Open Journal of Psychiatry
Vol.3 No.4(2013), Article ID:36503,7 pages DOI:10.4236/ojpsych.2013.34037
Dissociative decline of spatial learning and recall with aging in male CD-1 mice
![]()
Department of Mental Health & Old Age Psychiatry, Psychiatry II, Ulm University, Günzburg, Germany
Email: *matthias.riepe@uni-ulm.de
Copyright © 2013 Katharina L. Neumeister et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 30 May 2013; revised 29 June 2013; accepted 5 July 2013
Keywords: Spatial Learning; Maze; CD-1; Middle-Aged; Old; Recall; Retrieval; Acquisition; Encoding; Memory; Running Speed
ABSTRACT
On aging, spatial memory declines to some degree in both, rodents and humans. It is unknown, however, whether aging brings about a decline of encoding and retrieval of spatial information in parallel. The present study examined spatial encoding and retrieval in male CD-1 mice at 4, 9, and 12 months of age in a complex dry-land maze. The total time to reach the goal zone was age-dependent favoring mice at 4 months of age. We showed previously that moving time represents encoding of spatial information and resting time represents recall of previously learned spatial information. The average moving time decreased from 69.8 ± 5.3 s (mean ± SEM), 69.7 ± 8.0 s, and 78.9 ± 4.9 s to 17.0 ± 2.3 s (p < 0.001 ), 24.7 ± 2.7 s (p < 0.001), and 31.0 ± 3.5 s (p < 0.001) at 4, 9, and 12 months of age, respectively. The average resting time decreased from 34.9 ± 5.6 s, 22.2 ± 4.2 s, and 41.7 ± 5.3 s to 3.6 ± 1.2 s (p < 0.001), 5.3 ± 1.8 s (p = 0.009), and 22.7 ± 4.9 s (p = 0.007) at 4, 9, and 12 months of age, respectively. We conclude that age-related deficits of spatial memory in mice manifest with an encoding deficit prior to a retrieval deficit.
1. INTRODUCTION
Spatial learning in experimental animals is assessed using mazes since the beginning of the 19th century [1]. While dry-land mazes have been used in early studies, the use of water mazes has become the mainstay of research on spatial memory in rodents for about 30 years [2-5]. Recently, it has been argued that use of dry-land mazes may offer additional insight into the mechanisms of spatial learning. Thus, the number of animal studies using dry-land mazes has increased over the last couple of years. Spatial learning in water mazes is subject to the influence of non-cognitive processes such as fear which may blur the insight into the cognitive processes at work [6,7]. Furthermore, use of dry-land mazes allows separation of different cognitive processes performed during resting and moving time in the maze [8]. This is not possible in water mazes since animals need to move continuously.
On moving around in an unknown or familiar environment, the localization information is encoded [8-10] and animals build an internal representation of the space around them, and the cognitive map [11,12]. During resting times, animals update and recall the localization information and compare currently experienced sequence information with stored sequences [8,13,14].
Aging is associated with mild impairments in spatial learning and memory in humans [15-18] and rodents [3,19,20]. In humans, aged adults are impaired in multiple aspects of spatial memory, from navigating through a recently learned environment [15,16] to recalling where an item was located [17,18]. Likewise, aged rats are impaired in encoding [21-25] and recall [26] of spatial information.
Encoding and retrieval of spatial information is under the control of the frontal cortex [27-32] and in addition requires integrity of the hippocampal region and the medio-temporal cortex [33-35]. Both, frontal and mediotemporal networks are subject to age-associated changes in humans [32,36,37]. Likewise, humans and rodents exhibit age-related alterations in the hippocampus [2,38- 44]. The onset of these age-associated changes is assumed to be in middle-age in rodents [5,45,46].
It was the goal of the present study to assess age-related spatial memory performance in a complex dryland maze in male CD-1 mice.
2. MATERIALS AND METHODS
All animal experiments were performed according to institutional guidelines and the “Principles of laboratory animal care” (NIH publication No. 86-23, revised 1985).
2.1. Animals
Three groups of male CD-1 mice (Charles River) from different age cohorts, i.e., young (4 months, n = 9), middle-aged (9 months, n = 4), and aged (12 months, n = 9) mice were used in this study. Previous reports in humans [47] and experimental animals [48] show a clear-cut gender dependence of spatial orientation with males relying on hippocampal integrity to a much greater extent than females. In addition, hippocampus-dependent spatial learning in females may be influenced by the stage of the estrus cycle [49]. At the given age of the transgenic animals used in this study, hippocampal impairment is predominant. Therefore, the present study was restricted to male animals. Four or five animals were housed in a cage and were maintained on a 12 h light/dark cycle in a temperature (22˚C ± 2˚C) and humidity (55% ± 5%) controlled room, with water ad libitum available.
