Open Journal of Animal Sciences
Vol.07 No.02(2017), Article ID:75446,17 pages
10.4236/ojas.2017.72010
Bovine Endometrial Cells Mount Innate Immune Response to the Intracellular Ligands CL097 and Poly(dA:dT) Indicating Roles against Uterine Viruses
Chike F. Oguejiofor1*, Zhangrui Cheng1, Ali A. Fouladi-Nashta2, D. Claire Wathes1
1Department of Production and Population Health, Royal Veterinary College, North Mymms, UK
2Department of Comparative Biomedical Sciences, Royal Veterinary College, North Mymms, UK

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 23, 2017; Accepted: April 14, 2017; Published: April 17, 2017
ABSTRACT
Uterine infection and endometritis cause infertility and economic losses in the cattle industry. The innate immune response of the endometrium is critical in the elimination of pathogenic organisms that invade the uterus in postpartum cows. This study investigated the response of bovine endometrium to synthetic intracellular ligands which activate innate immunity by stimulating similar receptors to those used to recognise the presence of some viruses. Mixed primary epithelial and stromal cell cultures were treated with 5 μg/ml of CL097 (a TLR7/8 ligand) or 2 μg/ml of poly(dA:dT) (a DNA analogue) for either 6 h or 24 h. Cellular responses were assessed by the mRNA expression of 18 immune-related genes and 3 endogenous reference genes by conventional PCR followed by qRT-PCR from four replicate experiments. Bovine endometrial cells expressed the cytosolic pattern recognition receptors (PRRs) DDX58 (RIG-I), IFIH1 (MDA5) and LRRFIP1 which act as intracellular nucleic acid sensors. Neither ligand altered the expression of the extra-cytosolic pattern recognition receptors (PRRs) TLR3, TLR4, TLR7 or TLR8 whereas poly(dA:dT) treatment increased the expression of IFIH1 and DDX58. Treated cells also responded to CL097 or poly(dA:dT) with a differential up-regulation of genes involved in innate immune response including type I interferon/antiviral response (MX1, IFNAR1), antimicrobial activity (MUC1, SLPI) and cytokine activity (TNF, IL1B, IL8). Bovine endometrial cells therefore express both cytosolic and extra-cytosolic intracellular PRRs and are able to mount an innate immune response upon stimulation with intracellular ligands. This suggests an important role for these cells in the defence against viruses that may be present in the uterus in postpartum cows.
Keywords:
Endometrium, Innate Immunity, Intracellular Ligands, Gene Expression

1. Introduction
Uterine infection and endometritis cause infertility and economic losses in the cattle industry. In addition to several bacteria including Escherichia coli and Trueperella pyogenes [1] , viral pathogens such as bovine herpesvirus 4 (BoHV-4), a dsDNA virus, and bovine viral diarrhoea virus (BVDV), an ssRNA virus, have been associated with reproductive disease and infertility in cows [2] [3] .
The innate immune system of the endometrium thus has an important role in the elimination of pathogens that invade the bovine uterus [4] . Specialised immune cells including macrophages, neutrophils and dendritic cells (DCs) possess pattern recognition receptors (PRRs) such as toll-like receptors (TLRs), nucleotide oligomerisation domain (NOD)-like receptors (NLRs), retinoic acid inducible gene I (RIG-I)-like receptors (RLRs) and cytosolic DNA sensors (CDS). These PRRs have an important role in recognising pathogen-associated molecular patterns (PAMPs) such as lipopolysaccharide (LPS) and nucleic acids (RNA and DNA) from bacterial and viral pathogens, leading to the activation of an innate immune response [5] [6] . Viral RNA or DNA are either recognised outside the cytoplasm of infected cells by TLRs located in intracellular compartments such as endosomes and endolysosomes, or within the cytoplasm by several DNA sensors and the RLRs such as DExD/H-box helicase 58 (DDX58, also known as RIG-I) and interferon induced with helicase C domain 1 (IFIH1, also known as MDA5) [6] [7] .
A number of natural (microbial) ligands/PAMPs and synthetic ligands have been shown to stimulate the same PRRs in different cells. These synthetic ligands have been used to stimulate innate and adaptive immunity against infectious organisms such as viruses and bacteria, and in the study of immune response in a variety of mammalian cells [8] [9] . The synthetic molecule CL097 is a derivative of the imidazoquinoline compounds which, similar to ssRNA, are recognised by TLR7 and TLR8 [10] . Ligand binding to TLR7/8 activates human dendritic cells (DCs) to differentiate and to produce pro-inflammatory or type I IFN cytokines [11] [12] . Poly(dA:dT) is a repetitive dsDNA sequence of poly(dA-dT)∙poly(dA-dT) which functions as a synthetic analogue of B-form DNA. Poly(dA:dT) is recognised directly in the cytoplasm, in a cell-type specific manner, by several DNA sensors including DNA-dependent activator of interferon-regulatory-factors (DAI) [13] , leucine rich repeat (in FLII) interacting protein 1 (LRRFIP1) [14] , absent in melanoma 2 (AIM2) [15] and indirectly by DDX58 (RIG-I) via the activity of the enzyme RNA polymerase III [16] . Activated cells respond by the production of type I IFNs and inflammatory cytokines [17] .
The epithelial and underlying stromal cells of the endometrium constitute the first line of defence to pathogens that invade the uterus in postpartum cows. These cells express PRRs such as TLRs [18] and are able to mount an innate immune response to bacteria [19] or bacterial ligands such as LPS [20] [21] . They also respond directly to viruses including BoHV-4 [22] and BVDV [23] . Although the epithelial and stromal cells are present in large numbers in the bovine endometrium relative to immune cells, there is lack of information on their role in the endometrial response to intracellular pathogens such as viruses or viral PAMPs. Therefore, the purpose of this study was to examine the ability of bovine endometrial epithelial and stromal cells (BEESCs) to recognise and mount an immune response to the synthetic intracellular ligands CL097 and poly(dA:dT). The significance is to provide information on the possible contribution of these cells to endometrial innate immune response to uterine viruses.
