but as stated above 32.26% trees/shrubs species, falls under “poor” and “not regenerat-
Figure 6. The regeneration status of woody plant in the Berbere forest.
ing” status. Such condition might have been occurred due to existing disturbance in the study site like, over grazing, firewood collection and poor biotic potential of tree species which either affect the fruiting or seed germination or successful conversion of seedling to sapling stage. Moreover, individuals in young stages of any species are more vulnerable to any kind of environmental stress and anthropogenic disturbance ( Tesfaye et al., 2002 ; Nagamatsu et al., 2002 ). Hence, such human-induced activity may alter the future structure and composition of the Berbere forest
4. Conclusion and Recommendations
The general variability in population structure and regeneration status indicates the history of the past disturbance to that species and the environment. The pattern of DBH and Height class distribution of woody plant species in the Berbere forest showed an inverted J-shaped distribution. Overall analysis of population structure of most common species of trees and shrubs revealed different patterns of population structure, indicating a high variation among species population dynamics within the forest. The regeneration status of the tree species of the study site is satisfactory at community level which shows “good” regeneration status but 32.26% trees/shrubs species, falls under “poor” and “not regenerating” status. Therefore it needs urgent great conservation priority and management. The following recommendations were made; in woody plants which are currently not regenerating themselves as well as depleted tree population, it is better to think of enrichment planting of indigenous tree species and understanding the natural regeneration potential of these forests is crucial to have detailed information on soil seed bank in the forest; in addition, fourteen tree species need detailed regeneration study and conservation priority, since they have no seedling or saplings. These are Buddleja polystachya Caesalpinia volkensii, Combretum aculeatum, Cordia africana, Diospyros abssinica, Erythrina brucei, Fagaropsis angolensis, Ficus vasta, Nuxia congesta, Olea welwitschii, Pouteria adolfi-friede- ricii, Syzygium guineense, Tamarindus indica and Warburgia ugandensis.
Acknowledgements
Madda Walabu University highly acknowledged for funding the study. We would like to extend our deepest thanks to Oromia Forest and Wild life Agency of Bale zone Branch and Berbere Woreda branch office for their valuable information about the forest and providing us materials for data collection.
Cite this paper
Bogale, T., Datiko, D., & Belachew, S. (2017). Structure and Natural Regeneration Status of Woody Plants of Berbere Afromontane Moist Forest, Bale Zone, South East Ethiopia; Implication to Biodiversity Conservation. Open Journal of Forestry, 7, 352-371. https://doi.org/10.4236/ojf.2017.73021
References
- 1. Adamu, W. H., Bekele, T., & Dalle, G. (2012). Floristic Diversity, Regeneration Status and Vegetation Structure of Woodlands in Metema Area, Amhara National Regional State, North-Western Ethiopia. Journal of Forestry Research, 23, 391-398.
https://doi.org/10.1007/s11676-012-0275-z [Paper reference 1] - 2. Aliyi, K., Hundera, K., & Dalle, G. (2015). Floristic Composition, Vegetation Structure and Regeneration Status of Kimphe Lafa Natural Forest, Oromia Regional State, West Arsi, Ethiopia. Research & Reviews: Journal of Life Sciences, 5, 19-32. [Paper reference 2]
- 3. Ayalew, A., Bekele, T., & Demissew, S. (2006). The Undifferentiated Afromontane Forest of Denkoro in the Central Highland of Ethiopia: A Floristic and Structural Analysis. Ethiopian Journal of Science, 29, 45-56. [Paper reference 2]
- 4. Bekele, T. (1993). Vegetation Ecology of Remnant Afromonten Forests on the Central Plateau of Shewa, Ethiopia. Acta Phytogeographica Suecica, 79, 1-59. [Paper reference 1]
- 5. Bekele, T. (1994). Phyosociology and Ecology of Humid Afromontane Forest on the Central Plateau of Ethiopia. Journal of Vegetation Science, 5, 87-98.
https://doi.org/10.2307/3235642 [Paper reference 4] - 6. Bekele, T., Gunther, H., & Soromessa, T. (1999). Forest Genetic Resources of Ethiopia: Status and Proposed Actions. In S. Edwards, A. Demissie, T. Bekele, & H. Gunther (Eds.), Proceedings of Forest Genetic Resources Conservation: Principles, Strategies and Actions (pp. 39-48). Addis Ababa: Institute of Biodiversity Conservation and Research with GTZ. [Paper reference 1]
- 7. Bharali, S., Paul, A., Khan, M. L., & Singha, L. B. (2012). Impact of Altitude on Population Structure and Regeneration Status of Two Rhododendron Species in a Temperate Broad Leaved Forest of Arunachal Pradesh, India. International Journal of Ecosystem, 2, 19-27.
