Open Journal of Inorganic Chemistry
Vol.2 No.3(2012), Article ID:19766,9 pages DOI:10.4236/ojic.2012.23009
Metal-organic frameworks derived from divalent metals and 4-(1H-1,2,4-triazol-1-yl)benzoic acid
![]()
Institute of Chemistry for Functionalized Materials, College of Chemistry and Chemical Engineering, Liaoning Normal University, Dalian, China
Email: *yqtian@lnnu.edu.cn
Received 24 March 2012; revised 25 April 2012; accepted 13 May 2012
Keywords: Coordination Polymers; 4-(1H-1,2,4-triazol-1-yl)benzoic Acid; Crystal Structure; Cadmium; Copper
ABSTRACT
Two inorganic-organic coordination polymers, [Cd(TBA)2]·3H2O (1) and [Cu(TBA)2]·2H2O (2) have been synthesized by reaction of the metal ions of Cd(II) and Cu(II) with the ligand HTBA [HTBA = 4- (1H-1,2,4-triazol-1-yl)benzoic acid] under solvothermal condition. The compounds were characterized by single-crystal X-ray diffraction, elemental analysis, IR spectroscopy, X-ray powder diffraction (XRPD), and thermo gravimetric analysis (TGA). The structural analyses reveal the two independent polymers exhibit different structures. Complex 1 exhibits a 3D framework with 1D channels, and complex 2 shows a 2-fold interpenetrating 3D diamond net.
1. INTRODUCTION
The reticular design and synthesis of metal-organic frameworks (MOFs) are currently the hot topic for discovery of the materials with potential applications in magnetism [1-6], catalysis [7-10], luminescence and chemical sensing [11-15], as well as gas adsorption and/or separation [16-18]. However, any of a successful construction of the target MOFs shall practically depend on a deliberate design or a judicious selection of the organic ligand incurporating with a proper geometrical metal. For example, the MOFs with zeolitic or zeolite-like structures are constructed by using imidazole and its derivates incorporating with tetrahedral coordinated metals [19-20]. To create novel MOFs having the 4-connected zeolitic structures, the MOFs based on imidazole derivate of 4-(1Himidaozol-1-yl)benzoic acid (HIBA) and divalent sixcoordinated metals have been reported in our previous work [21,22], where the ligand presents two ((2.21)syn-syn and (2.11)syn) of the several probable coordination models (Scheme 1).
The ligand of 4-(1H-1,2,4-triazol-1-yl)benzoic acid (HTBA) is structurally similar to HIBA and can be facilely prepared by the following procedure: So far we know that the use of 4-(1H-1,2,4-triazol-1-yl)benzoic acid (HTBA) to construct a coordination polymer remains still unexplored until this work. Compared with the ligand of HIBA, HTBA shows only small structural difference with one carbon being replaced by a nitrogen atom. Thus, HTBA shall also form the various probable coordination modes shown in Scheme 1. By virtue of this consideration, novel MOF structures of HTBA ligand were expected. In our current work, we successfully obtained two novel 3D coordination polymers formulated as [Cd(TBA)2]·3H2O (1), [Cu(TBA)2]·2H2O (2), which are characterized by X-ray single crystal analysis, elemental analysis, IR spectroscopy, X-ray powder diffraction (XRPD), and thermo gravimetric analysis (TGA).
2. EXPERIMENTAL SECTION
2.1. Materials and Methods
All chemicals and solvents used in the syntheses were commercially available and used without further purification. The elemental analyses were carried out on a Perkin-Elmer 2400 elemental analyzer. The IR spectra were recorded (400 - 4000 cm–1) on a FT-IR spectrometer Bruker TENSOR 27. X-ray powder diffraction (XRPD) analyses were carried out on a Bruker D8 Advance and thermal gravimetric analyses (TGA) was performed under static air atmosphere with a heating rate of 10˚C/min by using a Perkin-Elmer Diamond thermo gravimetric analyzer.
