Stem Cell Discovery
Vol. 2 No. 2 (2012) , Article ID: 18714 , 5 pages DOI:10.4236/scd.2012.22006
The CD133/1+ cell subset from human subcutaneous adult fat retains hemogenic potential
![]()
1Experimental Medicine Program, Research Coordination, National Cancer Institute of Rio de Janeiro, Rio de Janeiro, Brazil; *Corresponding author: csondermannf@yahoo.com.br
2Plastic Surgery, Cancer Hospital I, National Cancer Institute of Rio de Janeiro, Rio de Janeiro, Brazil
3Pedro Ernesto Hospital, Rio de Janeiro State University, Rio de Janeiro, Brazil
4Bone Marrow Transplantation Unit, National Cancer Institute of Rio de Janeiro, Rio de Janeiro, Brazil
Received 23 December 2011; revised 25 January 2012; accepted 28 February 2012
Keywords: Adipose Tissue-Derived Stem Cells; CD133; Hematogenesis; Hemangioblast; Pericyte
ABSTRACT
Research has shown that cells from adult fat tissue can effect long-term blood reconstitution. Fat-derived multipotentiality was ascribed to CD34+ perivascular populations from its prominent microvasculature, that represent mostly non-hemogenic, mesenchymal cells, although this tissue contains a CD34+45+ subset committed to a hemogenic fate. Here, in order to analyze cell subsets presenting hemogenic capabilities within fat, CD133/1+ cells and pericytes, the latter defined by CD140b (PDGFRb, Platelet-Derived Growth Factor Receptor Beta) expression, were immunomagnetically selected from stromal-vascular fractions (SVF). In vitro Colony Forming Unit (CFU) assays were negative for CD140b+ pericytes and positive for CD133/1+ cells when a prolonged CFU assay was performed, revealing fat as another store of primitive progenitors that retain hemogenic potential.
1. INTRODUCTION
Adult tissues aside from bone marrow (BM) figure as alternative sources of multipotent cells, representing complex mixtures of stem and progenitor subsets, either committed or not to particular differentiation fates [1,2]. In this context, subcutaneous fat contains large quantities of cells with multipotential capabilities [3], available for numerous therapeutic purposes [4], including long-term blood reconstitution, as was shown in mouse models [5,6].
Fat-derived multipotentiality has been ascribed to CD34+ pericytes [7], among a multiplicity of cell subsets surrounding microvessels [8,9]. However, in this tissue, only the committed CD34+45+ subset presents hemogenic potential, the majority of the CD34+ pool representing non-hemogenic mesenchymal stem cells [10], resembling the CD34+ stroma of fetal bone marrow [11].
In its glycosylated form, surface expressed CD133/1 (AC133) is considered a marker of “stemness” [12,13]. Among progenitors from BM and umbilical cord blood (UCB), CD133 marks a primitive hemogenic subset [14,15], as well as microvasculature-forming endothelial precursor cells (EPC) [16]. Bipotent hemogenic and endothelial progenitors in adult tissues were reported as “adult hemangioblasts” [17], putatively reminiscent from development. The vascular origin of hematogenesis in the embryo has been shown to represent a transdifferentiation event, the blood cells arising directly from endothelial cells of the dorsal aorta [18]. In adults, circulating EPC coexpress CD133, CD34 and VEGFR (Vascular Endothelium Growth Factor Receptor) [19]. It seemed plausible that in fat, which plasticity requires constant neovasculogenesis, EPC at diverse stages would be strongly represented, some of which are putatively CD133+.
In fact, the presence of bipotent hemogenic and endothelial progenitors in fat has been reported, defined by VEGFR expression. Surprisingly, however, these did not relate to CD34 nor CD133 expression [20]. On the other hand, development models point to pericytes as derived from EPC, which are committed under the control of VEGF [21].
Taken together, these findings raise some ambiguity among the potentialities of fat-derived adult progenitors. Here, our group intended to search for cells presenting hemogenic properties within fat subcutaneous tissues, focusing on two cell subsets defined using the surface markers CD133/1 and CD140b (PDGFRb), the latter a marker of pericytes [7,22].
