Paper Menu >>
Journal Menu >>
 J. Biomedical Science and Engineering, 2010, 3, 977-985 JBiSE doi:10.4236/jbise.2010.310128 Published Online October 2010 (http://www.SciRP.org/journal/jbise/). Published Online October 20 10 in SciRes. http://www.scirp.org/journal/jbise Global pattern of pairwise relationship in genetic network Ao Yuan, Qingqi Yue, Victor Apprey, George E. Bonney National Human Genome Center, Howard University, Washington DC, USA. Email: yuanao@hotmail.com; ayuan@howard.edu Received 6 April 2010; revised 22 April 2010; accepted 10 May 2010. ABSTRACT In recent times genetic network analysis has been found to be useful in the study of gene-gene inter- actions, and the study of gene-gene correlations is a special analysis of the network. There are many methods for this goal. Most of the existing methods model the relationship between each gene and the set of genes under study. These methods work well in applications, but there are often issues such as non-uniqueness of solution and/or computational difficulties, and interpretation of results. Here we study this problem from a different point of view: given a measure of pair wise gene-gene relationship, we use the technique of pattern image restoration to infer the optimal network pair wise relationships. In this method, the solution always exists and is unique, and the results are easy to interpret in the global sense and are computationally simple. The regulatory relationships among the genes are in- ferred according to the principle that neighboring genes tend to share some common features. The network is updated iteratively until convergence, each iteration monotonously reduces entropy and variance of the network, so the limit network represents the clearest picture of the regulatory relationships among the genes provided by the data and recoverable by the model. The method is illus- trated with a simulated data and applied to real data sets. Keywords: Convergence, Gene-Gene relationship, Neigh- borhood, Pattern analysis, Relationship measure. 1. INTRODUCTION A gene regulatory network (also called a GRN or genetic regulatory network) is a collection of DNA segments in a cell which interact with each other (indirectly through their RNA and protein expression products) and with other substances in the cell, thereby governing the rates at which genes in the network are transcribed into mRNA. From methodology point of view, genetic net- works are models that, in a simplified way, describe some biological phenomenon from interactions between the genes. They provide a high-level view and disregard most details on how exactly one gene regulates the ac- tivity of another. The gene-gene pair wise relationships provide a special insight of the network and are of inter- est in the study. Our work is closely related to that of genetic network analysis, and we first give a brief review of the methods. Some methods are deterministic, such as differential (difference) equation models [1-3], which may not be easy to solve nor have unique solutions. Since the ge- netic network is a complex system, any artificial model can only explain part of its mechanism; the unexplained parts are random noises, so we prefer a stochastic model. Existing stochastic methods for this problem including the linear models [4,5] or generalized linear models [6,7], the Bayesian network [8,9] etc. All these methods have their pros and cons, but have the common disadvantage that the solution may not be unique and the results are not easy to interpret. Also, when the network size ex- ceeds that of the data, these methods break down. In genetic work the pair wise regulatory relationships among the genes are important. For such data, it is of interest to investigate the underlying patterns that may have biologic significance, in particular those arising from pair wises regulatory relationships among the genes. Here we study this problem from a different point of view. Given a measure of pair wise gene-gene rela- tionship, we compute the measures from the data, and use the technique of pattern recognition and image res- toration