2.2. X-Ray Single-Crystal Structure Determination
Suitable single crystals of two complexes 1 and 2 were mounted on glass fibers for X-ray measurement. Crys-
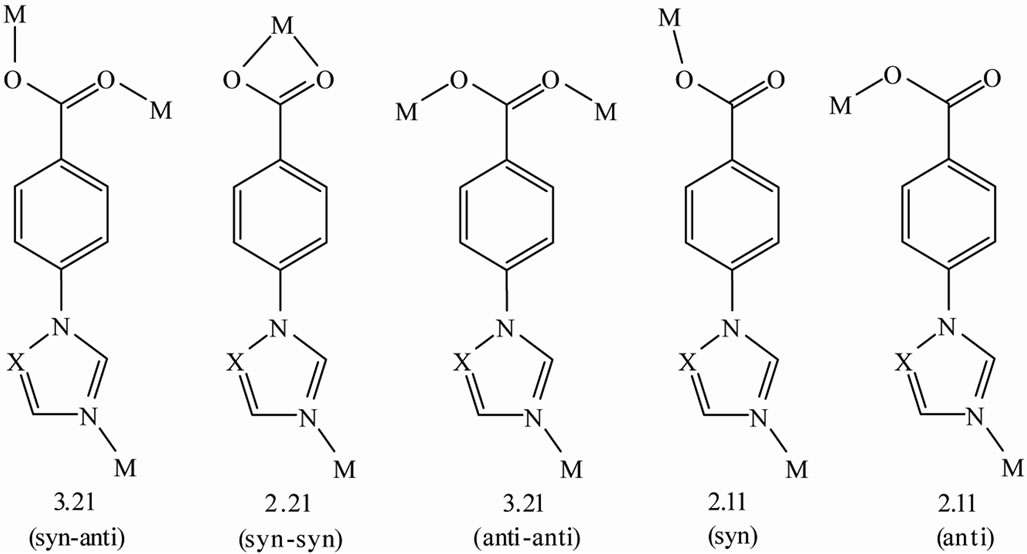
Scheme 1. The probable coordination modes of ligand (X=C or N atom).
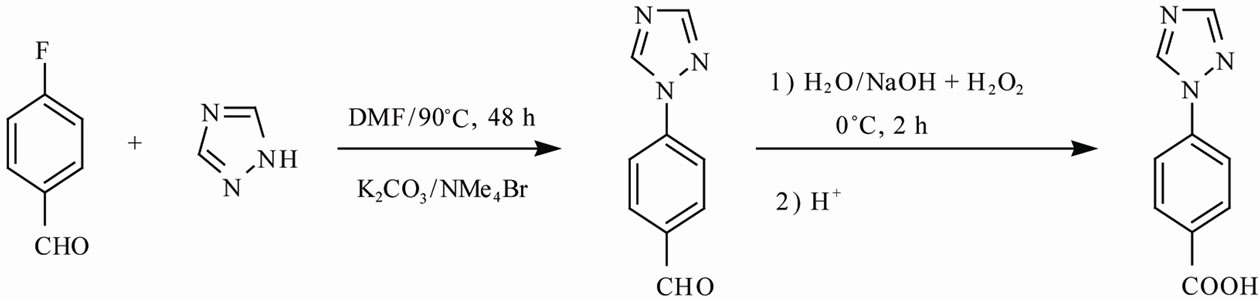
Scheme 2. Synthesis pathway of the ligand.
tallographic measurements for 1 & 2 were carried out on a Bruker SMART-Apex-II CCD diffractometer with graphite-monochromatized MoKα radiation (λ = 0.71073 Å) at 293(2) K. All absorption corrections were performed using the SADABS program [23]. Crystal structures were solved by the direct method. All non-hydrogen atoms were refined anisotropically. Hydrogen atoms of carbon atoms were fixed at calculated positions with isotropic thermal parameters, while the free lattice water molecules were located by difference Fourier maps. All calculations were performed using the SHELX-97 program [24]. However, hydrogen atoms on O1W and O2W in 1 are refined isotropically and hydrogen atom of O1W, O2W in 2 could not be located. Crystal data and details of the data collection and the structure refinement are given in Table 1. The selected bond lengths and bond angles of complexes 1 and 2 are listed in Table 2.
2.3. Synthesis of the Organic Ligands
4-(1H-1,2,4-triazol-1-yl)-benzaldehyde: a mixture of 4- fluorobenzaldehyde (5.58 g, 45 mmol), 1,2,4-triazol (2.05 g, 30 mmol), anhydrous K2CO3 (4.14 g, 30 mmol) and tetramethylammonium bromide (0.159 g, 0.6 mmol) in anhydrous DMF (50 mL) was stirred under nitrogen atmosphere at 90˚C for 48 h. After the reaction was completed, the solvent was removed by vacuum distillation. Then, ice water was added into the residue and the white needle product was obtained by filtration. Yield: 4.2 g (96%).
4-(1H-1,2,4-triazol-1-yl)benzoic acid (HTBA): 4-(1H-1, 2,4-triazol-1-yl)-benzaldehyde (3.46 g, 20 mmol) and 0.8 g NaOH (10 mmol) were placed in a 200 mL flask, stirred at 0˚C, then 30% H2O2 was added dropwisely. After the mixture was stirred at 0˚C for 2 h and refluxed for 12 h, it was filtrated. The filtrate was then adjusted to pH 6.0 with dilute hydrochloric acid to obtain solid product. The product was washed with water and dried to obtain a white solid of 4-(1H-1,2,4-triazol-1-yl)benzoic acid (Scheme 2). Yield: 3.02 g (80%).
