Open Journal of Obstetrics and Gynecology
Vol.3 No.9(2013), Article ID:39149,5 pages DOI:10.4236/ojog.2013.39118
Effects of gestational weight gain on the outcome of labor at the Yaounde central hospital maternity, Cameroon
![]()
1Department of Obstetrics and Gynecology, Faculty of Medicine and Biomedical Sciences, University of Yaounde 1, Cameroon
2Maternity Unit, Central Hospital, Yaounde, Cameroon
Email: *rembu2000@yahoo.com
Copyright © 2013 Robinson E. Mbu et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 23 July 2013; revised 20 August 2013; accepted 28 August 2013
Keywords: Obesity Rate; Gestational Weight Gain; Pregnancy
ABSTRACT
Obesity rates are increasing in Cameroon. Obstetric literature has recently focused on the rising incidence of complications with increases in weight gain in pregnancy. Some of these complications include gestational diabetes, hypertensive disorders, operative deliveries, genital tract lacerations and fetal birth trauma. Examining the effects of excess weight gain during the course of pregnancy could help identify weight gain limits. The Institute of Medicine (IOM) was recommended by the World Health Organization (WHO) to develop guidelines for weight gain during pregnancy and we designed this study in order to determine delivery outcomes when weight is gained above these guidelines. We also sought to know if these guidelines are applicable in our environment. In this cross-sectional analytic design, pre-pregnancy and intra-partum BMIs were calculated for all the parturients who consented. They were classified into normal weight gain and excessive weight gain based on IOM recommendations. Those in the normal weight gain group were women with BMIs that ranged between 18.5 kg/m² and 30 kg/m² and who gained 9 - 16 kgs. Those who gained weight above these range were considered as having gained excessive weight during pregnancy. They were all followup in labor using the partogram. We compared prepartum, intra-partum and post-partum outcomes in the two groups by calculating odds ratios (ORs), 95% confidence intervals and p values. One hundred and ten (110) overweight women were matched against the same number of women who had normal weight gain. There was no significant difference between social status, marital status as well as level of educational and weight gain in the two groups. Under- weight (BMI < 18.5), was strongly associated with excessive weight gain (OR 2.5, 95% CI 1 - 6 and p = 0.048). Women who gained weight above the recommended range suffered from preeclampsia 18.2% vs. 6.4% (OR 3.2, 95% CI 1.3 - 8.0, p = 0.014), higher cesarean section rates 27.3% vs. 10% (OR 3.3, 95% CI 1.5 - 7.1, p = 0.002), higher rates of induced labor 19.1% vs. 9.0% (OR 2.4, 95% CI 1.0 - 5.2, p = 0.05), prolonged labor 43.6% vs. 16.4% (OR 4.0, 95% CI 2.1 - 7.4, p = 0.000), postpartum hemorrhage 10% vs. 1.8% (OR 6.2, 95% CI 1.3 - 9.2, p = 0.002). There were also higher rates of fetal mal-presentation, 11.8% vs. 3.6% (OR 4.0, 95% CI 1.31 - 11.9, p = 0.004), macrosomia 30.9% vs. 6.4% (OR 7.0, 95% CI 2.7 - 15.6, p < 0.0001), large for gestational age babies 32.7% vs. 15.5% (OR 2.6, 95% CI 1.3 - 5.1, p = 0.0045) and birth trauma 10% vs. 1.8%. (OR 6.2, 95% CI 1.3 - 9.2, p = 0.023). Women who gained weight during pregnancy above the recommended range had increased risk of adverse obstetric and neonatal outcomes.
1. INTRODUCTION
The systematic review of outcomes of maternal weight gain during pregnancy, often referred to as gestational weight gain by health organizations is motivated by several trends in perinatal health that are of public health concern [1]. Gestational weight gain includes the products of conception, which comprise the fetus and placenta, increases in maternal fat stores, plasma volume, as well as uterine and breast tissue.
The prevalence of overweight and obesity is rapidly increasing in industrialized as well as developing countries. Studies from urban populations in Cameroon have shown that overweight and obesity are increasingly common among the adult population. The estimated prevalence of obesity, based on body mass index (BMI), was 17.1% in women in urban Cameroon by 2002 [2]. There is also an increasing prevalence of obesity in women of child-bearing age [3]. Pregnant women constitute an important subpopulation with elevated risk for developing obesity due to excess weight gain during pregnancy, and hence obesity-related health problems [4].
