 Psychology 2013. Vol.4, No.4, 389-395 Published Online April 2013 in SciRes (http://www.scirp.org/journal/psych) http://dx.doi.org/10.4236/psych.2013.44056 Copyright © 2013 SciRes. 389 Modulation of the Startle Reflex during Brief and Sustained Exposure to Emotional Pictures Aimee Mavratzakis1,2, Elaine Molloy1, Peter Walla1,2* 1Faculty of Science and Information Technology, School of Psychology, University of Newcastle, Callaghan, Australia 2Centre for Translational Neuroscience and Mental Health Research, University of Newcastle, Callaghan, Australia Email: *peter.walla@newcastle.edu.au Received December 6th, 2012; revised January 7th, 2013; accepted February 5th, 2013 Copyright © 2013 Aimee Mavratzakis et al. This is an open access article distributed under the Creative Com- mons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, pro- vided the original work is properly cited. Previous investigations using pictures to elicit an emotional response have shown that the startle reflex habituates over time due to decreased excitation in the obligatory startle processing pathway, an effect that is independent of emotion modulation aspects of the startle response. However, in some instances, startle magnitude has been selectively potentiated during sustained exposure to passively viewed un- pleasant pictures. This study assessed startle modulation during brief, alternating and sustained exposure to emotional pictures. Self-reported ratings of emotion were collected online with picture viewing to de- termine if any change in startle magnitude was observable in explicit emotional responses. Self-reported ratings of pleasantness and arousal were no different across the brief and sustained picture presentations. However, a significant main effect (independent from emotion category) of presentation condition was found for startle magnitudes, showing that, contrary to previous research involving passive picture view- ing, mean startle magnitudes during sustained exposure were reduced relative to brief exposure. These findings are likely the result of a general habituation of the startle reflex in the obligatory pathway. The findings are also discussed in terms of the effect of the concurrent emotion rating task, which may have differently affected the cumulative effects of emotion exposure compared to passive picture viewing. Keywords: Emotion; Explicit; Implicit; Brief Exposure; Sustained Exposure Introduction The startle reflex is an indicator of the brains aversive moti- vational system via its structural connection to motivation sys- tems at the synaptic level (Yeomans & Frankland, 1995). It is a sudden involuntary movement involving the whole body (mus- cle contractions), and is thought to serve as a mechanism for quickly withdrawing the whole organism from harm. The reflex is activated in response to an intense unexpected stimulus with a rapid rising time and can be elicited via any of the senses (e.g. loud noise, flash of light, air puff). Reflexes are by definition automatic, however it is also known that activation patterns of reflexes vary as a function of psychological factors (Filion, Dawson, & Schell, 1998). Previous investigations of the neural circuitry influencing acoustic startle magnitude suggest input from two distinct cir- cuits. The primary obligatory circuit projects from the cochlear nucleus to the spinal cord and the secondary modulatory circuit, which includes the amygdala connects with the primary obliga- tory circuit causing its modulation (Yeomans & Frankland, 1995). The obligatory circuit is thought to be responsible for the early excitatory startle reactivity seen in initial startles, which rapidly habituates after about six trials (Blumenthal et al., 2005). Bradley and colleagues demonstrated habituation of the obligatory pathway in a study investigating startle responses to repeated presentations of the same unpleasant, pleasant and neutral pictures over time (Bradley, Lang, & Cuthbert, 1993). They showed that while the overall strength of the reflex gra- dually reduced, blink magnitude continued to discriminate be- tween negative and positive stimuli, which shows that habitua- tion of the startle reflex is more sensitive to factors related to the obligatory pathway than to psychological factors. In terms of understanding the temporal effects of a sustained emotion context on the startle response, a variety of stimulus ex- posure conditions have been used to manipulate emotional states. Some of these approaches involved increasing exposure time to a single picture (Codispoti, Bradley, & Lang, 2001; Sutton, Davidson, Donzella, Irwin, & Dottl, 1997), repetitive presenta- tions of identical pictures (Ferrari, Bradley, Codispoti, & Lang, 2010), rapid presentations of a series of pictures shown at vary- ing rates (Smith, Löw, Bradley, & Lang, 2006) as well as exa- mining persistence of emotion effects into the interstimulus in- terval (Bradley, Cuthbert, & Lang, 1996). While many of these studies implicate factors such as novelty and attention, limiting generalisability of the results to the specific effects of emotio- nal valence, a prominent finding nonetheless has been that con- sistently activating a negative emotional state heightens or main- tains defensive responses across time. Using a passive picture viewing paradigm, Smith, Bradley *Corresponding author.  