 Journal of Behavioral and Brain Science, 2013, 3, 74-84 http://dx.doi.org/10.4236/jbbs.2013.31008 Published Online February 2013 (http://www.scirp.org/journal/jbbs) Protective Effect of Curcumin on Anxiety, Learning Behavior, Neuromuscular Activities, Brain Neurotransmitters and Oxidative Stress Enzymes in Cadmium Intoxicated Mice Gasem M. Abu-Taweel1, Jamaan S. Ajarem2, Mohammad Ahmad3* 1Department of Biology, College of Education, Dammam University, Dammam, KSA 2Department of Zoology, College of Science, King Saud University, Riyadh, KSA 3Department of Medical Surgical Nursing, College of Nursing, King Saud University, Riyadh, KSA Email: *mbadshah@ksu.edu.sa Received November 13, 2012; revised December 15, 2012; accepted December 22, 2012 ABSTRACT Cadmium (Cd) exposure can induce acute lethal health-related threats to humans since it has an exceptional ability to accumulate in living organisms and cause toxicological effects. Curcumin (Cur) on the other hand has a wide variety of biological activities and several animal studies have suggested for a potential therapeutic or preventive effects against several ailments and infections. To study the effect of Cur on the toxicity of Cd, sixty Swiss-Webster strain male mice were divided into 6 groups of ten each at random. Group-1 served as the naïve control and received no treatment. Group-2, 3 and 4 were the experimental controls and were administered once a day with a single oral dose of 50% di- methyl sulphoxide (DMSO), Cur (300 mg/kg) or Cd (100 mg/kg) respectively, for 2 weeks. Group-5 and 6 received Cur and Cd in combination once a day orally for 2 weeks except that Cur in a dose of 150 and 300 mg/kg to group 5 and 6 respectively, was administered one hour before Cd (100 mg/kg) administration to both groups. After treatment period, the animals were subjected to behavioral tests and thereafter, the animals were sacrificed for the estimation of neuro- transmitters like serotonin (5-HT), dopamine (DA) and it’s metabolite 3,4-dihydroxyphenylacetic acid (DOPAC) as well as oxidative stress enzymes like lipid peroxides in the form of thiobarbituric acid–reactive substances (TBARS) and total glutathione (GSH) in the forebrain tissue. Cd reduced significantly the body weight gain, the locomotor activ- ity, anxiety behavior in the plus maze and the learning capability (cognitive effect) in the shuttle-box test. Biochemical analysis further revealed that Cd exposure significantly altered the brain neurotransmitters and the oxidative stress en- zymes. However, administration of Cur along with Cd had an ameliorating effect on all the behavioral and biochemical parameters studied herein and reduced the toxicity of Cd significantly and dose-dependently. Thus, Cur may be benefi- cial for anxiety, neuromuscular, and cognitive problems and protect from Cd intoxication. Keywords: Curcumin; Cadmium; Male Mice; Anxiety; Cognitive Behaviors; Neurotransmitters; Oxidative Stress 1. Introduction Cadmium (Cd) represents one of the most toxic and car- cinogenic heavy metal [1]. It is considered as a serious environmental and industrial pollutant and may represent as a serious health hazard to humans and other animals [2-4]. Some important sources of Cd exposure for hu- mans can be emissions from industries of batteries, metal plating, pigments, plastics, toys and alloy, cigarette smo- king and through dietary consumption [5-7]. Exposure to Cd may cause lesions in many organs such as the liver, kidney and testis [8-11], leading to various possible pa- thological conditions such as hepatic, renal and testicular dysfunction, respiratory and nervous system disorders [12,13]. Cd is reported to induce the generation of reac- tive oxygen species (ROS), and this oxidative stress was found to result in mitochondrial dysfunction and apop- tosis, both in vivo and in vitro [14,15]. The oxidative damage within the tissues and DNA damages is consid- ered to be an early manifestation of Cd toxicity and car- cinogenicity [16,17]. Curcumin (Cur) is a well known biologically active natural phytochemical phenolic compound (diferuloylme- thane) found as a major component in turmeric, a yellow curry spice, extracted from the rhizome of Curcuma longa L. (family Zingiberaceae). Cur is well absorbed in the body system and has exceedingly low toxicity [18]. It *Corresponding author. C opyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 75 possesses many beneficial activities in the body and is effective in several disorders including anorexia, coryza, cough, hepatic diseases, and sinusitis [19,20]. Recent studies provide scientific evidence regarding the poten- tial pharmacological, prophylactic or therapeutic use of Cur, as anti-inflammatory, anticarcinogenic, antiviral, an- tifungal, antiparasitic, antimutagen, antiinfectious and an- tioxidant compound [21-26]. The multiple beneficial ef- fects of Cur have also been elaborated in neurogenesis process which in turn has been reported for its neuropro- tective effects in age-related neurodegenerative diseases [27]. Several studies have shown that Cur exhibits pro- tective effects against oxidative damage and has antioxi- dant property exerting powerful oxygen free radical sca- venging effects and increased intracellular glutathione concentration, thereby protecting lipid peroxidation [28- 32]. Commercial Cur contains 77% curcumin, 17% de- methoxycurcumin and 3% bisdemethoxycurcumin [33] and virtually all these three components in Cur are bio- logically active and possess protective properties [34]. In the light of the above information it appears that Cur may prove beneficial in several ways for Cd toxici- ties and this aspect needs more and more research work. Thus, the present study was undertaken to explore the ef- fects of Cur against the Cd induced behavioral deficits