https://doi.org/10.5923/j.ije.20120201.04 [Paper reference 2] - 8. Burju, T., Hundera, K., & Kelbessa, E. (2013). Floristic Composition and Structural Analysis of Jibat Humid Afromontane Forest, West Shewa Zone, Oromia National Regional State, Ethiopia. Ethiopian Journal of Education and Science, 8, 11-33. [Paper reference 3]
- 9. Cain, S. A., & Castro, G. M. O. (1959). Manual of Vegetation Analysis. New York: Harper & Brothers. [Paper reference 1]
- 10. Chauhan, D. S., Dhanai, C. S., Singh, B., Chauhan, S., Todaria, N. P., & Khalid, M. A. (2008). Regeneration and Tree Diversity in Natural and Planted Forests in a Terai-Bhabhar Forest in Katarniaghat Wildlife Sanctuary, India. Tropical Ecology, 49, 53-67. [Paper reference 2]
- 11. Denu, D. (2006). Floristic Composition and Ecological Study of Bibita Forest (Gura ferda), Southwest Ethiopia (pp. 26-44). MSc. Thesis (Unpublished), Addis Ababa: Addis Ababa University. [Paper reference 2]
- 12. Dhaulkhandi, M., Dobhal, A., Bhatt, S., & Kumar, M. (2008). Community Structure and Regeneration Potential of Natural Forest Site in Gangotri, India. Journal of Basic and Applied Sciences, 4, 49-52. [Paper reference 2]
- 13. Dibaba, A., Soromessa, T., Kelbessa, E., & Tilahun, A. (2014). Diversity, Structure and Regeneration Status of the Woodland and Riverine Vegetation of Sire Beggo in Gololcha District, Eastern Ethiopia. Momona Ethiopian Journal of Science, 6, 70-96. [Paper reference 4]
- 14. Didita, M., Nemomissa, S., & Woldemariam, T. (2010). Floristic and Structural Analysis of the Woodland Vegetation around Dello Menna, Southeast Ethiopia. Journal of Forestry Research, 21, 395-408.
https://doi.org/10.1007/s11676-010-0089-9 [Paper reference 1] - 15. Duchok, R. K., Kent, A. D., & Khumbongmayum, A. (2005). Population Structure and Regeneration Status of Medicinal Tree Illicium griffithii in Relation to Disturbance Gradients in Temperate Broad-Leaved Forest of Arunachal Pradesh. Current Science, 89, 673-676. [Paper reference 1]
- 16. Enkossa, W. (2008). Floristic Analysis of Alata-Bolale Forest in Gudaya Bila Wereda East Welega Zone, Oromia Regional State, West Ethiopia. Unpublished MSc Thesis, Addis Ababa: Addis Ababa University. [Paper reference 2]
- 17. Gebre-Egziabher, T. B. (1991). Diversity of Ethiopian Flora. In J. M. M. Engels, J. G. Hawkes, & M. Worede (Eds.), Plant Genetic Resources of Ethiopia (pp. 75-81). Cambridge: Cambridge University Press. [Paper reference 1]
- 18. Gebrehiwot, K., & Hundera, K. (2014). Species Composition, Plant Community Structure and Natural Regeneration Status of Belete Moist Evergreen Montane Forest, Oromia Regional State, Southwestern Ethiopia. Momona Ethiopian Journal of Science, 6, 97-101. [Paper reference 6]
- 19. Grubb, P. J., Lloyd, J. R., Penigton, T. D., & Whitmore, T. C. (1963). A Comparison of Montane and Lowland Rainforests in Ecuador. Journal of Ecology, 51, 567-601.
https://doi.org/10.2307/2257748 [Paper reference 1] - 20. Gurmessa, F., Soromessa, T., & Kelbessa, E. (2012). Structure and Regeneration status of Komto Afromontane Moist Forest, East Wellega Zone, West Ethiopia. Journal of Forestry Research, 23, 205-216.
