Open Journal of Physical Chemistry
Vol.3 No.2(2013), Article ID:31776,8 pages DOI:10.4236/ojpc.2013.32011
Methane Steam Reforming on Supported Nickel Based Catalysts. Effect of Oxide ZrO2, La2O3 and Nickel Composition
1Laboratory of Chemistry of Natural Gas, Faculty of Chemistry (USTHB), Algiers, Algeria
2Laboratory of Storage and Valorisation of Renewable Energies, Faculty of Chemistry (USTHB), Algiers, Algeria
3Université M’Hamed Bougara de Boumerdès, U.M.B.B., Boumerdès Department, Boumerdès, Algeria
Email: *sarakila@yahoo.fr
Copyright © 2013 Akila Belhadi et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received December 24, 2012; revised January 20, 2013; accepted February 20, 2013
Keywords: Methane steam reforming; nickel; coke; supported catalysts
ABSTRACT
The catalytic properties of Ni (4 and 10 wt%) supported on both La2O3 and ZrO2 were investigated for the methane steam reforming reaction between 475˚C and 700˚C at atmospheric pressure. The catalysts were prepared by the impregnation method and characterized by several techniques (atomic absorption, BET method, X-ray diffraction and TG-TPO). The catalytic activity of Ni/support systems strongly depends on both of the nature and physico-chemical properties of the support. No deactivation was observed in catalytic systems, whatever the reaction temperature indicating high stability of the catalyst.
1. Introduction
The valorization of the natural gas via the conversion of methane presents attracting interest because of the abundance of natural gas and its low cost. The methane conversion to synthesis gas (mixture of CO and H2) can be realized by different processes like the methane steam reforming (MSR) with H2O [1-4], dry methane reforming with CO2 [5-6] and methane oxidation with molecular oxygen [7-8]. The most catalysts are usually nickel-based systems because of their thermal stability and low cost [2, 9-11].
One of the major problems of these processes is the catalyst deactivation caused by carbon deposits formed on the surface which is related to high temperatures needed to activate the stable methane molecule. It has been reported in several works that the coke formation on Ni-based catalysts is sensitive to the acido basic character of support, metal-support interactions and metal crystalline structure. Supports with strong Lewis basicity as TiO2, ZrO2 [12-14] strong interactions between Ni and support lead to the formation of small Ni crystallites [15], and Ni in a spinel structure as NiAl2O4 [16] can minimize carbon deposit. Moreover, the Ni/Al2O3 systems with additives such as alkaline oxides (CaO, MgO) have also shown to be more resistant to coke formation [17-18].
However, some studies have shown that the deactivation of the catalyst is quite sensitive to the nature of carbon formed on the catalyst surface. Thus, the comparative study on two Ni/γ-Al2O3 and Ni/SiC systems showed that the carbon as nanofilament formed with Ni/γ-Al2O3, is originally the catalyst deactivation by blocking the active sites while the carbon in the nucleation form and growth observed on Ni/SiC favors the conversion of methane. The formation of different structures of carbon from the nickel surface was attributed to the existence of various metal-support interactions which modified the exposed faces of the metal [19].
In MSR reaction over supported Pt based systems, it was observed an enhancement of the catalytic activity that has been attributed to the high amount of carbon deposits around or near the Ni metal particles [20]. It has been underlined that the decomposition of CH4 may take place on the metal particle, resulting in the formation of carbon and hydrogen and that carbon formed can partially reduce the support near the metal particles. Thus, the increase of carbon deposits around or near the particle metal should favor the methane conversion and the active sites are not encapsulated by carbon deposited. The aim of the present work is to report the catalytic behaviour of Ni/ZrO2 and Ni/La2O3 with strong Lewis basicity of support and different loading of Ni (4 and 10 wt%), in the MSR reaction in the temperature range 475˚C - 700˚C. The catalysts have been characterized by BET, atomic absorption, X-ray diffraction (XRD) and TG-TPO techniques.
2. Experimental
2.1. Sample Preparation
ZrO2 was prepared as described by Boulayt et al. [21] from zirconium n-propylate in propane-2-ol (70%). The ZrO2-based precipitate was filtered off, washed several times, dried overnight at 110˚C and then calcined at 300 ˚C/2h and 500˚C/2h with a rate of 2˚C/min under oxygen flow (1.2 L/h). The supported systems, Ni/MO [MO= La2O3 (Merck) and ZrO2] were prepared by impregnation of the support MO with Ni(NO3)2·6H2O aqueous solution (1 M). The mixture was stirred during 2 h at 80˚C. The samples were dried at 80˚C/12h, and calcined under air flow (1.2 L/h) at 300˚C/2h and 500˚C/2h with a rate of 4˚C/min. The catalysts were then sieved to have a particle diameter less than 0.16 mm.