2.4. Synthesis of the Two Complexes
[Cd(TBA)2]·3H2O (1): 0.0410 g (0.2 mmol) of HTBA was dissolved in 7 mL of DMF, and 0.0308 g (0.1 mmol) of Cd(NO3)2·4H2O was added and stirred at room temperature for 1 h. The reaction mixture was sealed in a 25 mL Teflon-lined stainless autoclave, heated at 120˚C under autogenous pressure for 48 h, and then slowly cooled down to room temperature. The colorless block crystals formed were collected, washed with ethanol, and dried at room temperature; Yield, 0.0195 g; 36% (based on Cd). Formula of the compound (molecular mass): C 40.95 (calc. 41.88); H 3.45 (3.49); N 15.44 (16.29)%.


Table 1. Crystallographic data for complexes 1 and 2.
IR spectrum (KBr, cm–1): 3415(s), 3117(s), 1602(s), 1535(vs), 1381(vs), 1278(w), 1148(w), 974(w), 787(m). (see Figure S5 in supporting information).
[Cu(TBA)2]·2H2O (2): 0.0410 g (0.2 mmol) of HTBA was dissolved in 7 mL of DMF, and 0.02625 g (0.1 mmol) of Cu(ClO4)2·6H2O was added and stirred at room temperature for 1 h. The reaction mixture was sealed in a 25 mL Teflon-lined stainless autoclave, heated at 100˚C for 48 h under autogenous pressure, and slowly cooled to room temperature. The blue block crystals formed were collected, washed with ethanol, and dried at room temperature; Yield, 0.0216 g; 43.7% (based on Cu). Formula of the compound (molecular mass): C 45.64 (calc.45.39); H 3.71 (3.36); N 16.91 (17.65)%.
IR spectrum (KBr, cm–1): 3428(m), 3098(m), 1673(m), 1609(s), 1552(vs), 1393(vs), 1281(m), 980(m), 850(m), 779(m), 669(w) (see Figure S6 in supporting information).
3. RESULTS AND DISCUSSION
3.1. Synthesis of the Compounds of 1 and 2
The two compounds were prepared under solvothermal conditions in DMF. The metal ions employed in the synthesis are divalent six-coordinated transition metal cations because we hope that they can incorporate with the ligands of HTBA just as with the ligand of HIBA to form the neutral four-connected frameworks. So far, only two MOFs of 1 and 2, representing two different structural types, have been obtained by reaction of Cd(II) and Cu(II) with the ligand of HTBA. Their crystal structures are determined and their X-ray powder diffraction patterns on the bulk samples were experimentally obtained that were in agreement with those simulated from their X-ray single-crystal data, revealing that the structures of the single crystal 1 and 2 represent those of the bulk samples.
However, the other Cd and Cu group compounds failed to obtain, such as Zn and Ag.
3.2. Structural Description of Complexes 1 and 2
The complex 1 crystallizes in monoclinic, space group C2/c. The asymmetry unit of 1 contains half a cadmium ion, one TBA− ligand and one highly disordered lattice water molecule. The coordination environment of the cadmium ion (Cd) is depicted in Figure 1. In the structure of 1, each of the metal Cd(II) ion is six-coordinated by two nitrogen atoms (N1 and N1#3) from two different TBA− ligands and four carboxylate oxygen atoms (O1#4, O1#5, O2#1, O2#2) from another four different TBA−

Table 2. Selected bond distances (Å) and angles (deg) of complexes 1 and 2.

Figure 1. The coordination environment of Cd(II) in complex 1 (The lattice water molecules and hydrogen atoms have been omitted for clarity. Symmetry codes: #1 -x + 3/2, -y + 3/2, -z + 2; #2 x - 1/2, y + 1/2, z; #3 -x + 1, -y + 2, -z + 2; #4 -x + 3/2, y + 1/2, -z + 3/2; #5 x - 1/2, -y + 3/2, z + 1/2).
ligands; Each TBA− ligand shows a dihedral angle of 16.8˚ between triazole and benzoyl rings and bridges three Cd(II) ions, adopting the (3.21 )syn-anti coordination mode (Scheme 1). Therefore, all the metal ions in the structure exhibit distorted octahedral coordination geometry with the equatorial positions occupied by the carboxylate oxygens donors and the apical positions taken up by the nitrogen atoms.
The N-Cd-O angles slightly deviate from 90˚, revealing the nearly regular octahedron with the equatorial Cd-O distances ranging from 2.303(3) to 2.343(2) Å and the two apical Cd-N distances are identical of 2.343(3) Å. The Cd-N and Cd-O bond distances are similar to that observed in other Cd(II) octahedral coordination polymer [25].
In order to understand the structure thoroughly, the coordinate relationship between the metals and the ligands of complex 1 are analyzed further as follows: We regard that the structure are composed of Cd(II) chains, which are bridged by carboxylate groups of TBA− ligands along [001] direction (Cd···Cd distance of 4.617(1) Å) (Figure 2(b)). And then, these chains are linked by the connected six-five-rings of TBA− ligands into a 3D network with 1D channels. (Cd···Cd contact of 12.738(6) Å, Cd-Cd-Cd angle of 102.24(0)˚ and 77.76(0)˚ respectively) (Figures 2(a), (c)).