2. Materials and Methods
2.1. Bovine Endometrial Cell Culture
Apparently healthy bovine uteri in the early luteal phase of the oestrous cycle were obtained from four heifers slaughtered at the local abattoir. The cycle stage was determined by the presence of a newly-formed corpus haemorrhagicum in one of the ovaries. The uteri were used in the preparation of primary mixed endometrial epithelial and stromal cell cultures by following the methods described previously [20] [21] . Briefly, endometrial tissue was stripped off and chopped into 1 mm3 followed by digestion for 90 min at 37˚C in media containing 100 mg of bovine serum albumin (BSA; Sigma, Steinheim, Germany), 50 mg of trypsin III (Worthington, New Jersey, USA) and 50 mg of collagenase A (Roche, Mannheim, Germany) per 100 mL of Hanks’ balanced salt solution (HBSS; Sigma, Ayrshire, UK). Digested tissue was filtered through 100 µm cell strainers (BD Biosciences, Erembodegem, Belgium) and then washed by re-suspending in HBSS containing 10% fetal bovine serum (FBS; PAA Laboratories GmbH, Pasching, Austria) and 3 µg/mL of trypsin inhibitor (Sigma, Steinheim, Germany). After centrifugation at 100 × g and 10˚C for 10 min, the cell sediment was re-suspended and the wash step repeated twice. Cell sediments were pooled together for each cow sample and cell count/cell viability evaluated by staining with trypan blue (Sigma, Steinheim, Germany). The isolated cells were re-sus- pended in growth media (GM) comprising MEM (PAA Laboratories GmbH, Pasching, Austria) containing 10% FBS and 1% antibiotic solution (100 IU/ml penicillin + 100 µg/ml streptomycin; Sigma, St. Louis, MO, USA). Cells were allocated at 5 × 105 cells/well to sterile 24-well plates (Nalge Nunc, Rochester, USA) and then cultured for 8 days in a humidified incubator at 37˚C and 5.0% CO2 while changing the GM every 48 h.
2.2. Validation of Endometrial Cell Culture Model
The proportions of cells in the mixed cultures were assessed using specific fluorescent immunocytochemical staining (Figure 1) as described previously [20] [21] . Epithelial cells were identified by a positive staining for cytokeratin, stromal
Figure 1. Micrographs of mixed bovine endometrial cells after 8 days of culture. Endometrial cells were characterised by fluorescent immunostaining (a)-(c). Green fluorescence represents positive staining for cytokeratin in epithelial cells (a) or vimentin in stromal cells (b). Isotype-matched control of the primary antibody (c) shows only the nuclei (blue) labelled with Hoechst. Phase contrast micrograph (d) showing endometrial epithelial cells (ES) and stromal cells (SC) in culture.
cells stained positive for vimentin and immune cells (e.g. macrophages and granulocytes) stained positive for CD172. The relative proportions of each cell type in the cultures on day 8 (before the start of treatment) were determined using ImageJ software, version 1.44 (Research Services Branch, NIMH/NIH, Bethesda, USA). The temporal differences in growth characteristics allow for a predominance of epithelial cells relative to the stromal cells after day 6 of culture as previously reported [20] [21] . Therefore, individual cultures derived from each of four cows had an epithelial cell population of approximately 90% (range, 85% - 93%) prior to treatment. There was a negligible presence of immune cells (<0.001%) in all the cultures used for the study. Prior to treatment with the ligands at Day 8 of culture, the number of endometrial cells per culture well was determined for each culture batch. Replicate cultures were treated with trypsin to release the adherent cells and the total cell count and percentage of viable cells was determined after staining with trypan blue.
2.3. Endometrial Cell Viability Assay
Endometrial cell viability was assessed following treatment with the synthetic ligands using the CellTiter 96 AQueous One Solution Cell Proliferation Assay kit (Promega, Southampton, UK). Briefly, about 5 × 104 mixed BEESCs were allocated per well in a 96-well plate and cultured for 8 days. Cultures were then treated in triplicate for 24 h with a range of four doses of each ligand: 2.5, 5, 10 and 25 μg/ml CL097 (Invivogen, San Diego, CA, USA) and 1, 2, 4 and 10 μg/ml poly(dA:dT) complexed with LyoVec, a transfection agent (Invivogen, San Diego, CA, USA). The dose ranges covered the typical doses of 5 μg/ml CL097 or 2 μg/ml poly(dA:dT) as recommended by the manufacturer, as well as doses from previous studies that utilized the ligands. Following treatment, 20 µL of the CellTiter 96 reagent was added to each well and incubated at 37˚C for 3 h. The absorbance at 490 nm was measured and the obtained values were directly proportional to the number of viable cells in the culture.
2.4. Cell Culture Treatments and RNA Isolation
Bovine endometrial cell cultures were specified as either control group (GM without CL097 and poly(dA:dT) for 0 h, 6 h and 24 h) or the exposed groups which were treated for 6 h and 24 h respectively with either 5 μg/ml CL097 or 2 μg/ml poly(dA:dT) in GM. There were initially six wells per treatment group and cultures from three wells were then pooled to provide two replicates per experiment. The experiment was repeated four times utilizing endometrial cultures from four different cows.
