New Journal of Glass and Ceramics
Vol.3 No.1(2013), Article ID:27340,5 pages DOI:10.4236/njgc.2013.31003
Sol-Gel Synthesis of SiO2-CaO-Na2O-P2O5 Bioactive Glass Ceramic from Sodium Metasilicate
![]()
1Department of Chemistry, Faculty of Science, University of Lagos, Lagos, Nigeria; 2Department of Chemical Sciences, Bells University of Technology, Ota, Nigeria; 3Department of Chemistry, Prairie View A. & M. University, Prairie View, USA.
Email: *ladams@unilag.edu.ng
Received November 13th, 2012; revised December 13th, 2012; accepted December 23rd, 2012
Keywords: Bioative Glass; Sol-Gel; Crystalline; Sodium Metasilicate; Hydroxyapatite
ABSTRACT
Bioactive glass ceramic with SiO2-Ca2O-Na2O-P2O5 composition was prepared by the sol-gel method using sodium metasilicate (Na2SiO3) as silica source. The monolith obtained was sintered at 1000˚C for 2 hours after which X-ray diffraction (XRD) analysis showed presence of combeite (Na2Ca2Si3O9) as the crystalline phase. In vitro bioactivity test conducted on the material using simulated body fluid (SBF) showed the formation of carbonated hydroxyapatite on its surface. The material during the SBF test was observed to transform from a mechanically strong crystalline phase Na2Ca2Si3O9 to an amorphous phase after incubation for 14 days indicating that the material was biodegradable. Scanning electron microscopy (SEM) was used to investigate the surface morphology, while Fourier transform infrared (FTIR) spectroscopy facilitated the confirmation of hydroxyapatite (HA) formation. The monolith material obtained may be a good candidate for application in tissue engineering scaffolds.
1. Introduction
Despite the significant risks, autograft and allograft treatments for bone loss have achieved varying degrees of success in restoring form and function. While donor site morbidity is a common side effect of autograft [1], complications associated with allografts include disease transmission [2] and the possibility of compromised materials from the sterilization process. Another drawback for both is the limited supply of these materials. Consequently, as life expectancy increases there is also the growing and urgent need for an attractive alternative to bone grafts in form of a biocompatible osteoconductive synthetic scaffolds able to withstand mechanical loading [3], as a paradigm from replacement to regeneration [4].
Silica-based glasses with their interesting properties [5] have been explored for use as alternative second generation bone defects and soft tissue treatment biomaterials due to their biocompatibility [6]. For example, dissolution products from bioactive glasses up-regulate expression of genes that control osteogenesis [7,8], which explains their higher rate of bone formation in comparison to other inorganic ceramics such as hydroxyapatite [9].
The melt method is the traditional approach to produce bioactive glasses from mixtures of carbonates and phosphates at high temperatures before quenching to form a glass. Inclusion of Na2O or K2O offers reduced melting temperature [10] and increased solubility advantages of the materials in aqueous media thereby facilitating host tissue-material interaction, [8,11-14]. Furthermore, it is envisioned that inclusion of Na2O will facilitate crystallization of certain sodium calcium silicate phases, such as Na2Ca2Si3O9 [15] which ultimately will improve their mechanical properties. This approach however, often leads to chemically heterogeneous materials with incipient crystallization and some degree of possible contamination from chemicals used, during the cooling or grinding procedures [16].
A viable alternative is the sol-gel method with several advantages in comparison to the melt and solidification route showing higher purity and homogeneity, and a wider range of compositions [17,18]. In this regards synthesis of sodium-containing quaternary SiO2-CaO-Na2O-P2O5 bioactive glasses are well documented [16,19] based on alkoxysilanes, such as tetraethyl orthosilicate (TEOS) and tetramethyl orthosilicate (TMOS) as precursors. However, alkoxides are hazardous and expensive [20-23] and are therefore not suitable for large-scale production. Our objective in this work is to prepare sodium containing quaternary bioactive glass-ceramic material using sodium metasilicate as cheap silica source.