to infer the underlying network relationships. The pair wise regulatory relationships among the genes are inferred according to the principle that neighboring genes tend to share some common features, as neigh- boring genes tend to be co-regulated by some enhancers because of their close proximity [10]. In this method, the solution is unique and computationally simple, the re- sults are easy to interpret and the network can be of any size. In the following we describe our method, study its  978 A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE basic properties, and illustrate its application. This me- thod is used to reveal the true relationships of structured high dimensional data array [11-14]. 2. MATERIALS AND METHODS The gene expression data are generally time dependent, as in Iyer et al. [15]. Let () ij X t (1,..., ;1,imj ...,;1,... )nt k be the observed gene expression re- sponse for subject i, gene j at time t. Denote 1 ( )(( ),...,( ))' ii in x txtxt be the observations across all the genes for subject i, and we use () x t to denote a general sample of the () i x t’s. Often for this type of data, m and k are in the low tens, and n in the tens to thousands. The commonly used differential equation model for genetic network analysis is a set of first order homogeneous differential equations with constant coefficients, in the simple case, has the form () () dx tWx t dt , where () ij Ww is the n × n matrix of unknown regu- latory coefficients to be solved. This type of models and its more specific and complicated variations characterize well the dynamic of the network over time. The base solution of the above equation set is the matrix exponen- tial 1 0 :/!:(( ),...,( ))' tWr r n r etWrvtvt , and the general solution of it has the form 1 () () n ijj j x tcvt x_{i} (t), (i = 1,...,m), where the j c’s are constants to be deter- mined by initial conditions from the data. So there are in total n² + n = n(n + 1) coefficients, n2 of them from W and n from the j c’s, to be determined from a total of mnk data points. When mnk < n(n + 1) these coefficients can not be determined; when mnk ≥ n(n + 1) they may be uniquely or non-uniquely determined, or may still be not determined. For differential (difference) equation models more complicated than this, solutions are more difficult to get. The commonly used stochastic model is the multi- variate linear model (1)() , iii xt Wxt () 0, i E (i = 1, …, m; t = 1, …, k-1) where 1 (,...,)' iin is the random deviations unex- plained by the model. Denote () ( ()) ij X txt, if X′(t)X(t) is non-singular, the least-squares solution of the above model is W = X’(t + 1) X(t) (X’(t) X(t))-¹, and it may have multiple solutions for different t. For '( )( ) rr X tXt to be non-singular, one must have n ≤ m. Even for n < m, '( )() rr X tXt may not necessarily be non-singular. This puts an immediate restriction on the size of the network to be analyzed. Also, the solution of the above model may not be unique due to different time points. For these reasons, we study the problem from a dif- ferent point of view; by analyze the pair wise gene-gene relationships in the network. In the following we de- scribe our model in which there is always an unique so- lution, the result is easy to interpret, and there is no re- striction on the size of the network. Since the pattern in the genetic network is based on the principle of neigh- boring similarity, the order of the genes matters in the study, and generally we assume the genes are arranged in their chromosome order. First we need a measurement for the relationship be- tween any pair of genes, and the network can be repre- sented by the matrix of the pair wise relationships. For large network, linear relationship is not adequate to use, as most of the coefficients will be very small. Also, as mentioned above, such model in this case has no solu- tion because of the small sample size. Pearson’s correla- tion is a good choice for this purpose, other choices in- cluding Kendal’s tau and Spearman’s rho, etc. Here we illustrate the method with Pearson’s correlation, and our goal