Isolation of total RNA from the cell cultures was performed using the RNeasy Mini spin column method (Qiagen, Hilden, Germany) following the supplied protocol. The concentration and purity of isolated RNA samples were determined using a NanoDrop spectrophotometer (ND-1000; NanoDrop Technologies, Wilmington, DE, USA). All RNA samples had concentrations of ≥230 ng/µL and 260/280 absorbance ratios within the range 1.9 to 2.0. The integrity of the RNA samples was validated using agarose gel electrophoresis as previously described [24] .
2.5. Conventional PCR and Quantitative Reverse Transcription-PCR (qRT-PCR)
Specific oligonucleotide primers (Supplementary Table 1) were designed for the genes of interest using Primer3 version 4.0 (http://primer3.ut.ee) with reference sequence (RefSeq) templates obtained from GenBank (http://www.ncbi.nlm.nih.gov/). Primer specificity to target gene was verified using Primer-BLAST (http://www.ncbi.nlm.nih.gov/tools/primer-blast/). The quality of primer pairs was evaluated using Oligo Analyzer version 3.1 (Integrated DNA Technologies; http://eu.idtdna.com/analyzer/applications/oligoanalyzer/) before the primers were synthesized (Eurofins MWG Operon, Ebersberg, Germany).
The expression of 18 candidate immune-related genes and 3 endogenous reference genes: RN18S1, ACTB and GAPDH were assessed by conventional PCR using the Qiagen multiplex PCR kit (Qiagen, Hilden, Germany) and the G-Storm thermal cycler (Gene Technologies, Essex, United Kingdom). PCR products were visualised by agarose gel electrophoresis and the intensity (expression) of each gene product was digitally captured and quantified by densitometric analysis using the Alpha Ease FC software, version 3.1.2 (Alpha Innotech, San Leandro, CA, USA). The quantity of expression of each gene was calculated as the intensity density value (IDV). The stability of the endogenous reference genes under the experimental conditions was evaluated. Treatment with CL097 or poly(dA:dT) did not alter the expression of the reference genes RN18S1, ACTB and GAPDH compared to the control cultures at the different time points, therefore gene expression values were then normalized relative to the reference genes.
Based on these preliminary results, absolute quantification of the mRNA expression of 12 selected candidate genes and the three reference genes was next performed using the KAPA SYBR FAST qPCR kit (Kapa Biosystems, Boston, MA, USA) and the CFX96 Real-time thermal cycler (Bio-Rad, Hercules, CA, USA). All qRT-PCR assays were first optimized and then performed in duplicate using the same amount of reaction template (50 ng of reverse-transcribed RNA) with all samples included in the same assay to avoid inter-assay variation. Experimental samples were assayed together with 10 known concentrations of the respective standard (ranging from 1 × 101 to 1 × 10−8 ng/ml) and the no-template control. The absolute gene expression values were calculated in femtogram (fg) per microgram of total RNA using the standard curve and the Bio-Rad CFX Manager software ver. 3.1.
2.6. Data Analysis
The gene expression data were analysed using analysis of variance (ANOVA) with repeated measures via a linear mixed-effects model built in SPSS, version 20 (IBM Corp, Armonk, NY, USA). Values were compared relative to the control at 6 h and 12 h post-exposure to the ligands. Cell cultures from each of four cows contained all treatment groups. Treatments, time and their interactions were taken as the fixed effects and cows as the subject for repeated measure. Where ANOVA showed significant difference, differences between the means were confirmed using Fisher’s LSD multiple comparison. Results were considered significant when p < 0.05.
3. Results
Endometrial cell types were characterised by fluorescent immunostaining after 8 days of culture (Figure 1). At this stage both epithelial cells and stromal cells were present. Prior to treatment with the ligands at Day 8 of culture, the average number of endometrial cells per culture well ranged from 178,000 to 184,000 (mean ± SD: 180,813 ± 2511) and the percentage of viable cells ranged from 89% to 93% (mean: 91%; SD: 1.8) for the four different batches of cell culture from the four cows. The cells had attained a confluence of between 80% - 90%.
3.1. Endometrial Cell Viability after Treatment with Synthetic Ligands
Dose responses for the respective ligands were tested for potential toxicity using a colorimetric method to evaluate the proportion of metabolically active (viable) cells. The absorbance values at 490 nm were compared between the control and the respective ligand treatment groups. Treatment of BEESCs for 24 h with the different doses of the ligands CL097 or poly(dA:dT) did not significantly alter (p > 0.05) the proportion of viable cells in culture compared to the control (Table 1). The doses used in the main experiment were therefore those recommended by the manufacturers, which were 5 μg/ml of CL097 or 2 μg/ml of poly(dA:dT).
3.2. Quantitation of Gene Expression Changes in Bovine Endometrial Cells Treated with CL097or Poly(dA:dT)
Preliminary analysis using conventional PCR showed that all selected genes were detectable in the culture system used. The two treatments did not alter the expression of any of the three reference genes selected (RN18S1, ACTB and GAPDH). Neither ligand appeared to alter the expression of TLR3, TLR4, TLR7, TLR8, leucine rich repeat (in FLII) interacting protein 1 (LRRFIP1), IL6, macrophage migration inhibitory factor (MIF), intercellular adhesion molecule 1 (ICAM1) or transforming growth factor, beta 1 (TGFB1) at either time point (Supplementary Figure 1). Based on these results, twelve candidate genes were selected for analysis by qRT-PCR together with the three reference genes. Evaluation of the mean absolute mRNA expression of the studied genes at the baseline (control samples, 0 h) revealed that two were constitutively expressed by BEESCs at relatively higher levels (1400 - 1700 fg/µg RNA; the antimicrobial gene mucin 1, cell surface associated (MUC1) and interferon (alpha, beta and omega) receptor 1 (IFNAR1), some at moderate levels (130 - 600 fg/µg RNA; TLR4, IL8, myxovirus (influenza virus) resistance 1 (MX1) and IFIH1) whereas others were expressed at much lower levels (10 - 70 fg/µg RNA; TLR7, TLR8, IL1B, TNF, secretory leukocyte peptidase inhibitor (SLPI) and DDX58) (Table 2). It should be noted that relative mRNA expression cannot be assumed to equate to proportional synthesis in protein expression as it does not account for differential downstream processing.