and biochemical toxicity in the brain of male mice. 2. Materials and Methods 2.1. Experimental Animals Sixty male Swiss-Webster strain mice (8 - 10 weeks old) were housed in opaque plastic cages under hygienic con- ditions in the animal facility of the Zoology Department, King Saud University, Riyadh, Saudi Arabia. All animals were maintained under reversed lighting conditions with white lights on from 22.00 to 10.00 hours local time. The ambient temperature was regulated between 20˚C and 22˚C. Food (Pilsbury’s Diet) and water were available ad libitum, unless otherwise indicated. All procedures were carried out in accordance with the ethical guidelines for care and use of laboratory animals, and all protocols were approved by the local Ethics and Care of Experi- mental Animals Committee. All animals were divided into six different groups with ten animals in each. Group I consisted of untreated mice and served as naïve controls. Group II was treated with 50% DMSO (solvent of Cur). Group III was treated with 300 mg/kg Cur dissolved in 50% DMSO. Group IV was treated with Cd (100 mg/kg). Groups V and VI consisted of mice administered with Cur as well as Cd in combina- tion in the doses of 150 + 100 and 300 + 100 mg/kg re- spectively. All exposures were through oral administra- tion, once a day, for two weeks, except that in groups V and VI, Cur was administered one hour before Cd expo- sure. 2.2. Cur and Cd Administration Cur of analytical grade, Sigma Chemical Company, USA, was dissolved in 50% DMSO to give a dose of 150 and 300 mg/kg body weight and diluted further with drinking water in 1.0 ml volume and was administered orally once a day. Cd was also administered orally once a day in the form of cadmium chloride (analytical grade, Riedel de Haen, Germany) dissolved in drinking tap water at a dose of 100 mg/kg body weight in 1.0 ml volume. In the fifth and sixth groups of animals where Cur and Cd were ad- ministered together orally once a day, Cur was adminis- tered one hour before Cd administration. The naïve con- trol group received 1.0 ml plain tap water only. The doses of Cur and Cd used in this study are at par with the effective doses reported in the literature for such studies. The factor for the possibility of presence of Cd traces in food and tap water was not taken into account for calcu- lating the daily Cd intake. However, this factor was mi- nimized by giving the same source of food and tap water to all experimental groups including the controls. 2.3. Body Weight The body weight of all animals from each experimental group was recorded on day 1 of the treatment and on days 5, 10 and 15 of the total treatment period. Thereaf- ter, the animals were subjected to behavioral tests and subsequently were sacrificed for the isolation of fore- brain tissue for the biochemical estimations. 2.4. Behavioral Studies Anxiety, learning capability and locomotor behavior in all animals were measured in the same order in plus maze, shuttle-box and in automated activity meter respectively. 2.4.1. Anxiety Behavior in the Elevated Plus-Maze Test The elevated plus-maze (with 2 opened and 2 enclosed arms) is frequently used as a measure for evaluating the risk assessment and anxiety behavior of an ethologically derived animal model [35]. The plus-maze was elevated to a height of 80 cm above the floor. The mice were in- dividually placed onto the central platform facing one of the open arms and were observed for 5 min while freely exploring the maze. The animal was considered to have entered an arm when all four limbs were inside the arm. Duration of time spent in open and enclosed arms and number of entries in open and enclosed arms were meas- ured during the test period. On completion of the test, the maze was cleaned with a 10% ethanol solution to control Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 76 for any possible olfactory cues. 2.4.2. Learning Capability in the Shuttle-Box Test (Active Avoidance Responses) The active avoidance responses were measured in the animals using an automatic reflex conditioner “shuttle box” (Ugo Basile, Comerio-Varese, Italy). The rectangu- lar shaped shuttle-box was divided into two chambers of equal size by a stainless steel partition with a gate pro- viding access to the adjacent compartment. Before start- ing the trial sessions, each animal was allowed to adapt and acquaint itself with the shuttle box for 2 min without any stimulus. A light bulb (21 W) for 6 s duration and a buzzer (670 Hz and 70 dB) was switched on consecu- tively and used as a conditioned stimulus (CS). The CS preceded the onset of the unconditioned stimulus (US) by 5 s. The US was an electric scrambler shock (1 mA for 4 s) applied to the metallic grid floor. If the animal avoided the US by running into the other compartment within 5 s after the onset of the CS, the microprocessor recorder unit of the shuttle box recorded an avoidance response and this was considered as conditioned avoidance response to avoid the electric shock. Each animal was given 50 trials with a fixed inter trial interval of 15 s. During the 50 tri- als session of the individual animal, the total number of avoidance was measured. The total time taken until the animal entered the other compartment to avoid the shock treatment (latency of avoidance response or escape la- tency in seconds) was also measured for each animal. The recorder unit of the automated shuttle box continu- ously recorded these parameters during the whole expe- rimental period (50 trials) of each animal. 2.4.3. Motor Activity Test in Automated Activity Meter Motor activity was measured using automated electronic activity meter (Ugo Basile, Comerio-Varese, Italy). The horizontal and vertical motor activities were detected by arrays of infrared beams located above the floor of the testing arena. Each interruption of the beams on the x or y axis generated an electric impulse which was recorded on a digital counter. Each animal was tested separately and the motor activity was recorded for a period of 2 min in the activity meter. 