The complex 2 crystallizes in orthorhombic, space group Pnna. The asymmetric unit of complex 2 consists of half a copper atom, one TBA− ligand and a free water molecule. In the structure of complex 2, each of Cu(II) ions is six-coordinated by four oxygen atoms and two nitrogen atoms from four different TBA− ligands. Due to Jahn-Teller effect, the Cu(II) ions display a distorted octahedral coordination geometry (Figure 3) with Cu-O1 bond distance of 1.975(4) Å, Cu-O2 bond distance of 2.516(0) Å and Cu-N bond distance of 1.994(5) Å, which are close to those reported in literature [21,22]. Each TBA− ligand in complex 2 links two Cu(II) and shows dihedral angle of 18.7˚ between triazole and phenyl rings, adopting (2.11)syn coordination mode (Scheme 1).
Thus, each metal center is coordinated by four TBA− ligands and each ligand in turn links two Cu(II) ions,

Figure 2. The framework structure of complex 1: (a) View the whole structure along [001] to show the 1D channels; (b) The Cd(II) chains bridged by carboxylate groups oriented along [010]; (c) View the whole structure along [010] to show the connectivity of the chains with the six-five-rings of the TBA− ligands.
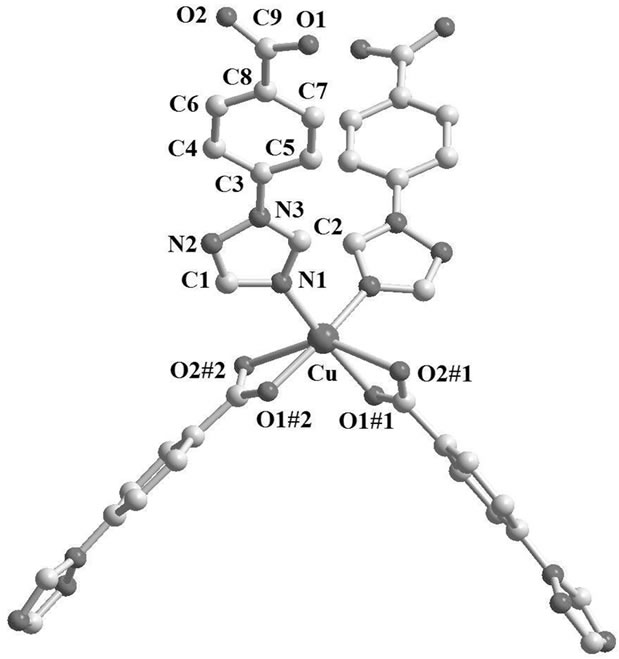
Figure 3. The coordination environment of the Cu(II) in complex 2 (The lattice water molecules and hydrogen atoms have been omitted for clarity. Symmetry codes: #1 x – 1/2, –y + 1/2, z + 1/2 ; #2 –x, y + 1/2, z + 1/2; #3 –x – 1/2, –y + 1, z for 2).
which shows a 4-connected net with diamondoid structure with the adjacent Cu···Cu distance of 11.979(1) Å and the Cu-Cu-Cu angles ranging from 86.35˚ to 131.18˚, deviating significantly from the standard value of 109.5˚ for diamond network (Figure 4(a)). Two set of such nets interpenetrate and form the structures of compound 2 (Figure 4(b)), which show 1D channels of 6.0 × 3.0 Å window (Van der Waals radii was considered) (Figure 4(c)).
3.3. Thermogravimetric and X-Ray Powder Diffraction Analysis
Thermo gravimetric analysis (TGA) was carried out for investigating the thermal stabilities of the coordination polymers. TGA curve of complex 1 exhibits three main steps of weight losses (see Figure S1 in supporting information). The first weight loss starts at 42˚C and completes at 175˚C, which corresponds to the release of three free water molecule per molecule of complex. The observed weight loss of 9.7% is close to the calculated value (9.9%). The second weight loss region occurs in the range of 175˚C - 430˚C. The weight loss is about 33.5% that is corresponding to the decomposition of the framework into CdCO3 and TBA− ligands (calculated 31.77%). The third weight loss region occurs in the range of 520˚C - 630˚C. The weight loss is about 33.5% that is corresponding to the complete decomposition of the TBA− ligands and CdCO3. The final residue is CdO because the TGA curve does not change with the temperature increase above 630˚C (34.67%).