2. METHODS
2.1. Processing Human Lipoaspirates
Human subcutaneous fat tissues were obtained following informed consent, in accordance with the Ethics Committees from both institutions involved (CEP-HUPE 2311, CEP-INCA 42/09). Nonobese women (median weight 62 kg) submitted to elective aesthetic surgeries were donors of abdomen or hip lipoaspirates, extracted following the injection of saline plus epinephrine 1:500,000, without local anesthetic, but under general anesthesia or spinal block. Aspirates from 60 mL sterile syringes were extensively washed by centrifugation with phosphate-buffered saline (PBS), during which fat tissue pieces floated while blood contaminants pelleted and were discarded. Tissue was digested with collagenase type 1A (Sigma-Aldrich, USA) at a final concentration of 0.25 mg/mL in PBS, for 1h at 37˚C, blocked with complete culture medium (DMEM with 10% FBS, GIBCO, Life Tech, USA), and centrifuged at 800 × g for 10 min, in order to collect the pelleted stromal-vascular fraction (SVF).
Fresh SVF cell suspensions were submitted to positive selection through immunomagnetic columns (Miltenyi Biotech, Germany) after incubation with the following Miltenyi bead-conjugated antibodies (Ab): anti-human CD133/1; anti-human CD34; anti-human CD45; goat anti-mouse IgG, as a secondary Ab following primary purified anti-human CD140b (Pharmingen, BD Biosciences, USA).
2.2. CFU Assays
The selected cells (104 - 105) were suspended in Iscove’s Medium (GIBCO) with 2% FBS (Hyclone SH30070.03, Thermo Sci, USA), mixed with semisolid medium with hematopoietic cytokines (Methocult GF 04434, StemCell Tech, USA) (containing Bovine Serum Albumin, 2-Mercaptoethanol, Recombinant Human (rh) Stem Cell Factor, rh GM-CSF, rh IL-3, and rh Erythropoietin), and dispensed onto 35 mm plastic culture plates for the CFU assay. After 2 - 3 weeks colonies were counted, and classified according to standard morphologic parameters as erythroid progenitors (BFU-E), granulocyte-macrophage progenitors (CFU-GM) and mixed granulocyte, erythroid, macrophage and megakaryocyte progenitors (CFU-GEMM). Positive controls for this assay were run in parallel, using CD34+ positively-selected cells from UCBs, obtained following informed consent of the parents of neonates at the Laranjeiras Perinatal Clinic, Rio de Janeiro, after project approval by the Board of the institution, in accordance with the ethical regulations. UCB mononuclear cells were separated by density gradient (Histopaque 1077, Hybri-Max, Sigma), suspended in 12.5% DMSO in complete medium and stored in liquid nitrogen at –196˚C until use.
Colonies from each experiment were harvested as a pool, cells washed with PBS contanining 2% FCS and processed for cytofluorometric analysis as follows. After incubation with FcR Blocking Reagent Human (Miltenyi) for 15 min at 8˚C, plus 20 min with the monoclonal antibodies Phycoerithrin (PE)-conjugated anti-human Glycophorin A, and Fluorescein Isothiocyanate (FITC)-conjugated anti-human CD71 (Transferrin Receptor) (BD Biosciences, USA), cells were 2 times washed and assessed for surface staining in a BD Biosciences FACScan. Data were analyzed using the CellQuest software (BD Biosciences).
Alternatively, they were cytospined in order to access the morphology of individual cells following May-Grunwald-Giemsa staining, using an Olympus Imaging Corp. (Japan) Bx41TF optical microscope. The images were acquired using a digital camera, model C7070WZ, in HQ mode and processed using the Corel Photo-Paint 9 software to adjust brightness/contrast/intensity.