2.5. Biochemical Studies Immediately after completing the behavioral tests, the animals were sacrificed by decapitation, the brains were dissected on ice, the fore brain areas (containing the hip- pocampus and striatum areas) were removed and frozen in liquid nitrogen and stored at −70˚C for determination of monoamines, lipid peroxides (TBARS) and glutathi- one content. 2.5.1. Determination of Monoamines The monoamines were estimated using the modified me- thod of Patrick et al. [36]. A 10% homogenate of fore brain tissue was prepared by homogenizing the tissues for 10 s in 0.1 M HClO4 containing 0.05% EDTA, cen- trifuged at 17,000 rpm at 4˚C for 5 min. The supernatants were filtered using 0.45 µm pore filters and analyzed by high performance liquid chromatography (HPLC). The mobile phase consisted of 32 mM citric acid monohy- drate, 12.5 mM disodium hydrogen orthophosphate, 7% methanol, 1 mM octane sulphonic acid and 0.05 mM EDTA. The mobile phase was filtered through 0.22 µm filter and degassed under vacuum before use. µBondpak C18 column was used at a flow rate of 1.2 ml/min and the injection volume of the sample was 20 µl. The levels of dopamine (DA), DOPAC and serotonin or 5-hydroxy- tryptamine (5-HT) were calculated using a calibration curve and results were expressed as ng/mg tissue weight. 2.5.2. Determination of Lipid Per oxides Lipid peroxides (LP) in fore brain tissue were determined spectrophotometrically as thiobarbituric acid-reactive sub- stances (TBARS) according to the method of Ohkawa et al. [37]. The tissue samples were homogenized in 1.15% cold KCl with an Ultraturax homogenizer. After centrifu- gation at 3000× g for 5 min, an aliquot of supernatant was mixed with 2 ml of reaction mixture (containing 15% trichloroacetic acid and 0.375% thiobarbituric acid solution in 0.25 N HCl) and heated for 5 min in a boiling water bath. The tubes were cooled at room temperature and centrifuged at 1000× g for 10 min. The absorbance of supernatant was read at 535 nm against a blank that contained all reagents except homogenate. Tissue lipid peroxide levels were quantified using extinction coeffi- cient of 1.56 × 105 m−1·cm−1 and expressed as nanomoles of TBARS formed per g tissue weight. 2.5.3. Determination of Glutathione Total glutathione (GSH) level in fore brain tissue was measured enzymatically in the brain tissues by a slightly modified method of Mangino et al. [38]. Briefly, about 50 mg of isolated brain tissues were homogenized with 1 ml 0.1 M perchloric acid plus 0.005% EDTA. The ho- mogenates were centrifuged at 4000 rpm for 10 min and the supernatants were used for GSH assay. The reaction mixture consisted of the following three freshly prepared solutions: solution I, 0.3 mM NADPH; solution II, 6 mM 5,5’-dithio-bis(2-nitrobenzoic acid) and solution III, 50 U/ml glutathione (all chemicals from Sigma). All three solutions were prepared with a stock buffer consisting of 125 mM NaH2PO4 and 6.3 mM EDTA at pH 7.5. At the time of glutathione assay, 800 µl of solution I, 100 µl of solution II, and 10 µl of solution III were mixed in a quartz cuvette and placed in a dual beam UV-VIS spec- Copyright © 2013 SciRes. JBBS 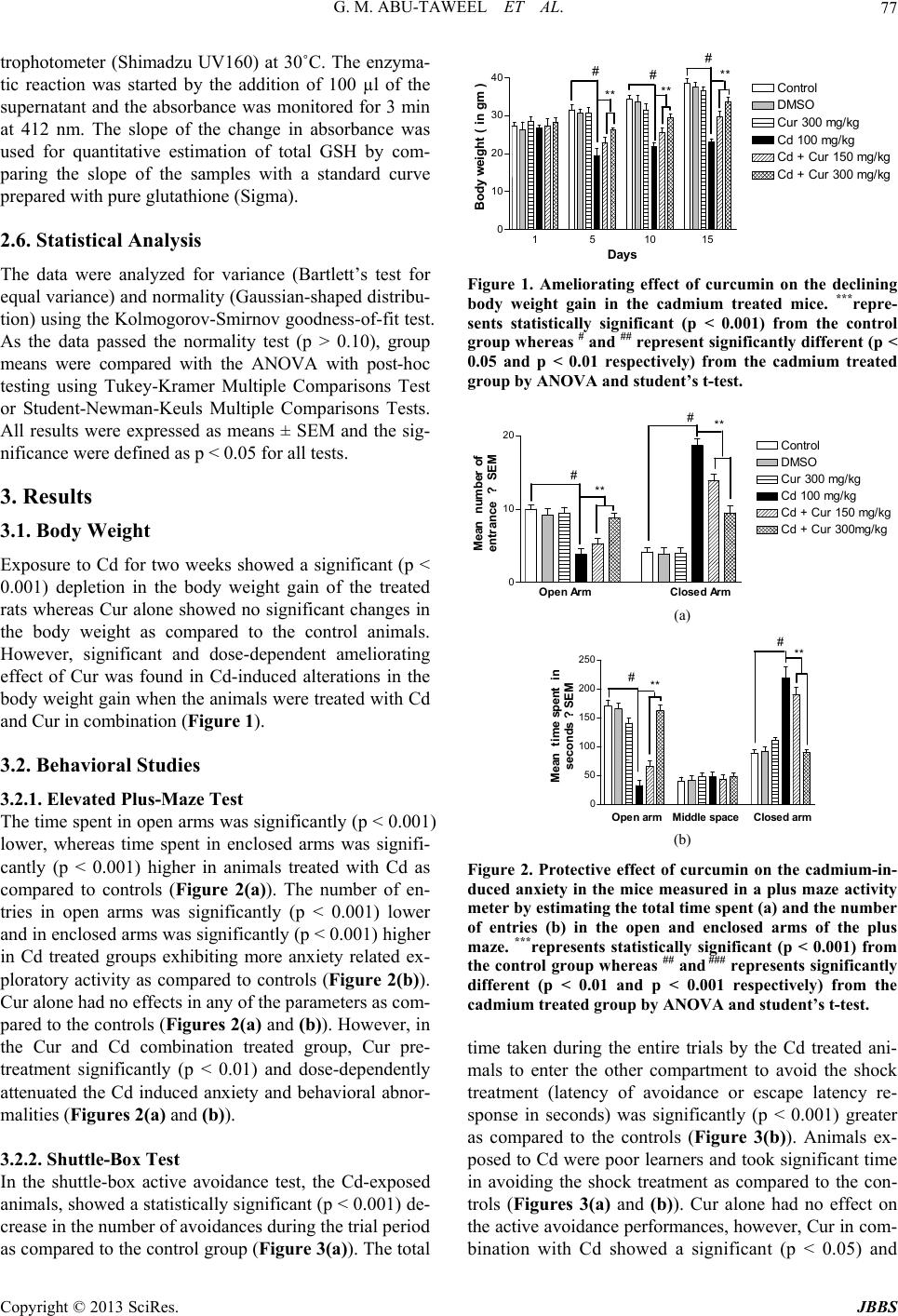 G. M. ABU-TAWEEL ET AL. 77 trophotometer (Shimadzu UV160) at 30˚C. The enzyma- tic reaction was started by the addition of 100 µl of the supernatant and the absorbance was monitored for 3 min at 412 nm. The slope of the change in absorbance was used for quantitative estimation of total GSH by com- paring the slope of the samples with a standard curve prepared with pure glutathione (Sigma). 2.6. Statistical Analysis The data were analyzed for variance (Bartlett’s test for equal variance) and normality (Gaussian-shaped distribu- tion) using the Kolmogorov-Smirnov goodness-of-fit test. As the data passed the normality test (p > 0.10), group means were compared with the ANOVA with post-hoc testing using Tukey-Kramer Multiple Comparisons Test or Student-Newman-Keuls Multiple Comparisons Tests. All results were expressed as means ± SEM and the sig- nificance were defined as p < 0.05 for all tests. 3. Results 3.1. Body Weight Exposure to Cd for two weeks showed a significant (p < 0.001) depletion in the body weight gain of the treated rats whereas Cur alone showed no significant changes in the body weight as compared to the control animals. However, significant and dose-dependent ameliorating effect of Cur was found in Cd-induced alterations in the body weight gain when the animals were treated with Cd and Cur in combination (Figure 1). 3.2. Behavioral Studies 3.2.1. Elevated Plus-Maze Test The time spent in open arms was significantly (p < 0.001) lower, whereas time spent in enclosed arms was signifi- cantly (p < 0.001) higher in animals treated with Cd as compared to controls (Figure 2(a)). The number of en- tries in open arms was significantly (p < 0.001) lower and in enclosed arms was significantly (p < 0.001) higher in Cd treated groups exhibiting more anxiety related ex- ploratory activity as compared to controls (Figure 2(b)). Cur alone had no effects in any of the parameters as com- pared to the controls (Figures 2(a) and (b)). However, in the Cur and Cd combination treated group, Cur pre- treatment significantly (p < 0.01) and dose-dependently attenuated the Cd induced anxiety and behavioral abnor- malities (Figures 2(a) and (b)). 