2.2. Maze and Behavioural Testing
To assess learning we used a complex maze that has been described previously [50-52] (Figure 1). The complex
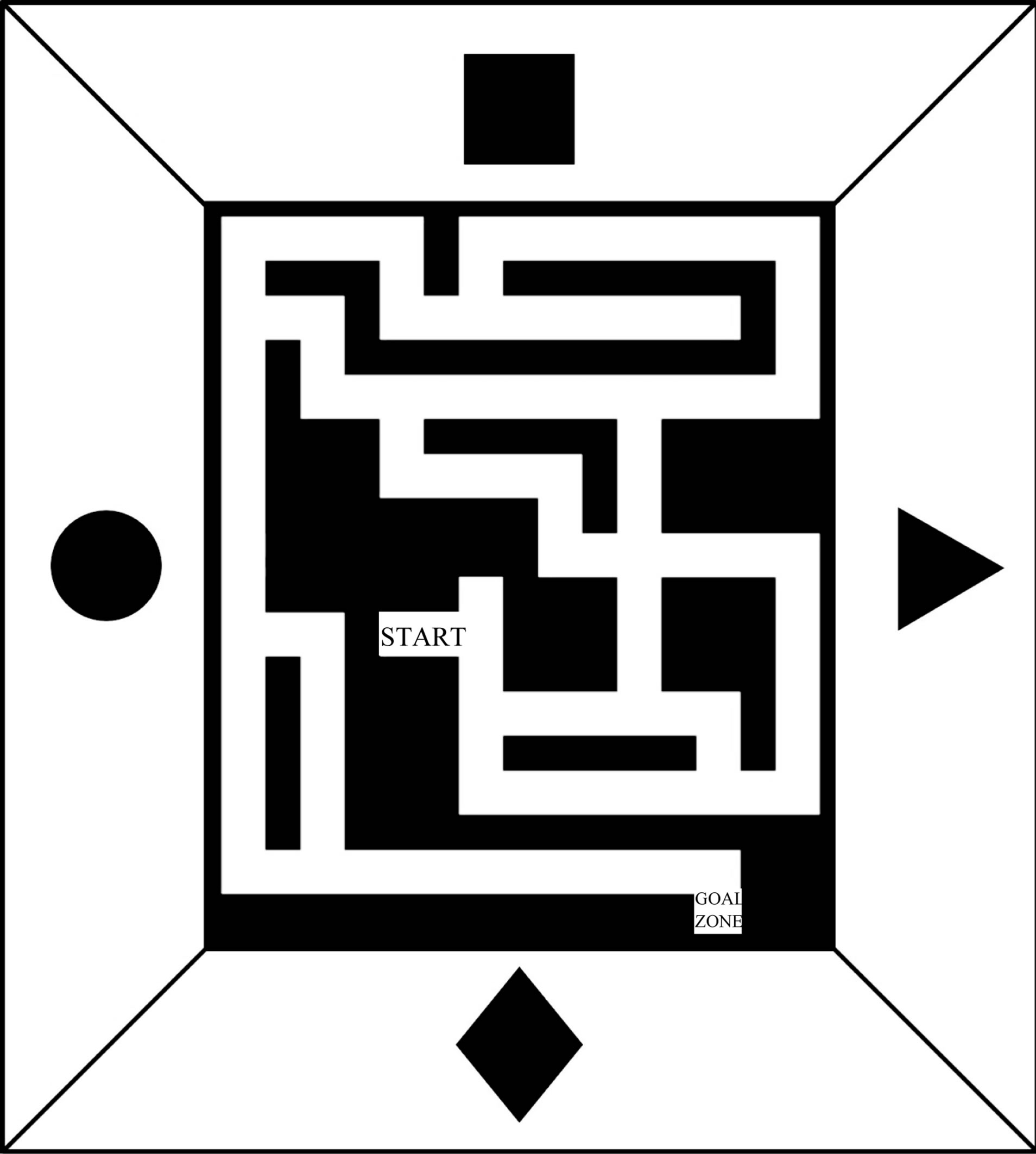
Figure 1. Aerial view of the complex maze with curtain and geometrical cues. Animals were placed in the start zone. A video-tracking system (cf. methods) registered the location of the animals’ position at a frequency of 1 Hz.
maze was constructed of gray rigid PVC and the overall size was 200 × 180 cm with 10 cm high, opaque walls and 10 cm wide, red alleys the mice traveled on. The maze has a starting point, crossings, t-junctions, blind alley and a goal zone and was surrounded by a curtain with optical geometric cues at four cardinal directions. Animals were trained five times a day for eight consecutive days. They had a maximum time of 300 s to find the goal zone, where they were rewarded with a food pellet. The behavioural observation was made between 10:00 AM and 4:00 PM. Experiments were recorded by a tracking system (Multitrack, Accuscan, USA).
2.3. Statistical Analysis
All statistical analyses were carried out using the statistics program SPSS (SPSS 17.0 for Windows, SPSS Inc. IL 60606, Chicago). Statistical significance was accepted at p < 0.05.
3. RESULTS
To assess spatial learning we analyzed total time to reach the goal zone. A two-way ANOVA with Fisher LSD multiple comparison testing and factors trial and age shows a significant effect of trial (F39,1120 = 6.937, p < 0.001) and age (F2,1120 = 45.448, p < 0.001) (Figure 2, Table 1).
Previously we argued, that separate analysis of moving and resting time allows distinction of encoding the spatial information and retrieving it [8]. Hence, we analyzed moving and resting time in a similar fashion. All age-groups improved their performance during repeated exposure to the complex maze as shown by a significant effect of trial on moving time (two-way ANOVA; F39,1120 = 9.206, p < 0.001) and resting time (two-way ANOVA; F39,1120 = 2.797, p < 0.001) (Figure 2). Likewise, a significant effect of age was found for moving time (twoway ANOVA; F2,1120 = 27.251, p < 0.001) and resting time (two-way ANOVA; F2,1120 = 41.994, p < 0.001) (Table 1). Compared to mice at an age of 4 months, moving time was increased in animals at the age of 9 and 12 months. Resting time, however, did not increase until the age of 12 months.