The qPCR analysis confirmed that neither CL097 nor poly(dA:dT) altered the expression of either the reference genes or the PRR genes TLR4, TLR7 or TLR8 in comparison with the controls (Table 2). Similarly CL097 did not change the expression of the RLR family genes IFIH1 and DDX58 but poly(dA:dT) up-regulated both IFIH1 and DDX58 significantly at 24 h by 1.9 and 2.3 fold respectively. The two ligands also had different effects on the expression of the IFN and antiviral response gene MX1 as this was significantly up-regulated by
Table 1. Assessment of bovine endometrial cell viability after treatment for 24 h with CL097 or poly(dA:dT)#.
#Data are the mean ± SEM of three samples compared with the control (ANOVA) and represent the absorbance at 490 nm, which was directly proportional to the number of viable cells in the culture. Values did not differ from the control. +TD: typical dose. These were 5 μg/ml CL097 or 2 μg/ml poly(dA:dT), as recommended by the manufacturers.
Table 2. The expression of candidate genes in bovine endometrial cells after treatment for 6 h and 24 h with CL097 or poly(dA:dT) assessed by qRT-PCR#.
#Gene expression levels in fg per µg total RNA. Data are presented as mean ± SEM and represent samples from four individual cows. Significantly different from controls: *p < 0.05, **p < 0.01, ***p < 0.001; linear mixed-effects analysis.
7.2-fold at 24 h by poly(dA:dT) while CL097 had no effect. IFNAR1 was up-regulated at 6 h by CL097 and at both 6 h and 24 h by poly(dA:dT) treatment, all by about 2-fold. The expression of antimicrobial genes showed a general increase following treatment with the ligands: MUC1 was significantly up-regulated at both 6 and 24 h by both ligands (1.7- to 2-fold increases) while SLPI was up-regulated at 6 h (4.4-fold) and 24 h (3.1-fold) after CL097 treatment only. Analysis of the effects on genes with cytokine function showed that CL097 up-regulated TNF by 5.5-fold at 6 h and increased IL8 and IL1B by 5-fold and 16-fold respectively at 6 h, with increases remaining significant at 24 h. In contrast, the expression levels of IL8 and IL1B and TNF were numerically higher by 1.5- to 2.8-fold at 6 h after treatment with poly(dA:dT) but the differences failed to achieve significance and had decreased to baseline by 24 h.
4. Discussion
The innate immune response of bovine endometrial cells against extracellular bacteria including E. coli and bacterial ligands such as LPS has been described in some detail but there is much less information on the response of these cells to intracellular pathogens such as viruses and intracellular bacteria or their ligands. In this experiment, the intracellular immune stimulants CL097 and poly(dA:dT) were used to study innate immune response in bovine endometrial cells in vitro. CL097 is recognised by TLR7 and TLR8. These receptors are also activated by ssRNA as found in viruses such as BVDV, whereas poly(dA:dT) is a repetitive dsDNA sequence which functions as a synthetic analogue of B-form DNA as found in dsDNA viruses such as bovine herpesvirus 4 (BoHV-4) [6] [17] [25] {Diebold, 2008 #21636} . Both the natural (microbial) and the synthetic ligands have been shown previously to stimulate the same pattern recognition receptors [6] . We do not imply from our results that the subsequent immune response is identical for both natural and synthetic stimulants but we are able to infer that the endometrial cells have the potential to participate in the innate immune protection of the endometrium using these pathways. The two time points of 6 h and 24 h after treatment were selected to provide the maximum amount of information based on a previous time course study into aspects of the innate immune response in bovine endometrium [20] . It is possible that changes to some genes such as TNF, which are known to be up-regulated at an early stage [20] would have been greater if an earlier time point had also been included. This was not feasible due to the large number of replicates of all the treatments and times and genes of interest within the same batch of primary cells derived from a single endometrium.
The observed cellular responses can reliably be attributed to endometrial epithelial and stromal cells, because cell cultures used for the study had insignificant carry-over of immune cells (<0.001%) from endometrial tissue. The use of mixed endometrial epithelial and stromal cell culture could be justified by several factors. Epithelial cells are the luminal layer of cells first encountered by pathogens that contaminate the postpartum uterus. However, placental separation at calving disrupts the caruncular epithelium allowing microbial access to the underlying endometrial stroma and connective tissue. Viral infections are also likely to access the uterus via the bloodstream, so first encountering the stromal cells. Therefore, stromal cells may also have innate protective function and also express PRRs. Moreover, stromal cells secrete growth factors that promote transepithelial resistance and barrier function [26] , indicating an important functional and physiologic interaction between the two cell types. The culture system used is well established and has the advantage of minimizing the initial handling during isolation (which can cause damage) and then allowing for the functional interaction between both cell types.
The study has provided clear evidence that BEESCs could mount a significant, but differing, innate immune response to both intracellular ligands, with possible responses illustrated in Figure 2. The two ligands showed temporal differences in the peak induction of genes in different signalling pathways. CL097 induced a peak up-regulation of cytokines (TNF, IL8 and IL1B) in the early period (6 h) whereas poly(dA:dT) induced a later peak increase (24 h) in the IFN- stimulated genes (MX1 and IFNAR1).