3. RESULTS AND DISCUSSION
As shown in Table 1, among fat-derived cell subsets, only the CD133/1+ selected cells were able to form colonies under the influence of hematopoietic cytokines in CFU assays. The colonies were not as abundant as those from UCB-derived CD34+ progenitors (Figure (1a)), but showed typical myeloid morphology, comprising white (CFU-GM), red (BFU-E) and mixed (CFU-GEMM) colonies (Figure (1b)), with the rate and frequency varying according to the sample (Table 1). The aspect of individual cells recovered from fat-derived CFUs and stained with May-Grunwald-Giemsa seemed compatible with blood forming, although, compared with those from UCB-derived CFUs, they presented a higher frequency of eosinophilic granulocytes and macrophage-like morphologies. Also, a percentage of the cells recovered from fat-derived CFUs revealed, under cytofluorometric analysis, a positive staining for the markers of erythroid development Glycophorin-A and CD71 (Transferrin Receptor) (Figure 2), the first being expressed on cell surface when proerythroblasts first appear [23]. The observed differentiation bias toward eosinophilic granulocytes and macrophage-like morphologies may reflect the previous influence of fat microenvironment, still under analysis.
Compared to CD34+-UCB-derived CFUs, CD133/1+- fat-derived CFUs took longer to reveal as colonies, three weeks instead of the two required by the former. This suggests that they represent primitive hemogenic stem cells present in the subcutaneous fat, and that they possibly share with the UCB-derived CD133/1+ cells the qui-
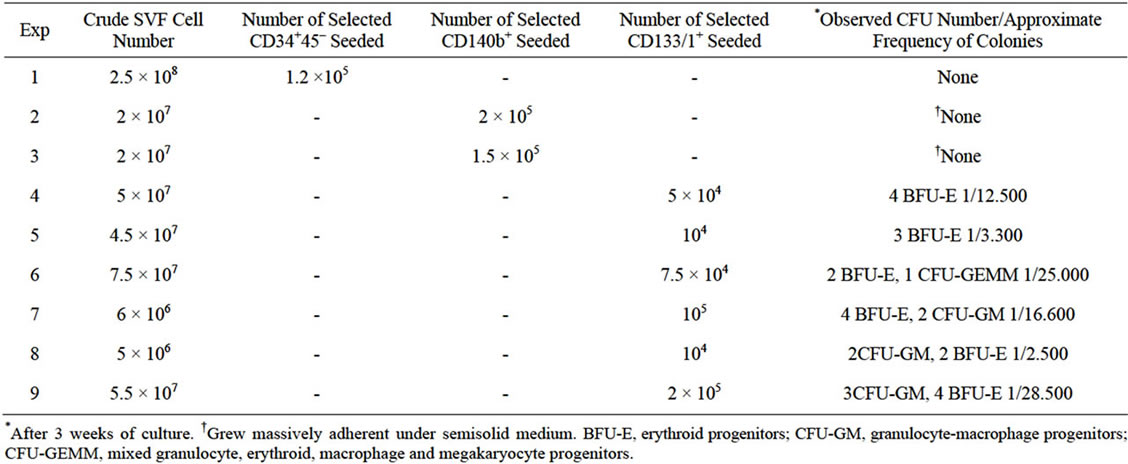
Table 1. Colony Forming Unit assays with cell subsets from stromal-vascular fractions (SVF) of human lipoaspirates.