2.2. Characterization
Solid composition was determined by atomic absorption with a spectrometer type Perkin-Elmer 1100B. The specific surface area was determined by the BET method using nitrogen gas as absorbate on a surface analyzer (Coultronics 2100E) after pre-treatment of samples under vacuum at 200˚C for 1 h (5˚C/min) to have a clean surface.
X-ray diffraction powder patterns were recorded with a θ/2θ diffractometer (CGF) using Mo Kα radiation (λ = 0.70930 Å). The apparent sizes of nickel oxide and metal nickel particles were calculated from the Scherrer formula, L = 0.9λ/βcosθ, where β is the width of the most intense peak at half-height and θ the corresponding diffraction angle [22].
2.3. Activity Measurements
The MSR reaction carried out in a quartz fixed bed tubular reactor (L = 65 cm, f = 1 cm) under atmospheric pressure in the temperature range (450˚C - 700˚C), with on-line TCD chromatograph analysis (Hewlett-Packard 5730) using carbosieve B column, 100 - 200 mesh, of 2 m length and hydrogen as vector gas. A thermocouple was installed within the reactor, in contact with catalyst bed. Calcined Ni/MO sample (100 mg) was activated in situ overnight at 500˚C under hydrogen flow (1.2 L/h). The gas feed consisted of methane and water in a ratio H2O/CH4 = 3.3. CH4 (10%)/Ar was introduced to the reaction zone by flowing through a water saturator maintained at 65˚C with a flow rate of 1.2 L/h. Before each analysis, the reactants and products pass through a water-trap at 0˚C to remove water. The conversion of CH4 and product selectivities is calculated using following formulas:
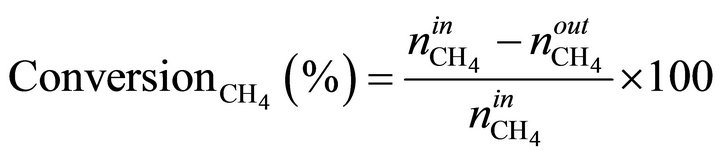
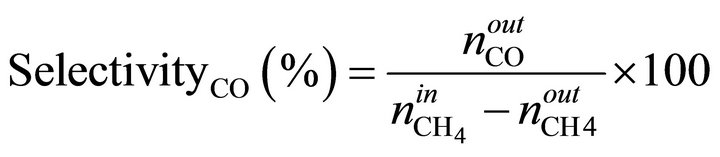

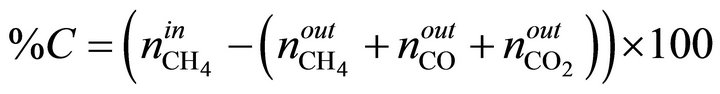
n˚: number of moles.
2.4. Coke Oxidation
In order to determine the carbon amount, the oxidation of coked catalyst was performed using MTB 10 - 8 Setaram Microbalance with relative and absolute sensitivities of 4.10 - 8 and 4.10 - 7 g respectively. The microbalance is linked to a computer via a Cobra interface. 30 mg of coked sample was pretreated at 50˚C under vacuum (10 - 3 Pa) until stabilization of the weight. Molecular oxygen was introduced at a pressure of 200 mbar. The oxidation temperature rose to 550˚C (5˚C/min) and the sample was maintained at this temperature during 6 h.
3. Results and Discussion
3.1. Catalytic Systems Characterization
The physical characteristics of the solids are summarised in Table 1 and XRD patterns of 4% Ni/La2O3 in Figure 1. The atomic absorption analysis shows that the composition of different systems is very close to the theoretical one.
The crystallite size was calculated from X-ray line broadening of NiO and that of Ni peaks (2θ = 19.5˚ and 20˚ respectively) using the Scherrer equation. The results show that the support influence significantly the average size of Ni particles with ca. 12 nm and ca. 22 nm for Ni/La2O3 and Ni/ZrO2 respectively, while for the NiO particles, the value is ca. 36 nm for both carriers. The formation of small Ni particles observed in presence ofLa2O3 can be associated to the stronger interactions between Ni, NiO and La2O3, more basic than ZrO2, as observed in the case of Ni/MgO catalyst [23]. It is noted that the particle size of NiO and Ni appear to be independent of the deposited amount of active phase.