To rule out that total or moving time are modulated by speed in an age-dependent manner we analyzed running speed. Running speed was lowest in animals at 9 months of age and higher at both, 4 and 12 months. To analyze the effect of age and trial on running speed we performed a two-way ANOVA with factors trial and age and found significant effects for both, trial (F39,1120 = 15.570, p < 0.001) and age (F2,1120 = 32.193, p < 0.001) (Figure 3, Table 1). Regardless of this effect of age on speed, the length of the path that animals use on their way to the goal zone shows an age-dependent increase (Figure 3, Table 1). This indicates that the cognitive processes of
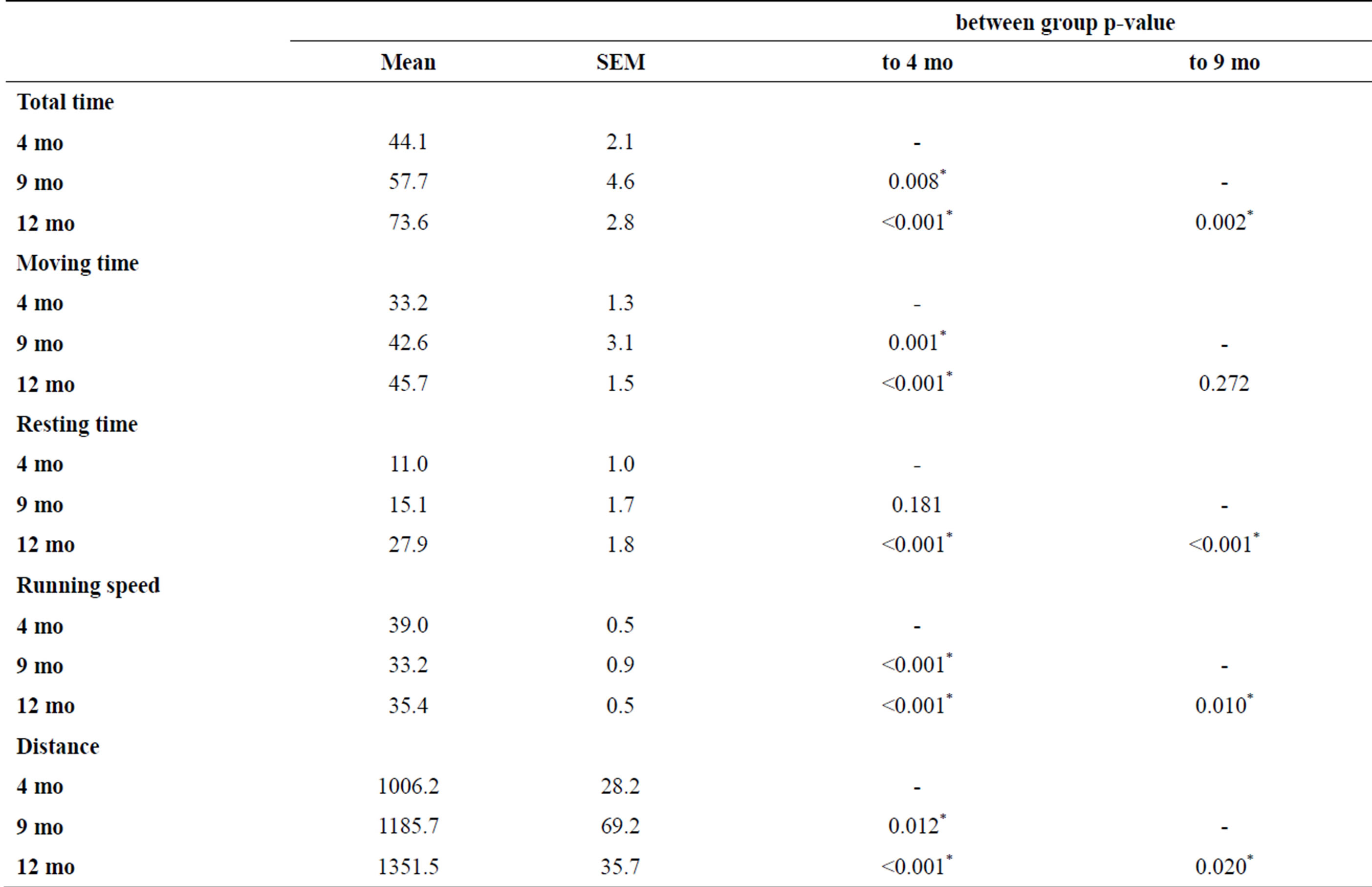
Table 1. Two-way ANOVA for male CD-1 mice in age of 4 months (4 mo), 9 months (9 mo), and 12 months (12 mo) in the complex maze. Post hoc multiple comparison testing (Fisher LSD) with p-values for comparison of differences between groups. *indicate statistical significance.
encoding and retrieval of the spatial information is more successful in 4 than in 12 months old animals.
4. DISCUSSION
The present study examined spatial learning in a complex dry-land maze in mice at 4, 9, and 12 months of age. Overall, spatial memory performance declined with aging. This finding is in good harmony with a rich literature on decline of spatial memory as assessed with different setups of water mazes [2-5] and dry-land mazes [19,20,53]. To our knowledge, separate analyses of different cognitive processes while the animals rest and move have only been performed, recently [8-10,13,14].
4.1. Running Speed
Running speed is a confounding variable in using escape latency from start zone to the goal zone in dry-land mazes. In harmony with a previous study in female CD-1 mice [54], we observed a u-shape-like running speed with highest speed in mice at 4 and 12 months of age and slowest speed in animals 9 months of age. Regardless of the modulation of moving and total time by speed we also observed a difference in the length of the path from start zone to goal zone being lowest in the youngest animals and highest in the oldest animals. We interpret the latter finding such as to show that the overall performance for spatial learning declines steadily with aging.
4.2. Encoding and Recall of Spatial Information
Spatial information is encoded while animals move in the maze [8-10]. Older animals in the present study encoded the spatial map of the maze less successfully than younger animals and needed more time and a longer path length to reach the goal zone. This is in good accordance with the literature showing a decline of encoding performance in middle-aged and aged humans [15,16,55,56] and experimental animals [21-25]. While the spatial encoding performance is worse in 9 and 12 months old animals compared to 4 months old animals, no further decline is observed between 9 and 12 months.