A. MAVRATZAKIS ET AL. and Lang (2005) attempted to separate potential effects of emo- tion (the psychological pathway) from general habituation ef- fects (the obligatory pathway) by analysing startle magnitudes occurring early and late within three successive blocks of pic- tures, and then comparing startles during the first block to star- tles during the last block. Within blocks, they found potentia- tion of startles occurring later compared to startles occurring ear- lier for unpleasant pictures, but not for pleasant or neutral pic- tures, indicating selective potentiation of aversive motivatio- nal processes during sustained emotion exposure. When startle responses were compared across rather than within the three blocks however, there was no change in magnitude for unplea- sant or neutral pictures, but there was a decrease in startle mag- nitude for pleasant pictures, indicating selective attenuation of appetitive motivational processes after a longer induction of emo- tional states. They further suggest that this was not due to habi- tuation of the obligatory pathway since the rate of reduction of the startle magnitude was not consistent across valence conditions. In the current experiment, rather than assessing the cumula- tive effects of sustained emotion exposure on the startle re- sponse early and late in a block of emotional pictures, the aim was to investigate how startle responses differ during brief, al- ternating exposure to emotional pictures compared to during sus- tained exposure. In addition, this study aimed to compare startle responses with online emotional response rating for each pic- ture to see whether any effect of sustained exposure to emotio- nal pictures could be observed in changes in explicit selfreports of emotional responses. This is of special interest, because pre- vious studies demonstrated that some aspects of affective infor- mation processing modify startle reflex amplitudes while leav- ing explicit rating unchanged (Walla et al., 2010; Geiser et al., 2011; Grahl et al., 2012; Walla et al., 2013). This phenomenon gives reason to believe that self reported emotion reflects only a fraction of ongoing raw affective processing (see Walla & Pank- sepp, 2013), an idea that deserves more attention than it has pre- viously got. In this experiment, two modified picture viewing paradigms were used—one in which consistently-valanced stimuli were presented over an extended amount of time and one in which the valence of the presented stimuli was randomised and there- fore continually alternating. The design, therefore, involved six response conditions and was analysed using a 2 (Presentation order: Block, Mix) by 3 (Picture valence: Unpleasant, Neutral, Pleasant) within-subjects design. To do this, 90 novel pictures from the IAPS were used, 45 in each paradigm and 15 per va- lence condition (unpleasant, pleasant and neutral). Pictures from each valence category were chosen so that all categories were matched on intensity of valence and arousal based on pre-eva- luations of the IAPS picture collection. Pictures high in arousal were chosen to accentuate valence effects in the startle response (Cuthbert, Bradley, & Lang, 1996). Self-reported arousal rat- ings were collected to verify these conditions as well as autono- mic physiological arousal indices by recording skin conduc- tance changes. Self-reported pleasantness and arousal ratings were assessed using the Self-Assessment Manikin (SAM), a pic- torial rating system accompanying the IAPS collection, which has demonstrated reliability for predicting emotional responses to these picture stimuli, and is widely used in startle research. In addition, mood (The Affect Grid) was assessed prior to the ex- periment in order to reference ratings to a baseline affective state. Consistent with previous findings, we expected that picture presentations in the mixed and blocked viewing paradigms would result in emotion-dependent modulation of the startle re- sponse magnitude, specifically that startle responses would be largest when viewing unpleasant pictures compared to neutral and smallest when viewing pleasant pictures compared to neu- tral. Consistent with findings reported by Smith, Bradley and Lang (2005), we expected startle responses to be largest during blocked unpleasant pictures, reflecting a potentiation effect dur- ing sustained exposure to unpleasant stimuli. Since current the- ory holds that the emotion-modulated startle response is an on- line indicator of an internal emotional state, it was hypothesis- ed that self-reported pleasantness ratings would also reflect the direction of startle magnitude, specifically that pleasantness ra- tings would be lowest for blocked unpleasant pictures compar- ed to be mixed. Method Ethics Statement The ethics committee of the University of Newcastle appro- ved this study (H-2010-1061). Participants The present study is based on 17 consenting participants. They were all undergraduate students of the University of New- castle (16 females). Mean age was 21.53 years (SD = 3.48, range 18 - 29 years). Participants were right-handed in accordance with Oldfield’s criteria (Oldfield, 1971), non-smokers with nor- mal hearing and normal or corrected