According to the American College of Obstetricians and Gynecologists (ACOG), pregnancy complications associated with excess weight gain put the mother at risk of gestational diabetes, pre-eclampsia/eclampsia, cesarean delivery, prolonged labor, and their offspring at risk of macrosomia, stillbirths, preterm births, fetal distress, poor Apgar scores and neonatal mortality. These trends pointed to the need for guidelines to address optimal weight gain for all women during pregnancy [5].
In the 1930s, the recommended weight gain for all pregnant women, irrespective of pregravid weight status, was 6.8 kg. This recommendation to restrict weight gain to decrease the risk of pregnancy complications continued until 1970 [6].
Over the next two decades, accumulating research suggested that one guideline was unlikely to say it all about morbid weight gain in pregnancy. This recognition led the World Health Organization (WHO) and other committees to ask the Institute of Medicine (IOM) committee, a nonprofit organization in the United States of America that works outside the government to provide unbiased and authoritative advice to decision makers and the public, to issue guidelines that offered standardized guidance to providers and to the general public based on a woman’s pregravid weight status in 1990. Due to increase in gestational weight gain (GWG) and the associated health complications, the committee was asked to re-examine and modify pregnancy weight guidelines and this was in 2009 [7].
The IOM recommended that the amount of weight a woman gains during pregnancy be guided by her prepregnancy BMI using World Health Organization (WHO) classification for BMI; those with higher BMIs prior to pregnancy were advised to gain less weight during pregnancy than those with lower BMIs [8].
These guidelines were: for women with BMIs < 18.5 kg/m², weight gain of 12.5 - 18 kg was recommended; with BMIs between 18.5 - 24.9 kg/m², weight gain of 11.5 - 16 kg was recommended; with BMIs between 25.0 - 29.9 kg/m², weight gain of 7 - 11.5 kg was recommended and with BMIs > 30 kg/m², weight gain of 5 - 9 kg was recommended. Health Canada as well as many developing countries uses the new IOM 2009 guidelines to counsel women with respect to appropriate weight gain during pregnancy. These guidelines are based on WHO cut-off points for the BMI categories and include a specific, relatively narrow range of recommended weight gain for obese women [8].
A few earlier studies have examined the effects of varying gestational weight gain on obstetric outcome in women and there are indications that weight gain within the 2009 IOM’s recommendations could improve both maternal and fetal outcomes in developed and developing countries [9]. Though few studies have been carried out in our setting, the knowledge of increased weight gain during pregnancy to morbid levels will constitute a solid base for maternal and fetal morbidity and mortality risk evaluation. This study was designed to determine pregnancy (fetal and maternal) outcomes in relation to gestational weight gain away from the 2009 IOM recommended guidelines.
2. MATERIALS AND METHODS
The design was cross sectional and analytic over a period of four years (2009-2012 inclusive) at the maternity of the Yaounde central hospital. Following ethical clearance No. 288/CNE/SE/2008 and verbal or written consent formalities, pregnant women in early labor who consented were enrolled. A review of pregnancy histories was made and eligibility was based on pre-pregnancy or early pregnancy recorded weights from where booking BMIs were calculated. Measurement of second weight and BMI estimation for every woman was at the onset of labor. The women were then divided into two groups based on IOM recommendations: normal weight gain (BMI 18.5 kg/m² - 30 kg/m² and weight gained 9 - 16 kg), abnormal weight gain (BMIs and weights gained above this range). Each group had 110 women.
Age, marital status, educational level, employment status were obtained by interview and compared in the two groups. Labor was spontaneous, induced or augmented and was monitored using the partogram. Each baby delivered was examined completely and the third stage of labor was managed actively. Maternal and fetal outcomes were compared.
Data were entered into Epi Info 7 spreadsheet and analyzed. Continuous variables were presented as means and categorized variables were presented as frequencies. We used the Chi-square test to determine associations and differences between proportions. Odds Ratios (ORs) and 95% confidence intervals (95% CI) were calculated for each variable compared and significance level between differences in proportions was placed at 5%.