is to infer the triangular correlation matrix 1 () ijijn Rr from the observed data, where ij r is the Pearson’s correlation coefficient between genes i and j. As usually the number m of individuals is small (some- times as few as 2), estimate the correlations using the data at each time point alone is inadequate. So we use all the data to estimate them. An empirical initial version of these correlations are () 11 (( )( ))(()()) 1 (())(()) mk rii sjj o ij rs ij x txtxtxt rmk Var xtVar xt , ((1 )ijn (1) where 1 1 () (), n iri r x txt m 2 1 1 (())( ()()), n irii r Varx txtx t m (i = 1,...,n; s=1,...,k). here the () ri x t’s are not i.i.d. over the time t’s, and the sample size mk is often not large, so the above empirical correlations are very crude evaluations of the true corre- lations ij r’s. The initial table (0) (0) (:1 ) ij Rr ijn is used as the raw data for the next step analysis. For each fixed i the observations () i x ts at different time conditions reduced the common features in the data, this table is biased as an estimate of R. We need to restore their values according to the basic property of the ge- netic regulatory system. Many reports have shown that nearby genes tend to have similar expression profiles [16-19], thus nearby pairs of genes tend to have similar relationships, and their correlations tend to be close. This  A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE 979 is just the same principle as used in image restoration of data arrays of any size. In the following we use this technique to reduce the bias and improve the estimate of R based on the observation (0) R. Meloche and Zammar [14] considered a method for image restoration of binary data, here we adopt their idea and revise their method to gene expression analysis for continuous data. We assume the following model (0) , ijij ij rr ij ~ 2 (0, )N , (1 ≤ i < j ≤ n) (2) for some unknown 2 > 0, where the ij ’s represent the part of measurements unexplained by the true reg- ulatory relationships in the model. Define the neighbor (0) ij R of (0) ij r to be the collection of the nine immedi- ate members of (0) ij R of (0) ij r = {(0) :|| 1, ab rai ||1bj}, which includes (0) ij r itself at the center. For (0) ij r’s on the boundary of (0) Rthe definition is modified accordingly. For example, (0) 1,2 R and (0) 1,nn R has only three members, (0) 1, j R (3 < j < n-1) has six members, etc. Larger neighbors of different shapes can also be considered; here we only illustrate using the above neighbor systems. We assume the (0) ij r’s only depend on their neighbors (0) ij R’s. The aim is to provide estimates ij r ’s for the true ij r’s based on the records (0) R. We assume the estimates have the form for some function h( ⋅ ) to be specified. The per- formance of the estimates will be measured by the aver- age conditional mean squared error. (0) () ij ij rhR, (1≤ i < j ≤ n), (3) 2(0) 1 2[() |] (1)ij ij ijn Er rR nn (4) The optimal set of estimates is the one which mini- mizes (4). Although ij r is deterministic, we may view it as a realization of the random variable I J r with (I, J) uniformly distributed over the integer set {(,) :1}Sijijn. So (4) can be rewritten as 2(0) 2(0) (0)2 (0) [() |][() |] [( ())|] IJIJ IJIJ IJ IJ EErr RErr R EhRr R Thus by (3), the minimizer of (4) is achieved by * (0)(0)(0) :()(| )(|) IJIJIJIJ IJ rhRErRErR , and so *(0) (0) ()(|). ij ijIJ rhR ErR To evaluate the above conditional expectation, we need a bit more preparation. Note 2 is estimated by 2(0)2 (, ) 2() (1) ij ij S rr nn , (0) (, ) 2 (1) ij ij S rr nn . Denote 2 (| )t the normal density function with mean 0 and variance 2 . Denote ij S as the collection of indices for (0) ij R. Given (0) ij R, for (,)ij I JS , view (0) IJ r as a random vector over indices (I,J). We define the conditional distribution of (0) I J r as (0)(0) (0) (0) (0) (|) {#} 1 || || IJuv ij ij uv ij ij PrrR member inRr SS In the above we used the fact that the (0) uv rare con- tinuous random variables, so the collection {members in (0) ij R= (0) uv r}={ (0) uv r} almost surely. The correspond- ing conditional probability