Retrieval of spatial information in the complex maze take place predominantly during resting time [8,13,14]. Resting time increases with age in the present study. This is in good accordance with the literature showing a decline of retrieval performance in aged humans [17,18] and experimental animals [26]. In contrast to encoding of
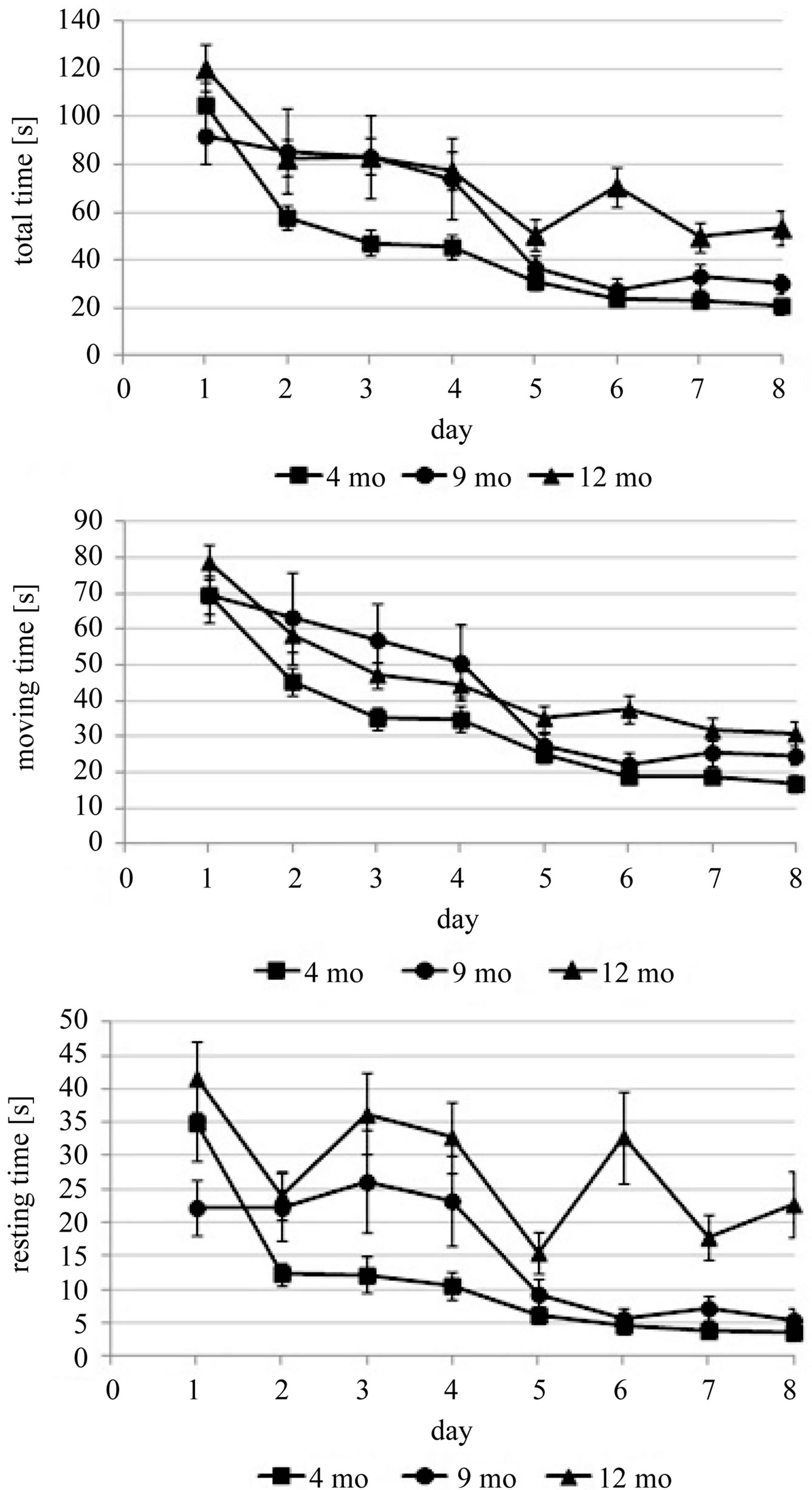
Figure 2. Learning curves for total, moving and resting time that male animals needed to perform the complex maze over the whole experimental period. Values represent means ± standard errors for male 4 (squares), 9 (circles), and 12 months old (triangles) mice.
spatial information with similar performance of 9 and 12 months old animals there is further decline of spatial retrieval performance between 9 and 12 months.