2. Materials and Experimental Procedures
2.1. Materials
The chemicals used for synthesizing the bioactive glass were: sodium metasilicate, Na2SiO3 with composition SiO2 24.9, Na2O 20.9 and H2O 54.2 wt% (obtained locally), nitric acid (Fluka, Germany), phosphorus pentoxide (BDH Laboratory, 98%) and calcium nitrate tetrahydrate (LOBA, 98%).
2.2. Experimental Procedures
Bioactive glass having the composition (mol%); 50SiO2:25Na2O:21CaO:4P2O5 was synthesized through the sol-gel process from sodium metasilicate, Na2SiO3 according to the following procedure. The Na2SiO3 (5.0 g) was stirred in a beaker using a magnetic stirrer in deionised water (15.0 ml) followed by addition of ethanol (2.5 ml) to give a clear solution. To the stirred mixture was added 2M HNO3 dropwise, stirring was continued further for 1 hour to allow complete hydrolysis. P2O5 (0.5 g) and Ca(NO3)2∙4H2O (4.1 g) were added in sequence under constant stirring. Each reagent was allowed 45 minutes to react before adding the next reagent, finally the mixture was stirred for 1 hour after the last addition. The resulting gel was aged at room temperature for 5 days, dried at 60˚C for 72 hours to give the monolith. The monolith was further heat treated as follows; 200˚C (40 hours), 600˚C (5 hours), 800˚C (3 hours) and 1000˚C (2 hours).
2.3. Methods of Characterization
The sintered specimen was analysed by X-ray diffraction (XRD, Mini Diffractometer MD-10) before and after immersion in SBF using CuKα radiation source operated at 40 kV and 40 mA. The diffraction patterns were obtained in the 2θ range from 10˚ - 75˚. The surface morphology of the material before and after incubation in SBF was assessed by SEM (EVO/MA10) at an accelerating voltage of 15 kV. Fourier transform infrared (FTIR, Perkin Elmer Spectrum X) spectroscopy employing KBr pellets operating in a reflectance mode with a 4 cm−1 resolution was used to monitor the types of bonds present in the glass network before and after immersion in SBF.
2.4. Evaluation of Bone Bonding Ability in Simulated Body Fluid (SBF)
The ability of the material to bond to host bone involves the formation of a carbonated HA layer on the surface of the material, either when implanted or placed in contact with biological fluids [6,23]. This was assessed in vitro in SBF by monitoring the formation of HA on the monolith surface using standard procedure [24]. Solution of analytical grade reagents; NaCl, KCl, K2HPO4, MgCl2, CaCl2 and Na2SO4 of ion concentrations (Table 1) were prepared in deionised water and buffered to pH 7.4 at 37 ˚C with tris (hydroxymethyl) aminomethane and hydrochloric acid (HCl). The dimension of the monolith sample was 10 × 10 × 10 mm which was soaked in an SBF containing flask and kept in an incubator for 14 days. The sample was removed from the incubator and rinsed gently in 100% ethanol, then deionised water, and finally left to dry at ambient temperature in a dessicator. The dried sample was investigated with XRD, SEM and FTIR.
3. Results and Discussion
3.1. Morphology of the Sintered Bioactive Glass
The porous microstructures SEM images of the quarternary bioactive glass are illustrated in Figures 1(a)-(b) at two different magnifications. The cross-section of the sample is coarse and consists of interconnected pores (with average diameter ~3 μm). The presence of small pores and less sidestep surfaces on the sample suggests a crystalline and less residual glass phase. The effect of sintering shows thicker pore struts observed at a higher magnification in Figure 1(b) indicating a nearly fully densified material under the sintering condition (1000˚C for 2 hours).
3.2. XRD Analysis of the Sintered Bioactive Glass
XRD pattern of the bioactive glass sintered at 1000˚C for 2 hours is shown in Figure 2. Both angular location and intensity of the peaks was of good match to the standard PDF #22.1455 by indicating formation of the crystalline phase of Na2Ca2Si3O9. Similar phase has been previously identified [25,26]. The extensive densification and fine crystallinity of Na2Ca2Si3O9 confers on the scaffold good mechanical property [27].
