International Journal of Organic Chemistry
Vol.2 No.3A(2012), Article ID:25102,3 pages DOI:10.4236/ijoc.2012.223041
Quantitative Determination of Click Reaction in the Presence of Glycine Derivatives and in Dilute Solution
Graduate School of Agriculture, Hokkaido University, Sapporo, Japan
Email: *hasimoto@abs.agr.hokudai.ac.jp
Received August 15, 2012; revised September 21, 2012; accepted October 2, 2012
Keywords: Click Chemistry; Quantitative Determination; Propargylglycine; Cu(I); Buffer
ABSTRACT
The click reaction is one of the latest techniques for the functional analysis of bioactive compounds and the analysis makes novel concepts and strategies for medicinal chemistry. N-methylated glycine derivatives have inhibitory activity for the click reaction in direct Cu(I) system because of decrease of Cu(I) concentration. The Cu(I) concentration recovered effectively by sodium ascorbate. Quantitative determination of click reaction at various ligand concentrations revealed that the decrease in reaction yields was observed in a substrate concentration-dependent manner.
1. Introduction
The functional analysis of bioactive compounds is important for medicinal chemistry and drug discovery. The chemical modification of bioactive compounds is one of the major strategies to study biochemical aspects. There are many advantages of introduction of unnatural functional groups to bioactive compounds because it is very difficult to obtain reaction selectivity when the common functional groups of biomolecules were used for exogenous labeling. Click chemistry is the study of highly efficient addition reactions. The most well-known click reaction is the Cu(I) catalyzed azide-alkyne cycloaddition reaction, which forms a triazole ring linkage [1]. Both azide and alkyne, which are small unnatural functional groups, are available for the selective chemical modification of bioactive compounds. We have recently reported on inhibition of click reaction with a-amino acid skeletons (glycine and propargylglycine) because the Cu(I) concentration in direct Cu(I) system was decreased in the presence of N-unsubstituted a-amino acids. We also found sodium ascorbate could recover the Cu(I) concentration in the reaction mixture [2]. No report has been published in substitution effects of a-amino acids. Furthermore it has been reported that the click reactions are characterized by rapid and quantitative conversion from starting materials to products [1]. The typical organic reaction efficiency was decreased dramatically at the low reactant concentrations. There are only few reports on quantitative determination of click reaction in dilute solution [3]. In this paper, we report the quantitative determination of click reaction with inhibition of N-methylated glycine derivatives and effects on yields in low reactant concentrations.
2. Results and Discussion
2.1. Inhibitory Effects of N-Substituted Glycine Derivatives for Click Reaction
We have been reported that glycine (4) inhibited click reaction of N-Boc-propargylglycine (1) and benzyl azide (2) in the presence of CuBr at room temperature [2]. Colorimetric assay with bathocuproine disulphonic acid [4-6] during the reaction revealed the Cu(I) concentration was decreased in a time dependent manner. N-unprotected glycine inhibited the reaction, but on the other hand N-Boc protected a-amino acid did not inhibit the reaction. These results indicated that the amide (urethane) bond did not cause decrease the Cu(I) concentration. The click reaction mixture of 1 and 2 in the presence of glycine (4) and N-methyl substituted glycine derivatives (sarcosine 5, N,N-dimethylglycine 6 and betaine 7, 1 eq) were subjected to colorimetric assay for Cu(I) determination. Each glycine derivatives and CuBr were dissolved in t-BuOH-H2O and the mixtures were subjected to colorimetric assay every hour. All additives promoted to decrease Cu(I) concentration in a time-dependent manner, but slow decrease was observed in the mixture with quaternary ammonium salt (betaine, 7). Compounds 1 and 2 were added to the reaction mixture after 3 hours. It was observed no significant differences for the Cu(I) concentration after click substrate addition. The click product was detected by addition of quaternary ammonium salt (betaine, 7). These results suggested that concentration of Cu(I) was relatively high in the quaternary ammonium salt mixture than others, and the click reaction proceeded partially. But there was not enough Cu(I) concentration to react completely. Finally sodium ascorbate (0.2 eq) was added the reaction mixtures. Concentration of the Cu(I) recovered and the click reaction was completed within an hour in all reaction mixtures (Table 1). It was observed the same decrease of Cu(I) concentration by glycine derivatives when the all substrates were added in the solution at same time and addition of sodium ascorbate recovered the Cu(I) concentration.