4.3. Sequence of Decline of Spatial Learning Processes with Aging—A Synthesis Considering the Literature
The effects of age on memory performance are determined by both the level of information processing at the encoding phase and the degree of environmental support during the retrieval phase. Possible memory mechanism failures in the elderly include poor encoding or acquisition of new information, faulty storage or retention of
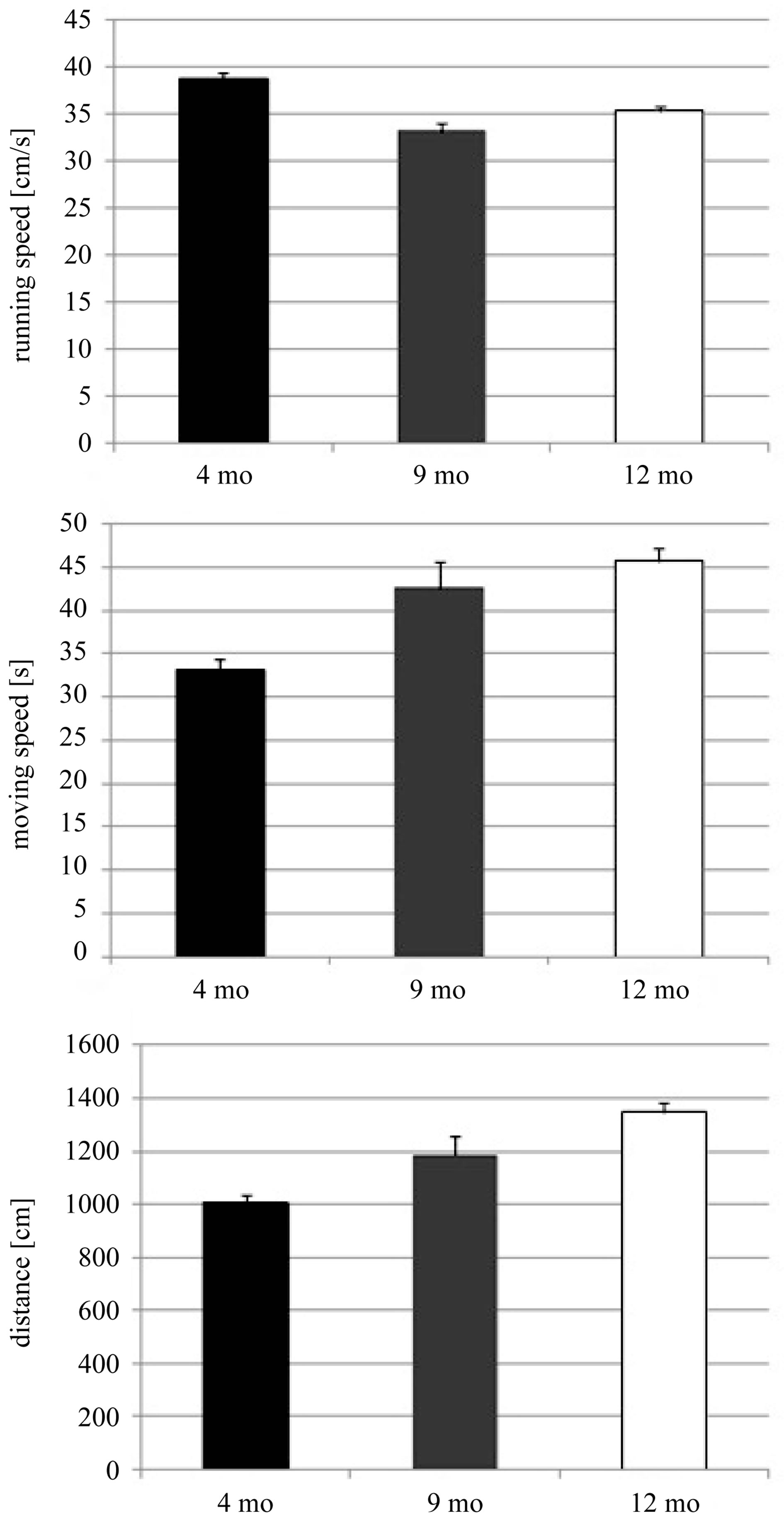
Figure 3. Average running speed, total time and distance that male animals needed to perform the complex maze over the whole experimental period using two-way ANOVA with Fisher LSD multiple comparison testing. Values represent means ± standard errors for male 4 (black bar, 4 mo), 9 (gray bar, 9 mo), and 12 months old (white bar, 12 mo) mice.
information in memory, and/or an inability to decode or retrieve material that has been adequately encoded and stored [57].
Overall, we observe a progressive decline of spatial retrieval performance with increasing age from 4 and 9 to 12 months in the present study. In contrast, spatial memory encoding performance declines from 4 to 9 months but remains alike between 9 and 12 months. We interpret this pattern such as to indicate that spatial memory encoding performance reaches a floor level already at or prior to 9 months of age in male CD-1 mice in the current paradigm while spatial memory retrieval is less sensitive to aging and further decline of performance is observed between 9 and 12 months in the current paradigm. This interpretation is supported by previously reported results on encoding and retrieval in humans with assessment of source memory, i.e. the ability to remember contextual information present at the moment an event occurs.
The failure to retrieve memory content correctly may arise from a failure to encode the representation sufficiently strongly [58]. “Encoding” can be assessed by means of investigating “subsequent memory effects” [59]. Source memory accuracy differed between young, middle-aged, and old adults [60] for reasons of less efficient encoding mechanisms. Brain regions which participate in the successful encoding of source information for persons in their 50 s are different than those utilized by young adults and change between middle age and old age [60].
In humans, recall performance between young and middle-aged groups has been reported to be alike while it declined in old age. Middle-aged adults may have preserved retrieval capacities, and/or enhanced encoding capacities. Old subjects enhanced but failed to normalize their retrieval performance, suggesting only partially successful compensatory mechanisms [61]. In verbal recall the old subjects scored lower than both the young and middle-aged subject with young and middle-aged subjects performing alike [62,63].
5. CONCLUSION
We conclude that age-related deficits of spatial memory manifest with an encoding deficit prior to a retrieval deficit in aging rodents. Future studies need to further investigate the neurobiological and neuroanatomical reasons for this age-specific decline in different spatial memory processes. Since there are indications towards sex differences in spatial memory [47-49], female mice should be included in follow-up studies.
6. ACKNOWLEDGEMENTS
This work was supported by a grant from the Deutsche Forschungsgemeinschaft (M. W. R. Ri 583/2-3).