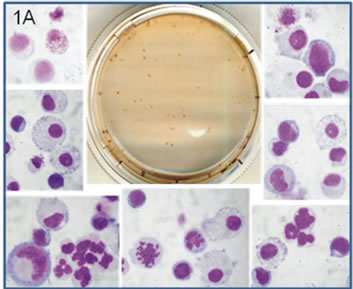
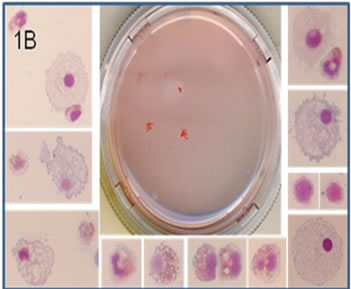
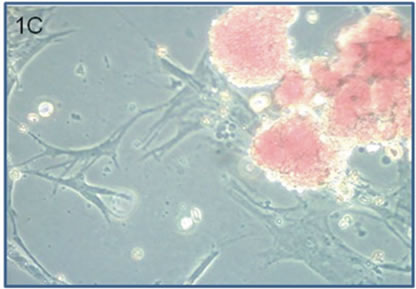
Figure 1. Colony forming unit assay with fat-derived CD133/1+ cells, using umbilical cord blood-derived CD34+ as the positive control. (1A) CD34+ cells from mononuclear fraction of umbilical cord blood, and (1B) CD133/1+ cells from the stromal—vascular fraction of human lipoaspirates, were positively selected and cultured in semisolid medium containing hematopoietic cytokines. After 2 (in 1A) or 3 weeks (in 1B), the colonies were harvested as pools, the cell suspensions were cytospined, and individual cell types were observed following staining with May-Grunwald-Giemsa, using an Olympus Bx41TF optical microscope and a digital camera model C7070WZ in High-Quality mode (original magnification ×400 for individual cells in 1A and 1B). Images were adjusted for brightness/contrast/intensity using the Corel Photo-Paint 9 software. (1C) Using a NIKON TMS-F inverted phase microscope (Japan), some fat-derived CD133/1+ cells were observed adhering to the plates, under the semisolid medium, presenting aspect of mesenchymal cells (original magnification ×300). Shown one experiment typical of six.
escent phenotype [15], thus requiring a longer period of incubation with the cytokines. The presently observed fat-derived primitive progenitors would putatively take part in the long-term hematopoietic reconstitution previously reported in a mouse model [5], besides the committed CD34+45+ progenitors to which it was ascribed.
Neither CD34+45– nor CD140b+ selected cells produced colonies in response to hematopoietic cytokines (Table 1). The latter were observed growing massively under the semisolid medium, adherent to the plates, and displayed typical mesenchymal morphology (not shown). Similarly, in the CD133/1+ selected pool, a few adherent cells grew under the semisolid medium (Figure (1c)), though their characteristics were not accessed in this work. They could represent either mesenchymal contaminants in the selected suspension or true CD133/1+ cells acquiring an adherent phenotype [19,24]. In this respect, Pozzobon et al. [25] reported that BM-derived CD133+ cells possess the ability to adhere to plastic surfaces, acquiring mesenchymal differentiation potentialities, however without the loss of hemogenic or endothelial potential [25]. In contrast, unselected BM-derived mesenchymal cells expanded by adherence did not display hemogenic nor endothelial potential [25], suggesting a mesenchymal commitment not yet achieved by the CD133+ pool, or a hierarchy.
Concluding, in regard to fat-derived cells under the conditions presented, comparison between CD133/1+ and CD140b+ subsets revealed segregation of the hemogenic potential to cells in the CD133/1+ subpopulation, unravelling the ambiguity of potentialities, and pointing to the presence, in adult human fat, of a hemogenic progenitor with a more primitive character than that previously reported. Considering developmental decisions, pericytes

Figure 2. Erythroid development assessed by cytofluorometric analysis of cell suspensions from fat-derived CFUs. Colonies were harvested as pools and washed cell suspensions incubated with FcR Blocking Reagent followed by fluorochrome-conjugated antibodies (anti-Glycophorin A-PE/Gly and anti-CD71- FITC). High left, dot plot shows the analysis windows depicting three cell subpopulations (R1, R2, R3) based on their size (FSC) versus granulosity (SSC). R1, R2 and R3 dot plots show the fluorescence intensity of staining in each subpopulation. R1 (small and smooth cells) comprised Gly negative (neg) cells, either CD71 neg or intermediately (int) positive (pos). R2 (large and smooth cells) comprised mainly Gly pos, CD71 int cells. R3 (both small and large granular cells) comprised mainly Gly neg, CD71 high cells, which probably precedes R2 as an early developmental stage [23]. Shown one of two experiments.
presented commitment to a mesenchymal fate revealed by the unresponsiveness to hemogenic cytokines and massive adherent growth. These findings further punctuate the diversity of stem/progenitor subsets present in adult human fat, still poorly characterized.