3. RESULTS
3.1. Maternal Outcomes with Respect to Weight Gain (Table 1)
Maternal outcomes with respect to weight gain were broken down into pre-partum, intra-partum and post-partum. Women who gained weight above the recommended range were three times more likely to develop preeclampsia (OR = 3.7, 95% CI = 1.32 - 8.09, p = 0.014) and had 2 folds the odds of being induced (OR = 2, 95% CI = 1 - 5, p = 0.003). The odds of having prolonged labor was about 4 times among them (OR = 3.957, 95% CI = 2.11 - 7.43, p = 0.000). They were more likely to be delivered instrumentally (OR 3.57, 95 % CI 1.26 - 10.13, p = 0.012), to have episiotomies (OR 3.13, 95% CI 1.38 - 7.09, p = 0.008) and to be delivered by caesarean section (OR 3.38, 95% CI 1.59 - 7.15, p = 0.002). The odds of developing post partum hemorrhage and post partum eclampsia was 6 and 3 respectively (Table 1).
3.2. Fetal Outcomes with Respect to Weight Gain
Though not statistically significant, cases of fetal distress and fetal demise were recorded in greater proportions among women who gained weight excessively (ORs 1.43 and 2.03).
The odds of fetal mal-presentation was almost 4 among these women (OR 3.68, 95% CI 1.13 - 11.97, p = 0.004). There were more cases of poor Apgar scores among them (OR 2.37, 95% CI 1.37 - 4.08, p = 0.002). The babies delivered by these women had 6 folds the odds of having traumas at birth (OR 6.02, 95% CI 1.31 - 9.2, p = 0.023). Fetal macrosomia was high (OR 6.58, 95% CI = 2.77 - 15.65, p < 0.001) as well large for gestational (OR = 2.66, 95% CI 1.39 - 5.11, p = 0.0045) (Table 2).
4. DISCUSSION