The TGA data of complex 2 indicates three steps of weight lost (See Figure S2 in supporting information). It firstly loses one and a half of crystaline water molecules and two free water molecule per molecule of complex, the in the temperature range of 50˚C - 190˚C (observed 15.2%); Then, the second weight loss occurs at the range of 248˚C - 300˚C. The weight loss is about 14.65% that is corresponding to the decomposition of the framework 480˚C. The weight loss is about 54.5% that is corresponding to the complete decomposition of the TBA− ligands and CuCO3. The final residue is CuO because the
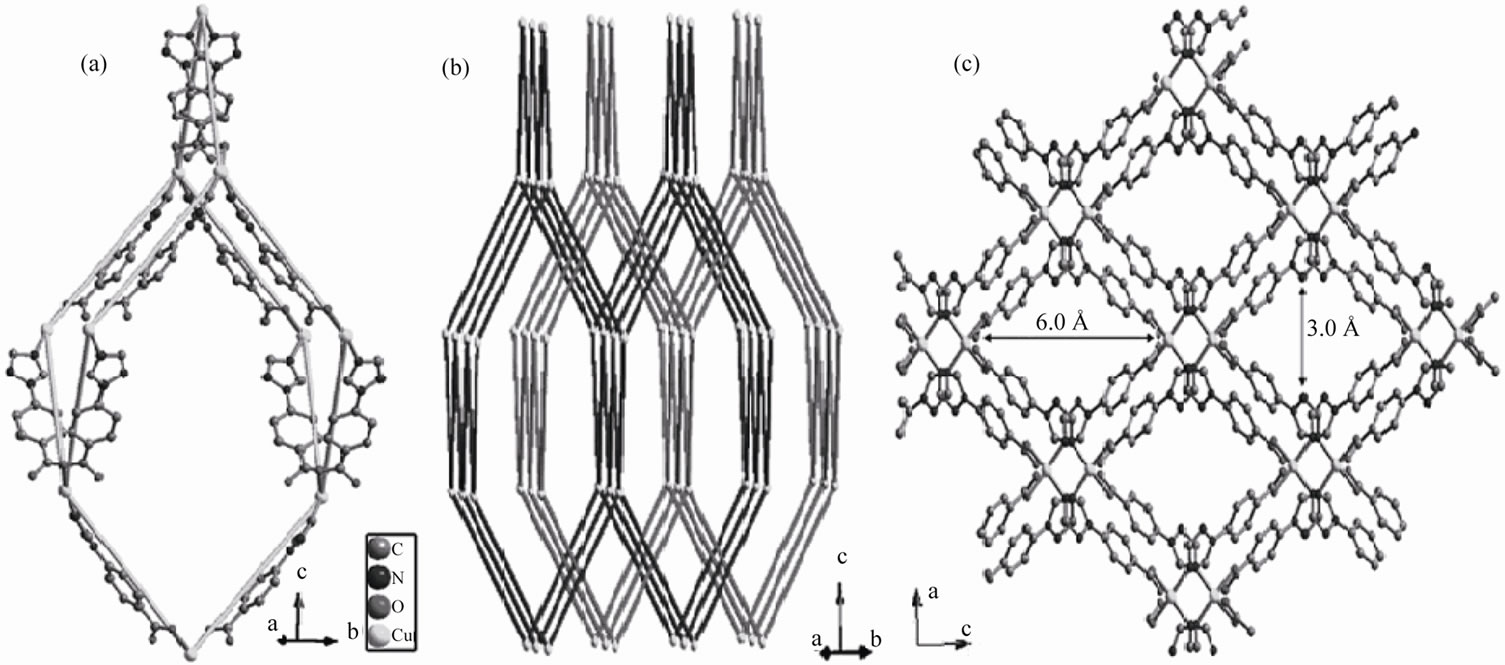
Figure 4. The structure of compound 2: (a) A diamantane unit of the diamond network; (b) The ball-and-stick diagram to display the 2-fold interpenetrating diamondoid net (balls: metals, sticks: TBA− ligands); (c) View the structure along [010] to show the 1D channels.
into CuCO3 and TBA− ligands (calculated 14.58%). The third weight loss region occurs in the range of 350˚C— TGA curve does not change with the temperature increase above 480˚C (56.67%).
The Powder X-ray diffraction (PXRD) was used to confirm the phase purity of the bulk samples of complexes 1 and 2 (see Figures S3 and S4 in the supporting information). All the peaks presented in the measured patterns closely match with the simulated patterns generated from single crystal diffraction data.
4. CONCLUSIONS
In conclusion, by use of 4-(1H-1,2,4-triazol-1-yl) benzoic acid (HTBA), two novel 3D coordination polymers of [Cd(TBA)2]·3H2O (1) and [Cu(TBA)2]·2H2O (2) have been successfully synthesized for the first time under solvothermal conditions. The TBA− ligand exhibits two different coordination modes in the two compounds. Compound 1 has a tubular net consisting of metal-carboxylate chains linked by TBA− ligands, which presents (3.21)syn-anti coordination mode that was not seen in the MOFs constructed by HIBA. Compound 2 demonstrates a 4-connected diamond net of 2-fold interpenetrating structure where the metals serve as the nodes and the TBA− as linkers, which demonstrates (2.11)syn coordination mode that also exists in the MOF constructed by HIBA, but with a two dimensional (4, 4)-network [21,22]. Compared with the numbers of the MOFs constructed by the ligand HIBA, why only two of MOFs could have been produced for the ligand of HTBA is till an open question.