is defined as (0) (0)(0) 2(0)(0) (0)(0) 2(0)(0) (,) ((,)(,) |) (|)(|) (|)(|) ij ij uv ijuvij uv ijuvij uv S PIJuv R rrPr R rrPr R , (u,v) ij S By (2), we deduce (0) (0) (| ,(,)(,)) IJ IJuv Er RIJuvr, so we have (0) (0) (,) (0) (| )(| ,(,) (,))((,)(,)|) ij ijIJijIJ ij uv S ij rErRErR IJ uvPI JuvR (0)(0)(0) 2(0)(0) (,) (0)(0) 2(0)(0) (,) (|)(|) (|)(|) ij ij uvuv ijuvij uv S uv ijuvij uv S rrr PrR rrPrR (0)(0)(0) 2(0)(0)(0) 2 (,) (,) (0)(0) 2(0)(0) 2 (,) (,) (|)(|) (|) (|) ij ij ij ij uvuv ijuvuv ij uv Suv S uv ijuv ij uv Suv S rrr rrr rr rr , (i, j)ij S (5) The matrix () ij Rr is our one-step restored esti- mate of the genetic correlation network R, we also de- note it by (1) (1) () ij RR r . Denote F(⋅) the operator given in (5), as (1) (1) () ij ij rFR, and denote (1) (1) () ij ij rFR (1) (0)(0) ()(|)RFR ERR . We view F(⋅) as a filter for the noises, so (1) R is a smoothed version of (0) R. Let I J Rr be the random variable of the ij r’s over the random index (I,J) and the variation of possible values of the ij r’s, with density p(⋅), its uncertainty can be characterized by variance and en- tropy, which is defined as H(p) = –E[log p(R)]= –p(r)log p(r)dr. It is maximized or most uncertain when R is uni- formly distributed, and has smaller value when the dis- tribution of r is more certain. It has some relationship with variance. The former depends on more innate fea- tures, such as moments, of the distribution than the latter, which only measures the disparity from the mean. When  980 A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE p(⋅) is a normal density with variance 2 , then 2 () 12Hp . For many commonly used parametric distributions, entropy and variance agree with each other, i.e. an increase in one of them implies so for the other. But this is not always true and a general closed form relationship between variance and entropy does not exists. Variance is more popular in practice because of its sim- plicity. Although generally, in the image restoration context, R is estimated by just applying F once, a natural question is what will happen if we use the operator F repeatedly? i.e. let (1) (1)()() ()()(|) kk kk ij Rr FRERR for k ≥ 0. To investigate this question, we impose the model ()() , kk ij ijij rr ()k ij ~ ),0( )(2 k N , (1 ≤ i < j ≤ n) (6) The estimators ()k ij r ‘s are obtained by minimizing 2() 1 2[() |] (1) k ij ij ijn Er rR nn (7) and are given by () () (| ) kk ijIJ ij rErR. similarly 2()k is estimated by 2( )( )( )2 (, ) 2() (1) kkk ij ij S rr nn , () () (, ) 2 (1) kk ij ij S rr nn . Since n is usually large, 2( )k is a good estimator of 2( )k . Corresponding to (5), we have (1)()() (|) kkk ijIJ ij rErR ()(0) (0)2 (,) (0)(0) 2 (,) ( )(0)(0)2() (,) (0)(0)2() (,) (|) (|) (|) (|) ij ij ij ij k uvuv ij uv S uv ij uv S kk uvuv ij uv S k uv ij uv S rrr rr rrr rr , (i,j) ij S , k1 (8) In the above we do not replace the (0) ij r’s by the ()k ij r’s in (|) but with 2(0) replaced by the step k estimator 2()k , only for the reason of simplicity in the proof of the Proposition below. Finally, 2( )k is re- placed by 2()k in actual computation. Although few density functions are convex, many of them are log-convex. For example, the normal, exponen- tial (in fact any quadratic exponential families), Gamma, Beta, chisquare, triangle, uniform distributions. But some are not, such as the T and Cauchy distributions. Condition A) does not require all the () () k p’s to belong to the same parametric family, nor even to be parametric. Condition B) is satisfied for almost all parametric fami- lies as few parametric families require more than the first two moments to determine. The only restriction we make is that all the () () k p ’s belong to the same para- metric family. View ()k r as a random realization of the ()k ij r’s and as of (0) R, let () () k p be the density function of ()k r. To study the property of the algorithm, we say a non-negative function f(⋅) is log-convex if log f( ⋅ ) is convex, and assume the following conditions A) () () k p is log-convex for all k. B) All the () () k p ’s belong to a parametric family which is determined by the fist two moments. Our algorithm has the following desirable property (see Appendix for the proof) Proposition. 