The specific surface areas of 4wt% and 10wt% Ni/ ZrO2 are similar with 86 and 88 m2/g, slightly lower than
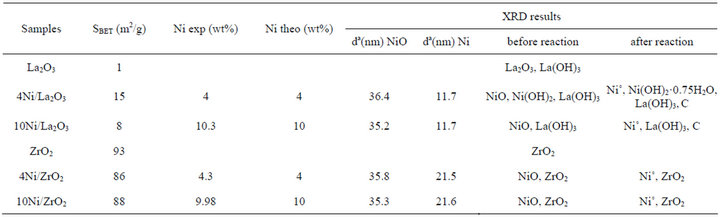
Table 1. Characteristics of Ni/support systems.

Figure 1. XRD patterns of support (a) La2O3 and catalysts 4(%) Ni/La2O3 (b) before and (c) after reaction ![]() : La(OH)3; *: La2O3;
: La(OH)3; *: La2O3; ![]() : Nio;
: Nio; ![]() : Ni(OH)2;
: Ni(OH)2; ![]() : Ni(OH)2 0.75H2O;
: Ni(OH)2 0.75H2O; ![]() : Ni˚,
: Ni˚, ![]() Camorphe.
Camorphe.
that of the ZrO2 support (93 m2/g). La2O3 support has a very low surface area (1 m2/g) that increases to 8 and 15 m2/g after impregnation of 10 and 4 wt% Ni respectively. This increase may be due to the presence of both La(OH)3 and Ni(OH)2 phases examined by XRD analysis. Contrarily to Ni/ZrO2 system, in presence of La2O3, the specific surface area decreases from 15 to 8 m2/g with increasing of Ni percentage from 4 wt% to 10 wt%. This decrease could be explained by the formation of agglomerates on the support surface.
After calcination at 500˚C, the XRD pattern of 4 wt% Ni/La2O3 (Figure 1) shows peaks assigned to Ni(OH)2, La(OH)3 and NiO and no peak corresponding to La2O3 is observed. It is well known that La2O3 is highly hygroscopic at room temperature.
After MSR reaction, the presence of Ni˚ metallic species and that of support are visible in the patterns of Ni/ZrO2 (Table 1), whereas in the case of Ni/ La2O3, in addition to the presence of Ni˚ metallic species, there appear peaks attributed to carbon, Ni(OH)2 and La(OH)3. The absence of peaks corresponding to carbon in presence of Ni/ZrO2 catalyst could be due to an amorphous form of carbon.
3.2. Methane Steam Reforming Reaction
The catalytic performances of supported Ni systems in the MSR reaction were examined in the temperature range (475˚C - 700˚C), after reduction pretreatment under hydrogen flow at 500˚C (1.2 L/h) for 12 h [24]. The MSR reaction over the catalysts leads to the formation of CO, CO2, H2 and carbon and the results are reported in Figures 2, 3 and Tables 2, 3.
3.2.1. Effect of Calcination Temperature
For the preparation of 4%Ni/ZrO2 sample, two calcinations temperatures (500˚C and 700˚C) [25] were used to examine their effect on the catalytic performance. Figure 2 shows methane conversion and CO selectivity as a function of reaction temperature. After reduction pretreatment (H2/500˚C/overnight), 4%Ni/ZrO2 system leads to similar evolution of the methane conversion with reaction temperature for calcinations temperatures 500˚C and 700˚C. When the catalyst is calcined at 500˚C, the CO formation is favoured at low reaction temperatures (<650˚C) and from 650˚C; the CO selectivity is the same for both calcinations temperatures. This result shows that the used calcination temperature, during the catalyst preparation, does not have a significant effect on the conversion whereas CO formation is favored when the calcination temperature is 500˚C. So, the calcination temperature was fixed at 500˚C for all studied systems.
3.2.2. Steady-State Performance
The catalytic activity of 4%wt Ni/support in the MSR reaction was examined in the temperature range (475˚C - 700˚C) after in situ pre-treatment of the catalyst under hydrogen flow at 500˚C overnight (Figure 3).