3.2.2. Shuttl e-Box Test In the shuttle-box active avoidance test, the Cd-exposed animals, showed a statistically significant (p < 0.001) de- crease in the number of avoidances during the trial period as compared to the control group (Figure 3(a)). The total 1510 15 0 10 20 30 40 Contr ol DMSO Cur 300 mg/kg Cd 100 mg/kg Cd + C ur 150 mg/kg ## # ** ** ** Days Body weight ( i n gm ) Cd + C ur 300 mg/kg Figure 1. Ameliorating effect of curcumin on the declining body weight gain in the cadmium treated mice. ***repre- sents statistically significant (p < 0.001) from the control group whereas # and ## represent significantly different (p < 0.05 and p < 0.01 respectively) from the cadmium treated group by ANOVA and student’s t-test. 0 10 20 Control # # ** ** Mean number of entrance ? SE M [A] DMSO Cur 300 mg/ kg Cd 100 mg/kg Cd + Cur 150 mg/kg Cd + C ur 30 0mg/kg Open Arm Closed Arm (a) 0 50 100 150 200 250 O en arm Midd le s ace Closed arm Me an time spent in seconds ?SEM #** #** [B] (b) Figure 2. Protective effect of curcumin on the cadmium-in- duced anxiety in the mice measured in a plus maze activity meter by estimating the total time spent (a) and the number of entries (b) in the open and enclosed arms of the plus maze. ***represents statistically significant (p < 0.001) from the control group whereas ## and ### represents significantly different (p < 0.01 and p < 0.001 respectively) from the cadmium treated group by ANOVA and student’s t-test. time taken during the entire trials by the Cd treated ani- mals to enter the other compartment to avoid the shock treatment (latency of avoidance or escape latency re- sponse in seconds) was significantly (p < 0.001) greater as compared to the controls (Figure 3(b)). Animals ex- posed to Cd were poor learners and took significant time in avoiding the shock treatment as compared to the con- trols (Figures 3(a) and (b)). Cur alone had no effect on the active avoidance performances, however, Cur in com- bination with Cd showed a significant (p < 0.05) and Copyright © 2013 SciRes. JBBS 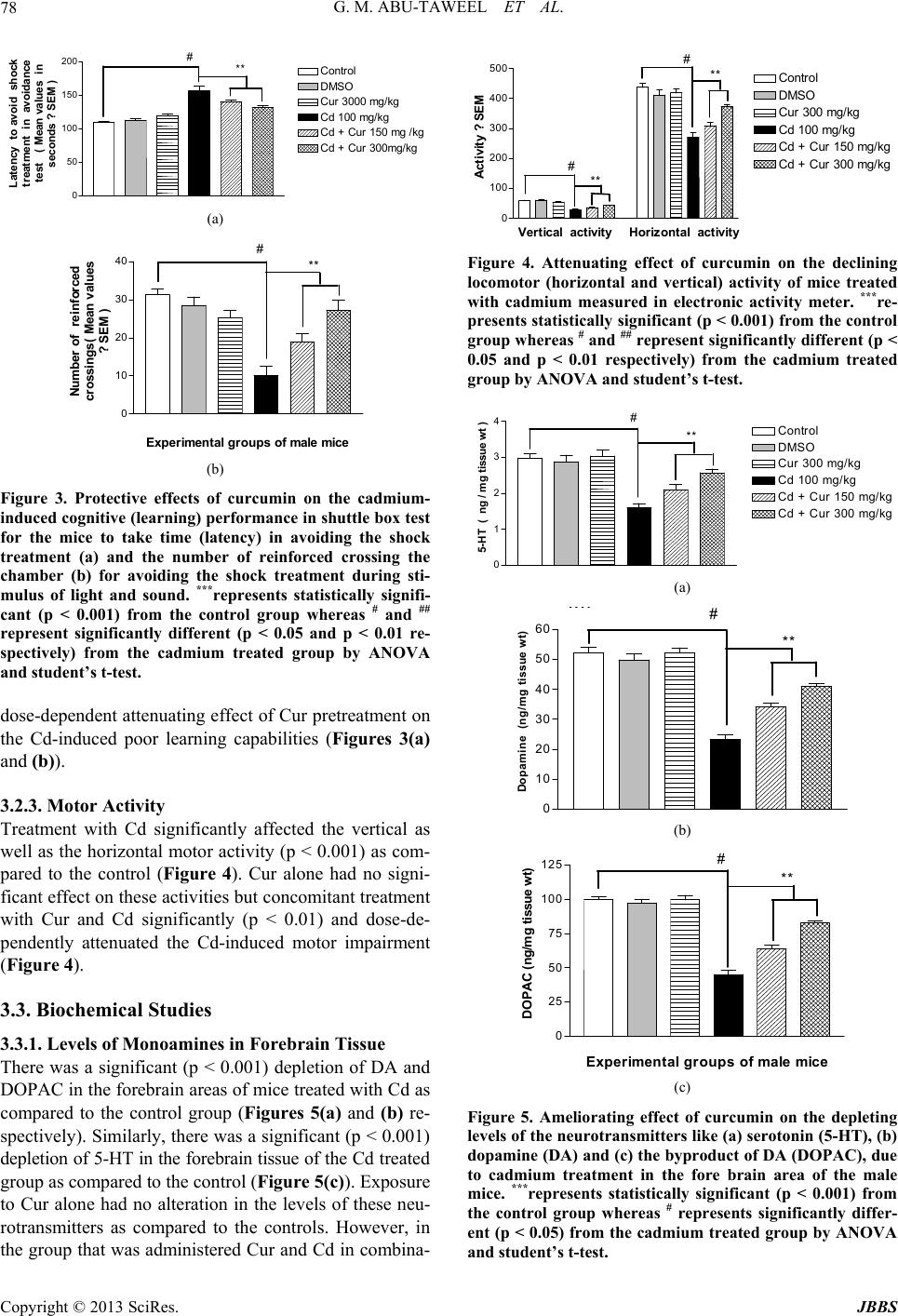 G. M. ABU-TAWEEL ET AL. 78 0 50 100 150 200 Contr ol DMSO Cur 3 000 mg/kg Cd 100 mg/kg Cd + Cur 150 mg /kg Cd + Cur 300mg/kg #** Latency to avoid shock t reat ment i n avoi dance t est ( M ean values i n seconds ?SEM ) (a) 0 10 20 30 40 Exper imental gr oups o f male mice #** Number of reinforced crossings( M ean valu es ?SEM ) (b) Figure 3. Protective effects of curcumin on the cadmium- induced cognitive (learning) performance in shuttle box test for the mice to take time (latency) in avoiding the shock treatment (a) and the number of reinforced crossing the chamber (b) for avoiding the shock treatment during sti- mulus of light and sound. ***represents statistically signifi- cant (p < 0.001) from the control group whereas # and ## represent significantly different (p < 0.05 and p < 0.01 re- spectively) from the cadmium treated group by ANOVA and student’s t-test. dose-dependent attenuating effect of Cur pretreatment on the Cd-induced poor learning capabilities (Figures 3(a) and (b)). 3.2.3. M otor Activi t y Treatment with Cd significantly affected the vertical as well as the horizontal motor activity (p < 0.001) as com- pared to the control (Figure 4). Cur alone had no signi- ficant effect on these activities but concomitant treatment with Cur and Cd significantly (p < 0.01) and dose-de- pendently attenuated the Cd-induced motor impairment (Figure 4). 3.3. Biochemical Studies 3.3.1. Levels of Monoamines in Forebrain Tissue There was a significant (p < 0.001) depletion of DA and DOPAC in the forebrain areas of mice treated with Cd as compared to the control group (Figures 5(a) and (b) re- spectively). Similarly, there was a significant (p < 0.001) depletion of 5-HT in the forebrain tissue of the Cd treated group as compared to the control (Figure 5(c)). Exposure to Cur alone had no alteration in the levels of these neu- rotransmitters as compared to the controls. However, in the group that was administered Cur and Cd in combina- 0 100 200 300 400 500 Control DMSO Cur 300 mg/ kg Cd 100 mg/kg Cd + C ur 15 0 mg/kg #** #** Ver t ical activity Activity ?SEM Hor iz ont al activity Cd + C ur 30 0 mg/kg Figure 4. Attenuating effect of curcumin on the declining locomotor (horizontal and vertical) activity of mice treated with cadmium measured in electronic activity meter. ***re- presents statistically significant (p < 0.001) from the control group whereas # and ## represent significantly different (p < 0.05 and p < 0.01 respectively) from the cadmium treated group by ANOVA and student’s t-test. 0 1 2 3 4Control DMSO Cur 300 mg/kg Cd 100 mg/kg Cd + Cur 150 mg/kg Cd + Cur 300 mg/kg # ** 5-HT ( ng / mg tissue wt ) (a) 0 10 20 30 40 50 60 Dopamine (ng/mg tissue wt) # ** (b) 0 25 50 75 100 125 Experimental groups of male mice # ** DOPAC (ng/mg tissue wt) (c) Figure 5. Ameliorating effect of curcumin on the depleting levels of the neurotransmitters like (a) serotonin (5-HT), (b) dopamine (DA) and (c) the byproduct of DA (DOPAC), due to cadmium treatment in the fore brain area of the male mice. ***represents statistically significant (p < 0.001) from the control group whereas # represents significantly differ- ent (p < 0.05) from the cadmium treated group by ANOVA and student’s t-test. Copyright © 2013 SciRes. JBBS 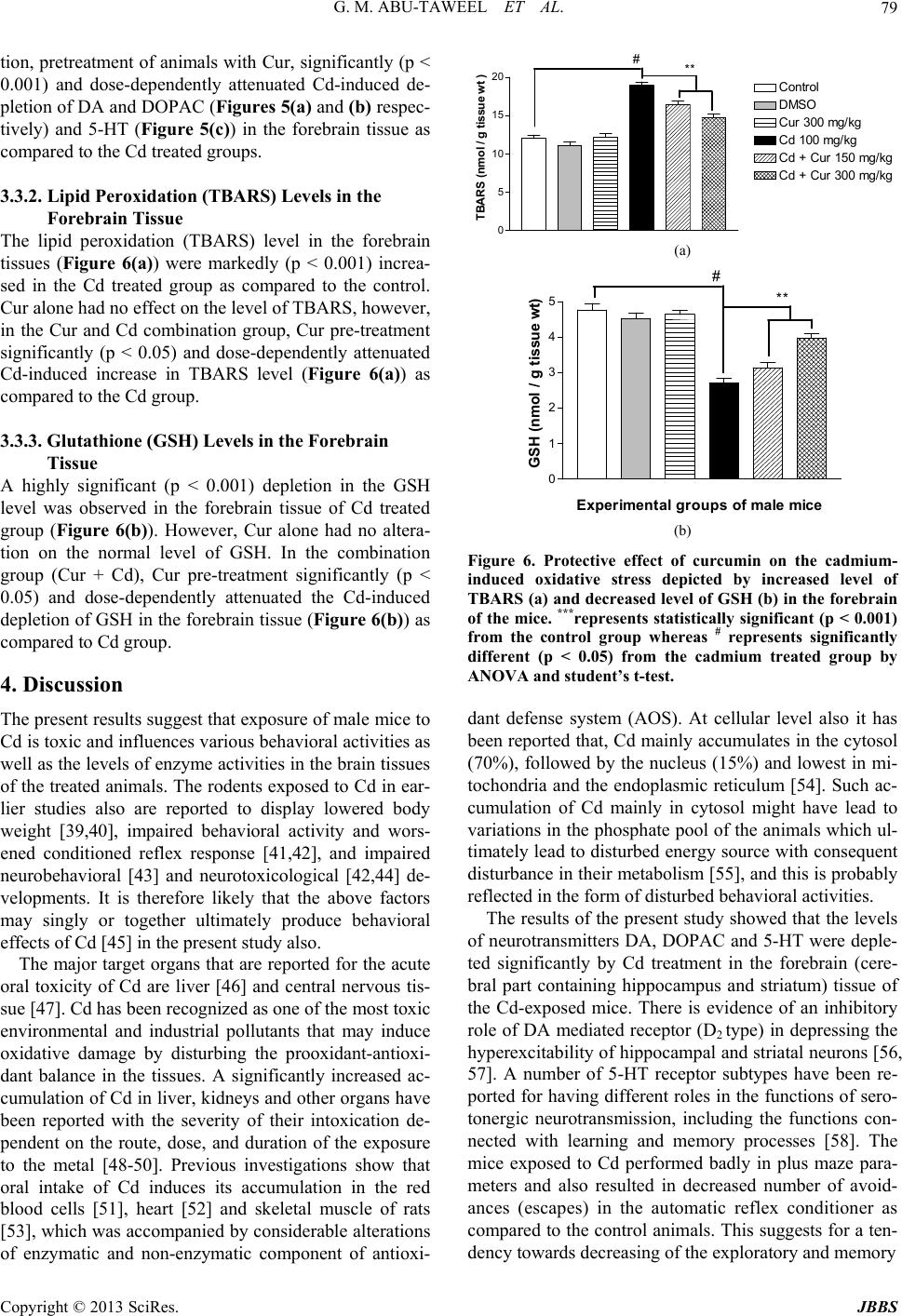 G. M. ABU-TAWEEL ET AL. 79 tion, pretreatment of animals with Cur, significantly (p < 0.001) and dose-dependently attenuated Cd-induced de- pletion of DA and DOPAC (Figures 5(a) and (b) respec- tively) and 5-HT (Figure 5(c)) in the forebrain tissue as compared to the Cd treated groups. 3.3.2. Lipid Peroxidation (TBARS) Levels in the Forebrain Tissue The lipid peroxidation (TBARS) level in the forebrain tissues (Figure 6(a)) were markedly (p < 0.001) increa- sed in the Cd treated group as compared to the control. Cur alone had no effect on the level of TBARS, however, in the Cur and Cd combination group, Cur pre-treatment significantly (p < 0.05) and dose-dependently attenuated Cd-induced increase in TBARS level (Figure 6(a)) as compared to the Cd group. 