vision and had no history of neuropathology. Assessments and Measures Current Mood Ongoing mood can influence decision making behaviour and alter startle baseline reactivity (Grüsser, Wölfling, Mörsen, Ka- thmann, & Flor, 2007). Therefore, immediately following the consenting procedure participant’s mood was assessed using the Affect Grid (Russell, Weiss, & Mendelsohn, 1989). The Af- fect Grid is a single item two dimensional bipolar assessment of pleasure-displeasure and arousal-calm. Users are instructed to place a cross within the two dimensional grid, which is then converted to two individual scores ranging from one to nine for each axis. Self-Reported Valence and Arousal Ratings A computerised version of the Self-Assessment Manikin (SAM); (Bradley & Lang, 1994) was used to assess affect along valence and arousal dimensions using two nine-point likert scales (1 = negative valence or low arousal, respectively; 9 = positive valence or high arousal, respectively). Each SAM scale includes a non-verbal pictorial assessment of a human-like stimulus with visible emotional expressions. Using five figures for each scale, SAM permits the assessment of core valence and arousal di- mensions of emotional experience. This is a standard assess- ment technique for evaluating affective responses for the IAPS collection and is widely used in startle research. Physiological Measures NeXus-10 (produced by Mind Media BV) wireless recording and Bio-trace + software was used to measure and process both Copyright © 2013 SciRes. 390  A. MAVRATZAKIS ET AL. skin conductance and eye blink responses. Skin conductance was recorded at a rate of 32 samples/s with a Nexus-10-SC/ GSR sensor (two finger sensor) connected to the Nexus-10 re- cording system with a 24 bit resolution which is able to reg- ister changes of less than .0001 microsiemens. We attached one sensor to the middle finger and the other sensor to the ring fin- ger of the left hand. Eye blinks were elicited by a repeatedly presented startle stimulus (see below for details). With bipolar electromyography (EMG) carried out with the Nexus-10 muscle potential changes of the musculus orbicularis oculi (which elicit eye blinks) of the left eye of every study participant were measured and stored on the hard drive of a lap top computer. We used a dual channel electrode cable with carbon coating and active shielding tech- nology for low noise and an additional ground electrode cable. EMG sampling rate was 2048 per s. A band pass filter from 20 Hz to 500 Hz was applied during online recording. Raw EMG data were then recalculated by using the root mean square (RMS) method to transform EMG signals into amplitudes. The resulting amplitudes were then subject to statistical analysis. Stimuli Visual Stimuli The emotion eliciting stimuli were 90 pictures (30 neutral, 30 unpleasant and 30 pleasant) from the International Affective Picture System collection (IAPS); (Lang, Bradley, & Cuthbert, 2005). Pictures were selected based on pre-evaluated ratings of picture pleasantness and arousal. Pictures rated high for valence (unpleasant and pleasant) and arousal (unpleasant, neutral and pleasant) dimensions were selected. An Analysis of Variance (ANOVA) and t-tests were used to confirm respective criteria matched across picture categories. The means and standard de- viations of the pictures for this experiment are displayed in Ta- ble 1. The pictures were presented in colour against a black background on a computer monitor screen. Acoustic Startle Stimuli Our startle stimulus was a 50 ms burst of acoustic white noise at 105 dB sound pressure level delivered through profes- sional headphones fully covering both ears. Sound pressure le- vel was measured with a mobile measuring device (produced by Voltcraft). To achieve the respective loudness a commercial headphone pre-amplifier was used (Behringer; MicroAMP HA400). Design Pictures were randomly allocated to the mixed or blocked Table 1. Mean values of visual picture stimuli based on pre-evaluations using the Self Assessment Manikin (SAM) rating system. Rating Pleasantness Arousal M SD M SD Unpleasant 2.65 .45 6.58 .36 Neutral 4.52 .23 5.41 .62 Pleasant 7.28 .46 6.60 .38 presentation session using a random number generator. Pictures in the mixed presentation session were further quasi-randomly arranged to ensure constant variation in picture valence. To minimise the potential for order effects, five uniquely random- ised lists of pictures were used interchangeably throughout the recruiting period. Hence, each new list contained a different combination of pictures in each viewing session, and all six ex- perimental conditions were fully counterbalanced. Four of the 15 pictures in each valence category were simultaneously asso- ciated with a startle-eliciting stimulus at the fifth second of view- ing. Pictures associated with the startle-eliciting stimulus were also varied. The interval between two startle-eliciting stimuli was randomised to occur at approximately 35, 57 or 79 seconds to prevent learning effects. For both sessions, each picture trial lasted approximately 22 seconds and included a one second cross fixation screen, a six second presentation of the lead pic- ture stimulus, a user-paced SAM pleasantness rating screen and a user-paced SAM arousal