Figure 2. Summary diagram showing the suggested induction of an innate immune response in bovine endometrial cells after treatment with the synthetic ligands CL097 or poly(dA:dT). (a) Uptake of CL097 or (b) poly(dA:dT)/dsDNA by BEESC. (c) CL097 is recognised by TLR7/8 in the endosomal compartments whereas (d) dsDNA is transferred to the cytoplasm and is recognised by several cytosolic DNA sensors (CDSs) including DDX58. (e) Ligand recognition triggers downstream signalling leading to the activation of the transcription factors IRF3/7 and NFkB. (f) Activated transcription factors translocate to the nucleus: IRF3/7 drives the expression of IFN-stimulated genes, NFkB drives the expression of inflammatory cytokines [6] [40] . aGenes up-regulated by CL097; bGenes up-regulated by poly(dA:dT).
The BEESCs expressed the extra-cytosolic PRRs TLR3, TLR4, TLR7 and TLR8 in agreement with previous findings [18] [20] , as well as the cytosolic PRRs IFIH1 [21] , DDX58 and LRRFIP1. These receptors have important roles in detecting both extracellular and intracellular pathogens during innate immunity. Stimulation of TLR7 or the RLRs DDX58 (RIG-I) and IFIH1 (MDA5) induces the expression of pro-inflammatory cytokines and type I IFN genes [6] . LRRFIP1 recognises both dsRNA and dsDNA and triggers increased production of type I IFN [14] . While these various receptors were present in the BEESC, only DDX58 and IFIH1 were up-regulated by poly(dA:dT) at 24 h. This is consistent with previous observations in which the stimulation of known PRRs induced TLR-mediated responses in bovine macrophages without influencing the mRNA concentrations of relevant TLR members [27] . The up-regulation of DDX58 and IFIH1 may indicate the importance of these receptors in viral recognition and the establishment of antiviral innate immune response.
The ligands altered the expression of MX1 and IFNAR1 differently. MX1 is an IFN-induced antiviral protein that typically blocks early viral replication [28] . The type I IFN response modulates the innate and adaptive immune response [29] . The receptor IFNAR1 mediates the type I IFN response to induce the transcription of a large number of IFN-stimulated genes [30] . MX1 was one of the most highly up-regulated genes studied, with expression increasing over 7-fold. In agreement with our findings, the expression of MX1 was up-regulated by an ssRNA ligand in human endometrial endothelial cells [31] . Similarly, poly(dA:dT) increased the expression of several ISGs including MX1, and the production of IFNB in murine cells [17] . Infection with BVDV suppressed the expression of IFIH1 and MX1 in bovine endometrial cells, thus highlighting the importance of these genes in antiviral response [23] .
MUC1 showed high baseline expression of mRNA in the untreated endometrial cells. This cannot be taken to infer that protein expression is also high, as this will be influenced by mRNA stability and processing. It may, however, reflect the key role of the membrane-bound MUC genes in forming a protective mucous layer on the mucosal surface of the endometrium [32] [33] . Both ligands doubled the expression of MUC1 at both time points. Of the other antimicrobial peptides tested, CL097 significantly up-regulated SLPI. SLPI protects epithelial tissues from serine proteases and also has antibacterial and anti-inflammatory functions [34] .
With respect to the cytokines tested, CL097 significantly up-regulated TNF and IL8 by 5-fold at 6 h post-exposure while expression of IL1B was 16-fold higher at this time point. In contrast, treatment with poly(dA:dT) was followed by a numerical but not significant increase in these genes at 6 h only. We previously showed peak expression of TNF at 1 h after treatment of BEESC with LPS although the peak response to IL1B was at 6 h [20] so it is possible that a peak cytokine response to poly(dA:dT) had occurred earlier than the first time of assessment. These key inflammatory cytokines are an important component of the early innate immune defence [21] and also serve as a crucial link in the development of an adaptive immune response [33] . TLR7/8 agonists also up-regulated many cytokine genes and proteins in human DCs [35] . CL097 increased IL8 but not IL6 or TNFα proteins in human endocervical epithelial cells [36] . On the other hand in our experiment neither CL097 nor poly(dA:dT) altered the expression of the cytokines IL6, MIF, ICAM1 and TGFB1. This may be attributed to differences in cell-type response to the ligands.
While mounting a robust defence to endometrial pathogens is essential to maintain fertility, it is important to note that inflammatory and immune pathways have a complex inter-relationship with reproductive processes in mammals. In females, TLRs have been implicated in the regulation of ovulation, fertilization, gestation and parturition, as well as in pathological conditions such as endometritis and mastitis [37] . In fact, immune responses generated towards other cells and molecules could have adverse consequences for reproduction [38] [39] [40] . Inappropriate activation of TLRs and other innate immune mediators such as cytokines and eicosanoids may also disrupt reproductive functions in the endometrium or the development of the conceptus thereby decreasing fertility.
5. Conclusion
Innate immunity and inflammatory responses in the endometrium are crucial for the elimination of uterine pathogens in dairy cows. The large numbers of epithelial and stromal cells in the bovine endometrium, relative to the specialised immune cells, are likely to contribute significantly to innate immune function. Bovine endometrial cells express cytosolic and extra-cytosolic intracellular PRRs and are able to detect and mount a rapid innate immune response to the intracellular ligands CL097 and poly(dA:dT). These artificial ligands stimulate similar receptors to those activated by some viruses which are known to affect bovine fertility such as BoHV-4 and BVDV. This suggests important roles for these cells in the defence against viruses that may be present in the uterus in postpartum cows. A greater understanding of the role of viral infection and how the uterus responds to it may help us to develop better methods for diagnosing and treating these conditions in the cow, so potentially reducing the major economic losses caused by the adverse impact of uterine disease on fertility.
Acknowledgements
This study was supported by the Commonwealth Scholarship Commission in the United Kingdom. RVC manuscript approval number PPH_01167.