3.3.3. Glutathione (GSH) Levels in th e Fo rebrain Tissue A highly significant (p < 0.001) depletion in the GSH level was observed in the forebrain tissue of Cd treated group (Figure 6(b)). However, Cur alone had no altera- tion on the normal level of GSH. In the combination group (Cur + Cd), Cur pre-treatment significantly (p < 0.05) and dose-dependently attenuated the Cd-induced depletion of GSH in the forebrain tissue (Figure 6(b)) as compared to Cd group. 4. Discussion The present results suggest that exposure of male mice to Cd is toxic and influences various behavioral activities as well as the levels of enzyme activities in the brain tissues of the treated animals. The rodents exposed to Cd in ear- lier studies also are reported to display lowered body weight [39,40], impaired behavioral activity and wors- ened conditioned reflex response [41,42], and impaired neurobehavioral [43] and neurotoxicological [42,44] de- velopments. It is therefore likely that the above factors may singly or together ultimately produce behavioral effects of Cd [45] in the present study also. The major target organs that are reported for the acute oral toxicity of Cd are liver [46] and central nervous tis- sue [47]. Cd has been recognized as one of the most toxic environmental and industrial pollutants that may induce oxidative damage by disturbing the prooxidant-antioxi- dant balance in the tissues. A significantly increased ac- cumulation of Cd in liver, kidneys and other organs have been reported with the severity of their intoxication de- pendent on the route, dose, and duration of the exposure to the metal [48-50]. Previous investigations show that oral intake of Cd induces its accumulation in the red blood cells [51], heart [52] and skeletal muscle of rats [53], which was accompanied by considerable alterations of enzymatic and non-enzymatic component of antioxi- 0 5 10 15 20 Control DMSO Cur 300 mg/kg Cd 100 mg/kg Cd + Cur 150 mg/kg ** TBARS (nmol / g tissue wt ) Cd + Cur 300 mg/kg (a) # ** 5 4 3 2 1 0 Experimental groups of male mice GSH (nmol / g t issue wt) (b) Figure 6. Protective effect of curcumin on the cadmium- induced oxidative stress depicted by increased level of TBARS (a) and decreased level of GSH (b) in the forebrain of the mice. ***represents statistically significant (p < 0.001) from the control group whereas # represents significantly different (p < 0.05) from the cadmium treated group by ANOVA and student’s t-test. dant defense system (AOS). At cellular level also it has been reported that, Cd mainly accumulates in the cytosol (70%), followed by the nucleus (15%) and lowest in mi- tochondria and the endoplasmic reticulum [54]. Such ac- cumulation of Cd mainly in cytosol might have lead to variations in the phosphate pool of the animals which ul- timately lead to disturbed energy source with consequent disturbance in their metabolism [55], and this is probably reflected in the form of disturbed behavioral activities. The results of the present study showed that the levels of neurotransmitters DA, DOPAC and 5-HT were deple- ted significantly by Cd treatment in the forebrain (cere- bral part containing hippocampus and striatum) tissue of the Cd-exposed mice. There is evidence of an inhibitory role of DA mediated receptor (D2 type) in depressing the hyperexcitability of hippocampal and striatal neurons [56, 57]. A number of 5-HT receptor subtypes have been re- ported for having different roles in the functions of sero- tonergic neurotransmission, including the functions con- nected with learning and memory processes [58]. The mice exposed to Cd performed badly in plus maze para- meters and also resulted in decreased number of avoid- ances (escapes) in the automatic reflex conditioner as compared to the control animals. This suggests for a ten- dency towards decreasing of the exploratory and memory Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 80 effect of Cd under conditions of reduced functional ca- pacity of serotonergic neurotransmission as also reported earlier for aluminum toxicity [59]. Recently, a growing body of research has focused on the participation of se- rotonin (5-HT) in the neurochemical mechanisms of cog- nition and especially of learning and memory. Potential toxic mechanisms of action for Cd may include disrup- tion in serotonergic neurotransmission through disturbed levels of neurotransmitters in the brain hippocampus [60]. Other studies have also shown that Cd inhibits the ac- tivity of majority of enzymes involved in AOS [61-63] inducing an increased production of free radicals, lipid peroxidation, and destruction of cell membranes [51,54, 64]. Cd is also reported to inhibit the activities of many enzymes by binding to their sulfhydryl groups or by in- hibiting the protein synthesis [65,66]. Cur in the present study had a significantly ameliorat- ing effect on the Cd-induced deficits in the body weight, anxiety behavior, learning capability (cognitive effect) and muscular activity. Biochemical analysis in forebrain tissue also revealed that Cur significantly attenuated Cd- induced neurotransmitters (reportedly associated with lo- comotor and cognitive activities) and the Cd-induced oxidative stress related enzymes (associated with behav- ioral and cognitive deficits). Furthermore, the ineffective- ness of Cur alone to cause any behavioral and biochemi- cal deficits, clearly suggest that Cur alone is non-toxic and further supports for the ameliorating effect of Cur on the behavioral and biochemical toxicity induced by Cd. The biochemical damage may be due to the fact that Cd induces an oxidative stress that results in oxidative dete- rioration of biological macromolecules [40,67]. Cur re- portedly has potent antioxidant activities [68,69], anti-in- flammatory [70] and chemoprotective properties [71]. It has been shown to have a neuroprotective effect in mod- els of cerebral ischemia [72,73], ethanol induced brain damage [74] and reduced amyloid pathology in trans- genic mice of Alzheimer’s disease [75]. Lipid peroxidation (LP) is one of the main manifesta- tions of oxidative damage, which plays an important role in the toxicity of many xenobiotics [76,77]. Our results confirm that intoxication with Cd causes a significant in- crease of lipid peroxide concentration in forebrain tissue of mice. Since it causes LP in numerous tissues both in vivo and in vitro [6,51,62,65], it has been suggested that Cd may induce oxidative stress by producing hydroxyl radicals [78], superoxide anions, nitric oxide and hydro- gen peroxide [79,80]. Moreover, it has been shown that various antioxidants and AOS protect cells from Cd-in- duced toxicity [81-83]. Co-treatment with Cur in the present study was effec- tive in the prevention of oxidative damage induced by Cd, which resulted in significantly lower lipid peroxides con- centration in the form of TBARS in the forebrain tissue. This can be explained by the important role of Cur in preventing LP and in protection of integrity and func- tioning of tissues and cells. The prevention of LP is es- sential for all aerobic organisms and so the organism is well equipped with antioxidants that directly or indirectly protect cells against the adverse effects of xenobiotics, carcinogens and toxic radicals [84]. The role of antioxi- dants in reversing this oxidative stress has been a matter of deep interest to basic scientists and clinicians [85]. The decreased activity of GSH due to Cd exposure in the present study suggests for the disturbed oxidant and antioxidant system in the brain tissue. Furthermore, pres- ence of Cur along with Cd ameliorates significantly the GSH level and tries to increase the level of GSH indicat- ing for the role of Cur as an antioxidant. It has been well established that the antioxidants such as Vit E, Vit C and GSH protect the membrane from oxidative damage [64, 84,85]. In the present study, Cur reduced the cellular to- xicity caused by Cd-induced ROS and protected the brain antioxidant system. Thus, Cd accumulation in brain tis- sue is most likely due to chronic dietary intake of Cd, and it is associated with marked alteration of neurotrans- mitters and enzyme GSH of AOS. These results also sug- gest that LP is associated with Cd toxicity in brain tissue. Our results showed that the antioxidant Cur ameliorated oxidative stress and loss of cellular antioxidants and sug- gested that Cur efficiently protect forebrain from Cd-in- duced oxidative damage. However, for asserting this state- ment, further studies are needed to measure other enzy- matic components like superoxide dismutase (SOD), ca- talase (CAT), glutathione peroxidase (GSH-Px) and glu- tathione-S transferase (GST) and nonenzymatic compo- nents such as vitamin C (Vit C) and vitamin E (Vit E) of AOS. 5. Conclusion It is concluded from the present study that although Cur has many possible reported benefits, the full effects however, are not yet fully understood and more and more research work is needed. The results indicate that Cur possesses several multifold beneficial effects that may in- clude better cognitive performance, better muscular ac- tivity and protection from externally induced oxidative stress and neurotransmitters dysfunction in the brain. Thus, inclusion of Cur in our normal diet and its use as a nutritional supplement may have a tremendous potential for health improvement and protection from Cd intoxica- tion. 6. Acknowledgements The authors are grateful to the The Excellence Center of Science and Mathematics Education, King Saud Univer- Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 81 sity, Riyadh, for their support and encouragement. REFERENCES [1] IARC, “International Agency for Research on Cancer Mo- nographs. Cadmium,” IARC Press, Lyon, 1993, pp. 119- 238. [2] M. M. Brzoska and J. M. Jakoniuk, “Interactions between Cadmium and Zinc in the Organism,” Food and Chemical Toxicology, Vol. 39, No. 10, 2001, pp. 967-980. doi:10.1016/S0278-6915(01)00048-5 [3] M. M. Brzoska, K. Majewska and E. Kupraszewicz, “Ef- fects of Low, Moderate and Relatively High Chronic Ex- posure to Cadmium on Long Bones Susceptibility to Frac- tures in Male Rats,” Environmental Toxicology and Pharmacology, Vol. 29, No. 3, 2010, pp. 235-245. doi:10.1016/j.etap.2010.01.005 [4] J. L. Li, R. Gao, S. Li, J. T. Wang, Z. X. Tang and S. W. Xu, “Testicular Toxicity Induced by Dietary Cadmium in Cocks and Ameliorative Effect by Selenium,” Biometals, Vol. 23, No. 4, 2010, pp. 695-705. doi:10.1007/s10534-010-9334-0 [5] P. B. Hammond and E. C. Foulkes, “Metal Ion Toxicity in Man and Animals,” In: H. Sigel, Ed., Metal Ions in Biological Systems, Marcel Dekker, New York, 1986, pp. 157-200. [6] F. M. El-Demerdash, M. I. Yousef, F. S. Kedwany and H. H. Baghdadi, “Cadmium-Induced Changes in Lipid Per- oxidation, Blood Hematology, Biochemical Parameters and Semen Quality of Male Rats: Protective Role of Vi- tamin E and Beta-Carotene,” Food and Chemical Toxi- cology, Vol. 42, No. 10, 2004, pp. 1563-1571. doi:10.1016/j.fct.2004.05.001 [7] P. F. de Souza, M. A. Diamante and H. Dolder, “Testis Response to Low Doses of Cadmium in Wistar Rats,” In- ternational Journal of Experimental Pathology, Vol. 91, No. 2, 2010, pp. 125-131. doi:10.1111/j.1365-2613.2009.00692.x [8] M. Satoh, H. Koyama, T. Kaji, H. Kito and C. Tohyama, “Perspectives on Cadmium Toxicity Research,” Tohoku Journal of Experimental Medicine, Vol. 196, No. 1, 2002, pp. 23-32. doi:10.1620/tjem.196.23 [9] R. A. Goyer, J. Liu and M. P. Waalkes, “Cadmium and Cancer