1) Assume either A) or B), then (1)() ()(), kk Hp Hp k ≥0. 2) 2(1)2()kk , k ≥0. 3) As k →∞, the table ()k R converges in the component wise sense: () *k RR for some stationary array **(0) (,)RRRF. This Proposition tells us that, if the assumption of neighboring similarity is valid for (0) R, then the esti- mates ()k R become more and more clear (less entropy), and more and more accurate as an estimator of R (less variance). So (*) R is the sharpest picture the data (0) R provide and can be restored by the filter F, the innate regulatory relationships among the genes can be recov- ered by filter F and provided by the data (0) R under the ideal situation of no noise. Intuitively, this picture has some close relationship with the haplotype block struc- tures. As of small sample size (mk) and large number (n(n – 1)/2) of parameters, there is no way of talking about the consistency of R to R. So in general (*) R and R may not equal, however our algorithm enable us to do the best effort we can. Convergence of ()k R can be accessed by the distance criteria: for a given > 0 (usually =1/100 or 1/1000) (1) (1) ()() 1 2 (,)| | (1) k kk k ij ii ij dR Rrr nn or (1) (1)()()1/2 2 2 (,)(()) (1) k kk k ij ii ij dR Rrr nn .  A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE 981 Network at each time. We may also investigate the problem at each different time point t. In this case (1) is replaced by () 1 (()())(()()) 1 (( ))(( )) mrii rjj o ij rij x txtxtxt rmVar xtVar xt ,(1 )ijn where, 1 1 () (), m iri r x txt m 2 1 1 (())( ()()), m irii r Varx txtx t m (i = 1,...,n; t = 1,...,k) and (0) R(t) = ((0)() ij rt: 1 ≤ i < j ≤n) be the corre- sponding initial table at each t, and the neighborhood for (0) () ij rt is (0) () ij Rt = {(0) ab r: |a – i| ≤ 1, |b – j| ≤ 1}. In this case (6) is () () () ()(), kk ij ijij rtrt t ()() k ij t ~ 2( ) (0,( )) k Nt , (1 ≤ i < j ≤ n) and ()()() () (()|()). kkk ijIJ ij rtEr tRt let 2()( )( )2 (, ) 2 ()( ()()) (1) kkk ij ij S trtrt nn , () () (, ) 2 () () (1) kk ij ij S rt rt nn . (8) is now (1)()() () (()|) kkk ijIJ ij rtErtR ()(0)(0)2 (,) (0)(0) 2 (,) ()(0)(0) 2() (,) (0)(0)2( ) (,) () (()()|) (() ()|) () (()()|()) (()()|()) ij ij ij ij k uvuv ij uv S uv ij uv S kk uvuv ij uv S k uv ij uv S r trtrt rtrt r trtrtt rtrtt , (i,j) ij S , k1 The matrix ()() () (()) kk ij Rtr t is the k-step re- stored estimate of the genetic correlation network ()( ()) ij Rtr t at time t. The proposition is then hold for each fixed t. 3. SIMULATION STUDY We simulate 40 genes over 12 time conditions at time (hour) points 112 ( ,...,)tt = (1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12) for 6 individuals by mimicking the setting of the data analyzed in Iyer et al. [15]. We simulate the genes from 6 clusters, the numbers of genes in each cluster are given by the vector 16 ( ,...,)nn = (8, 6, 4, 4, 6, 12). The base- line values of the gene expressions over time t ∈ [0,12] in cluster k are generated by functions of the form 12 3 sinsin/2sin(/3) kk kk htat atat , (k = 1,....,6). Let h(t) be the vector of length 40, with first n₁ com- ponents given by h1(t), second n2 components by h2(t),...., last n6 components by h6(t). Denote 123 (, , ) kkkk aaaa. We arbitrarily choose the k a’s as a1 = (0.54, –0.18, 1.23), a2 = (–0.12, –0.25, 0.45), a3 = (1.0, –0.55, –0.15), a4 = (–0.32, –0.15, –0.65), a5 = (0.15, 0.25, 0.35) and a6 = (–0.52, –0.45, –0.55). First we need to simulate the ij r’s with coordinated patterns. We divide the 40 genes into the 6 clusters, and assume independence among the clusters. Then for given a covariance matrix 16 we generate the data using this and the time condi- tions, where k is the covariance matrix for the genes in cluster k. Directly specifying a high dimensional posi- tive matrix is not easy, we let each k has the structure ' kkk QQ , for some k Q non-singular, so that k is positive definite. Note that the 'kk QQ’s may not be correlation matrices, but they are covariance matrices, so is . Let k Q be upper