rating screen. A black screen of one second duration was used to transition between rating and pic- ture screens. Procedure Experimental testing was conducted within a two month re- cruiting period at the University of Newcastle Callaghan Cam- pus Functional Neuro-imaging laboratory in individual two hour sessions at regulated temperature (20.5˚C). Consenting parti- cipants completed assessments for demographics and pre-ex- perimental affect respectively before being seated in front of a computer monitor and connected to the physiological recording equipment by the experimenter. Participants were instructed to pay close attention to their feelings throughout the session and to quickly rate their immediate reaction to each picture for all trials by using a computer mouse to click on the SAM rating. Participants were also told to ignore any loud bursts of noise through the headphones. Prior to commencing the sessions par- ticipants practised using the rating system for three trials in- cluding one item of extreme valence from each category (un- pleasant and pleasant extremes and one neutral) which served as an anchor for subsequent picture rating. The mixed and blo- cked sessions were separated by a two minute break interval. Three pilot trials were conducted prior to official data collec- tion. Data Reduction Subjective reports of emotion on trials associated with the acoustic startle may be biased by the inherently startling nature of the probe. Therefore SAM ratings for each valence category were taken from four startle-unassociated trials. Similarly, other physiological measures can be augmented by the startle probe. To avoid this unwanted source of variation data for skin con- ductance were taken exclusively from the six second picture presentation interval on startle-associated trials for each valence category. Extracted values were then averaged to form mean physiological values for each of the six valence-viewing condi- tions. A trigger sound detector was used to identify onset of the acoustic probe in the data read out. This served as a reference for detecting the true onset of startle reactivity. The startle blink value was defined as the peak rise in EMG waveform on startle associated trials. Trials for which peak rise in EMG waveform was less than 5 μV were scored as missing. To decrease the Copyright © 2013 SciRes. 391 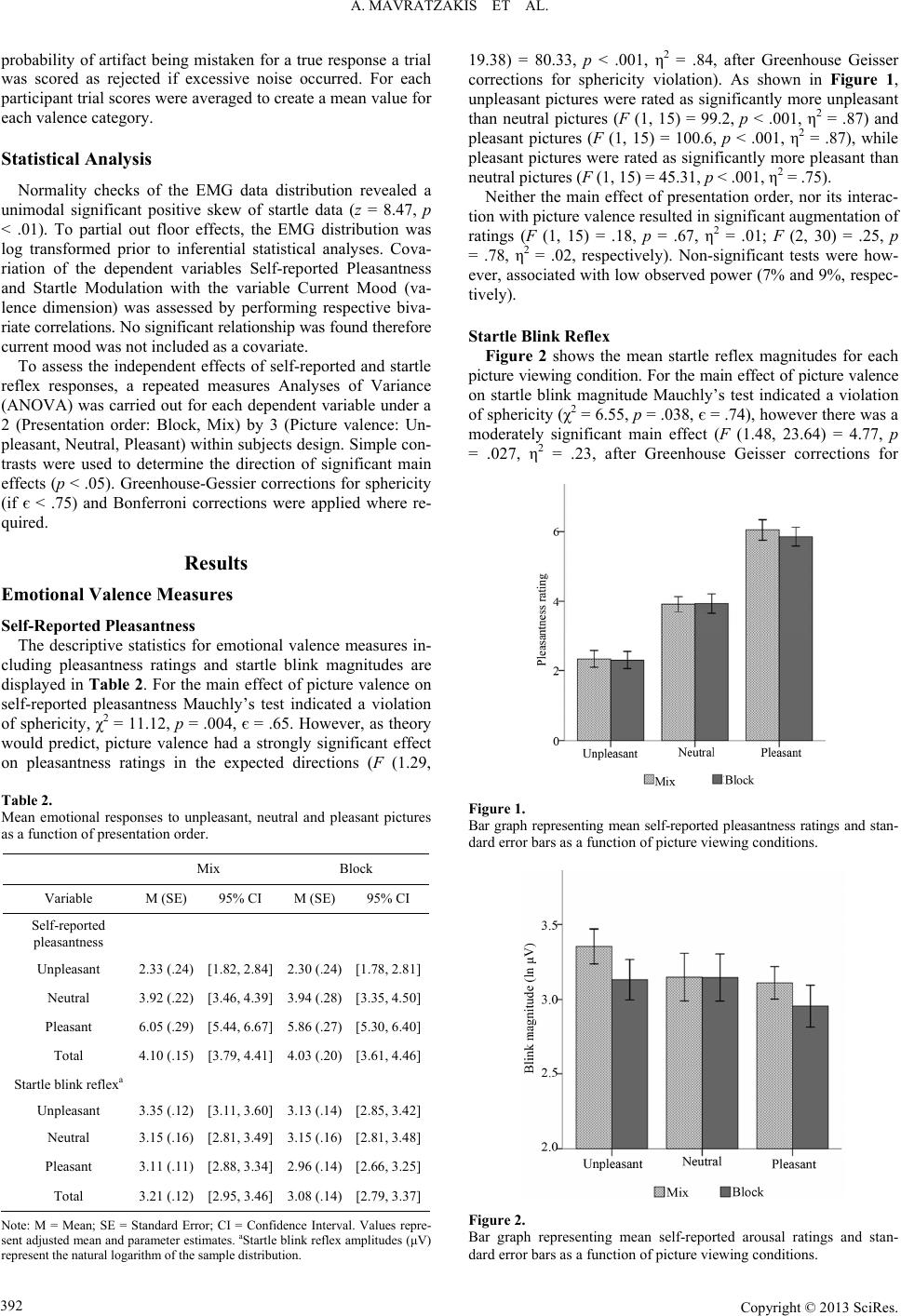 A. MAVRATZAKIS ET AL. probability of artifact being mistaken for a true response a trial was scored as rejected if excessive noise occurred. For each participant trial scores were averaged to create a mean value for each valence category. Statistical Analysis Normality checks of the EMG data distribution revealed a unimodal significant positive skew of startle data (z = 8.47, p < .01). To partial out floor effects, the EMG distribution was log transformed prior to inferential statistical analyses. Cova- riation of the dependent variables Self-reported Pleasantness and Startle Modulation with the variable Current Mood (va- lence dimension) was assessed by performing respective biva- riate correlations. No significant relationship was found therefore current mood was not included as a covariate. To assess the independent effects of self-reported and startle reflex responses, a repeated measures Analyses of Variance (ANOVA) was carried out for each dependent variable under a 2 (Presentation order: Block, Mix) by 3 (Picture valence: Un- pleasant, Neutral, Pleasant) within subjects design. Simple con- trasts were used to determine the direction of significant main effects (p < .05). Greenhouse-Gessier corrections for sphericity (if є < .75) and Bonferroni corrections were applied where re- quired. Results Emotional Valence Measures Self-Reported Pleasantness The descriptive statistics for emotional valence measures in- cluding pleasantness ratings and startle blink magnitudes are displayed in Table 2. For the main effect of picture valence on self-reported pleasantness Mauchly’s test indicated a violation of sphericity, χ2 = 11.12, p = .004, є = .65. However, as theory would predict, picture valence had a strongly significant effect on pleasantness ratings in the expected directions (F (1.29, Table 2. Mean emotional responses to unpleasant, neutral and pleasant pictures as a function of presentation order. Mix Block Variable M (SE) 95% CI M (SE) 95% CI Self-reported pleasantness Unpleasant 2.33 (.24) [1.82, 2.84] 2.30 (.24) [1.78, 2.81] Neutral 3.92 (.22) [3.46, 4.39] 3.94 (.28) [3.35, 4.50] Pleasant 6.05 (.29) [5.44, 6.67] 5.86 (.27) [5.30, 6.40] Total 4.10 (.15) [3.79, 4.41] 4.03 (.20) [3.61, 4.46] Startle blink reflexa Unpleasant 3.35 (.12) [3.11, 3.60] 3.13 (.14) [2.85, 3.42] Neutral 3.15 (.16) [2.81, 3.49] 3.15 (.16) [2.81, 3.48] Pleasant 3.11 (.11) [2.88, 3.34] 2.96 (.14) [2.66, 3.25] Total 3.21 (.12) [2.95, 3.46] 3.08 (.14) [2.79, 3.37] Note: M = Mean; SE = Standard Error; CI = Confidence Interval. Values repre- sent adjusted mean and parameter estimates. aStartle blink reflex amplitudes (μV) represent the natural logarithm of the sample distribution. 