5. SUPPORTING INFORMATION AVAILABLE
TGA, XRD, IR and CIF files for compounds 1 and 2 are available on the http://www.scirp.org/joural/ojic or from the author. The atomic coordinates, isotropic thermal parameters, and complete bond distances and angles have been deposited with the Cambridge Crystallographic Data Center. CCDC Nos. 859904 (1) and 859905 (2) contain the supplementary crystallographic data for 1 - 2. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html, (or from The Cambridge Crystallographic Data Centre, 12, Union Road, Cambridge CB21EZ, UK; E-mail: deposit@ccdc.cam.ac.uk).
6. ACKNOWLEDGEMENTS
We are thankful for financial support from the Foundation for the Author of National Excellent Doctoral Dissertation of PR China (FANEDD) (200733).
REFERENCES
- Xiao, D.R., Wang, E.B., An, H.Y., Li, Y.G., Su, Z.M. and Sun, C.Y. (2006) A bridge between pillared-layer and helical structures: A series of three-dimensional pillared coordination polymers with multiform helical chains. Chemistry—A European Journal, 12, 6528-6541. doi:10.1002/chem.200501308
- Lee, H.Y., Park, J., Lah, M.S. and Hong, J.I. (2008) One-dimensional double helical structure and 4-fold type [2 + 2] interpenetration of diamondoid networks with helical fashion. Crystal Growth & Design, 8, 587-591. doi:10.1021/cg7007232
- Wang, X.L., Qin, C., Wang, E.B., Xu, L., Su, Z.M and Hu, C.W. (2004) Interlocked and interdigitated architectures from self-assembly of long flexible ligands and cadmium salts. Angewandte Chemie International Edition, 43, 5036-5040. doi:10.1002/anie.200460758
- Fan, J., Yee, G.T., Wang, G. and Hanson, B.E. (2006) Syntheses, structures, and magnetic properties of inorganic-organic hybrid cobalt(II) phosphites containing bifunctional ligands. Inorganic Chemistry, 45, 599-608. doi:10.1021/ic051286h
- Fan, J., Slebodnick, C., Angel, R. and Hanson, B.E. (2005) New zinc phosphates decorated by imidazolecontaining ligands. Inorganic Chemistry, 44, 552-558. doi:10.1021/ic0487528
- Wu, T., Yi, B.H. and Li, D. (2005) Two novel nanoporous supramo-lecular architectures based on copper(I) coordination polymers with uniform (8, 3) and (8210) nets: In situ formation of tetrazolate ligands. Inorganic Chemistry, 44, 4130-4132. doi:10.1021/ic050063o
- Choi, H.J., Dinca, M. and Long, J.R. (2008) Broadly hysteretic H2 adsorption in the microporous metal-organic framework Co (1, 4-benzenedipyrazolate). Journal of the American Chemical Society, 130, 7848-7850. doi:10.1021/ja8024092
- Kaye, S.S., Dailly, A., Yaghi, O.M. and Long, J.R. (2007) Impact of preparation and handling on the hydrogen storage properties of Zn4O (1,4-benzenedicarboxylate)3 (MOF-5). Journal of the American Chemical Society, 129, 14176-14177. doi:10.1021/ja076877g
- Dinca, M. and Long, J.R. (2007) High-enthalpy hydrogen adsorption in cation-exchanged variants of the microporous metal-organic framework Mn3[(Mn4Cl)3(BTT)8(CH3- OH)10]2. Journal of the American Chemical Society, 129, 11172-11176. doi:10.1021/ja072871f
- Han, S.S., Furukawa, H., Yaghi, O.M. and William, A.G. III (2008) Covalent organic frameworks as exceptional hydrogen storage materials. Journal of the American Chemical Society, 130, 11580-11581. doi:10.1021/ja803247y
- Wang, Z.X., Shen, X.F., Wang, J., Zhang, P., Li, Y.Z., Nfor, E.N., et al. (2006) A sodalite-like framework based on octacyanomolybdate and neodymium with guest methanol molecules and neodymium octahydrate ions. Angewandte Chemie International Edition, 45, 3287- 3291. doi:10.1002/anie.200600455