These inhibitory activities of N-alkylglycine skeletons for the click reactions were observed due to decrease of Cu(I) concentration in the reaction mixture. There have been reported that some of tertially amine enhanced the click reactions [7]. The decrease of Cu(I) concentration derived from N-unsubstituted or N-alkylated a-amino acid skeletons. It was considered that quaternary ammo nium
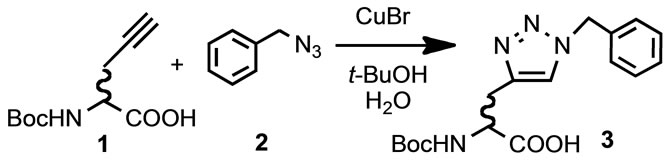

Table 1. Quantitative determination of Cu(I) with colorimetric assay at 485 nm with bathocuproine disulphonic acid and click product 3 in the presence of N-methyl glycine derivatives 4 - 7.
salt (betaine 7) weakest potential inhibitory activity.
2.2. Quantative Determination of Click Reaction in Dilute Solution
It has been reported that the click reaction proceeded quantitatively in various condition [1]. The effect of many organic reactions depends on reactant concentrations and low reactant concentration sometimes caused low yields. As we would expect, click reaction for functional analysis have to apply very low amount of target molecules. We would like to establish ligand concentration manner for the click reaction. The reaction of benzyl azide 2 (280 mM) and an equivalent of DL-propargylglycine 8 were proceeded in the presence of CuBr (0.3 eq) and sodium ascorbate (0.2 eq) in t-BuOH and H2O at 75˚C for a day. Some common buffers (acetate, Tris and phosphate) were subjected for click reaction solvents to apply the click reaction for functional analysis. The product 9 was determined by HPLC, which can quantify 9 in the range of 0.2 to 3 nmol. Phosphate buffer (pH 7.2) afforded highest yield for the click reaction of compounds 2 and 8 in the condition, but other buffers also provided enough results to apply functional analysis (Table 2).
Different concentrations of compound 8 were subjectted to click reaction with equivalent amount of 2 in 0.1 M phosphate buffer (pH 7.2). Concentration-dependent manner and drastic reduction in the click reaction yield at concentrations of 8 lower than 10 mM were observed (Table 3). The low yields did not improve when the 10 equivalents of 2 against 8 were subjected to the click reaction.
3. Experimantal
3.1. General Information
Bathocuproine disulphonic acid and N,N-dimethylglycine were purchased from Dojin (Kumamoto, Japan) and Wako Chemical (Osaka, Japan), respectively. Sarcosine and betaine anhydrous were purchased from Tokyo kasei
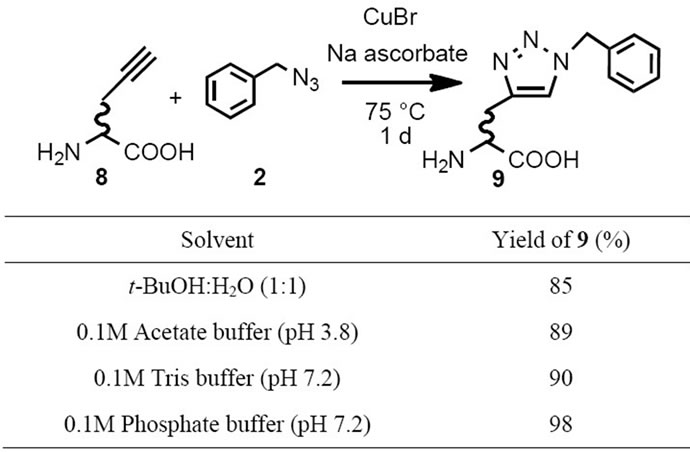
Table 2. Solvent effects on the click reaction for propargylglycine 8 and benzyl azide 2.
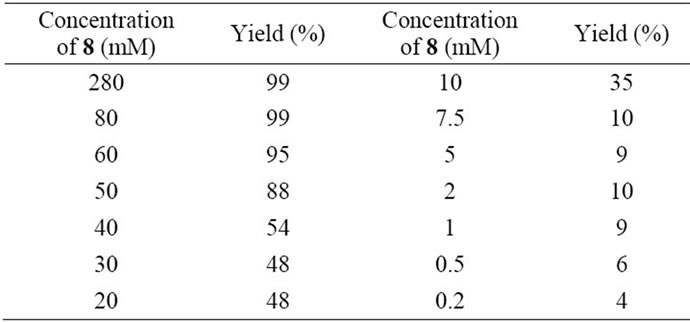
Table 3. Click reaction in substrate 8 concentration-dependent manner in phosphate buffer (pH 7.2).
(Tokyo, Japan). DL-propargylglycine was obtained from Sigma (St. Louis, USA). Chirobiotic T HPLC column was purchased from Supelco (Bellefonte, USA). All other reagents were commercially available and used without further purification.