Similar evolutions of the methane conversion as a

Figure 2. Methane conversion and CO selectivity for 4%Ni/ ZrO2 calcined at 500˚C and at 700˚C at different reaction temperatures, m = 0.1 g, Tred = 500˚C/H2/over-night, d = 1.2 L∙h−1, H2O/CH4 = 3.3.
function of time-on-stream were obtained at different temperatures for 4%wt Ni/ZrO2 catalyst. Steady-state was reached at the beginning of reaction for all temperatures indicating a good stability of this system. Unlike to 4%wt Ni/ZrO2, the catalytic activity of 4%wt Ni/La2O3 decreased with the reaction time and became stable in less than 3 h for reaction temperatures below 700˚C. The methane conversion increases from ca. 20 to ca. 85% in presence of 4%wt Ni/ZrO2 and from ca. 5 to ca. 90% for 4%wt Ni/La2O3 with increasing of reaction temperature from 500˚C to 700˚C, reflecting the endothermic nature of MSR reaction. It is important to note that the catalytic conversion remained unchanged up to 7 h on stream indicating the complete absence of deactivation under the reaction conditions. It is well known that this type of reaction leads to a large amount of carbon on the catalyst
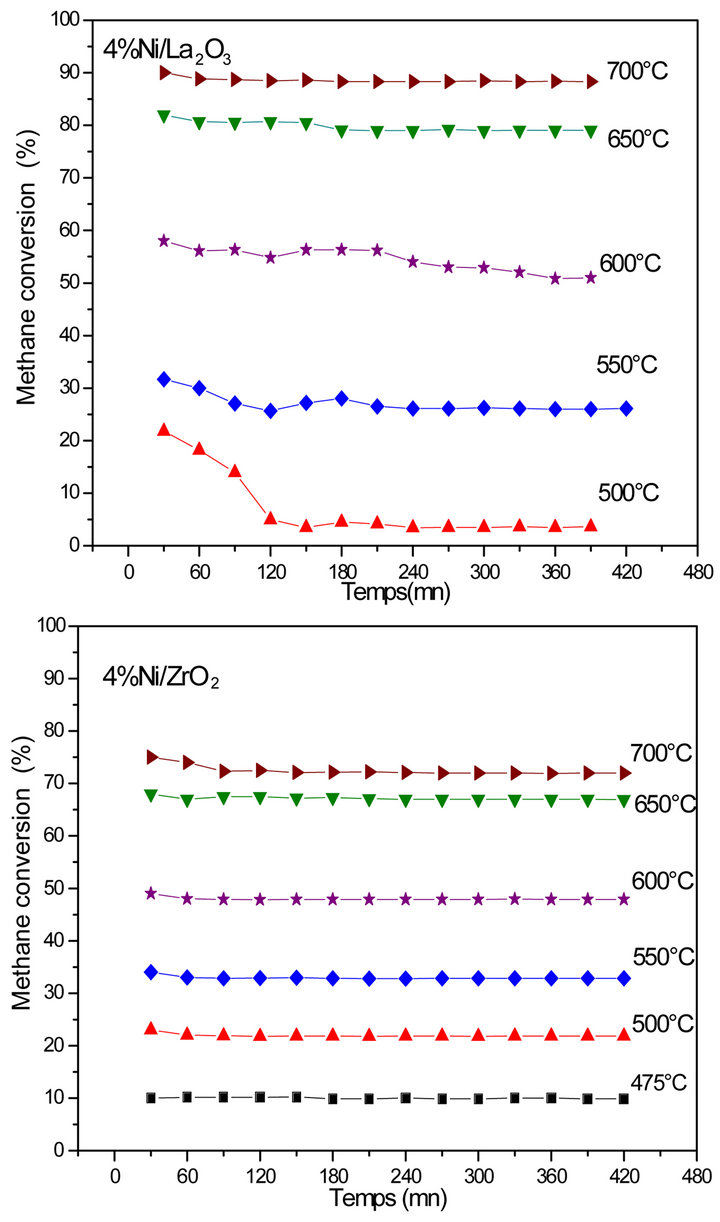
Figure 3. Methane conversion vs. reaction time, for catalysts 4% Ni/La2O3 and 4% Ni/ZrO2 at different temperatures, m = 0.1 g, Tred = 500˚C/H2/overnight, d = 1.2 L∙h−1, H2O/CH4 = 3.3.
surface inducing deactivation [19,24-26].
These results show the promotional effect of both La2O3 and ZrO2 supports on the stability of active sites namely Ni metal. This is probably due to strong metalsupport interactions related to the basicity of the support, responsible of the high activity and stability of the catalyst for the H2O reforming of CH4.