Cite this paper
Oguejiofor, C.F., Cheng, Z., Fouladi-Nashta, A.A. and Wathes, D.C. (2017) Bovine Endometrial Cells Mount Innate Immune Response to the Intracellular Ligands CL097 and Poly(dA:dT) Indicating Roles against Uterine Viruses. Open Jour- nal of Animal Sciences, 7, 110-126. https://doi.org/10.4236/ojas.2017.72010
References
- 1. Williams, E.J., Fischer, D.P., Pfeiffer, D.U., England, G.C., Noakes, D.E., Dobson, H. and Sheldon, I.M. (2005) Clinical Evaluation of Postpartum Vaginal Mucus Reflects Uterine Bacterial Infection and the Immune Response in Cattle. Theriogenology, 63, 102-117.
https://doi.org/10.1016/j.theriogenology.2004.03.017 - 2. Grooms, D.L. (2004) Reproductive Consequences of Infection with Bovine Viral Diarrhea Virus. Veterinary Clinics of North America Food Animal Practice, 20, 5-19.
https://doi.org/10.1016/j.cvfa.2003.11.006 - 3. Donofrio, G., Franceschi, V., Capocefalo, A., Cavirani, S. and Sheldon, I.M. (2009) Isolation and Characterization of Bovine Herpesvirus 4 (BoHV-4) from a Cow Affected by Postpartum Metritis and Cloning of the Genome as a Bacterial Artificial Chromosome. Reproductive Biology and Endocrinology, 7, 83.
https://doi.org/10.1186/1477-7827-7-83 - 4. Singh, J., Murray, R.D., Mshelia, G. and Woldehiwet, Z. (2008) The Immune Status of the Bovine Uterus during the Peripartum Period. The Veterinary Journal, 175, 301-309.
https://doi.org/10.1016/j.tvjl.2007.02.003 - 5. Medzhitov, R. (2007) Recognition of Microorganisms and Activation of the Immune Response. Nature, 449, 819-826.
https://doi.org/10.1038/nature06246 - 6. Kumar, H., Kawai, T. and Akira, S. (2011) Pathogen Recognition by the Innate Immune System. International Reviews of Immunology, 30, 16-34.
https://doi.org/10.3109/08830185.2010.529976 - 7. Berke, I.C., Li, Y. and Modis, Y. (2013) Structural Basis of Innate Immune Recognition of Viral RNA. Cellular Microbiology, 15, 386-394.
https://doi.org/10.1111/cmi.12061 - 8. Booth, J.S., Buza, J.J., Potter, A., Babiuk, L.A. and Mutwiri, G.K. (2010) Co-Stimulation with TLR7/8 and TLR9 Agonists Induce Down-Regulation of Innate Immune Responses in Sheep Blood Mononuclear and B Cells. Developmental and Comparative Immunology, 34, 572-578.
- 9. Mifsud, E.J., Tan, A.C. and Jackson, D.C. (2014) TLR Agonists as Modulators of the Innate Immune Response and Their Potential as Agents against Infectious Disease. Frontiers in Immunology, 5, 79.
https://doi.org/10.3389/fimmu.2014.00079 - 10. Vasilakos, J.P. and Tomai, M.A. (2013) The Use of Toll-Like Receptor 7/8 Agonists as Vaccine Adjuvants. Expert Review of Vaccines, 12, 809-819.
https://doi.org/10.1586/14760584.2013.811208 - 11. Gibson, S.J., Lindh, J.M., Riter, T.R., Gleason, R.M., Rogers, L.M., Fuller, A.E., Oesterich, J.L., Gorden, K.B., Qiu, X., Mckane, S.W., Noelle, R.J., Miller, R.L., Kedl, R.M., Fitzgerald-Bocarsly, P., Tomai, M.A. and Vasilakos, J.P. (2002) Plasmacytoid Dendritic Cells Produce Cytokines and Mature in Response to the TLR7 Agonists, Imiquimod and Resiquimod. Cellular Immunology, 218, 74-86.
https://doi.org/10.1016/s0008-8749(02)00517-8 - 12. Di Domizio, J., Blum, A., Gallagher-Gambarelli, M., Molens, J.P., Chaperot, L. and Plumas, J. (2009) TLR7 Stimulation in Human Plasmacytoid Dendritic Cells Leads to the Induction of Early IFN-Inducible Genes in the Absence of Type I IFN. Blood, 114, 1794-1802.
https://doi.org/10.1182/blood-2009-04-216770 - 13. Takaoka, A., Wang, Z., Choi, M.K., Yanai, H., Negishi, H., Ban, T., Lu, Y., Miyagishi, M., Kodama, T., Honda, K., Ohba, Y. and Taniguchi, T. (2007) DAI (DLM-1/ZBP1) Is a Cytosolic DNA Sensor and an Activator of Innate Immune Response. Nature, 448, 501-505.
https://doi.org/10.1038/nature06013 - 14. Yang, P., An, H., Liu, X., Wen, M., Zheng, Y., Rui, Y. and Cao, X. (2010) The Cytosolic Nucleic Acid Sensor LRRFIP1 Mediates the Production of Type I Interferon via a Beta-Catenin-Dependent Pathway. Nature Immunology, 11, 487-494.
https://doi.org/10.1038/ni.1876 - 15. Hornung, V., Ablasser, A., Charrel-Dennis, M., Bauernfeind, F., Horvath, G., Caffrey, D.R., Latz, E. and Fitzgerald, K.A. (2009) AIM2 Recognizes Cytosolic dsDNA and Forms a Caspase-1-Activating Inflammasome with ASC. Nature, 458, 514-518.
https://doi.org/10.1038/nature07725 - 16. Chiu, Y.H., Macmillan, J.B. and Chen, Z.J. (2009) RNA Polymerase III Detects Cytosolic DNA and Induces Type I Interferons through the RIG-I Pathway. Cell, 138, 576-591.