19.38) = 80.33, p < .001, η2 = .84, after Greenhouse Geisser corrections for sphericity violation). As shown in Figure 1, unpleasant pictures were rated as significantly more unpleasant than neutral pictures (F (1, 15) = 99.2, p < .001, η2 = .87) and pleasant pictures (F (1, 15) = 100.6, p < .001, η2 = .87), while pleasant pictures were rated as significantly more pleasant than neutral pictures (F (1, 15) = 45.31, p < .001, η2 = .75). Neither the main effect of presentation order, nor its interac- tion with picture valence resulted in significant augmentation of ratings (F (1, 15) = .18, p = .67, η2 = .01; F (2, 30) = .25, p = .78, η2 = .02, respectively). Non-significant tests were how- ever, associated with low observed power (7% and 9%, respec- tively). Startle Blink Reflex Figure 2 shows the mean startle reflex magnitudes for each picture viewing condition. For the main effect of picture valence on startle blink magnitude Mauchly’s test indicated a violation of sphericity (χ2 = 6.55, p = .038, є = .74), however there was a moderately significant main effect (F (1.48, 23.64) = 4.77, p = .027, η2 = .23, after Greenhouse Geisser corrections for Figure 1. Bar graph representing mean self-reported pleasantness ratings and stan- dard error bars as a function of picture viewing conditions. Figure 2. Bar graph representing mean self-reported arousal ratings and stan- dard error bars as a function of picture viewing conditions. Copyright © 2013 SciRes. 392 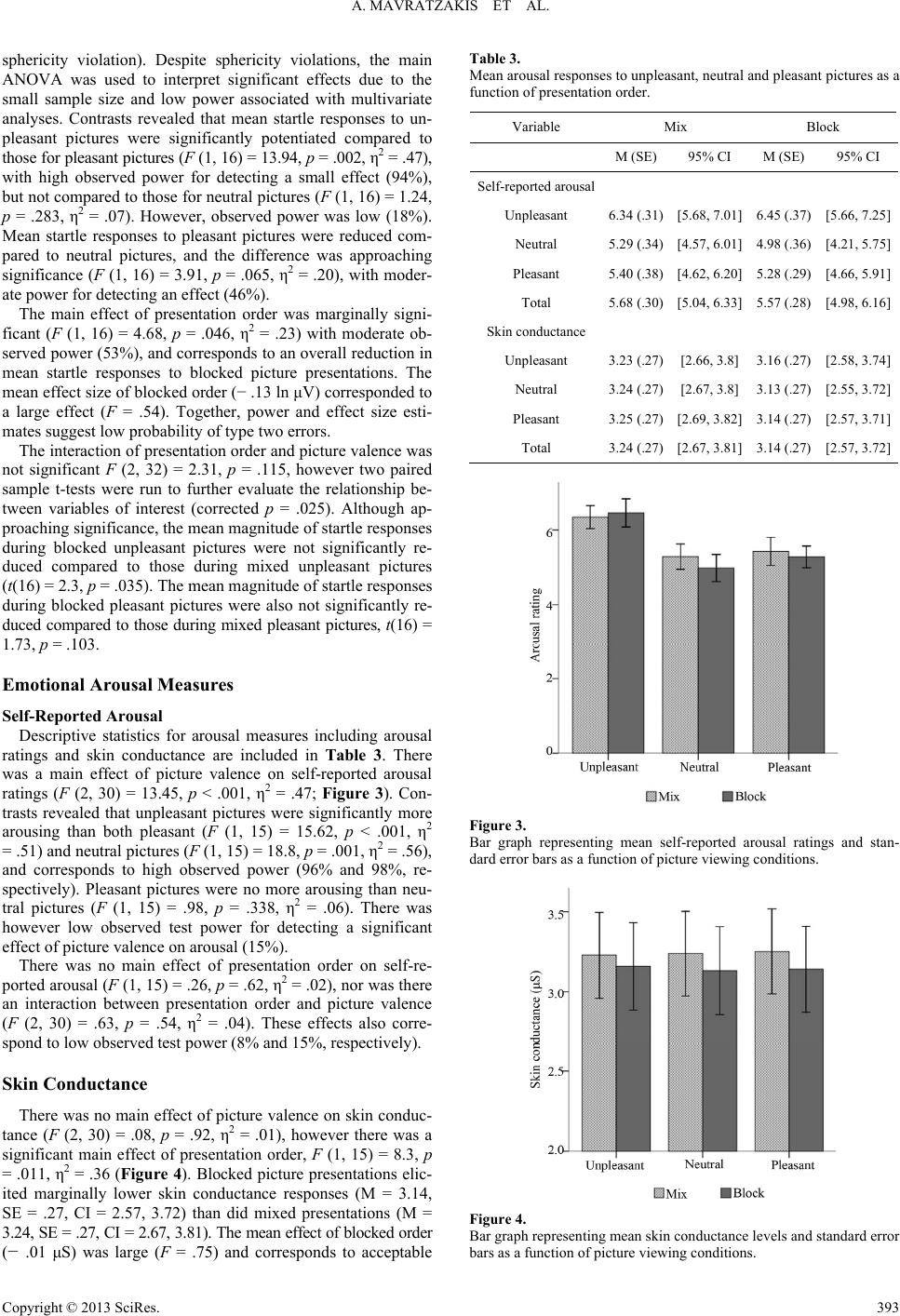 A. MAVRATZAKIS ET AL. sphericity violation). Despite sphericity violations, the main ANOVA was used to interpret significant effects due to the small sample size and low power associated with multivariate analyses. Contrasts revealed that mean startle responses to un- pleasant pictures were significantly potentiated compared to those for pleasant pictures (F (1, 16) = 13.94, p = .002, η2 = .47), with high observed power for detecting a small effect (94%), but not compared to those for neutral pictures (F (1, 16) = 1.24, p = .283, η2 = .07). However, observed power was low (18%). Mean startle responses to pleasant pictures were reduced com- pared to neutral pictures, and the difference was approaching significance (F (1, 16) = 3.91, p = .065, η2 = .20), with moder- ate power for detecting an effect (46%). The main effect of presentation order was marginally signi- ficant (F (1, 16) = 4.68, p = .046, η2 = .23) with moderate ob- served power (53%), and corresponds to an overall reduction in mean startle responses to blocked picture presentations. The mean effect size of blocked order (− .13 ln μV) corresponded to a large effect (F = .54). Together, power and effect size esti- mates suggest low probability of type two errors. The interaction of presentation order and picture valence was not significant F (2, 32) = 2.31, p = .115, however two paired sample t-tests were run to further evaluate the relationship be- tween variables of interest (corrected p = .025). Although ap- proaching significance, the mean magnitude of startle responses during blocked unpleasant pictures were not significantly re- duced compared to those during mixed unpleasant pictures (t(16) = 2.3, p = .035). The mean magnitude of startle responses during blocked pleasant pictures were also not significantly re- duced compared to those during mixed pleasant pictures, t(16) = 1.73, p = .103. Emotional Arousal Measures Self-Reported Arousal Descriptive statistics for arousal measures including arousal ratings and skin conductance are included in Table 3. There was a main effect of picture valence on self-reported arousal ratings (F (2, 30) = 13.45, p < .001, η2 = .47; Figure 3). Con- trasts revealed that unpleasant pictures were significantly more arousing than both pleasant (F (1, 15) = 15.62, p < .001, η2 = .51) and neutral pictures (F (1, 15) = 18.8, p = .001, η2 = .56), and corresponds to high observed power (96% and 98%, re- spectively). Pleasant pictures were no more arousing than neu- tral pictures (F (1, 15) = .98, p = .338, η2 = .06). There was however low observed test power for detecting a significant effect of picture valence on arousal (15%). There was no main effect of presentation order on self-re- ported arousal (F (1, 15) = .26, p = .62, η2 = .02), nor was there an interaction between presentation order and picture valence (F (2, 30) = .63, p = .54, η2 = .04). These effects also corre- spond to low observed test power (8% and 15%, respectively). Skin Conductance There was no main effect of picture valence on skin conduc- tance (F (2, 30) = .08, p = .92, η2 = .01), however there was a significant main effect of presentation order, F (1, 15) = 8.3, p = .011, η2 = .36 (Figure 4). Blocked picture presentations elic- ited marginally lower skin conductance responses (M = 3.14, SE = .27, CI = 2.57, 3.72) than did mixed presentations (M = 3.24, SE = .27, CI = 2.67, 3.81). The mean effect of blocked order (− .01 μS) was large (F = .75) and corresponds to acceptable Table 3. Mean arousal responses to unpleasant, neutral and pleasant pictures as a function of presentation order. Variable Mix Block M (SE)95% CI M (SE) 95% CI Self-reported arousal Unpleasant 6.34 (.31)[5.68, 7.01] 6.45 (.37) [5.66, 7.25] Neutral 5.29 (.34)[4.57, 6.01] 4.98 (.36) [4.21, 5.75] Pleasant 5.40 (.38)[4.62, 6.20] 5.28 (.29) [4.66, 5.91] Total 5.68 (.30)[5.04, 6.33] 5.57 (.28) [4.98, 6.16] Skin conductance Unpleasant 3.23 (.27)[2.66, 3.8] 3.16 (.27) [2.58, 3.74] Neutral 3.24 (.27)[2.67, 3.8] 3.13 (.27) [2.55, 3.72] Pleasant 3.25 (.27)[2.69, 3.82] 3.14 (.27) [2.57, 3.71] Total 3.24 (.27)[2.67, 3.81] 3.14 (.27) [2.57, 3.72] Figure 3. Bar graph representing mean self-reported arousal ratings and stan- dard error bars as a function of picture viewing conditions. Figure 4. Bar graph representing mean skin conductance levels and standard error bars as a function of picture viewing conditions. Copyright © 2013 SciRes. 