3.2.3. Effect of Reaction Temperature
The methane conversion and product distribution on the MSR reaction over different Ni/support systems in the temperature range 475˚C - 700˚C with a H2O/CH4 molar ratio of 3.3 are shown in tables 2 and 3. The testing results show that catalytic performances of Ni/ZrO2 are very sensitive to Ni content. In the 500˚C - 700˚C temperature range, 4%wt Ni/ZrO2 is more active than 10%wt Ni/ZrO2 with conversions of 22% - 82% against 22% -

Table 2. Catalytic activities of Ni/ZrO2 catalyst.

Table 3. Catalytic activities of Ni/La2O3 catalyst.
72% and more selective toward CO with 30% - 46% against 29% - 40%. It is also noted that 10%wt Ni/ZrO2 shows no activity at 475˚C contrary to 4%wt Ni/ZrO2 that displays a conversion of 22%. On the other hand, over 4%wt Ni/ZrO2 catalyst, the amount of hydrogen did not change markedly with temperature (17.0 - 17.5 mmol/g·h), while on 10%wt Ni/ZrO2, the hydrogen quantity increases substantially from 25.0 to 76.0 mmol/g·h.
CO2 is reaction product at low temperature with 42% of selectivity over 4 wt% Ni/ZrO2 at 475˚C and 30% in the case of 10 wt% Ni/ZrO2 at 500˚C. These values decrease with increasing temperature until they reach zero value above 600˚C. On the contrary, the carbon is the major product at 550˚C with selectivity varying between 48% - 59% for 4%Ni/ZrO2 and from 500˚C with selectivity varying between 41% - 62% for 10%Ni/ZrO2. These results show that the increase of carbon deposits favored the methane conversion. Moreover, the selectivities toward CO and H2 have practically not changed during reaction beyond 550˚C.
The comparison of the results of tables 2 and 3 shows that the nature of support has an effect on catalytic performance of Ni based catalyst, particularly on the product distribution. In contrast to Ni/ZrO2 system, the amount of Ni supported on La2O3 has no effect on the activity of solid. Similar evolution conversions from 20% - 26% to 88% with increasing the reaction temperature from 550˚C to 700˚C, were observed for 4 wt% Ni/La2O3 and 10 wt% Ni/La2O3 while the effect of Ni content on the product distribution is more pronounced. Thus on 4 wt% Ni/La2O3, high CO selectivities were obtained at low reaction temperature (550˚C and 600˚C), 50 and 52% against 20% and 34% respectively for 10 wt% Ni/La2O3. The formation of hydrogen favored on 4 wt% Ni/La2O3 with a amount varying between 26.1 - 80.2 against 0.0 - 40. 4 mmol/g·h for 10 wt% Ni/La2O3. It is noteworthy that 4 wt% Ni/ La2O3 is more selective than Ni/ZrO2 catalyst.
The carbon is the major product beyond 650˚C with selectivities of 63% - 69% for Ni/La2O3 catalysts while the CO2 product is observed only in trace amounts (2% of selectivity) in the presence of 10 wt% Ni/La2O3 at 550˚C. These results show also that the increase of carbon deposits favored the methane conversion as in the case of Ni/ZrO2.
The enhancement of the activity (tables 2 and 3) and the stability (Figure 2) of both Ni/ZrO2 and Ni/La2O3 on the MSR reaction can be attributed to the high amount carbon deposits around or near the Ni metal particles. These results are in agreement with those obtained by other authors on Pt/Al2O3, Pt/ZrO2 and Pt/Ce-ZrO2 systems [7-8,27-28]. It has been reported in these works that the decomposition of CH4 takes place on the metal particle, resulting in the formation of carbon and hydrogen and that carbon formed can partially reduce the support near the metal particles. Thus, the increase of carbon deposits near the metal particles favors the methane conversion and the active sites (Ni metal) were not encapsulated by carbon deposited.
It was underlined that the catalyst deactivation is also sensitive to the nature of the support. Thus, the comparative study performed on two Ni/γ-Al2O3 and Ni/SiC systems showed that the carbon as nanofilament was originally the catalyst deactivation by blocking the active sites while the carbon in the nucleation and growth form favored the conversion of methane. The formation of different structures of carbon from the nickel surface was attributed to the existence of different metal-support interactions which modified the exposed faces of the metal [19].