4. ACKNOWLEDGEMENTS
FAPERJ, INCA-FIOCRUZ, Laranjeiras Perinatal Clinic Board and Obstetrics Team, Pedro Ernesto Hospital Surgery Team, Experimental Medicine and Pediatric Oncohematology Research Programs of the National Cancer Institute, RJ, Brazil.
REFERENCES
- Freitas, C.S. and Dalmau, S.R. (2006) Multiple sources of non-embryonic multipotent stem cells: processed lipoaspirates and dermis as promising alternatives to bone marrow-derived cell therapies. Cell and Tissue Research, 325, 403-411. doi:10.1007/s00441-006-0172-x
- da Silva Meirelles, L., Chagastelles, P.C. and Nardi, N.B. (2006) Mesenchymal stem cells reside in virtually all postnatal organs and tissues. Journal of Cell Science, 119, 2204-2213. doi:10.1242/jcs.02932
- Zuk, P.A., Zhu, M., Ashjian, P., De Ugarte, D.A., Huang, J.Y., et al. (2002) Human adipose tissue is a source of multipotent stem cells. Molecular Biology of the Cell, 13, 4279-4295. doi:10.1091/mbc.E02-02-0105
- Fraser, J.K., Wulur, I., Alfonso, Z. and Hedrick, M.H. (2006) Fat tissue: an underappreciated source of stem cells for biotechnology. Trends in Biotechnology, 24, 150-154. doi:10.1016/j.tibtech.2006.01.010
- Cousin, B., André, M., Arnaud, E., Pènicaud, L. and Casteilla, L. (2003) Reconstitution of lethally irradiated mice by cells isolated from adipose tissue. Biochemical and Biophysical Research Communications, 301, 1016-1022. doi:10.1016/S0006-291X(03)00061-5
- Han, J., Koh, Y.J., Moon, H.R., Ryoo, H.G., Cho, C.H., et al. (2010) Adipose tissue is an extramedullary reservoir for functional hematopoietic stem and progenitor cells. Blood, 115, 957-964. doi:10.1182/blood-2009-05-219923
- Traktuev, D.O., Merfeld-Clauss, S., Li, J., Kolonin, M., Arap, W., et al. (2008) A population of multipotent CD34- positive adipose stromal cells share pericyte and mesenchymal surface markers, reside in a periendothelial location, and stabilize endothelial networks. Circulation Research, 102, 77-85. doi:10.1161/CIRCRESAHA.107.159475
- Lin, G., Garcia, M., Ning, H., Banie, L., Guo, Y.L., et al. (2008) Defining stem and progenitor cells within adipose tissue. Stem Cells and Development, 17, 1053-1063. doi:10.1089/scd.2008.0117
- Zimmerlin, L., Donnenberg, V.S., Pfeifer, M.E., Meyer, E.M., Péault, B., et al. (2010) Stromal Vascular Progenitors in Adult Human Adipose Tissue. Cytometry Part A, 77, 22-30. doi:10.1002/cyto.a.20884
- Sengenes, C., Lolmede, K., Zakaroff-Girard, A., Busse, R. and Bouloumié, A. (2005) Preadipocytes in the human subcutaneous adipose tissue display distinct features from the adult mesenchymal and hematopoietic stem cells. Journal of Cellular Physiology, 205, 114-122. doi:10.1002/jcp.20381
- Waller, E.K., Huang, S. and Terstappen, L. (1995) Changes in the growth properties of CD34+, CD38– bone marrow progenitors during human fetal development. Blood, 86, 710-718.