diagonal with dimension k n. The non-zero elements of Q1 are drawn from U(0.5,0.8); those for Q2 from U(0.2,0.4); those for Q3 from U(0.2,0.6), those for Q4 from U(–0.3, –0.1); those for Q5 from U(0.6,0.9); and those for Q6 from U(–0.8, –0.6). Then let 1/2 16 QQ . The 6 individuals are i.i.d, so we only need to describe the simulation of observa- tion 11 {():1,...,40;1,...,12} j xxtj t of the first indi- vidual. Note for each fixed t, 1111,40 ( )((),...,( )) x txtxthas covariance matrix . We first generate 140 ( ,...)yyy with the components i.i.d. N(0,1), then 1/2 1() ()() x thty t is the desired sample, where for each fixed t, 140 ( )((),...,( ))tt t is the noise, with the () it ’s i.i.d N(0,1) and independent over t. Convert the covariance matrix () ij to a correlation matrix() ij Rr as / ijijii jj r only for i < j. Using the data (()) ij X xt , we compute the ()k R’s from (8) then use perspective plots to compare the restored correlations after convergence at step k, ()k R, the one-step restored (1) R, the initial estimated (0) R and the true simulated correlations R. After computation, the algorithm meets the conver- gence criterion at iteration 14 with = 10–4. The distances between the observed, first step estimate and last step estimate are: d1((0) R, R) = 0.125, d1((1) R,R) = 0.108 and d1((14) R, R) = 0.094. We see that the estimate after con- vergence is closest to the true correlations. The results are displayed in Figure 1. We only display the correla- tions ij r for j > i. Those values for ij r is 1’s, and those for ij r (i > j) are set to zero’s, which can be obtained by symmetry.  982 A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE From this figure we see that the correlations computed from the raw data, panel (b), are very noisy, the true pat- tern, panel (a), in the network is messed up. The one-step estimate, panel (c), gives some limited sense, while the final estimates, panel (d), recover the true pic- ture with reasonably well. Considering the large number of 40(40+1)/2 = 820 parameters and the small number of 15 individuals on 40 genes, the last step estimates are quite a success. Large number of simulations yield simi- lar results, the convergence criterion is met with 10 to 15 iterations. Note we only used networks of 40 genes, as large networks are not easy to display graphically. The com- putations of a network with n genes is in the order n(n-1)/2, so there should be no computational problem for ordinary computer using this method to restore even the whole genome. 4. RESULTS We use the proposed method to analyze the data with 30 microarray chips from the Stanford microarray database: http:// smd.stanford.edu/cgi-bin/search/QuerySetup.pl. The Category is Normal tissue and the subcategory is PBMC, the following 30 files are the Raw data in the database: 19430.xls, 19438.xls, 19439.xls, 19446.xls, 19447.xls, 19448.xls, 19449.xls, 19450.xls, 19451.xls, 19500.xls, 19505.xls, 19506.xls, 19507.xls, 21407.xls, 21408.xls, 21409.xls, 21410.xls, 21411.xls, 21412.xls, 21413.xls, 21414.xls, 21415.xls, 21416.xls, 21424.xls, 21425.xls, 21426.xls, 21427.xls, 21428.xls, 21429.xls, 21430.xls. The data we used are the overall intensity (mean), the 67th column in the 30 excel files. We choose three subsets of genes on the 30 arrays: set I is genes 0-49, set II is genes 1000-1049 and set III is genes 5000-5049 from the original data set. There are 80 variable for each array. We choose the intensity from normal people for our analysis. The initial correlation coeffi- cients among the genes computed from the raw data in each set, and those estimated after convergence by our algorithm are shown in Figures 2-4. Clearly the initial correlations are noisy and difficult to see any patterned Figure 1. Network Correlations: (a). Simulated R, (b) Initial (0) R, (c) One-step Restored (1) R, (d) k-step Converged ()k R 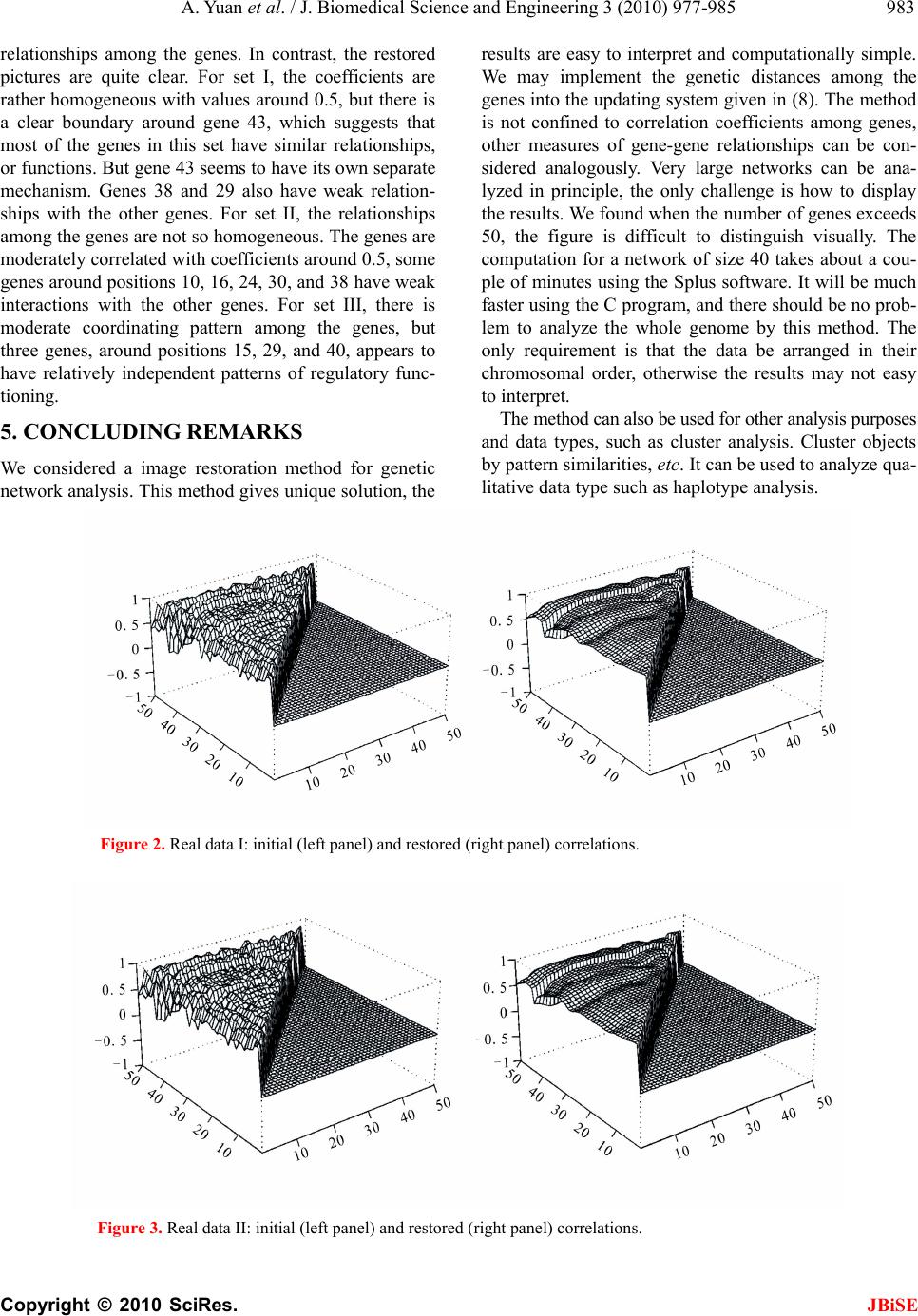 A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE 983 relationships among the genes. In contrast, the restored pictures are quite clear. For set I, the coefficients are rather homogeneous