- Kajiwara, T., Nakano, M., Kaneko, Y., Takaishi, S., Ito, T., Yamashita, M., et al. (2005) A single-chain magnet formed by a twisted arrangement of ions with easy-plane magnetic anisotropy. Journal of the American Chemical Society, 127, 10150-10151. doi:10.1021/ja052653r
- Liu, T.F., Fu, D., Gao, S., Zhang, Z.Y., Sun, H.L., Su, G. and Liu, Y.J. (2003) An azide-bridged homospin singlechain magnet: [Co(2, 2’-bithiazoline)(N3)2]n. Journal of the American Chemical Society, 125, 13976-13977. doi:10.1021/ja0380751
- Freeman, D.E., Jenkins, D.M., Lavarone, A.T., Freedman, D.E., Jenkins, D.M., Iavarone, A.T. and Long, J.R. (2008) A redox-switchable single-molecule magnet incorporating [Re(CN)7]3–. Journal of the American Chemical Society, 130, 2884-2885. doi:10.1021/ja077527x
- Ohba, M., Kaneko, W., Kitagawa, S. Maeda, T. and Mito, M. (2008) Pressure response of three-dimensional cyanide-bridged bimetallic magnets. Journal of the American Chemical Society, 130, 4475-4484. doi:10.1021/ja7110509
- Kou, H.Z., Jiang, Y.B. and Cui, A.L. (2005) Two-dimensional coordination polymers exhibiting antiferromagnetic Gd(III)-Cu(II) coupling. Crystal Growth & Design, 5, 77-79. doi:10.1021/cg049897s
- Wang, X.F., Lv, Y., Okamura, T.-A., Kawaguchi, H., Wu, G., Sun, W.Y. and Ueyama, N. (2007) Structure variation of mercury(II) halide complexes with different imidazole-containing ligands. Crystal Growth & Design, 7, 1125-1133. doi:10.1021/cg060814c
- Wu, G., Wang, X.F., Okamura, T., Sun, W.Y. and Ueyama, N. (2006) Syntheses, structures, and photoluminescence properties of metal(II) halide complexes with pyridine-containing flexible tripodal ligands. Inorganic Chemistry, 45, 8523-8532. doi:10.1021/ic060493u
- Zhao, X.X., Ma, J.P., Dong, Y.B., Huang, R.Q. and Lai, T.S. (2007) Construction of metal-organic frame-works (M = Cd(II), Co(II), Zn(II), and Cu(II)) based on semirigid oxadiazole bridging ligands by solution and hydrothermal reactions. Crystal Growth & Design, 7, 1058- 1068. doi:10.1021/cg060583+
- Qin, J., Ma, J.P. and Liu, L.L. (2009) A novel two-dimensional frame-work based on unprecedented cadmium(II) chains. Acta crystallographica, 65, 66-68. doi:10.1107/S0108270109000341
- Cui, K.H., Yao, S.Y., Li, H.Q., Li, Y.T., Zhao, H.P., Jiang, C.J. and Tian, Y.Q. (2011) Acentric and chiral four-connected metal-organic frameworks based on the racemic binaphthol-like chiral ligand of 4-(1-H(or methyl)-imidaozol-1-yl) benzoic acid. Crystal Engineering Communication, 13, 3432-3437. doi:10.1039/C0CE00789G
- Zhang, J.Z., Cao, W.R., Pan, J.X. and Chen, Q.W. (2007) A novel two-dimensional square grid cobalt complex: Synthesis, structure, luminescent and magnetic properties. Inorganic Chemistry Communication, 10, 1360-1364. doi:10.1016/j.inoche.2007.08.016
- Sheldrick, G.M. (1996) SADABS. Program for empirical absorption correction for area detector data. University of Göttingen, Göttingen.
- Sheldrick, G.M. (1997) SHELXS 97. Program for crystal structure refinement. University of Göttingen, Göttingen.
- Seidel, R.W., Goddard, R., Zibrowius, B. and Oppel, I.M. (2011) A molecular antenna coordination polymer from cadmium(II) and 4,4’-bipyridine featuring three distinct polymer strands in the crystal. Polymers Chemistry Journal, 3, 1458-1474. doi:10.3390/polym3031458
Supporting Information
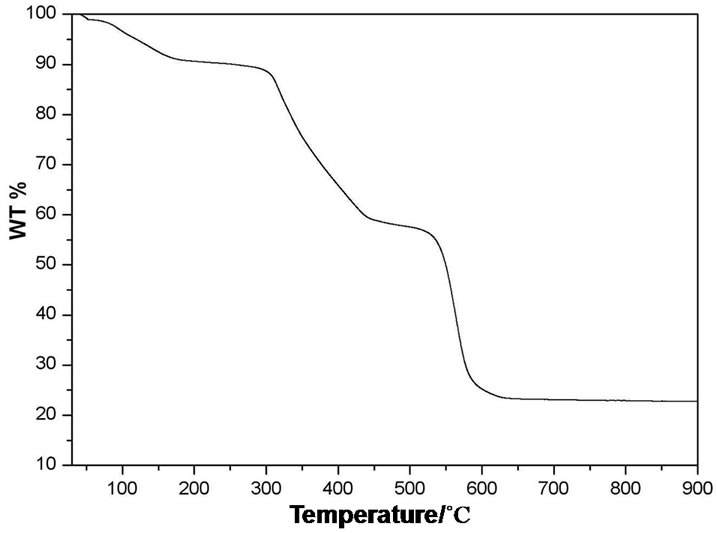
Figure S1. Thermogravimetric analysis curve of compound 1.