of Prostate and Testis,” Biometals, Vol. 17, No. 5, 2004, pp. 555-558. doi:10.1023/B:BIOM.0000045738.59708.20 [10] L. C. Xu, H. Sun, S. Y. Wang, L. Song, H. C. Chang and X. R. Wang, “The Roles of Metallothionein on Cadmium- Induced Testes Damages in Sprague Dawley Rats,” En- vironmental Toxicology and Pharmacology, Vol. 20, No. 1, 2005, pp. 83-87. doi:10.1016/j.etap.2004.10.008 [11] S. Amara, H. Abdelmelek, C. Garrel, P. Guiraud, T. Douki, J. L. Ravanat, A. Favier, M. Sakly and K. Ben Rhouma, “Preventive Effect of Zinc against Cadmium-Induced Stress in the Rat Testis,” Journal of Reproductive Devel- opment, Vol. 54, No. 2, 2008, pp. 129-134. doi:10.1262/jrd.18110 [12] J. Thompson and J. Bannigan, “Cadmium: Toxic Effects on the Reproductive System and the Embryo,” Reproduc- tive Toxicology, Vol. 25, No. 3, 2008, pp. 304-315. doi:10.1016/j.reprotox.2008.02.001 [13] B. I. Ognjanovic, S. D. Markovic, N. Z. Ethordevic, I. S. Trbojevic, A. S. Stajn and Z. S. Saicic, “Cadmium-In- duced Lipid Peroxidation and Changes in Antioxidant De- fense System in the Rat Testes: Protective Role of Coen- zyme Q(10) and Vitamin E,” Reproductive Toxicology, Vol. 29, No. 2, 2010, pp. 191-197. doi:10.1016/j.reprotox.2009.11.009 [14] S. Jimi, M. Uchiyama, A. Takaki, J. Suzumiya and S. Hara, “Mechanisms of Cell Death Induced by Cadmium and Arsenic,” Annals of New York Academy of Sciences, Vol. 1011, No. 1, 2004, pp. 325-331. doi:10.1196/annals.1293.032 [15] R. Sen Gupta, J. Kim, C. Gomes, S. Oh, J. Park, W. B. Im, J. Y. Seong, R. S. Ahn, H. B. Kwon and J. Soh, “Effect of Ascorbic Acid Supplementation on Testicular Steroido- genesis and Germ Cell Death in Cadmium-Treated Male Rats,” Molecular and Cellular Endocrinology, Vol. 221, No. 1-2, 2004, pp. 57-66. doi:10.1016/j.mce.2004.03.012 [16] M. P. Waalkes, T. P. Coogan and R. A. Barter, “Toxico- logical Principles of Metal Carcinogenesis with Special Emphasis on Cadmium,” Critical Review of Toxicology, Vol. 22, No. 3-4, 1992, pp. 175-201. doi:10.3109/10408449209145323 [17] M. P. Waalkes, “Cadmium Carcinogenesis in Review,” Journal of Inorganic Biochemistry, Vol. 79, No. 1, 2000, pp. 241-244. doi:10.1016/S0162-0134(00)00009-X [18] National Toxicology Program, “NTP Toxicology and Car- cinogenesis in Studies of Turmeric Oleoresin (CAS No. 8024-37-1) (Major Component 79% - 85% Curcumin, CAS No. 458-37-7) in F344/N Rats and B6C3F1 Mice (Feed Studies),” National Toxicology Program Technical Reproductive Services, Vol. 427, 1993, pp. 1-275. [19] I. Rahman, S. K. Biswas and P. A. Kirkham, “Regulation of Inflammation and Redox Signaling by Dietary Poly- phenols,” Biochemical Pharmacology, Vol. 72, No. 11, 2006, pp. 1439-1452. doi:10.1016/j.bcp.2006.07.004 [20] N. Tirkey, G. Kaur, G. Vij and K. Chopra, “Curcumin, a Diferuloylmethane, Attenuates Cyclosporine-Induced Re- nal Dysfunction and Oxidative Stress in Rat Kidneys,” BMC Pharmacology, Vol. 5, 2005, pp. 1-15. doi:10.1186/1471-2210-5-15 [21] N. Khanna, “Turmeric: Nature’s Precious Gift,” Current Science, Vol. 76, No. 10, 1999, pp. 1351-1356. doi:10.1007/s10495-006-6715-5 [22] J. Chen, X. Q. Tang, J. L. Zhi, Y. Cui, H. M. Yu, E. H. Tang, S. N. Sun, J. Q. Feng and P. X. Chen, “Curcumin Protects PC12 Cells against 1-Methyl-4-phenylpyridinium Ion-Induced Apoptosis by bcl-2-Mitochondria-ROS-iNOS Pathway,” Apoptosis, Vol. 11, No. 6, 2006, pp. 943-953. [23] L. Perez-Arriaga, M. L. Mendoza-Magana, R. Cortes-Za- rate, A. Corona-Rivera, L. Bobadilla-Morales, R. Troyo- Sanroman and M. A. Ramirez-Herrera, “Cytotoxic Effect of Curcumin on Giardia Lamblia Trophozoites,” Acta Tro- pica, Vol. 98, No. 2, 2006, pp. 152-161. [24] R. S. Ramsewak, D. L. DeWitt and M. G. Nair, “Cyto- Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 82 toxicity, Antioxidant and Anti-Inflammatory Activities of Curcumins IIII from Curcuma Longa,” Phytomedicine, Vol. 7, No. 4, 2000, pp. 303-308. doi:10.1016/S0944-7113(00)80048-3 [25] B. B. Aggarwal, C. Sundaram, N. Malani and H. Ichi- kawa, “Curcumin: The Indian Solid Gold,” Advances in Experimental Medical Biology, Vol. 595, 2007, pp. 1-75. doi:10.1007/978-0-387-46401-5_1 [26] O. Ciftci, S. Tanyildizi and A. Godekmerdan, “Protective Effect of Curcumin on Immune System and Body Weight Gain on Rats Intoxicated with 2,3,7,8-Tetrachlorodibenzo- p-dioxin (TCDD),” Immunopharmacology and Immuno- toxicology, Vol. 32, No. 1, 2010, pp. 99-104. doi:10.3109/08923970903164318 [27] G. M. Cole, B. Teter and S. A. Frautschy, “Neuroprotec- tive Effects of Curcumin,” Advances in Experimental Me- dicine and Biology, Vol. 595, 2007, pp. 197-212. doi:10.1007/978-0-387-46401-5_8 [28] A. Kuhad, S. Pilkhwal, S. Sharma, N. Tirkey and K. Chopra, “Effect of Curcumin on Inflammation and Oxida- tive Stress in Displatin-Induced Experimental Nephroto- xicity,” Journal of Agricultural and Food Chemistry, Vol. 55, No. 25, 2007, pp. 10150-10155. doi:10.1021/jf0723965 [29] O. Ciftci, I. Ozdemir, S. Tanyildizi, S. Yildiz and H. Ogu- zturk, “Antioxidative Effects of Curcumin, β-Myrcene and 1,8-Cineole against 2,3,7,8-Tetrachlorodibenzo-p-dio- xin-Induced Oxidative Stress in Rats Liver,” Toxicology and Industrial Health, Vol. 27, No. 5, 2011, pp. 447-453. doi:10.1177/0748233710388452 [30] O. Ciftci, A. Beytur, O. Cakir, N. Gurbuz and N. Vardi, “Comparison of Reproductive Toxicity Caused by Cispla- tin and Novel Platinum-N-Heterocyclic Carbine Complex in Male Rats,” Basic and Clinical Pharmacology and To- xicology, Vol. 109, No. 5, 2011, pp. 328-333. doi:10.1111/j.1742-7843.2011.00737.x [31] O. Ciftci, M. Aydin, I. Ozdemir and N. Vardi, “Quercetin Prevents 2,3,7,8-Tetrachlorodibenzo-p-dioxin-Induced Tes- ticular Damage in Rats,” Andrologia, Vol. 44, No. 3, 2012, pp. 164-173. doi:10.1111/j.1439-0272.2010.01126.x [32] O. Ciftci, I. Ozdemir, M. Aydin and A. Beytur, “Benefi- cial Effects of