This study evaluated the maternal and fetal outcomes with respect to variations in gestational weight gain based on women’s pregravid status. We wanted to know if weight gained during pregnancy above the USA revised Institute of Medicine recommendations for gestational weight gain had an effect on pregnancy outcomes and what these outcomes were in our milieu. These guidelines were set up in 2009 and were based on WHO BMI classification of weight. At the end of the study we realized that weight gained during pregnancy above the recommended ranges had an increased risk of adverse obstetrics as well as neonatal outcomes.
The prevalence of obesity is rapidly increasing in Cameroon [10] and most pregnancies are unplanned. Overweight and obese women may not have the opportunity to normalize their BMIs prior to pregnancy [11].
In 2009, IOM revised guidelines which were published in 1990 because the initial guidelines did not use WHO BMI classification, and they only considered maternal outcomes leaving out fetal outcomes. The new guidelines did not have these shortcomings. In our study women who gained weight within the recommended
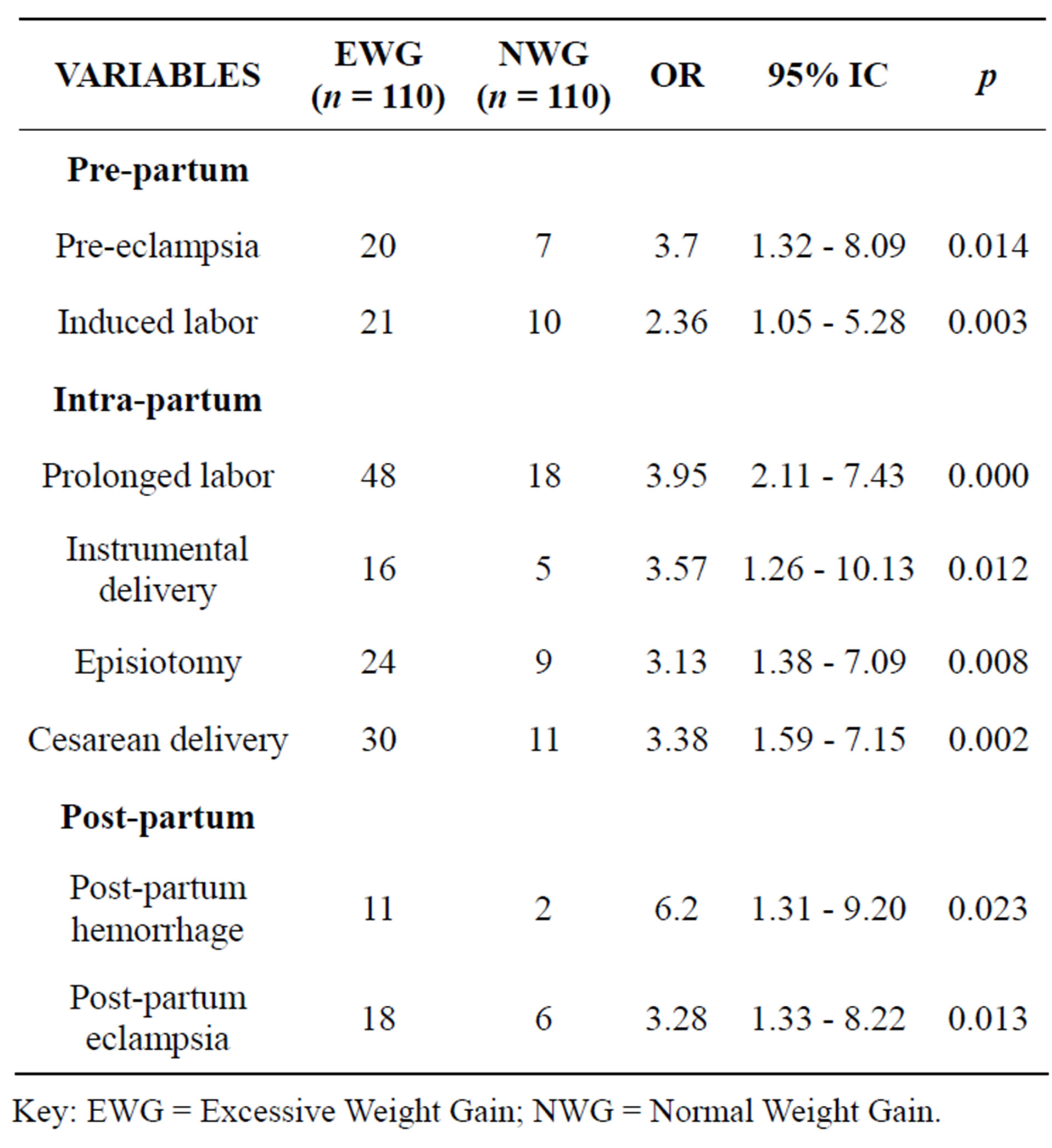
Table 1. Maternal outcomes with respect weight gain.
Key: EWG = Excessive Weight Gain; NWG = Normal Weight Gain.
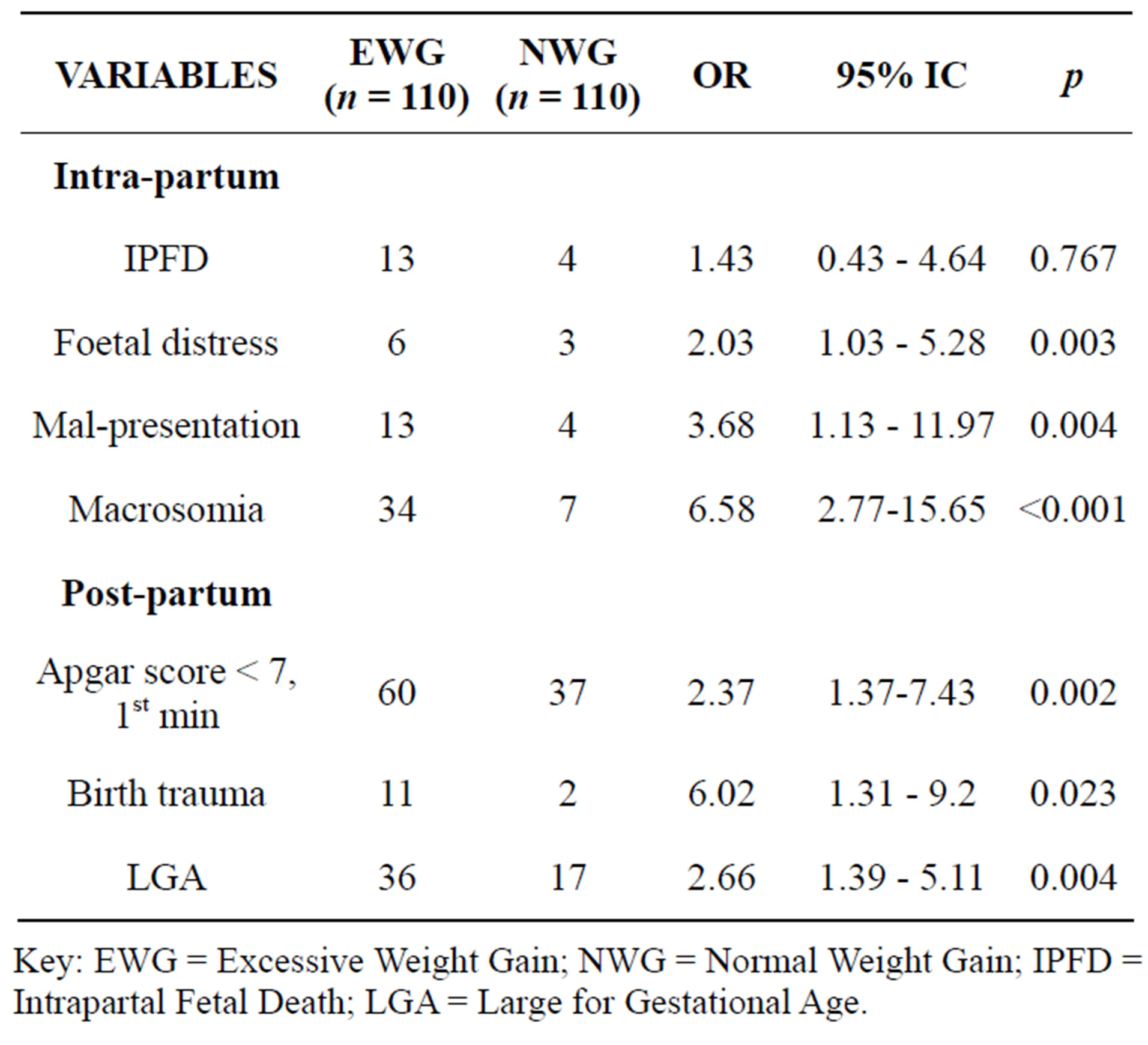
Table 2. Fetal outcomes with respect weight gain.
Key: EWG = Excessive Weight Gain; NWG = Normal Weight Gain; IPFD = Intrapartal Fetal Death; LGA = Large for Gestational Age.
range were matched against women who gained weight above their respective recommended ranges for outcomes. Although these guidelines were made for the American population, other countries including under developed countries like South Africa, Brazil, Malaysia and China have started adopting them [12,13].
Maternal BMI and gestational weight gain reflect nutritional status before and during pregnancy. Some evidence has considered abnormal weight gain to have a significant relationship with poor pregnancy outcomes [14].