- Florek, M., Haase, M., Marzesco, A.-M., Freund, D., Ehninger, G., et al. (2005) Prominin-1/CD133, a neural and hematopoietic stem cell marker, is expressed in adult human differentiated cells and certain types of kidney cancer. Cell and Tissue Research, 319, 15-26. doi:10.1007/s00441-004-1018-z
- Gallacher, L., Murdoch, B., Wu, D.M., Karanu, F.N., Keeney, M., et al. (2000) Isolation and characterization of human CD34.2Lin2 and CD34.1Lin2 hematopoietic stem cells using cell surface markers AC133 and CD7. Blood, 95, 2813-2820.
- Meregalli, M., Farini, A., Belicchi, M. and Torrente, Y. (2010) CD133+ cells isolated from various sources and their role in future clinical perspectives. Expert Opinion on Biological Therapy, 10, 1521-1528.
- Summers, Y.J., Heyworth, C.M., de Wynter, E.A., Hart, C.A., Chang, J., et al. (2004) AC133+ G0 cells from cord blood show a high incidence of long-term culture-initiating cells and a capacity for more than 100 million-fold amplification of colony-forming cells in vitro. Stem Cells, 22, 704-715. doi:10.1634/stemcells.22-5-704
- Aranguren, X.L., Luttun, A., Clavel, C., Moreno, C., Abizanda, G., et al. (2007) In vitro and in vivo arterial differentiation of human multipotent adult progenitor cells. Blood, 109, 2634-2642. doi:10.1182/blood-2006-06-030411
- Loges, S., Fehse, B., Brockmann, M.A., Lamszus, K., Butzal, M., et al. (2004) Identification of the adult human hemangioblast. Stem Cells and Development, 13, 229-242. doi:10.1089/154732804323099163
- Yokomizo, T. and Dzierzak, E. (2010) Three-dimensional cartography of hematopoietic clusters in the vasculature of whole mouse embryos. Development, 137, 3651-3661. doi:10.1242/dev.051094
- Peichev, M., Naiyer, A.J., Pereira, D., Zhu, Z., Lane, W.J., et al. (2000) Expression of VEGFR-2 and AC133 by circulating human CD34+ cells identifies a population of functional endothelial precursors. Blood, 95, 952-958.
- Miñana, M.D., Carbonell-Uberos, F., Mirabet, V., Marín, S. and Encabo, A. (2008) IFATS collection: Identification of hemangioblasts in the adult human adipose tissue. Stem Cells, 26, 2696-2704. doi:10.1634/stemcells.2007-0988
- Hagedorn, M., Balke, M., Schmidt, A., Bloch, W., Kurz, H., et al. (2004) VEGF coordinates interaction of pericytes and endothelial cells during vasculogenesis and experimental angiogenesis. Developmental Dynamics, 230, 23-33. doi:10.1002/dvdy.20020
- Amos, P.J., Shang, H., Bailey, A.M., Taylor, A., Katz, A.J., et al. (2008) IFATS collection: The role of human adipose-derived stromal cells in inflammatory microvascular remodeling and evidence of a perivascular phenotype. Stem Cells, 26, 2682-2690. doi:10.1634/stemcells.2008-0030
- Rogers, C.E., Bradley, M.S., Palsson, B.O. and Koller, M.R. (1996) Flow cytometric analysis of human bone marrow perfusion cultures: erythroid development and relationship with burst-forming units-erythroid. Experimental Hematology, 24, 597-604.
- Handgretinger, R., Gordon, P.R., Leimig, T., Chen, X., Buhring, H.J., et al. (2003) Biology and plasticity of CD133+ hematopoietic stem cells. Annals of the New York Academy of Sciences, 996, 141-151. doi:10.1111/j.1749-6632.2003.tb03242.x
- Pozzobon, M., Piccoli, M., Ditadi, A., Bollini, S., Destro, R., et al. (2009) Mesenchymal stromal cells can be derived from bone marrow CD133+ cells: implications for therapy. Stem Cells and Development, 18, 497-510. doi:10.1089/scd.2008.0003