https://doi.org/10.1007/s11676-012-0242-8 [Paper reference 1] - 21. Gurmessa, F., Soromessa, T., & Kelbessa, E. (2013). Floristic Composition and Community Analysis of Komto Afromotane Moist Forest, East Wollega Zone, West Ethiopia. Journal of Science, Technology and Arts Research, 2, 58-69. [Paper reference 1]
- 22. Hedberg, I., Friis, I., & Person, E. (2009). General Part and Index to Vol. 1-7. Flora of Ethiopia and Eritrea Volume 8. Addis Ababa and Uppsala: The National Herbarium. [Paper reference 1]
- 23. Henle, K., Davies, K. F., Kleyer, M., Margules, C., & Settele, J. (2004). Predictors of Species Sensitivity to Fragmentation. Biodiversity and Conservation, 13, 207-251.
https://doi.org/10.1023/B:BIOC.0000004319.91643.9e [Paper reference 2] - 24. Hundera, K., Bekele, T., & Kelbessa, E. (2007). Floristic and Phytogeographic Synopsis of a Dry Afromontane Coniferous Forest in Bale Mountains, Ethiopia: Implication to Biodiversity Conservation. SINET: Ethiopian Journal of Science, 30, 1-12. [Paper reference 4]
- 25. Kelbessa, E., & Soromessa, T. (2008). Interfaces of Regeneration, Structure, Diversity and Uses of Some Plant Species in Bonga Forest: A Reservoir for Wild Coffee Gene Pool. SINET: Ethiopian Journal of Science, 31, 121-134. [Paper reference 3]
- 26. Kelbessa, E., Demissew, S., Woldu, Z., & Edwards, S. (1992). Some Threatened Endemic Plants of Ethiopia. NAPRECA Monograph, 2, 35-55. [Paper reference 3]
- 27. Kenea, F. (2008). Remnant Vegetation and Population Structure of Woody Species of Jima Forest, Western Ethiopia. Unpublished M.Sc. Thesis, Addis Ababa: Addis Ababa University. [Paper reference 1]
- 28. Kent, M., & Coker, R. (1992). Vegetation Description and Analysis: A Practical Approach (p. 363). New York, NY: John Wiley and Sons. [Paper reference 4]
- 29. Khan, M. L., Rai, J. P. N., & Tripathi, R. S. (1987). Population Structure of Some Tree Species in Disturbed and Protected Subtropical Forests of Northeast India. Acta Oecologica Oecologia Applicata, 8, 247-255. [Paper reference 2]
- 30. Lamprecht, H. (1989). Silviculture in Tropics (p. 296). Berlin: Technical Cooperation of Federal Republic. [Paper reference 2]
- 31. Lulekal, E., Kelbessa, E., Bekele, T., & Yineger, H. (2008). PLANT Species Composition and Structure of the Mana Angetu Moist Montane Forest, South-Eastern Ethiopia. Journal of East African Natural History, 97, 165-185.
https://doi.org/10.2982/0012-8317-97.2.165 [Paper reference 3] - 32. Mueller-Dombois, D., & Ellenberge, H. (1974). Aims and Methods of Vegetation Ecology (p. 304). New York, NY: John Wiley and Sons. [Paper reference 2]
- 33. Nagamatsu, D., Kenji, S., & Akiko, S. (2002). Seedling Establishment of Deciduous Trees in Various Topographic Positions. Journal of Vegetation Science, 13, 35-44.
https://doi.org/10.1111/j.1654-1103.2002.tb02021.x [Paper reference 1] - 34. Peters, C. M. (1996). The Ecology and Management of Non-Timber Forest Resources (p. 322). Washington DC: World Bank Technical Paper.
https://doi.org/10.1596/0-8213-3619-3 [Paper reference 2] - 35. Putz, F. E., & Holbrook, N. M. (1989). Strangler Fig Rooting Habits and Nutrient Relations in the Llanos of Venezuela. American Journal of Botany, 76, 781-788.
https://doi.org/10.2307/2444534 [Paper reference 1] - 36. Saxena, A. K., & Singh, J. S. (1984). Tree Population Structure of Certain Himalayan Forest Associations and Implications Concerning Their Future Composition. Vegetatio, 58, 61-69.