For instance, our data indicates that, not up to half the subjects 91 (41.6%) had prepregnancy BMIs between 18.5 - 24.9 kg/m2 considered as normal. Furthermore, 43.8% of women with normal BMIs did not ultimately achieve the recommended weight gain. Abnormal weight gain was also seen in more than half of the obese women (BMIs ≥ 30 kg/m2). Overall, there was no significant difference between the initial BMIs and percentage of desired gestational weight gain as has been reported earlier in Iran [14].
Our results showed that weight gain in pregnancy was lower when pre-pregnancy BMI was higher. In particular, women who were overweight or obese before pregnancy generally gained less weight during pregnancy and very few had excessive weight gain. We also found out that underweight was strongly associated with excess weight gain. It has been described that maternal weight gain during pregnancy decreased with increasing pre-pregnancy BMI [15]. This relationship has been well established but the reason for this is not obvious. It can be speculated that medical management and/or personal efforts for not gaining too much weight could be responsible for a part of this association. However, during pregnancy, fat is stored to secure energy supply during fetal growth and lactation. In obese women, no additional storage is necessary, which suggests that pregnancy weight gain could be restricted because of physiological mechanisms [16].
Since the amount of total weight gain is widely variable among women with good pregnancy outcomes, and the perinatal outcomes of interest are multifactorial in origin, it should not be expected for weight gain alone to be utilized as a perfect diagnostic or screening tool [17].