REFERENCES
- Small, W.S. (1900) An experimental study of the mental processes of the rat. The American Journal of Psychology, 11, 133-165. http://dx.doi.org/10.2307/1412267
- Fischer, W., Chen, K.S., Gage, F.H. and Bjorklund, A. (1992) Progressive decline in spatial learning and integrity of forebrain cholinergic neurons in rats during aging. Neurobiology of Aging, 13, 9-23. http://dx.doi.org/10.1016/0197-4580(92)90003-G
- Frick, K.M., Baxter, M.G., Markowska, A.L., Olton, D.S. and Price, D.L. (1995) Age-related spatial reference and working memory deficits assessed in the water maze. Neurobiology of Aging, 16, 149-160. http://dx.doi.org/10.1016/0197-4580(94)00155-3
- Matzel, L.D., Light, K.R., Wass, C., Colas-Zelin, D., Man-Brice, A., Waddel, A.C. and Kolata, S. (2011) Longitudinal attentional engagement rescues mice from agerelated cognitive declines and cognitive inflexibility. Learning & Memory, 18, 345-356. http://dx.doi.org/10.1101/lm.2034711
- Shen, J. and Barnes, C.A. (1996) Age-related decrease in cholinergic synaptic transmission in three hippocampal subfields. Neurobiology of Aging, 17, 439-451. http://dx.doi.org/10.1016/0197-4580(96)00020-6
- Ehninger, D. and Kempermann, G. (2006) Paradoxical effects of learning the Morris water maze on adult hippocampal neurogenesis in mice may be explained by a combination of stress and physical activity. Genes, Brain and Behavior, 5, 29-39. http://dx.doi.org/10.1111/j.1601-183X.2005.00129.x
- Harrison, F.E., Hosseini, A.H. and McDonald, M.P. (2009) Endogenous anxiety and stress responses in water maze and Barnes maze spatial memory tasks. Behavioural Brain Research, 198, 247-251. http://dx.doi.org/10.1016/j.bbr.2008.10.015
- Neumeister, K.L. and Riepe, M.W. (2012) Synergistic effects of antidementia drugs on spatial learning and recall in the APP23 transgenic mouse model of Alzheimer’s disease. Journal of Alzheimer’s Disease, 30, 245- 251. http://dx.doi.org/10.3233/JAD-2012-111643
- Lee, I., Rao, G. and Knierim, J.J. (2004) A double dissociation between hippocampal subfields: differential time course of CA3 and CA1 place cells for processing changed environments. Neuron, 42, 803-815. http://dx.doi.org/10.1016/j.neuron.2004.05.010
- Hasselmo, M.E., Bodelon, C. and Wyble, B.P. (2002) A proposed function for hippocampal theta rhythm: Separate phases of encoding and retrieval enhance reversal of prior learning. Neural Computation, 14, 793-817. http://dx.doi.org/10.1162/089976602317318965
- Tolman, E.C. (1948) Cognitive maps in rats and men. Psychological Review, 55, 189-208. http://dx.doi.org/10.1037/h0061626
- O’Keefe, J. and Nadel, L. (1978) The hippocampus as a cognitive map. Oxford University Press, Oxford.
- Carr, M.F., Jadhav, S.P. and Frank, L.M. (2011) Hippocampal replay in the awake state: A potential substrate for memory consolidation and retrieval. Nature Neuroscience, 14, 147-153. http://dx.doi.org/10.1038/nn.2732
- Karlsson, M.P. and Frank, L.M. (2009) Awake replay of remote experiences in the hippocampus. Nature Neuroscience, 12, 913-918. http://dx.doi.org/10.1038/nn.2344
- Wilkniss, S.M., Jones, M.G., Korol, D.L., Gold, P.E. and Manning, C.A. (1997) Age-related differences in an ecologically based study of route learning. Psychology and Aging, 12, 372-375. http://dx.doi.org/10.1037/0882-7974.12.2.372
- Newman, M.C. and Kaszniak, A.W. (2000) Spatial memory and aging: Performance on a human analog of the morris water maze. Aging, Neuropsychology, and Cognition, 7, 86-93. http://dx.doi.org/10.1076/1382-5585(200006)7:2;1-U;FT086
- Uttl, B. and Graf, P. (1993) Episodic spatial memory in adulthood. Psychology and Aging, 8, 257-273. http://dx.doi.org/10.1037/0882-7974.8.2.257
- Chalfonte, B.L. and Johnson, M.K. (1996) Feature memory and binding in young and older adults. Memory & Cognition, 24, 403-416. http://dx.doi.org/10.3758/BF03200930
- Barnes, C.A. (1979) Memory deficits associated with senescence: A neurophysiological and behavioral study in the rat. Journal of Comparative & Physiological Psychology, 93, 74-104. http://dx.doi.org/10.1037/h0077579
- Oler, J.A. and Markus, E.J. (1998) Age-related deficits on the radial maze and in fear conditioning: Hippocampal processing and consolidation. Hippocampus, 8, 402-415. http://dx.doi.org/10.1002/(SICI)1098-1063(1998)8:4<402::AID-HIPO8>3.0.CO;2-I
- Tanila, H., Sipila, P., Shapiro, M. and Eichenbaum, H. (1997) Brain aging: Impaired coding of novel environmental cues. The Journal of Neuroscience, 17, 5167-5174.