393  A. MAVRATZAKIS ET AL. observed power (77%). There was no interaction between pre- sentation order and picture valence (F (2, 30) = .67, p = .52, η2 = .04). Three paired sample t tests (corrected p = .017) were carried out on variables of interest. The t-tests revealed that mean skin conductance levels were significantly reduced during blocked compared to mixed neutral pictures (t(15) = 2.83, p = .013), but not during blocked compared to mixed pleasant (t(15) = 2.45, p = .027) or unpleasant (t(15) = 1.88, p = .08) pictures. Discussion The question addressed in the current study was whether emo- tional responses are differently affected by sustained exposure to emotional stimuli compared to brief exposure, as measured by self-reported emotional responses and the affective startle- blink reflex. Previous research using a passive picture viewing paradigm has demonstrated that cumulative exposure can po- tentiate startles during negative conditions (Smith et al., 2005). Therefore, it was hypothesised that sustained exposure to un- pleasant emotional pictures would potentiate startle responses. It was further hypothesised that self-reported emotional responses would reflect these changes in startle responses. The results do not support either hypothesis, and instead, suggest that sustain- ed exposure to emotional pictures caused a general attenuation of startle magnitudes across all valence conditions. In addition, this effect was not observed in selfreported emotional responses, which instead showed no difference in ratings across brief and sustained emotion exposure conditions. This study assessed the effects of sustained exposure to emo- tional stimuli while also recording immediate self-reported emo- tional reactions to the presented stimuli. This procedure differs to the procedure used by Smith, Bradley and Lang (2005) where participants were required to passively view emotional pictures without any additional rating task. Recently, Wangelin, Low, McTeague, Bradley, and Lang (2011) investigated the effects of a cognitive distractor task on startle responses during sustained aversive exposure. They found that embedded distractor tasks did eliminate potentiation of startles during sustained exposure to unpleasant pictures compared to when pictures were viewed passively. This suggests that sustained effects of emotion on the startle reflex are quite sensitive to cognitive and perceptual dis- tractions. This is also strengthened by additional data from an earlier experiment (Bradley et al., 1996) which showed that star- tle responses were maintained over time when a passive picture viewing task was combined with a concurrent auditory-based emotion word recognition task (which was used to ensure par- ticipants paid attention to the acoustic startle stimuli). Therefore a possible explanation for why startles were not potentiated as expected during unpleasant picture viewing was that the rating task itself inadvertently acted as a distraction, momentarily di- verting attention from the sustained emotion, eliminating or downgrading cumulative effects. However, to draw any conclu- sions in this area would require a deliberate comparison of star- tle responses to “actively” and “passively” viewed pictures in a single experiment. Alternatively, research examining perceptual differences re- lated to different emotional tasks has found differences in neu- ral activity extending to limbic regions as a function of task ins- tructions such rating an emotional reaction compared to recog- nising a specific emotion (e.g. Liberzon et al., 2000). Therefore, use of a rating task in the current study may have differently affected perceptual processing and subsequent motivational system activation compared to other studies of sustained emo- tion that used a recognition task or a simple passive viewing task. Said differently, it is possible that actively paying atten- tion to ones own emotions rather than simply observing emo- tional events can have a regulatory influence, impeding the for- mation of a stronger negative emotional state. Walla et al. (2013a) found that self referenced negative emotion images were rated more negative that un-referenced negative images and self-re- ferenced positive images were rated more positive than un-re- ferenced positive images. In their study, startle responses were enhanced as a result of self reference regardless of emotion va- lence. This finding was interpreted as being reflective of moti- vation as indicated by startle reflex modulation data. Thus, the notion arises that in the current study too motivation is reflected in the startle reflex modulation data and motivation is reduced over time if emotion valence is kept constant over longer peri- ods of time. A second important finding was that contrary to what would be predicted by past empirical investigations, startle