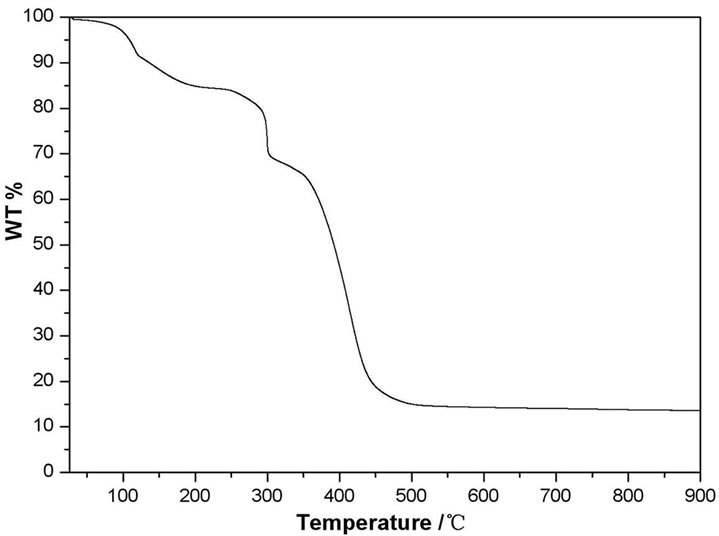
Figure S2. Thermogravimetric analysis curve of compound 2.
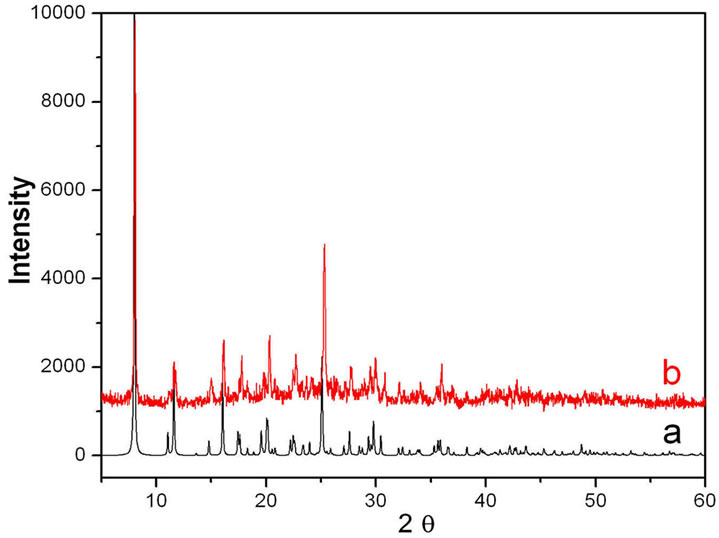
Figure S3. XRD powder patterns: (a) The simulated XPRD pattern calculated from single-crystal structure of complex 1; (b) Experimental XPRD for complex 1.
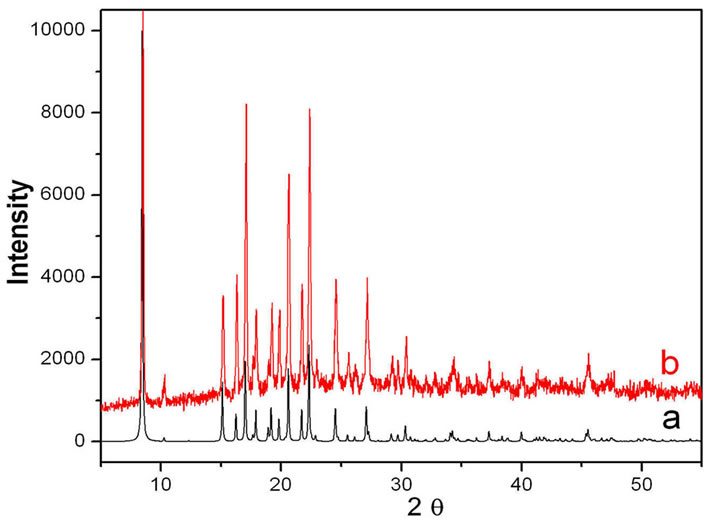
Figure S4. XRD powder patterns: (a) The simulated XPRD pattern calculated from single-crystal structure of complex 2; (b) Experimental XPRD for complex 2.

Figure S5. The infrared spectra for as-synthesized 1.

Figure S6. The infrared spectra for as-synthesized 2.
NOTES
*Corresponding author.