- Oler, J.A. and Markus, E.J. (2000) Age-related deficits in the ability to encode contextual change: A place cell analysis. Hippocampus, 10, 338-350. http://dx.doi.org/10.1002/1098-1063(2000)10:3<338::AID-HIPO14>3.0.CO;2-Y
- Wilson, I.A., Ikonen, S., McMahan, R.W., Gallagher, M., Eichenbaum, H. and Tanila, H. (2003) Place cell rigidity correlates with impaired spatial learning in aged rats. Neurobiology of Aging, 24, 297-305. http://dx.doi.org/10.1016/S0197-4580(02)00080-5
- Wilson, I.A., Ikonen, S., Gureviciene, I., McMahan, R.W., Gallagher, M., Eichenbaum, H. and Tanila, H. (2004) Cognitive aging and the hippocampus: How old rats represent new environments. The Journal of Neuroscience, 24, 3870-3878. http://dx.doi.org/10.1523/JNEUROSCI.5205-03.2004
- Wilson, I.A., Ikonen, S., Gallagher, M., Eichenbaum, H. and Tanila, H. (2005) Age-associated alterations of hippocampal place cells are subregion specific. The Journal of Neuroscience, 25, 6877-6886. http://dx.doi.org/10.1523/JNEUROSCI.1744-05.2005
- Barnes, C.A., Suster, M.S., Shen, J. and McNaughton, B.L. (1997) Multistability of cognitive maps in the hippocampus of old rats. Nature, 388, 272-275. http://dx.doi.org/10.1038/40859
- Owen, A.M., Milner, B., Petrides, M. and Evans, A.C. (1996) Memory for object features versus memory for object location: A positron-emission tomography study of encoding and retrieval processes. Proceedings of the National Academy of Sciences of USA, 93, 9212-9217. http://dx.doi.org/10.1073/pnas.93.17.9212
- McDermott, K.B., Buckner, R.L., Petersen, S.E., Kelley, W.M. and Sanders, A.L. (1999) Setand code-specific activation in frontal cortex: An fMRI study of encoding and retrieval of faces and words. Journal of Cognitive Neuroscience, 11, 631-640. http://dx.doi.org/10.1162/089892999563698
- Tulving, E., Kapur, S., Craik, F.I., Moscovitch, M. and Houle, S. (1994) Hemispheric encoding/retrieval asymmetry in episodic memory: Positron emission tomogramphy findings. Proceedings of the National Academy of Sciences of USA, 91, 2016-2020. http://dx.doi.org/10.1073/pnas.91.6.2016
- Nyberg, C.L., Cabeza, R. and Tulving, E. (1996) PET studies of encoding and retrieval: The HERA model. Psychonomic Bulletin and Review, 3, 135-148. http://dx.doi.org/10.3758/BF03212412
- Cabeza, R., Grady, C.L., Nyberg, L., McIntosh, A.R., Tulving, E., Kapur, S., Jennings, J.M., Houle, S. and Craik, F.I. (1997) Age-related differences in neural activity during memory encoding and retrieval: A positron emission tomography study. The Journal of Neuroscience, 17, 391-400.
- Buckner, R.L. (2004) Memory and executive function in aging and AD: Multiple factors that cause decline and reserve factors that compensate. Neuron, 44, 195-208. http://dx.doi.org/10.1016/j.neuron.2004.09.006
- Martin, A., Haxby, J.V., Lalonde, F.M., Wiggs, C.L. and Ungerleider, L.G. (1995) Discrete cortical regions associated with knowledge of color and knowledge of action. Science, 270, 102-105. http://dx.doi.org/10.1126/science.270.5233.102
- Martin, A., Wiggs, C.L., Ungerleider, L.G. and Haxby, J.V. (1996) Neural correlates of category-specific knowledge. Nature, 379, 649-652. http://dx.doi.org/10.1038/379649a0
- Kapur, S., Craik, F.I., Tulving, E., Wilson, A.A., Houle, S. and Brown, G.M. (1994) Neuroanatomical correlates of encoding in episodic memory: Levels of processing effect. Proceedings of the National Academy of Sciences of USA, 91, 2008-2011. http://dx.doi.org/10.1073/pnas.91.6.2008
- Lamar, M., Yousem, D.M. and Resnick, S.M. (2004) Age differences in orbitofrontal activation: An fMRI investigation of delayed match and nonmatch to sample. Neuroimage, 21, 1368-1376. http://dx.doi.org/10.1016/j.neuroimage.2003.11.018
- Raz, N. (2000) Aging of the brain and its impact on cognitive performance: Integration of structural and functional findings. In: Craik, F.I.M. and Salthouse, T.A., Eds., The Handbook of Aging and Cognition, Lawrence Erlbaum Associates, Mahwah, 1-90.
- Flood, D.G. and Coleman, P.D. (1988) Neuron numbers and sizes in aging brain: Comparisons of human, monkey, and rodent data. Neurobiology of Aging, 9, 453-463. http://dx.doi.org/10.1016/S0197-4580(88)80098-8
- West, M.J., Coleman, P.D., Flood, D.G. and Troncoso, J.C. (1994) Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. The Lancet, 344, 769-772. http://dx.doi.org/10.1016/S0140-6736(94)92338-8
- Barnes, C.A. (1988) Aging and the physiology of spatial memory. Neurobiology of Aging, 9, 563-568. http://dx.doi.org/10.1016/S0197-4580(88)80114-3
- Gallagher, M. and Pelleymounter, M.A. (1988) Spatial learning deficits in old rats: A model for memory decline in the aged. Neurobiology of Aging, 9, 549-556. http://dx.doi.org/10.1016/S0197-4580(88)80112-X
- Smith, T.D., Calhoun, M.E. and Rapp, P.R. (1999) Circuit and morphological specificity of synaptic change in the aged hippocampal formation. Neurobiology of Aging, 20, 357-358.