Previous studies have shown that pregnancy weight gain within the ranges recommended by IOM is associated with the best outcomes for both mothers and infants [18-21]. On the contrary, some studies that retrospectively assessed the sensitivity and specificity of this indicator, concluded that maternal weight gain alone is neither a sensitive nor a specific predictor of poor pregnancy outcomes.
Studies in China found out that excess weight gain was strongly associated with hypertensive disorders. But then, the cause and association for this effect is complicated. This is because preeclampsia causes edema which could contribute to increase weight gain and also, increase weight can cause preeclampsia. Erratically, high weight gain is likely to represent excessive fluid retention but the relationship between increased GWG and onset of hypertension is unclear.
Induced labor was associated with excessive weight gain in this study. In the Agency for Healthcare Research and Quality (AHRQ) review, five studies were reviewed related to an association between GWG and induction of labor. The strength of the evidence from these studies was rated weak for an association between high GWG and labor induction or failure of labor induction.
There was a significant increase in the length of labor with higher weight gain. Women with excessive weight gain had more primary cesarean deliveries as described in literature [22]. There are many theories to account for the increased risk of cesarean delivery in women who gain too much weight during pregnancy. This may be due to fetal macrosomia, increase in soft tissue in the pelvis narrowing the pelvic outlet, the negative effect of poor pelvic and abdominal tone on fetal position, the relatively higher dosages of oxytocin required for induction and or maintenance of adequate contractile forces, the technical difficulty in fetal monitoring, as well as the concerns by the physician about shoulder dystocia [22]. Excessive weight gain was also significantly associated with increased risk of post partum hemorrhage.
5. CONCLUSIONS
Gestational weight gain above IOM guidelines was not significantly associated with increased incidence of intrapartum fetal death (IPFD) in this study. We found a significant relationship between fetal distress and excess weight gain. There have been no prospective studies on the effect of weight gain on fetal heart rate patterns. It is also not well known if maternal habits or excessive weight gain causes a redistribution of placental blood or change in the normal physiological of pregnancy to affect fetal heart rate patterns.
There was a 4-fold odds in mal-presentation among women who gained excess weight. This could be explained by macrosomia and large for gestational age (LGA) found in them [22-24].
REFERENCES
- IOM (1990) Part I, weight gain. National Academy Press. Washington DC.
- Sobngwi, E., Mbanya, J., Unwin, N., Kengne, A., Fezeu, L., Minkoulou, E., et al. (2002) Physical activity and its relationship with obesity, hypertension and diabetes in urban and rural Cameroon. International Journal of Obesity, 26, 1009-1016.
- Yee, L.M., Cheng, Y.W., Inturrisi, M. and Caughey, A.B. (2011) Effect of gestational weight gain on perinatal outcomes in women with type 2 diabetes mellitus using the 2009 Institute of Medicine guidelines. American Journal of Obstetrics & Gynecology, 205, 257-261. http://dx.doi.org/10.1016/j.ajog.2011.06.028
- Ellen, A., Mireille, N., Poppel, V., Jacob, C., Seidell, P., Willem, M., et al. (2009) Correlates of Absolute and Excessive Weight Gain During Pregnancy. Journal of Women’s Health, 18, 1559-1560. http://dx.doi.org/10.1089/jwh.2008.1275
- Martin, J, Hamilton, B, Menacker, F, Sutton, P, Mathews, T. (2005) Preliminary births for 2004: Infant and maternal health. Health E-Stats, National Center for Health Statistics, Hyattsville.
- Eastman, N. and Jackson, E. (1968) Weight relationships in pregnancy I. The bearing of maternal weight gain and pre-pregnancy weight on birth weight in full term pregnancies. Obstetrical & Gynecological Survey, 23, 1003- 1025. http://dx.doi.org/10.1097/00006254-196811000-00001
- Rasmussen, K.M, Patrick, M. and Catalano, A.L. (2009) New guidelines for weight gain during pregnancy: What obstetricians/gynecologists should know. Current Opinion in Obstetrics and Gynecology, 21, 521-526. http://dx.doi.org/10.1097/GCO.0b013e328332d24e
- IOM (2009) Weight Gain during Pregnancy: Reexamining the Guidelines.
- Nana, P., Wandji, J., Fomulu, J., Mbu, R., Tonye, R., Ako, S., et al. (2009) Materno-foetal outcome of labor in obese women in Yaounde, Cameroon. Clinical Mother Child Health, 6, 989-993.