https://doi.org/10.1007/BF00044928 [Paper reference 1] - 37. Senbata, F. (2006). Biodiversity and Ecology of Afromontane Rainforests with Wild Coffea arabica L. Populations in Ethiopia. Ecology and Development Series No. 38. Bonn: Center for Development Research, University of Bonn. [Paper reference 2]
- 38. Shankar, U. (2001). A Case of High Tree Diversity Ina Sal (Shorea robusta)—Dominated Lowland Forest of Eastern Himalaya: Floristic Composition, Regeneration and Conservation. Current Science, 81, 776-786. [Paper reference 1]
- 39. Sharma, S., & Ahmad, J. (2014). Antropogenic Distrubances and Regeneration Status of Pinus roxburghil Sag. In Ponda Watershade, Rajouri Jammu and Kashmir. Journal of Biodiversity and Environmental Science, 4, 426-433. [Paper reference 1]
- 40. Shibru, S., & Balcha, G. (2004). Composition, Structure and Regeneration Status of Woody Plant Species in Dindin Natural Forest, Southeast Ethiopia: An Implication for Conservation. Ethiopian Journal of Biological Science, 3, 15-55. [Paper reference 2]
- 41. Steininger, M. K. (2000). Secondary Forest Structure and Biomass Following Short and Extended Land Use in Central and Southern Amazonia. Journal of Tropical Ecology, 16, 689-708.
https://doi.org/10.1017/S0266467400001656 [Paper reference 1] - 42. Teketay, D. (1997). Seedling Population and Regeneration of Woody Species in Dry Afromontane Forests of Ethiopia. Forest Ecology and Management, 98, 149-165. [Paper reference 1]
- 43. Teketay, D. (2005). Seed and Regeneration Ecology in Dry Afromontane Forests of Ethiopia: I. Seed Production-Population Structures. Tropical Ecology, 46, 29-44. [Paper reference 1]
- 44. Tesfaye, G., Teketay, D., & Fetene, M. (2002). Regeneration of 14 Tree Species in Harenna Forest, Southeast Ethiopia. Flora, 197, 461-474.
https://doi.org/10.1078/0367-2530-1210063 [Paper reference 4] - 45. Tilahun, A., Soromessa, T., Kelbessa, E., & Dibaba, A. (2011). Floristic Composition and Community Analysis of Menagesha Amba Mariam Forest (Egdu Forest) in Central Shewa, Ethiopia. Ethiopian Journal of Biological Science, 10, 111-136. [Paper reference 1]
- 46. Tiwari, K. P. G., Tadele, K., Aramde, F., & Tiwari, S. C. (2010). Community Structure and Regeneration Potential of Shorea robusta Forest in Subtropical Submontane Zone of Garhwal Himalaya, India. Nature and Science, 8, 70-74. [Paper reference 2]
- 47. Tripathi, R. S., & Khan, M. L. (2007). Regeneration Dynamics of Natural Forests—A Review, Proceedings of the Indian. National Science Academy, 73, 167-195. [Paper reference 1]
- 48. Vivero, J. L., Kelbessa, E., & Demissew, S. (2006). Progress on the Red List of Plants of Ethiopia and Eritrea: Conservation and Biogeography of Endemic Flowering Taxa. In S. A. Ghazanfar, & H. Beentje (Eds.), Taxonomy and Ecology of African Plants, Their Conservation and Sustainable Use (pp. 761-778). Kew: The Board of Trustees of the Royal Botanic Gardens. [Paper reference 1]
- 49. WCMC (World Conservation Monitoring Center) (1992). Global Biodiversity: Status of the Earth’s Living Resources (585 p.). London: Chapman and Hall. [Paper reference 1]
- 50. Woldemariam, T. M., Denich, M., Teketay, D., & Vlek, P. L. G. (2002). Human Impact on the Coffee Arabicagene Pool in Ethiopia and the Need for Its in Situ Conservation. In J. M. M. Engels, A. H. D. Brown, & M. T. Jackson (Eds.), Managing Plant Genetic Resources (pp. 237-247). Wallington DC: CAB International. [Paper reference 1]
- 51. Woldu, Z. (2008). The Population, Health and Environment Nexus, the Need for Integration and Networking (34 p.). A Background Paper for the Establishment and Launching of PHE. Addis Ababa: Addis Ababa University. [Paper reference 1]
- 52. Yeshitela, K., & Bekele, T. (2003). The Woody Species Composition and Structure of Masha-Andederacha Forest, Southwestern Ethiopia. Ethiopia Journal of Biological Science, 2, 31-48. [Paper reference 1]
- 53. Yineger, H., Kelbessa, E., Bekele. T., & Lulekal, E. (2008). Floristic Composition and Structure of the Dry Afromontane Forest at Bale Mountains National Park, Ethiopia. Ethiopian Journal of Science, 31, 103-120. [Paper reference 3]
Appendix 1. Regeneration Status of Woody Plants in the Berbere Forest Number of Seedling, Sapling and Mature Tree, Shrub, Tree/Shrub per Hectare