- Raz, N., Lindenberger, U., Rodrigue, K.M., Kennedy, K.M., Head, D., Williamson, A., Dahle, C., Gerstorf, D. and Acker, J.D. (2005) Regional brain changes in aging healthy adults: General trends, individual differences and modifiers. Cerebral Cortex, 15, 1676-1689. http://dx.doi.org/10.1093/cercor/bhi044
- O’Brien, J.L., O’Keefe, K.M., LaViolette, P.S., DeLuca, A.N., Blacker, D., Dickerson, B.C. and Sperling, R.A. (2010) Longitudinal fMRI in elderly reveals loss of hippocampal activation with clinical decline. Neurology, 74, 1969-1976. http://dx.doi.org/10.1212/WNL.0b013e3181e3966e
- Lynch, G., Rex, C.S. and Gall, C.M. (2006) Synaptic plasticity in early aging. Ageing Research Reviews, 5, 255-280. http://dx.doi.org/10.1016/j.arr.2006.03.008
- Kaczorowski, C.C. and Disterhoft, J.F. (2009) Memory deficits are associated with impaired ability to modulate neuronal excitability in middle-aged mice. Learning Memory, 16, 362-366. http://dx.doi.org/10.1101/lm.1365609
- Gron, G., Wunderlich, A.P., Spitzer, M., Tomczak, R. and Riepe, M.W. (2000) Brain activation during human navigation: Gender-different neural networks as substrate of performance. Nature Neuroscience, 3, 404-408. http://dx.doi.org/10.1038/73980
- Roof, R.L. and Stein, D.G. (1999) Gender differences in Morris water maze performance depend on task parameters. Physiology & Behavior, 68, 81-86. http://dx.doi.org/10.1016/S0031-9384(99)00162-6
- Warren, S.G. and Juraska, J.M. (1997) Spatial and nonspatial learning across the rat estrous cycle. Behavioral Neuroscience, 111, 259-266. http://dx.doi.org/10.1037/0735-7044.111.2.259
- Hellweg, R., Lohmann, P., Huber, R., Kuhl, A. and Riepe, M.W. (2006) Spatial navigation in complex and radial mazes in APP23 animals and neurotrophin signaling as a biological marker of early impairment. Learning Memory, 13, 63-71. http://dx.doi.org/10.1101/lm.2606
- Lohmann, P., Lange-Asschenfeldt, C., Huber, R. and Riepe, M.W. (2006) The stamp of ancestry: Roots of behavioral and neuronal impairment in adulthood. Experimental Neurology, 200, 104-111. http://dx.doi.org/10.1016/j.expneurol.2006.01.019
- Lange-Asschenfeldt, C., Lohmann, P. and Riepe, M.W. (2007) Hippocampal synaptic depression following spatial learning in a complex maze. Experimental Neurology, 203, 481-485. http://dx.doi.org/10.1016/j.expneurol.2006.08.025
- Bach, M.E., Barad, M., Son, H., Zhuo, M., Lu, Y.F., Shih, R., Mansuy, I., Hawkins, R.D. and Kandel, E.R. (1999) Age-related defects in spatial memory are correlated with defects in the late phase of hippocampal long-term potentiation in vitro and are attenuated by drugs that enhance the cAMP signaling pathway. Proceedings of the National Academy of Sciences of the United States of America, 96, 5280-5285. http://dx.doi.org/10.1073/pnas.96.9.5280
- Joyal, C.C., Beaudin, S. and Lalonde, R. (2000) Longitudinal age-related changes in motor activities and spatial orientation in CD-1 mice. Archives of Physiology and Biochemistry, 108, 248-256.
- Friedman, D., Nessler, D. and Johnson Jr., R. (2007) Memory encoding and retrieval in the aging brain. Clinical EEG and Neuroscience, 38, 2-7. http://dx.doi.org/10.1177/155005940703800105
- Ferreira, E.D., Romanini, C.V., Mori, M.A., de Oliveira, R.M. and Milani, H. (2011) Middle-aged, but not young, rats develop cognitive impairment and cortical neurodegeneration following the four-vessel occlusion/internal carotid artery model of chronic cerebral hypoperfusion. The European Journal of Neuroscience, 34, 1131-1140. doi: 10.1111/j.1460-9568.2011.07824.x
- Youngjohn, J.R. and Crook III, T.H. (1993) Learning, forgetting, and retrieval of everyday material across the adult life span. Journal of Clinical and Experimental Neuropsychology, 15, 447-460. http://dx.doi.org/10.1080/01688639308402570
- Stebbins, G.T., Carrillo, M.C., Dorfman, J., Dirksen, C., Desmond, J.E., Turner, D.A., Bennett, D.A., Wilson, R.S., Glover, G. and Gabrieli, J.D. (2002) Aging effects on memory encoding in the frontal lobes. Psychology and Aging, 17, 44-55. http://dx.doi.org/10.1037/0882-7974.17.1.44
- Friedman, D. and Johnson Jr., R. (2000) Event-related potential (ERP) studies of memory encoding and retrieval: A selective review. Microscopy Research and Technique, 51, 6-28. http://dx.doi.org/10.1002/1097-0029(20001001)51:1<6::AID-JEMT2>3.0.CO;2-R
- Cansino, S. (2009) Episodic memory decay along the adult lifespan: A review of behavioral and neurophysiological evidence. International Journal of Psychophysiology, 71, 64-69. http://dx.doi.org/10.1016/j.ijpsycho.2008.07.005
- Kalpouzos, G., Chetelat, G., Landeau, B., Clochon, P., Viader, F., Eustache, F. and Desgranges, B. (2009) Structural and metabolic correlates of episodic memory in relation to the depth of encoding in normal aging. Journal of Cognitive Neuroscience, 21, 372-389. http://dx.doi.org/10.1162/jocn.2008.21027
- Grady, C.L., Springer, M.V., Hongwanishkul, D., McIntosh, A.R. and Winocur, G. (2006) Age-related changes in brain activity across the adult lifespan. Journal of Cognitive Neuroscience, 18, 227-241. http://dx.doi.org/10.1162/jocn.2006.18.2.227
- Zelinski, E.M., Walsh, D.A. and Thompson, L.W. (1978) Orienting task effects on EDR and free recall in three age groups. Journal of Gerontology, 33, 239-245. http://dx.doi.org/10.1093/geronj/33.2.239
NOTES
*Corresponding author.