- Kamadjeu, R., Edwards, R., Atanga, J., Kiawi, E., Unwin, N. and Mbanya, J.-C. (2006) Anthropometry measures and prevalence of obesity in the urban adult population of Cameroon: An update from the Cameroon Burden of Diabetes Baseline Survey. BMC Public Health, 6, 228. http://dx.doi.org/10.1186/1471-2458-6-228
- Joan, M., Crane, M., Joanne, W., Murphy, P., Lorraine, B. and Donna, H. (2009) The effect of gestational weight gain by body mass index on maternal and neonatal outcomes. Journal of Obstetrics and Gynecology, 31, 28-35.
- Kruger, H. (2005) Maternal anthropometry and pregnancy outcomes: A proposal for the monitoring of pregnancy weight gain in outpatient clinics in South Africa. Curationis, 28, 40-49. http://dx.doi.org/10.4102/curationis.v28i4.1012
- Liu, Y., Dai, W., Dai, X. and Li, Z. (2012) Prepregnancy body mass index and gestational weight gain with the outcome of pregnancy: A 13-year study of 292,568 cases in China. Archives of Gynecology and Obstetrics, 286, 905-911. http://dx.doi.org/10.1007/s00404-012-2403-6
- Abrams, B., Altman, S. and Pickett, K. (2000) Pregnancy weight gain: Still controversial. The American Journal of Clinical Nutrition, 71, 1233S-1241S.
- Barbara, H., Valérie, G., Anne, F., Monique, K., Bernard, F., Michel, S., et al. (2012) Pre-pregnancy body mass index and weight gain during pregnancy: Relations with gestational diabetes and hypertension, and birth outcomes. Maternal and Child Health Journal, 16, 355-363. http://dx.doi.org/10.1007/s10995-011-0741-9
- Nohr, E., Vaeth, M., Baker, J., Sorensen, T., Olsen, J. and Rasmussen, K. (2009) Pregnancy outcomes related to gestational weight gain in women defined by their body mass index, parity, height, and smoking status. The American Journal of Clinical Nutrition, 90, 1288-1294. http://dx.doi.org/10.3945/ajcn.2009.27919
- Ehrenberg H., Milluzzi C. and Mercer B. (2002) Prevalence of maternal obesity in an urban center. American Journal of Obstetrics & Gynecology, 187, 1189-1193. http://dx.doi.org/10.1067/mob.2002.127125
- Ogunyemi, D., Hullett, S., Leeper, J. and Risk, A. (1998) Prepregnancy body mass index, weight gain during pregnancy, and perinatal outcome in a rural black population. Journal of Maternal-Fetal and Neonatal Medicine, 7, 190-193. http://dx.doi.org/10.1002/(SICI)1520-6661(199807/08)7:4<190::AID-MFM5>3.0.CO;2-D
- Boten, P. (2008) Effets du Gains de Poids Total pendant la Grossesse sur la Mere et le Fetus. Faculte de Medicine et des Sciences Biomedicales, 66-67.
- Kathleen, M. and Rasmussen, A.L.Y. (2009) Weight Gain during Pregnancy: Reexamining the Guidelines.
- Yee, L., Cheng, Y., Inturrisi, M. and Caughey, A. (2011) Effect of gestational weight gain on perinatal outcomes in women with type 2 diabetes mellitus using the 2009 Institute of Medicine guidelines. American Journal of Obstetrics and Gynecology, 205, e1-e6.
- Magriples, U., Kershaw, T., Rising, S., Westdahl, C. and Ickovics, J. (2009) The effects of obesity and weight gain in young women on obstetric outcomes. American Journal of Perinatology, 26, 365-371. http://dx.doi.org/10.1055/s-0028-1110088
- Yekta, Z., Ayatollahi, H., Porali, R. and Farzin, A. (2006) The effect of pre-pregnancy body mass index and gestational weight gain on pregnancy outcomes in urban care settings in Urmia-Iran. BMC Pregnancy Childbirth, 6, 15. http://dx.doi.org/10.1186/1471-2393-6-15
- Paul, C., et al. (1996) Williams Obstetrics. 20th Edition, Appleton & Lange, East Norwalk.
NOTES
*Corresponding author.