3.3. Biocompatibility Analysis in SBF
The XRD diffraction investigation of the bioglass after immersion in SBF for 14 days is shown in Figure 3. New peaks can be observed at 2θ29˚ and 32˚ assigned to (211) and (320) apatite reflections according to the standard JCPDS (09-0432). There is also an intense apatite
Table 1. Ion concentration of SBF in comparison with human plasma [24].


Figure 1. SEM micrographs of the bioactive glass sintered at 1000˚C for 2 hour at different magnifications.

Figure 2. XRD pattern of the bioactive glass sintered at 1000˚C for 2 hours showing major peaks of Na2Ca2Si3O9.
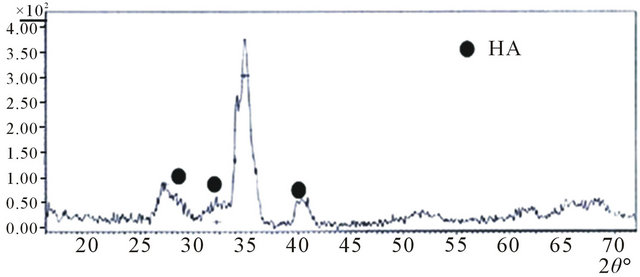
Figure 3. XRD analysis of the sintered bioactive glass after soaking in SBF for 14 days.
peak at 2θ40˚. Analysis of the SEM in Figure 4 shows evidence of an almost complete transition from Na2Ca2Si3O9 to an amorphous phase after soaking for 14 days [28], while the presence of Na2Ca2Si3O9 decreases the kinetics of apatite formation without inhibiting the growth of an apatite layer on the surfaces. It has been reported [27] that 28 days in SBF transforms Na2Ca2Si3O9 completely into possibly an amorphous calcium phosphate phase [5,29] ensuring biodegradability of the scaffold.
Figures 4(a) and (b) shows the surface morphology of the glass-ceramic specimen after incubation in SBF for 14 days. When compared for instance to the neat and SBF un-soaked sample (Figure 1(b)), there is evidence of more rounding and finer grains on the larger grains in Figure 4(b). This pattern is typical of HA precipitated in SBF, populating the surface of the glass-ceramics, Figures 4(a) and (b).
3.4. FTIR Analysis of the Bioactive Glass
The FTIR spectroscopy analysis of the neat and SBF soaked samples are shown in Figures 5(a) and(b) respectively. In Figure 5(a), the peak at 729 cm−1 is assigned to the symmetric Si-O-Si stretching vibration in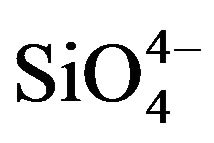 , typical of crystalline silicates. The 477 cm−1 peak is assigned to Si-O-Si bending vibration which is attributed to normal vibration modes of Si-O in the
, typical of crystalline silicates. The 477 cm−1 peak is assigned to Si-O-Si bending vibration which is attributed to normal vibration modes of Si-O in the 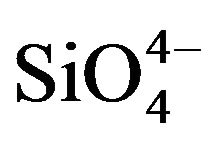 group indicating the presence of amorphous silicate [28]. The peak at 612 cm−1 can be attributed to the crystalline phase which corroborates the XRD result thereby suggesting the presence of crystalline phosphates [30].
group indicating the presence of amorphous silicate [28]. The peak at 612 cm−1 can be attributed to the crystalline phase which corroborates the XRD result thereby suggesting the presence of crystalline phosphates [30].
The intensity of the Si-O-Si bending vibration attributed to normal vibration modes of Si-O decreased from 477 cm−1 to 465 cm−1 after soaking for 14 days in SBF. Furthermore, a new absorption band due to bending mode of 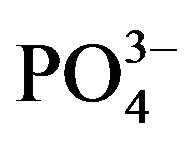 group and attributed to the formation of HA is seen at 575 cm−1 [31]. A very broad OH absorption band from 2500 - 3700 cm−1 and a weak water absorbed band around 1650 cm−1 appeared in the spectrum. The bands at 1420 and 1550 cm−1 were considered for the absorption of carbonate group (ν3). The peak around 900 to 1300 cm−1 were mainly attributed to the phosphate absorptions [16,32].
group and attributed to the formation of HA is seen at 575 cm−1 [31]. A very broad OH absorption band from 2500 - 3700 cm−1 and a weak water absorbed band around 1650 cm−1 appeared in the spectrum. The bands at 1420 and 1550 cm−1 were considered for the absorption of carbonate group (ν3). The peak around 900 to 1300 cm−1 were mainly attributed to the phosphate absorptions [16,32].