3.2. Quantitative Determination of Cu(I) in the Click Reaction with Bathocuproine Disulphonic Acid
CuBr (0.1 eq against compound DL-1) and amine (1 eq) were suspended in t-BuOH and H2O (1:1) at room temperature. After 3 hours, the compound DL-1 and 2 (1 eq) were added the suspension and the reaction mixture was stirred at same temperature for 4 hours. Finally, sodium ascorbate (0.2 eq) was added the reaction mixture. A part of the mixture (9 µL) at indicated time in Table 1 was pipetted out and added 17 mM bathocuproine disulphonic acid aqueous solution (15 µL) and sodium acetate solution (1.22 M, 840 µL). The assay mixture was adjusted to 1 mL with distilled water then subjected to absorption measurement at 485 nm with spectrophotometer. The click reaction mixtures were subjected to silicacolumn chromatography (CH2Cl2:CH3OH = 4:1, then 2:1) to afford 3 as pale yellow oil. The analytical data was identical to those of our previous report [2].
3.3. Click Reaction in a Substrate Concentration-Dependent Manner
Same equivalent of compound DL-8 and 2 at various concentrations were dissolved in the solvent. CuBr (0.3 eq) and sodium ascorbate (0.2 eq) were added to the mixture. The reaction mixture was heated at 75˚C for a day. The insoluble material was dissolved by addition of 1M HCl, then the reaction mixture was diluted to between 0.1 and 1.5 mM. The diluted samples were subjected to HPLC (Chirobiotic T 4.6 × 250 mm, CH3OH: H2O = 1:1, 1 mL/min, deteaction at 210 nm). The analysis of product 9 was identical to those described previously [2].
4. Conclusion
In conclusion, we have demonstrated that Cu(I) concentration of click reaction in direct Cu(I) system decreased in the presence of glycine derivatives and the a-amino acid moiety was important for the inhibition. The results indicated N-unsubstituted or N-methylated a-amino acid skeleton might promote oxidation of Cu(I) and sodium ascorbate could reduce the oxidized copper species to Cu(I) to proceed the click reaction. Furthermore, the click reaction yields are affected by substrate concentration-dependent manner at low concentrations. It needs scrupulous attention to the experiments of chemical modifications at low amount biomolecules with click chemistry. These findings will be very useful for functional analysis of bioactive compounds in the medicinal chemistry.
5. Acknowledgements
This research was partially supported by a Ministry of Education, Science, Sports and Culture Grant-in-Aid for Scientific Research (C), 19510210, 21510219.
REFERENCES
- H. C. Kolb, M. G. Finn and K. B. Sharpless, “Click Chemistry: Diverse Chemical Function from a Few Good Reactions,” Angewandte Chemie International Edition, Vol. 40, No. 11, 2001, pp. 2004-2021. doi:10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5
- Y. Ogasawara, Y. Murai, Y. Sakihama, Y. Hashidoko and M. Hashimoto, “Quantitative Analysis of Cu(I) Concentration in Click Chemistry—Biotinylation at Side Chain of Propargylglycine Using Click Chemistry under Heating Conditions,” Heterocycles, 2012. doi:10.3987/COM-12-S(N)32
- V. Hong, S. I. Presolski, C. Ma and M. G. Finn, “Analysis and Optimization of Copper-Catalyzed Azide-Alkyne Cycloaddition for Bioconjugation,” Angewandte Chemie International Edition, Vol. 48, No. 52, 2009, pp. 9879-9883. doi:10.1002/anie.200905087
- A. T. Faizullah and A. Townshend, “Spectrophotometric Determination of Copper by Flow Injection Analysis with an On-Line Reduction Column,” Analytica Chimica Acta, Vol. 172, No. 1, 1985, pp. 291-296. doi:10.1016/S0003-2670(00)82618-5
- W. G. Lewis, F. G. Magallon, V. V. Fokin and M. G. Finn, “Discovery and Characterization of Catalysts for AzideAlkyne Cycloaddition by Fluorescence Quenching,” Journal of American Chemical Society, Vol. 126, No. 30, 2004, pp. 9152-9153. doi:10.1021/ja048425z
- S. S. Gupta, J. Kuzelka, P. Singh, W. G. Lewis, M. Manchester and M. G. Finn, “Accelerated Bioorthogonal Conjugation: A Practical Method for the Ligation of Diverse Functional Molecules to a Polyvalent Virus Scaffold,” Bioconjugate Chemistry, Vol. 16, No. 6, 2005, pp. 1572-1579. doi:10.1021/bc050147l
- M. Meldal and C. W. Tornøe, “Cu-Catalyzed Azide-Alkyne Cycloaddition,” Chemical Reviews, Vol. 108, No. 8, 2008, pp. 2952-3015. doi:10.1021/cr0783479
NOTES
*Corresponding author.