with values around 0.5, but there is a clear boundary around gene 43, which suggests that most of the genes in this set have similar relationships, or functions. But gene 43 seems to have its own separate mechanism. Genes 38 and 29 also have weak relation- ships with the other genes. For set II, the relationships among the genes are not so homogeneous. The genes are moderately correlated with coefficients around 0.5, some genes around positions 10, 16, 24, 30, and 38 have weak interactions with the other genes. For set III, there is moderate coordinating pattern among the genes, but three genes, around positions 15, 29, and 40, appears to have relatively independent patterns of regulatory func- tioning. 5. CONCLUDING REMARKS We considered a image restoration method for genetic network analysis. This method gives unique solution, the results are easy to interpret and computationally simple. We may implement the genetic distances among the genes into the updating system given in (8). The method is not confined to correlation coefficients among genes, other measures of gene-gene relationships can be con- sidered analogously. Very large networks can be ana- lyzed in principle, the only challenge is how to display the results. We found when the number of genes exceeds 50, the figure is difficult to distinguish visually. The computation for a network of size 40 takes about a cou- ple of minutes using the Splus software. It will be much faster using the C program, and there should be no prob- lem to analyze the whole genome by this method. The only requirement is that the data be arranged in their chromosomal order, otherwise the results may not easy to interpret. The method can also be used for other analysis purposes and data types, such as cluster analysis. Cluster objects by pattern similarities, etc. It can be used to analyze qua- litative data type such as haplotype analysis. Figure 2. Real data I: initial (left panel) and restored (right panel) correlations. Figure 3. Real data II: initial (left panel) and restored (right panel) correlations.  984 A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE Figure 4. Real data III: initial (left panel) and restored (right panel) correlations. 6. ACKNOWLEDGEMENTS The research has been supported in part by the National Center for Research Resources at NIH grant 2G12 RR003048. REFERENCES [1] Goodwin, B.C. (1965) Oscillatory behavior in enzymatic control processes. In Weber, G., Ed., Advances in Enzyme Regulation, Pergamon Press, Oxford, 425-438. [2] Tyson, J.J. and Othmer, H.G. (1978) The dynamics of feedback cellular control circuits in biochemical path- ways. Progress in Biophysics, Academic Press, New York, 1-62. [3] Reinitz, J., Mjolsness, E. and Sharp, D.H. (1995) Model for cooperative control of positional information in Dro- sophila by bicoid and maternal hunchback. Journal of Experimental Zoology, 271(1), 47-56. [4] Wessels, L.F.A., Van Someren, E.P. and Reinders, M.J.T. (2001) A comparison of genetic network models. Pacific Symposium on Biocomputing, 6(4), 508-519. [5] D’haeseleer, P., Liang, S. and Somogyi, R. (1999) Gene expression data analysis and modeling. Pacific Sympo- sium on Biocomputating, Hawaii, USA. [6] Savageau, M.A. (1976) Biochemical System Analysis: A Study of Function and Design in Molecular Biology. Ad- dison-Wesley, Reading, Massachusetts. [7] Voit, E.O. (2000). Computational Analysis of Biochemi- cal Systems, Cambridge University Press, Cambridge. [8] Friedman, N., Linial, M., Nachman, I. and Pe’er, D. (2000) Using Bayesian network to analyze expression data. Journal of Computational Biology, 7(3-4), 601-620. [9] Zhang, B.T. and Hwang, K.B. (2003) Bayesian network classifiers for gene expression analysis. In: Berrar D.P., Dubitzky W. and Granzow M. Ed., A Practical Approach to Microarray Data Analysis, Kluwer Academic Publish- ers, Netherlands, 150-165. [10] Chen, L. and Zhao, H. (2005) Gene expression analysis reveals that histone deacetylation sites may serve as par- titions of chromatin gene expression domains. BMC Ge- netics, 6(1), 44. [11] Owen, A. (1984) A