3.2.4. Determination of Coke Deposited
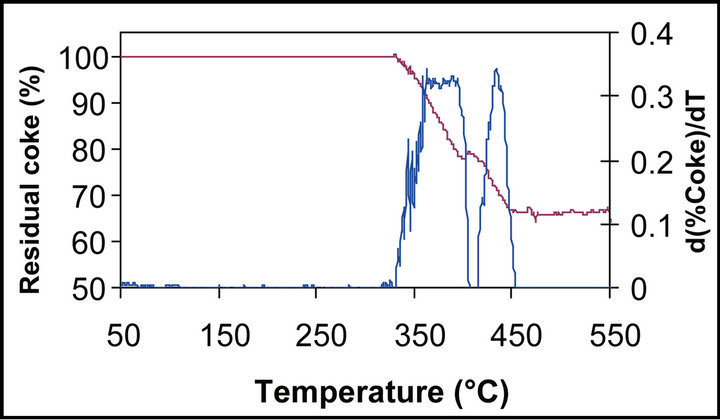
Temperature-programmed oxidation (TPO) coupled tothermogravimetric analysis (TG) was carried out on the Ni/support catalysts after 7 h of SRM reaction at 700˚C (Figure 4). The TG curves of 4% Ni/ZrO2 and 4% Ni/La2O3 (figure not shown) catalysts are similar with weight losses divided into two major events between 340˚C and 500˚C and between 350˚C and 550˚C respecttively attributed to CO2 release, coming probably from two different forms of carbon. It has been reported that graphitic carbon was ignited at a higher temperature of around 500˚C and reactive carbonaceous deposit or adsorbed carbon monoxide on the surface was ignited at a lower temperature below 400˚C [29]. The results also revealed the formation of a higher amount of carbon on 4% Ni/ZrO2 catalyst compared to the 4% Ni/La2O3 catalyst (35 wt% of CO2 against 10 wt%).
TPO-TG analysis shows a different behavior between 10 wt% Ni/support and 4 wt% Ni/support. Thus, with Ni content of 10 wt%, gradual weight losses of ca. 20 wt% from 350˚C to 550˚C for Ni/ZrO2 and of 65 wt% from 360˚C to 500˚C for Ni/La2O3 were observed.
These results indicate that the carbon formation depends on Ni loading and support nature. La2O3 support favored the carbon deposited when Ni content is high contrarily to ZrO2 support.
4. Conclusion
The obtained results showed that supported Ni (4 and 10 wt%) on ZrO2 and La2O3 support exhibited high catalytic activity (72% - 88% of methane conversion) at 700˚C and high stability for the steam reforming methane reaction to synthesis gas. The large amount of carbon depos-
 (a)
(a) (b)
(b)
Figure 4. Residual coke (%) and the DTG during the TPO of: (a)—4% Ni/ZrO2 , (b)—10% Ni/La2O3.
ited on the catalyst surface does not affect the activity of the Ni/ZrO2 and Ni/ La2O3 systems, probably due to a form of carbon that is not detrimental to catalyst activity. On the other hand, the catalysts seem to develop a self-stabilization process during the reaction.
5. Acknowledgements
We are thankful to Pr. Y. Boucheffa (UER de Chimie Appliquée, EMP) for his contribution for thermal analysis (TG, TPO).
REFERENCES
- B. Neumann and K. Jacob, “Equilibrium in Formation of Methane from Carbon Monoxide and Hydrogen, or from Carbon Dioxide and Hydrogen,” Schrift für Elektrochemie, Vol. 30, 1924, pp. 557-576.
- J. R. Rostrup-Nielsen, “Catalytic Steam Reforming,” In: J. R. Anderson and M. Boudart, Eds., Catalysis: Science and Technology, Springer-Verlag, New York, 1984, pp. 1-117. doi:10.1007/978-3-642-93247-2_1
- K. Hou and R. Hughes, “The Kinetics of Methane Steam Reforming over a Ni/a-Al2O Catalyst,” Chemical Engineering Journal, Vol. 82, No. 1-3, 2001, pp. 311-328. doi:10.1016/S1385-8947(00)00367-3
- S. Zhang, J. Wang and X. Wang, “Effect of Calcination Temperature on Structure and Performance of Ni/TiO2- SiO2 Catalyst for CO2 Reforming of Methane,” Journal of Natural Gas Chemistry, Vol. 17, No. 179, 2008, pp. 179- 183. doi:10.1016/S1003-9953(08)60048-1
- V. R Choudhary, B. S Uphade and A. S. Mamman, “Simultaneous Steam and CO2 Reforming of Methane to Syngas over NiO/MgO/SA-5205 in Presence and Absence of Oxygen,” Applied Catalysis A: General, Vol. 168, No. 1, 1998, pp. 33-46. doi:10.1016/S0926-860X(97)00331-1
- J. H. Kim, D. J. Suh, T. J. Park and K. L. Kim, “Effect of Metal Particle Size on Coking during CO2 Reforming of CH4 over Ni-Alumina Aerogel Catalysts,” Applied Catalysis A: General, Vol. 197, No. 2, 2000, pp. 191-200.