Figure 4. SEM micrographs showing the growth of hydroxyapatite on the surface of the glass-ceramic after incubation in SBF for 14 days at different magnifications.
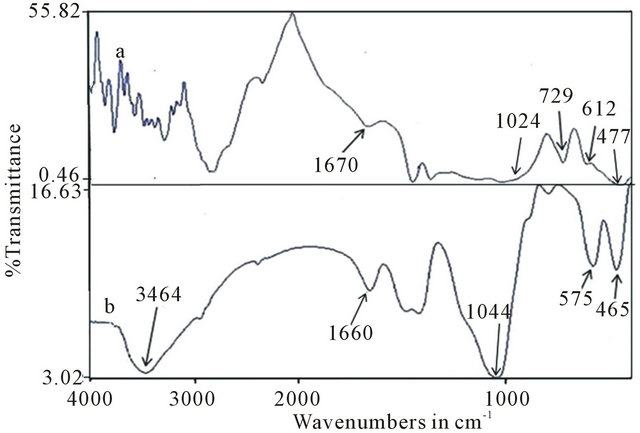
Figure 5. FTIR spectra of the bioactive glass, (a) before and (b) after soaking in SBF for 14 days.
4. Conclusion
SiO2-CaO-Na2O-P2O5-based glass-ceramics was synthesized through the sol-gel process using sodium metasilicate as a silica precursor. The material was sintered at 1000˚C for 2 hours resulting in the formation of the sodium calcium silicate crystal Na2Ca2Si3O9. The growth of carbonated hydroxyapatite on the material after it was soaked in SBF for 14 days and transformation of the crystalline Na2Ca2Si3O9 into almost complete amorphous phase of calcium phosphate showed that the material was highly biocompatible and biodegradable. We therefore envision that sodium metasilicate may be a low cost precursor for sol-gel synthesis of sodium containing quaternary bioactive glass-ceramic materials.
REFERENCES
- E. Nkenke, S. Schultze-Mosgau, M. Radespiel-Troger, F. Kloss and F. W. Neukam, “Morbidity of Harvesting of Chin Grafts: A Prospective Study,” Clinical Oral Implants Research, Vol. 2, No. 5, 2001, pp. 495-502. doi:10.1034/j.1600-0501.2001.120510.x
- S. H. Palmer, C. L. Gibbons and N. A. Athanasou, “Pathology of Bone Allograft,” Journal of Bone and Joint Surgery British, Vol. 81, No. 2, 1999, pp. 333-338. doi:10.1302/0301-620X.81B2.9320
- J. R. Woodward, A. J. Hilldore, S. K. Lan, C. J. Park, A. W. Morgan, J. A. C. Eurell, S. G. Clark, M. B. Wheeler, R. D. Jamison and A. J. W. Johnson, “The Mechanical Properties and Osteoconductivity of Hydroxyapatite Bone Scaffolds with Multi-Scale Porosity,” Biomaterials, Vol. 28, No. 1, 2007, pp. 45-54.