responses elicited across all unpleasant pictures were not significantly po- tentiated relative to neutral, nor were startle responses during pleasant pictures significantly reduced relative to neutral. This raises concern as to whether the lead stimuli used in this ex- periment were effective for inducing the desired emotional states. One possible explanation is that the trend towards atte- nuated startle responses may have washed out the potentiation effects for unpleasant pictures. However, this does not explain the null effect produced by pleasant pictures, which may alter- natively be the result of lower than expected arousal levels associated with pleasant pictures, which is apparent in the low self-reports of arousal for pleasant pictures. It should be noted that most of our participants were female and most of the pleasant pictures from the IAPS involve erotica, which are gen- erally rated as more pleasant by males than females. Thus, these unexpected findings were most likely caused by a gender bias in the sample. However, it should also be noted that the IAPS collection has to be treated with caution, because some of the images that are pre-evaluated as neutral may elicit affective valence, perhaps depending on cultural background. After all, startle reflex modulation has been repeatedly shown to be sen- sitive to raw affective processing in a variety of experiments us- ing many different lead stimuli. Urban neighbourhoods (Geiser et al., 2011), food intake (Walla et al., 2010), bottle shape (Grahl et al., 2012) and brand attitude (Walla et al., 2011) were shown to significantly modulate startle responses. A third and incidental finding was differences between self- reported arousal ratings and skin conductance levels. While there was no effect of mixed and blocked order on self-reported arou- sal ratings, skin conductance was reduced during blocked pre- sentations, with post hoc tests revealing that the effect was only significant for neutral pictures. Habituation of skin conductance levels over time has previously been documented by Bradley, Lang, & Cuthbert (1993), who also documented the relationship between arousal and the startle response (Cuthbert et al., 1996), showing that changes in arousal moderate the affective startle response such that a high level of arousal is necessary for va- lence-modulated differences to take effect. Therefore, it is like- ly that despite efforts to control for arousal across valence and order conditions, reduced startle response magnitudes seen in the current study were simply the result of habituated arousal levels. It should still be noted that several functional MRI studies have documented habituation of amygdala responses during re- Copyright © 2013 SciRes. 394  A. MAVRATZAKIS ET AL. Copyright © 2013 SciRes. 395 peated exposure to negative emotional stimuli such as pictures of fearful faces (Wright et al., 2001) and other unpleasant pic- tures (Liberzon et al., 2000). Zald (2003) reviewed studies of amygdala activation patterns during emotional evaluations of visual stimuli and concluded that habituation “may reflect a pro- cess through which stimuli are rapidly re-evaluated based on the lack of consequences arising from their previous exposure.” (Zald, 2003: p. 104). Given that the amygdala is a region im- plicated in the startle neural circuitry (Yeomans & Frankland, 1995), it is possible that reduced startle responses may be re- flecting activation related to amygdala circuitry. Conclusion Several important implications for emotion research have arisen from the current experiment. Firstly, neural pathways in- volved in the affective modulation of the startle response may be more sensitive to emotion exposure than can be observed via self-reported emotional responses. Hence, although the startle reflex is able to predict general directional differences in emo- tional states, a closer investigation suggests it is, at least in part, measuring emotion processing activity that is somewhat differ- ent to and inaccessible by self-reports. Second, this study pro- vides conflicting results compared to other studies investigating the effect of sustained exposure to emotional pictures, and sug- gests that certain conditions of picture viewing, specifically, ac- tive emotional rating compared to passive viewing, may diffe- rently affect the way emotional stimuli are processed. This find- ing may be relevant for research directed at understanding or developing cognitive strategies for optimised and adaptive emo- tional perception and comprehension, and may also be an im- portant factor to consider when deciding how to best design emotion experiments. Finally, the findings of this study may have useful implications for clinical populations (see Arthur- Kelly et al., 2013) as well as for advancing consumer neurosci- ence (Koller & Walla, 2012; Walla et al., 2013b). REFERENCES Arthur-Kelly, M., Lyons, G. S., & Walla, P. (2013). Toward improved ways of knowing children with Profound Multiple Disabilities (PMD): Introducing startle reflex modulation. Developmental Neurohabilita- tion, in press. Bradley, M., Cuthbert, B., & Lang, P. (1996). Picture media and emo- tion: Effects of a sustained affective context. Psychophysiology, 33, 662-670. doi:10.1111/j.1469-8986.1996.tb02362.x