https://doi.org/10.1016/j.cell.2009.06.015 - 17. Ishii, K.J., Coban, C., Kato, H., Takahashi, K., Torii, Y., Takeshita, F., Ludwig, H., Sutter, G., Suzuki, K., Hemmi, H., Sato, S., Yamamoto, M., Uematsu, S., Kawai, T., Takeuchi, O. and Akira, S. (2006) A Toll-Like Receptor-Independent Antiviral Response Induced by Double-Stranded B-Form DNA. Nature Immunology, 7, 40-48.
https://doi.org/10.1038/ni1282 - 18. Davies, D., Meade, K.G., Herath, S., Eckersall, P.D., Gonzalez, D., White, J.O., Conlan, R.S., O’farrelly, C. and Sheldon, I.M. (2008) Toll-Like Receptor and Antimicrobial Peptide Expression in the Bovine Endometrium. Reproductive Biology and Endocrinology, 6, 53.
https://doi.org/10.1186/1477-7827-6-53 - 19. Chapwanya, A., Meade, K.G., Doherty, M.L., Callanan, J.J. and O’Farrelly, C. (2013) Endometrial Epithelial Cells Are Potent Producers of Tracheal Antimicrobial Peptide and Serum Amyloid A3 Gene Expression in Response to E. Coli Stimulation. Veterinary Immunology and Immunopathology, 151, 157-162.
https://doi.org/10.1016/j.vetimm.2012.09.042 - 20. Swangchan-Uthai, T., Lavender, C.R., Cheng, Z., Fouladi-Nashta, A.A. and Wathes, D.C. (2012) Time Course of Defense Mechanisms in Bovine Endometrium in Response to Lipopolysaccharide. Biology of Reproduction, 87, 135.
https://doi.org/10.1095/biolreprod.112.102376 - 21. Oguejiofor, C.F., Cheng, Z., Abudureyimu, A., Fouladi-Nashta, A.A. and Wathes, D.C. (2015a) Global Transcriptomic Profiling of Bovine Endometrial Immune Response in Vitro. I. Effect of Lipopolysaccharide on Innate Immunity. Biology of Reproduction, 93, 100.
https://doi.org/10.1095/biolreprod.115.128868 - 22. Donofrio, G., Capocefalo, A., Franceschi, V., Price, S., Cavirani, S. and Sheldon, I.M. (2010) The Chemokine IL8 Is Up-Regulated in Bovine Endometrial Stromal Cells by the BoHV-4 IE2 Gene Product, ORF50/Rta: A Step Ahead toward a Mechanism for BoHV-4 Induced Endometritis. Biology of Reproduction, 83, 919-928.
https://doi.org/10.1095/biolreprod.110.086074 - 23. Oguejiofor, C.F., Cheng, Z., Abudureyimu, A., Anstaett, O.L., Brownlie, J., Fouladi-Nashta, A.A. and Wathes, D.C. (2015b) Global Transcriptomic Profiling of Bovine Endometrial Immune Response in Vitro. II. Effect of Bovine Viral Diarrhea Virus on the Endometrial Response to Lipopolysaccharide. Biology of Reproduction, 93, 101.
https://doi.org/10.1095/biolreprod.115.128876 - 24. Wieczorek, D., Delauriere, L. and Schagat, T. (2012) Methods of RNA Quality Assessment.
http://www.promega.co.uk/resources/pubhub/methods-of-rna-quality-assessment/ - 25. Diebold, S.S. (2008) Recognition of Viral Single-Stranded RNA by Toll-Like Receptors. Advanced Drug Delivery Reviews, 60, 813-823.
https://doi.org/10.1016/j.addr.2007.11.004 - 26. Wira, C.R., Fahey, J.V., Sentman, C.L., Pioli, P.A. and Shen, L. (2005) Innate and Adaptive Immunity in Female Genital Tract: Cellular Responses and Interactions. Immunological Reviews, 206, 306-335.
https://doi.org/10.1111/j.0105-2896.2005.00287.x - 27. Franchini, M., Schweizer, M., Matzener, P., Magkouras, I., Sauter, K.S., Mirkovitch, J., Peterhans, E. and Jungi, T.W. (2006) Evidence for Dissociation of TLR mRNA Expression and TLR Agonist-Mediated Functions in Bovine Macrophages. Veterinary Immunology and Immunopathology, 110, 37-49.
https://doi.org/10.1016/j.vetimm.2005.09.002 - 28. Haller, O., Stertz, S. and Kochs, G. (2007) The Mx GTPase Family of Interferon-Induced Antiviral Proteins. Microbes and Infection, 9, 1636-1643.
https://doi.org/10.1016/j.micinf.2007.09.010 - 29. Stetson, D.B. and Medzhitov, R. (2006) Type I Interferons in Host Defense. Immunity, 25, 373-381.
https://doi.org/10.1016/j.immuni.2006.08.007 - 30. Schoggins, J.W. and Rice, C.M. (2011) Interferon-Stimulated Genes and Their Antiviral Effector Functions. Current Opinion in Virology, 1, 519-525.
https://doi.org/10.1016/j.coviro.2011.10.008 - 31. Krikun, G., Potter, J.A. and Abrahams, V.M. (2013) Human Endometrial Endothelial Cells Generate Distinct Inflammatory and Antiviral Responses to the TLR3 Agonist, Poly(I:C) and the TLR8 Agonist, Viral ssRNA. American Journal of Reproductive Immunology, 70, 190-198.