- L. V. Mattos, E. R. de Oliveira, P. D. Resende, F. B. Noronha and F. B. Passos, “Partial Oxidation of Methane on Pt/Ce-ZrO2 Catalysts,” Catalysis Today, Vol. 77, No. 3, 2002, pp. 245-256. doi:10.1016/S0920-5861(02)00250-X
- F. B. Noronha, E. C. Fendley, R. R. Soares, W. E. Alvarez and D. E. Resasco, “Correlation between Catalytic Activity and Support Reducibility in the CO2 Reforming of Methane over Pt/CexZr1−xO2 Catalysts,” Chemical Engineering Journal, Vol. 82, No. 1-3, 2001, pp. 21-31. doi:10.1016/S1385-8947(00)00368-5
- H. S. Roh, K. W. Jun, W. S. Dong, J. S. Chang, S. E. Park and J. Yung-II, “Highly Active and Stable Ni/Ce-ZrO2 Catalyst for H2 Production from Methane,” Journal of Molecular Catalysis A: Chemical, Vol. 181, No. 1-2, 2002, pp. 137-142. doi:10.1016/S1381-1169(01)00358-2
- Y. Wang, Y. H. Chin, R. T. Rozmiarek, B. R. Johnson, Y. Gao, J. Watson, A. Y. L. Tonkovich and D. P. V. Wiel. “Highly Active and Stable Rh/MgO-Al2O3 Catalysts for Methane Steam Reforming,” Catalysis Today, Vol. 98, No. 4, 2004, pp. 575-581. doi:10.1016/j.cattod.2004.09.011
- T. Borowiecki, W. Gac and A. Denis, “Effects of Small MoO3 Additions on the Properties of Nickel Catalysts for the Steam Reforming of Hydrocarbons: III. Reduction of Ni-Mo/Al2O3 Catalysts,” Applied Catalysis A: General, Vol. 270, No. 1-2, 2004, pp. 27-36. doi:10.1016/j.apcata.2004.03.044
- T. Wu, Q. Yan and H. Wan, “Partial Oxidation of Methane to Hydrogen and Carbon Monoxide over a Ni/TiO2 Catalyst,” Journal of Molecular Catalysis A: Chemical, Vol. 226, No. 1, 2005, pp. 41-48. doi:10.1016/j.molcata.2004.09.016
- V. R. Choudhary, S. Banerjee and A. M. Rajput, “Hydrogen from Step-Wise Steam Reforming of Methane over Ni/ZrO2: Factors Affecting Catalytic Methane Decomposition and Gasification by Steam of Carbon Formed on the Catalyst,” Applied Catalysis A: General, Vol. 234, No. 1-2, 2002, pp. 259-270. doi:10.1016/S0926-860X(02)00232-6
- R. Takahashi, S. Sato, T. Sodesawa, M. Yoshida and S. Tomiyama, “Addition of Zirconia in Ni/SiO2 Catalyst for Improvement of Steam Resistance,” Applied Catalysis A: General, Vol. 273, No. 1-2, 2004, pp. 211-215. doi:10.1016/j.apcata.2004.06.033
- Z. W. Liu, K. W. Jun, H. S. Roh, S. C. Baek, S. E. Park, “Pulse Study on the Partial Oxidation of Methane over Ni/q-Al2O3 Catalyst,” Journal of Molecular Catalysis A: Chemical, Vol. 189, No. 2, 2002, pp. 283-293. doi:10.1016/S1381-1169(02)00365-5
- N. Sahli, C. Petit, A. C. Roger, A. Kiennemann, S. Libs and M. M. Bettahar; “Ni Catalysts from NiAl2O4 Spinel for CO2 Reforming of Methane,” Catalysis Today, Vol. 113, No. 3-4, 2006, pp. 187-193. doi:10.1016/j.cattod.2005.11.065
- A. S. AL-Ubaid, “The Activity and Stability of Nickel/Silica Catalysts in Water and Methane Reaction,” Industrial & Engineering Chemistry Research, Vol. 27, No. 5, 1988, pp. 790-795. doi:10.1021/ie00077a013
- M. V. Twigg, “Catalyst Handbook Mansson,” 2nd Edition, Manson Publishing, London, 1994.