- L. L. Hench and J. M. Polak, “Third Generation Biomaterials,” Science, Vol. 295, No. 5557, 2002, pp. 1014-1017. doi:10.1126/science.1067404
- G. Goller, H. Demirkiran, F. N. Oktar and E. Demirkesen, “Process and Characterization of Bioglass Reinforced Hydroxyapatite Composites,” Ceramics International, Vol. 29, No. 6, 2003, pp. 721-724. doi:10.1016/S0272-8842(02)00223-7
- L. L. Hench and J. Wilson, “Surface-Active Biomaterials,” Science, Vol. 226, No. 4675, 1984, pp. 630-636. doi:10.1126/science.6093253
- I. D. Xynos, A. J. Edgar, L. D. K. Buttery, L. L. Hench and J. M. Polak, “Gene-Expression Profiling of Human Osteoblasts Following Treatment with Ionic Products of Bioglass® 45S5 Dissolution,” Journal of Biomedical and Materials Research, Vol. 55, No. 2, 2001, pp. 151-157. doi:10.1002/1097-4636(200105)55:2<151::AID-JBM1001>3.0.CO;2-D
- L. L. Hench, “Genetic Design of Bioactive Glass,” Journal of European Ceramic Society, Vol. 29, No.7, 2009, pp. 1257-1265. doi:10.1016/j.jeurceramsoc.2008.08.002
- S. K. Ghosh, S. K Nandi, B. Kundu, S. Datta, D. K. De, S. K. Roy and D. Basu, “In Vivo Response of Porous Hydroxyapatite and Beta-Tricalcium Phosphate Prepared by Aqueous Solution Combustion Method and Comparison With Bioglass Scaffolds,” Journal of Biomedical and Materials Research Part B: Applied Biomaterials, Vol. 86B, No. 1, 2008, pp. 217-227. doi:10.1002/jbm.b.31009
- Q. C. Chen, Y. Li, L. Y. Jin, J. M. W. Quinn and P. A. Komesaroff, “A New Sol-Gel Process for Producing Na2O Containing Bioactive Glass Ceramics,” Acta Biomaterialia, Vol. 6, No. 10, 2010, pp. 4143-4153. doi:10.1016/j.actbio.2010.04.022
- L. L. Hench, “Bioceramics,” Journal of American Ceramic Society, Vol. 81, No. 7, 1998, pp. 1705-1728. doi:10.1111/j.1151-2916.1998.tb02540.x
- J. R. Jones, E. Gentleman and J. Polak, “Bioactive Glass Scaffolds for Bone Regeneration,” Elements, Vol. 3, No. 6, 2007, pp. 393-399. doi:10.2113/GSELEMENTS.3.6.393
- P. Sepulveda, J. R. Jones and L. L. Hench, “Characterization of Melt-Derived 45S5 and Gel-Derived Bioactive Glass,” Journal of Biomedical Materials Research, Vol. 58, No. 6, 2001, pp. 734-740. doi:10.1002/jbm.10026
- D. Arcos, D. C. Greenspan and M. Vallet-Regi, “A New Quantitative Method to Evaluate the in Vitro Bioactivity of Melt-Derived Silicate Glasses,” Journal of Biomedical Materials Research, Vol. 65A, No. 3, 2003, pp. 344-351. doi:10.1002/jbm.a.10503
- O. Peitl, E. D. Zanotto, G. P. Latorre and L. L. Hench, “Bioactive Ceramics and Method of Preparing Ceramics”, US Patent No. 041079, 1997.
- R. L. Siqueira, O. Peitl and E. D. Zanotto, “Gel-Derived SiO2-CaO-Na2O-P2O5 Bioactive Powders Synthesis and in Vitro Bioactivity,” Materials Science and Engineering C, Vol. 31, No. 5, 2011, pp. 983-991. doi:10.1016/j.msec.2011.02.018
- S. Sakka, “Handbook of Sol-Gel Science and Technology: Processing, Characterization and Applications,” Kluwer Academic Publishers, New York, 2005.