neighborhood based classifier for LANDSAT data. Canadian Journal of Statistics, 12(3), 191-200. [12] Ripley, B.D. (1986) Statistics, images, and pattern recog- nition. Canadian Journal of Statistics, 14(2), 83-111. [13] Besag, J. (1986) On the statistical analysis of dirty pic- tures. Journal of the Royal Statistical Society, 48(3), 259- 302. [14] Meloche, J. and Zammar, R. (1994) Binary-image resto- ration. Canadian Journal of Statistics, 22(3), 335-355. [15] Iyer, V.R., Eisen, M.B., Ross, D.T., Schuler, G., Moore, T., Lee, J.C., Trent, J.M., Staudt, L.M., Hudson J.J., Boguski, M.S., Lashkari, D., Shalon, D., Botstein, D. and Brown, P.O. (1999) The transcriptional program in the response of human fibroblast to serum. Science, 283(5398), 83-87. [16] Cohen, B.A., Mitra, R.D., Hughes, J.D. and Church, G.M. (2000) A computational analysis of whole-genome ex- pression data reveals chromosomal domains of gene ex- pression. Nature Genetics, 26(2), 183-186. [17] Caron, H., Schaik, B., Mee, M., Baas, F., Riggins, G., Sluis, P., Hermus, M.C., Asperen, R., Boon, K., Voute, P.A., et al. (2001) The human transcriptome map: clus- tering of highly expressed genes in chromosomal do- mains. Science, 291(5507), 1289-1292. [18] Lercher, M.J., Urrutia, A.O. and Hurst, L.D. (2002) Clustering of housekeeping genes provides a unified model of gene order in the human genome. Nature Ge- netics, 31(2), 180-183. [19] Spellman P.T., Rubin G.M. (2002) Evidence for large domains of similarly expressed genes in the Drosophila genome. Journal of Biology, 1(1), 5. [20] Ebrahimi, N., Maasoumi, E. and Soofi, E. (1999) Order- ing univariate distributions by entropy and variance. Journal of Econometrics, 90(2), 317-336.  A. Yuan et al. / J. Biomedical Science and Engineering 3 (2010) 977-985 Copyright © 2010 SciRes. JBiSE 985 Appendix Proof of the Proposition. Recall (1)(1) (1)() () kkkk IJ IJ rr rR is random in (I,J) and ()k R; for fixed (I,J)=(i,j), (1)(1) () () kkk ij ji rrR is ran- dom in ()k R; (1)(1)()(0) (|) kkk IJ IJ rrRR is random in (I,J) (discrete); also (1) ()()()() (|)[|] kkk kk IJUVIJ rErR ErR for random index (I,J) ∈ S and random index (, ) I J UV S. 1) We first prove the result under condition A). We have (1)(1)() (1) (1) (1)(1)() () log( )log( ) () ()log(||)0, () kk kk k kkk k Ep rEpr pr prdrDpp pr which is the relative entropy between (1) () k p and () () k p. It is known that (1) () (||)0 kk Dpp with “=” if and only if (1)() () () kk pp . Note log-convexity of () () k p imply, for each given ()k IJ R, ()()()() ()() log([| ])[log()| ] kkkkk k IJ IJ pErREprR. Thus by the above two inequalities we get ( 1)(1)(1)()( 1) ()() ()()() () ()log ()log() log([|])( [log(|]) kkkkk kkkkkk IJ IJ HpE prE pr EpErREEprR (1)()() log()(). kk k Ep rHp Under condition B), the result in Ebrahimi et al. [20] states that entropy and variance agree each other. i.e. one increase/decrease implies the other. Now the conclusion is immediate from 2). 2) By the total variance formula, we have () () () ()() () () [( |)] ([ |]) kk UV kk kk UV IJUV IJ Var r EVar rRVar ErR ()()() ()()(1)(1) [(|)]() [( |)] kk k UV IJIJ kk kk UV IJ EVar rRVarr EVar rR , (0,1, 2,...).k 3) We only need to prove the convergence of the component ()k ij r for any fixed (i,j). In fact from (8), for any integer m and k we have (1 )(0)(0)2(1) (,) () (0)(0)2( 1) (,) (1 ) (|) : (|) () ij ij km km uvuv ij uv S km ij km uv ij uv S km ij rrr rrr FR , (i,j) ∈ S, k,m = 0,1,2... Since (0) and () 0 k , by ii), we have () *k for some 0 ≤* < ∞. So if we let (1 )(0)(0)2* (,) () (0)(0) 2* (,) (1 ) (|) : (|) () ij ij km uvuv ij uv S km ij uv ij uv S km ij rrr rrr FR , (i,j) ∈ S, k,m = 0,1,2... then ()() (1) km km ij ij rro as k →∞, thus we only need to prove the convergence of {()km ij r }. Note (1 )2* (1 )(0)(0)2* (,) () (0 |): (|) ij km ij ij km ijuv ij uv S FR C rrr , We have 01 ij C , and for all m, (1)()(1)(1)()(0) ||| ||| ll kmkkm ijijij ijijij ijij rrCr rCrr 11 (1)()(1)(0) 00 (1) (0) || || ||, 1 mm kllkl ijijijijij ijij ll k ij ij ij ij Crr CCrr Crr C thus () 1,2 ,... {} km ij k r is a Cauchy sequence, and the con- vergence follows. |