https://doi.org/10.1111/aji.12128 - 32. Sando, L., Pearson, R., Gray, C., Parker, P., Hawken, R., Thomson, P.C., Meadows, J.R., Kongsuwan, K., Smith, S. and Tellam, R.L. (2009) Bovine Muc1 Is a Highly Polymorphic Gene Encoding an Extensively Glycosylated Mucin That Binds Bacteria. Journal of Dairy Science, 92, 5276-5291.
https://doi.org/10.3168/jds.2009-2216 - 33. Hickey, D.K., Patel, M.V., Fahey, J.V. and Wira, C.R. (2011) Innate and Adaptive Immunity at Mucosal Surfaces of the Female Reproductive Tract: Stratification and Integration of Immune Protection against the Transmission of Sexually Transmitted Infections. Journal of Reproductive Immunology, 88, 185-194.
https://doi.org/10.1016/j.jri.2011.01.005 - 34. Weldon, S. and Taggart, C.C. (2007) Innate Host Defense Functions of Secretory Leucoprotease Inhibitor. Experimental Lung Research, 33, 485-491.
https://doi.org/10.1080/01902140701756547 - 35. Birmachu, W., Gleason, R.M., Bulbulian, B.J., Riter, C.L., Vasilakos, J.P., Lipson, K.E. and Nikolsky, Y. (2007) Transcriptional Networks in Plasmacytoid Dendritic Cells Stimulated with Synthetic TLR 7 Agonists. BMC Immunology, 8, 26.
https://doi.org/10.1186/1471-2172-8-26 - 36. Radtke, A.L., Quayle, A.J. and Herbst-Kralovetz, M.M. (2012) Microbial Products Alter the Expression of Membrane-Associated Mucin and Antimicrobial Peptides in a Three-Dimensional Human Endocervical Epithelial Cell Model. Biology of Reproduction, 87, 132.
https://doi.org/10.1095/biolreprod.112.103366 - 37. Kannaki, T.R., Shanmugam, M. and Verma, P.C. (2011) Toll-Like Receptors and Their Role in Animal Reproduction. Animal Reproduction Science, 125, 1-12.
https://doi.org/10.1016/j.anireprosci.2011.03.008 - 38. Hansen, P.J. (2011) The Immunology of Early Pregnancy in Farm Animals. Reproduction in Domestic Animals, 46, 18-30.
https://doi.org/10.1111/j.1439-0531.2011.01850.x - 39. Gilbert, R.O. (2012) The Effects of Endometritis on the Establishment of Pregnancy in Cattle. Reproduction, Fertility and Development, 24, 252-257.
https://doi.org/10.1071/RD11915 - 40. Takeuchi, O. and Akira, S. (2010) Pattern Recognition Receptors and Inflammation. Cell, 140, 805-820.
https://doi.org/10.1016/j.cell.2010.01.022
Appendix
Supplementary Table 1. Details of the genes measured by conventional PCR and qRT-PCR after treatment of bovine endometrial cells with synthetic ligands.
Primers used for: *both conventional PCR and qRT-PCR, ^qRT-PCR only; bp: base pair.
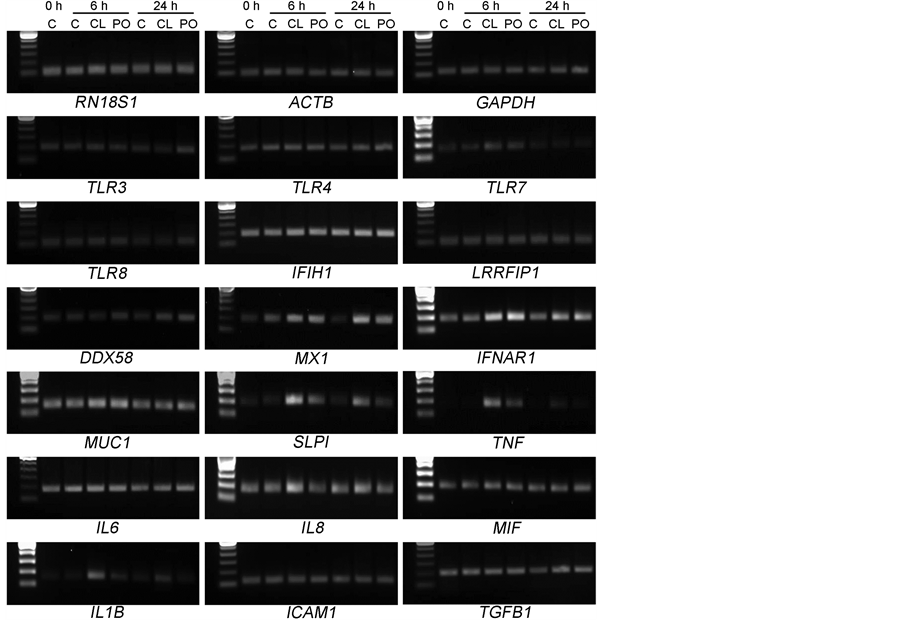
Supplementary Figure 1. The mRNA expression of candidate genes in bovine endometrial cells treated with synthetic ligands, assessed using conventional PCR. Cells were treated with either CL097 or poly(dA:dT) for 6 h and 24 h. Lanes represent: control (C), CL097 (CL) and poly(dA:dT) (PO).

Submit or recommend next manuscript to SCIRP and we will provide best service for you:
Accepting pre-submission inquiries through Email, Facebook, LinkedIn, Twitter, etc.
A wide selection of journals (inclusive of 9 subjects, more than 200 journals)
Providing 24-hour high-quality service
User-friendly online submission system
Fair and swift peer-review system
Efficient typesetting and proofreading procedure
Display of the result of downloads and visits, as well as the number of cited articles
Maximum dissemination of your research work
Submit your manuscript at: http://papersubmission.scirp.org/
Or contact ojas@scirp.org