- P. Leroi, B. Madani, C. Pham-Huu, M. J. Ledoux, S. Savin-Poncet and J. L. Bousquet, “Ni/SiC: A Stable and Active Catalyst for Catalytic Partial Oxidation of Methane,” Catalysis Today, Vol. 91-92, 2004, pp. 53-58. doi:10.1016/j.cattod.2004.03.009
- J. A. C. Ruiz, F. B. Passos, J. M. C. Bueno, E. F. SouzaAguiar, L. V. Mattos and F. B. Noronha, “Syngas Production by Autothermal Reforming of Methane on Supported Platinum Catalysts,” Applied Catalysis A: General, Vol. 334, No. 1-2, 2008, pp. 259-267. doi:10.1016/j.apcata.2007.10.011
- C. Lahousse, A. Aboulayt, F. Maugé, J. Bachelier and J. C. Lavalley, “Acidic and Basic Properties of ZirconiaAlumina and Zirconia-Titania Mixed Oxides,” Journal of Molecular Catalysis, Vol. 84, No. 3, 1993, pp. 283-297. doi:10.1016/0304-5102(93)85061-W
- C. R. Jung, J .Han, S. W. Nam, T. H. Lim, S. A. Hong and H. I. Lee, “Selective Oxidation of CO over CuOCeO2 Catalyst: Effect of Calcination Temperature,” Catalysis Today, Vol. 93-95, 2004, pp. 183-190. doi:10.1016/j.cattod.2004.06.039
- Y. H. Wang and B. Q. Xu, “Comparative Study of Atmospheric and High Pressure CO2 Reforming of Methane over Ni/MgO-AN Catalyst,” Catalysis Letters, Vol. 99, No. 1-2, 2005, pp. 89-96. doi:10.1007/s10562-004-0784-2
- A. Belhadi and O. Cherifi, “Effet des Ajouts métalliques sur les Catalyseurs à Base de Nickel Supportés sur Silice, Dans la réaction de Vaporeformage du méthane,” Journal de la Société Algérienne de Chimie, Vol. 19, No. 1, 2009, pp. 49-61.
- F. Fally, V. Perrichon, H. Vidal, J. Kaspar, G. Blanco, J. M. Pintado, S. Bernal, G. Colon, M. Daturi and J. C. Lavalley, “Modification of the Oxygen Storage Capacity of CeO2-ZrO2 Mixed Oxides after Redox Cycling Aging,” Catalysis Today, Vol. 59, No. 3-4, 2000, pp. 373- 386. doi:10.1016/S0920-5861(00)00302-3
- H. Vidal, J. Kaspar, M. Pijolat, G. Colon, S. Bernal, A. Cordón, V. Perrichon and F. Fally, “Redox Behavior of CeO2-ZrO2 Mixed Oxides: I. Influence of Redox Treatments on High Surface Area Catalysts,” Applied Catalysis B: Environmental, Vol. 27, No. 1, 2000, pp. 49-63. doi:10.1016/S0926-3373(00)00138-7
- S. M. Stagg-Williams and D. E. Resasco, “Effect of Promoters on Supported Pt Catalysts for CO2 reforming of CH4,” Studies in Surface Science and Catalysis, Vol. 119, 1998, pp. 813-818. doi:10.1016/S0167-2991(98)80532-6
- S. M. Stagg-Williams, F. B. Noronha, G. Fendley and D. E. Resasco, “CO2 Reforming of CH4 over Pt/ZrO2 Catalysts Promoted with La and Ce Oxides,” Journal of Catalysis, Vol. 194, No. 2, 2000, pp. 240-249. doi:10.1006/jcat.2000.2939
- D. Li, T. Shishido, Y. Oumi, T. Sano and K. Takehira, “Self-Activation and Self-Regenerative Activity of Trace Rh-Doped Ni/Mg(Al)O Catalysts in Steam Reforming of Methane,” Applied Catalysis A: General, Vol. 332, No. 1, 2007, pp. 98-109. doi:10.1016/j.apcata.2007.08.008
NOTES
*Corresponding author.