Bradley, M., & Lang, P. (1994). Measuring emotion: The self-asses- sment manikin and the semantic differential. Journal of Behavior Therapy and Experimental Ps yc h i at r y , 25, 49-59. doi:10.1016/0005-7916(94)90063-9 Bradley, M., Lang, P., & Cuthbert, B. (1993). Emotion, novelty, and the startle reflex: Habituation in humans. Behavioral Neuroscience, 107, 970-980. doi:10.1037/0735-7044.107.6.970 Codispoti, M., Bradley, M. M., & Lang, P. J. (2001). Affective reac- tions to briefly presented pictures. Psychophysiology, 38, 474-478. doi:10.1111/1469-8986.3830474 Cuthbert, B., Bradley, M., & Lang, P. (1996). Probing picture percep- tion: Activation and emotion. Psychophysiology, 33, 103-111. doi:10.1111/j.1469-8986.1996.tb02114.x Ferrari, V., Bradley, M., Codispoti, M., & Lang, P. (2010). Repetitive exposure: Brain and reflex measures of emotion and attention. Psy- chophysiology, 47, 1-8. Filion, D., Dawson, M., & Schell, A. (1998). The psychological sig- nificance of human startle eyeblink modification: A review. Biolo- gical Psychology, 47, 1-43. doi:10.1016/S0301-0511(97)00020-3 Geiser, M., & Walla, P. (2011). Objective measures of emotion during virtual walks through urban environments. Applied Sc i en c e s, 1, 1-11. Grahl, A., Greiner, U., & Walla, P. (2012). Bottle-shape elicits gender- specific emotion: A startle reflex modulation study. Psychology, 3, 548-554. doi:10.4236/psych.2012.37081 Grüsser, S., Wölfling, K., Mörsen, C., Kathmann, N., & Flor, H. (2007). The influence of current mood on affective startle modulation. Expe- rimental Brain Research, 177, 122-128. doi:10.1007/s00221-006-0653-x Koller, M., & Walla, P. (2012). Measuring affective information proc- essing in information systems and consumer research-introducing star- tle reflex modulation. ICIS Proceedings, 33rd International Confer- ence on Information Systems, Orlando, FL. http://aisel.aisnet.org/icis2012/proceedings/BreakthroughIdeas/1/ Lang, P., Bradley, M., & Cuthbert, B. (2005). International affective pic- ture system (IAPS): Instruction manual and affective ratings. Gaines- ville, FL: The Center for Research in Psychophysiology, University of Florida. Liberzon, I., Taylor, S. F., Fig, L. M., Decker, L. R., Koeppe, R. A., & Minoshima, S. (2000). Limbic Activation and Psychophysiologic Re- sponses to Aversive Visual Stimuli: Interaction with Cognitive Task. Neuropsychopharmacology, 23, 508-516. doi:10.1016/S0893-133X(00)00157-3 Oldfield, R. (1971). The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia, 9, 97-113. doi:10.1016/0028-3932(71)90067-4 Russell, J., Weiss, A., & Mendelsohn, G. (1989). Affect grid: A single- item scale of pleasure and arousal. Journal of Personality and Social Psychology, 57, 493-502. doi:10.1037/0022-3514.57.3.493 Smith, J. C., Bradley, M., & Lang, P. (2005). State anxiety and affec- tive physiology: Effects of sustained exposure to affective pictures. Biological Psychology, 69, 247-260. doi:10.1016/j.biopsycho.2004.09.001 Smith, J. C., Löw, A., Bradley, M. M., & Lang, P. J. (2006). Rapid picture presentation and affective engagement. Emotion, 6, 208. doi:10.1037/1528-3542.6.2.208 Sutton, S., Davidson, R., Donzella, B., Irwin, W., & Dottl, D. (1997). Manipulating affective state using extended picture presentations. Psychophysiology, 34, 217. doi:10.1111/j.1469-8986.1997.tb02135.x Walla, P., Rosser, L., Scharfenberger, J., Duregger, C., & Bosshard, S. (2013a). Emotion ownership: Different effects on explicit ratings and implicit responses. Psychology, 4, 213-216. Walla, P., Richter, M., Färber, S., Leodolter, U., & Bauer, H. (2010). Food evoked changes in humans: Startle response modulation and Event-Related Potentials (ERPs). Journal of Psychophysiology, 24, 25-32. doi:10.1027/0269-8803/a000003 Walla, P., & Panksepp, J. (2013). Neuroimaging helps to clarify brain affective processing without necessarily clarifying emotions, novel frontiers of advanced neuroimaging. http://www.intechopen.com/books/novel-frontiers-of-advanced-neur oiming/neuroimaging-helps-to-clarify-brain-affective-processing-with- out-necessarily-clarifying-emotions doi:10.5772/51761 Walla, P., Mavratzakis, A., & Bosshard, S. (2013b). Neuroimaging for the affective brain sciences, and its role in advancing consumer neu- roscience, novel frontiers of advanced neuroimaging. http://www.intechopen.com/books/novel-frontiers-of-advanced-neur oimaing/neuroimaging-for-the-affective-brain-sciences-and-its-role- in-advancing-consumer-neuroscience doi:10.5772/51042 Wangelin, B. C., Low, A., McTeague, L. M., Bradley, M. M., & Lang, P. J. (2011). Aversive picture processing: Effects of a concurrent task on sustained defensive system engagement. Psychophysiology, 48, 112-116. doi:10.1111/j.1469-8986.2010.01041.x Wright, C., Fischer, H., Whalen, P., McInerney, S., Shin, L., & Rauch, S. (2001). Differential prefrontal cortex and amygdala habituation to repeatedly presented emotional stimuli. Neu ror epo rt , 12, 379. doi:10.1097/00001756-200102120-00039 Yeomans, J., & Frankland, P. (1995). The acoustic startle reflex: Neu- rons and connections. Brain Research Reviews, 21, 301-314. doi:10.1016/0165-0173(96)00004-5 Zald, D. (2003). The human amygdala and the emotional evaluation of sensory stimuli. Brain Research Reviews, 41, 88-123. doi:10.1016/S0165-0173(02)00248-5
|