- J. R. Jones, “New Trends in Bioactive Scaffolds: The Importance of Nanostructure,” Journal of European Ceramic Society, Vol. 29, No. 7, 2009, pp. 1275-1281. doi:10.1016/j.jeurceramsoc.2008.08.003
- P. Saravanapavan, J. R. Jones, R. S. Pryce and L. L. Hench, “Bioactivity of Gel-Glass Powders in the CaO-SiO2 System: A Comparison with Ternary (CaO-P2O5-SiO2) and Quaternary Glasses (SiO2-CaO-P2O5-Na2O),” Journal of Biomedical Materials Research Part A, Vol. 66, No. 1, 2003, pp. 110-119. doi:10.1002/jbm.a.10532
- Z. Li, B. Hou, Y. Xu, D. Wu, Y. Sun, W. Hu and F. Deng, “Comparative Study of SolGel Hydrothermal and Sol-Gel Synthesis of Titania-Silica Composite Nanoparticles,” Journal Solid State Chemisty, Vol. 178, No. 5, 2005, pp. 1395-1405. doi:10.1016/j.jssc.2004.12.034
- E. Pabon, J. Retuert, R. Quijada and A. Zarate, “TiO2-SiO2 Mixed Oxides Prepared by a Combined Sol-Gel and Polymer Inclusion Method,” Microporous Materials, Vol. 67, No. 2-3, 2004, pp. 195-203. doi:10.1016/j.micromeso.2003.10.017
- M. Crisan, M. Raileanu, S. Preda, M. Zaharescu, A. M. Valean, E. J. Popovici, V. S. Teodorescu, V. Matejec and J. Mrazek, “Manganese Doped Sol-Gel Materials with Catalytic Properties” Journal of Optoelectronics and Advanced Materials, Vol. 8, No. 2, 2006, pp. 815-819.
- T. Kokubo, “Apatite Formation on Surfaces of Ceramics, Metals and Polymers in Body Environment,” Acta Materialia, Vol. 46, No. 7, 1998, pp. 2519-2527.
- T. Kokubo and H. Takadama, “How Useful Is SBF in Predicting in Vivo Bone Bioactivity?” Biomaterials, Vol. 27, No. 15, 2006, pp. 2907-2915. doi:10.1016/j.biomaterials.2006.01.017
- O. Peitl, E. D. Zanotto and L. L. Hench, “Highly Bioactive P2O5-Na2O-CaO-SiO2 Glass-Ceramics,” Journal of Non-Crystalline Solids, Vol. 292, No. 1-3, 2001, pp. 115- 126. doi:10.1016/S0022-3093(01)00822-5
- D. C. Clupper, J. J. Mecholsky, G. P. LaTorre and D. C. Greenspan, “Bioactivity of Tape Cast and Sintered GlassCeramics in Simulated Body Fluid,” Biomaterials, Vol. 23, No. 12, 2002, pp. 2599-2606. doi:10.1016/S0142-9612(01)00398-2
- Q. Z Chen, I. D. Thompson and A. R. Boccaccini, “45S5 Bioglass-Derived Glass-Ceramic Scaffolds for Tissue Engineering,” Biomaterials, Vol. 27, No. 11, 2006, pp. 2414-2425. doi:10.1016/j.biomaterials.2005.11.025
- O. Peitl, E. D. Zanotto, L. L. Hench, “Effect of Crystallization on Apatite Layer Formation of Bioactive Glass 45S5,” Journal of Biomedical Materials Research, Vol. 30, No. 4, 1996, pp. 509-514. doi:10.1002/(SICI)1097-4636(199604)30:4<509::AID-JBM9>3.0.CO;2-T
- J. Zhong and D. C. Greespan, “Processing and Properties of Sol-Gel Bioactive Glasses,” Journal of Biomedical Materials Research: Applied. Biomaterials, Vol. 53, No. 6, 2000, pp. 694-701.
- L. Lefebvre, J. Chevalier, L. Gremillard, R. Zenati, G. Thollet, D. Bernache-Assolant and A. Govin, “Structural Transformations of Bioactive Glass 45S5 with Thermal Treatments,” Acta Materialia, Vol. 55, No. 10, 2007, pp. 3305-3313. doi:10.1016/j.actamat.2007.01.029
- S. R. Radin and P. Ducheyne, “Plasma Spraying Induced Changes of Calcium Ceramic Characteristics and Effect on in Vitro Stability,” Journal of Materials Science: Materials in Medicine, Vol. 3, No, 1, 1992, pp. 33-42. doi:10.1007/BF00702942
- J. Wang, Q. Liu, J. G. C. Wolke, X. Zhang and K. De Groot, “Formation and Characteristics of the Apatite Layer on Plasma-Sprayed Hydroxyapatite Coating in Simulated Body Fluid,” Biomaterials, Vol. 18, No. 15, 1997, pp. 1027-1035. doi:10.1016/S0142-9612(97)00022-7
NOTES
*Corresponding author.