Chrysin on the Reproductive System of Adult Male Rats,” Andrologia, Vol. 44, No. 3, 2012, pp. 181-186. doi:10.1111/j.1439-0272.2010.01127.x [33] H. Ahsan, N. Parveen, N. U. Khan and S. M. Hadi, “Pro- Oxidant, Anti-Oxidant and Cleavage Activities on DNA of Curcumin and Its Derivatives Demethoxycurcumin and Bisdemethoxycurcumin,” Chemical and Biological Inter- action, Vol. 121, No. 2, 1999, pp. 161-175. doi:10.1016/S0009-2797(99)00096-4 [34] G. K. Jayaprakasha, L. J. Rao and K. K. Sakariah, “Anti- oxidant Activities of Curcumin, Demethoxycurcumin and Bisdemethoxycurcumin,” Food Chemistry, Vol. 98, No. 4, 2006, pp. 720-724. doi:10.1016/j.foodchem.2005.06.037 [35] P. M. Wall and G. Messier, “Methodological and Con- ceptual Issues in the Use of Elevated Plus-Maze as a Psy- chological Measurement Instrument of Animal Anxiety- Like Behavior,” Neuroscience and Biobehavior Review, Vol. 25, No. 3, 2001, pp. 275-286. doi:10.1016/S0149-7634(01)00013-6 [36] O. E. Patrick, M. Hirohisa, K. Masahira and M. Koreaki, “Central Nervous System Bioaminergic Responses to Me- chanic Trauma,” Surgical Neurology, Vol. 35, No. 4, 1991, pp. 273-279. doi:10.1016/0090-3019(91)90004-S [37] H. Ohkawa, N. Ohishi and K. Tgi, “Assay for Lipid Per- oxides in Animal Tissues by Thiobarbituric Acid Reac- tion,” Annals of Chemistry, Vol. 95, No. 2, 1979, pp. 351- 358. [38] M. J. Mangino, M. K. Murphy and G. G. Glabar, “Protec- tive Effects of Glycine during Hypothermic Renal Ische- mic Reperfusion Injury,” American Journal of Physiology, Vol. 261, No. 5, 1991, pp. F841-F848. [39] T. L. Sorell and J. H. Graziano, “Effect of Oral Cadmium Exposure during Pregnancy on Maternal and Fetal Zinc Metabolism in the Rat,” Toxicology and Applied Phar- macology, Vol. 102, No. 3, 1990, pp. 537-545. doi:10.1016/0041-008X(90)90048-Y [40] C. Claverie, R. Corbella, D. Martin and C. Diaz, “Protec- tive Effects of Zinc on Cadmium Toxicity in Rodents,” Biological Trace Element Research, Vol. 75, No. 1-3, 2000, pp. 1-9. doi:10.1385/BTER:75:1-3:1 [41] B. Baranski, “Effect of Exposure of Pregnant Rats to Ad- mium on Prenatal and Postnatal Development of the Young,” Journal of Hygiene, Epidemiology and Microbi- ology, Vol. 29, No. 3, 1984, pp. 253-262. [42] I. Desi, L. Nagymajtenyi and H. Schulz, “Behavioural and Neurotoxicological Changes Caused by Cadmium Treatment of Rats during Development,” Journal of Ap- plied Toxicology, Vol. 18, No. 1, 1998, pp. 63-70. doi:10.1002/(SICI)1099-1263(199801/02)18:1<63::AID-J AT475>3.0.CO;2-Z [43] G. Liu and J. Elsner, “Review of the Multiple Chemical Exposure Factors Which May Disturb Human Behavioral Development,” Sozial-und Praventivmedizin, Vol. 40, 1995, pp. 209-217. doi:10.1007/BF01354475 [44] L. Nagymajtenyi, H. Schulz and I. Desi, “Behavioural and Functional Neurotoxicological Change Caused by Cad- mium in a Three-Generational Study in Rats,” Human and Experimental Toxicology, Vol. 16, No. 12, 1997, pp. 691-699. doi:10.1177/096032719701601201 [45] K. Lehotzky, G. Ungvary, D. Polinak and A. Kiss, “Be- havioral Deficits Due to Prenatal Exposure to Cadmium Chloride in CFY Rat Pups,” Neurotoxicology and Tera- tology, Vol. 12, No. 2, 1990, pp. 169-172. doi:10.1016/0892-0362(90)90130-5 [46] S. Morita, “Defense Mechanisms against Cadmium Tox- icity I. A Biochemical and Histological Study of the Ef- fects of Pretratment with Cadmium on the Acute Oral Toxicity of Cadmium in Mice,” Japanese Journal of Phar- macology, Vol. 35, 1984, pp. 129-141. doi:10.1254/jjp.35.129 [47] M. Devi, D. A. Thomas, J. T. Barber and M. A. Finger- man, “Accumulation and Physiological and Biochemical Effects of Cadmium in a Simple Aquatic Food Chain,” Ecotoxicology and Environmental Saftey, Vol. 33, No. 1, 1996, pp. 38-43. doi:10.1006/eesa.1996.0004 Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. 83 [48] B. Ognjanovic, R. V. Zikic, A. Stajn, Z. S. Saicic, M. M. Kostic and V. M. Petrovic, “The Effects of Selenium on the Antioxidant Defense System in the Liver of Rats Ex- posed to Cadmium,” Physiological Research, Vol. 44, No. 5, 1995, pp. 293-300. [49] E. Casalino, C. Sblano and C. Landriscina, “Enzyme Ac- tivity Alteration by Cadmium Administration to Rats: The Possibility of Iron Involvement in Lipid Peroxidation,” Archives of Biochemistry and Biophysics, Vol. 346, No. 2, 1997, pp. 171-179. doi:10.1006/abbi.1997.0197 [50] A. Stajn, R. V. Zikic, B. Ognjanovic, Z. S. Saicic, S. Z. Pavlovic, M. M. Kostic and V. M. Petrovic, “Effect of Cadmium and Selenium on the Antioxidant Defense Sys- tem in Rat Kidneys,” Comparative Biochemistry and Phy- siology, Vol. 117C, No. 2, 1997, pp. 167-172. [51] M. M. Kostic, B. Ognjanovic, S. Dimitrijevic, R. V. Zikic, A. Stajn and G. L. Rosic, “Cadmium-Induced Changes of Antioxidant and Metabolic Status in Red Blood Cells of Rats: In Vivo Effects,” European Journal of Haematology, Vol. 51, No. 2, 1993, pp. 86-92. doi:10.1111/j.1600-0609.1993.tb01598.x [52] R. V. Zikic, A. S. Stajn, B. I. Ognjanovic, Z. S, Saicic, M. M. Kostic, S. Z. Pavloviv and V. M. Petrovic, “The Effect of Cadmium and Selenium on the Antioxidant Enzyme Activities in Rat Heart,” Journal of Environmental Patho- logy, Toxicology and Oncology, Vol. 17, No. 3-4, 1998, pp. 259-264. [53] S. Z. Pavlovic, B. I. Ognjanovic, A. S. Stajn, R. V. Zikic, Z. S. Saicic and V. M. Petrovic, “Antioxidant Defense System in Skeletal Muscle of Rats Treated with Cadmium. A Possible Protective Role of Coenzyme Q10,” Jugo- slavia Medical Biochemistry, Vol. 20, No. 4, 2001, pp. 229-235. [54] E. Casalino, C. Sblano and C. Landriscina, “Enzyme Ac- tivity Alteration by Cadmium Administration to Rats: The Possibility of Iron Involvement in Lipid Peroxidation,” Archives of Biochemistry and Biophysics, Vol. 346, No. 2, 1997, pp. 171-179. doi:10.1006/abbi.1997.0197 [55] J. G. Wilson, “Mechansims of Teratogenesis,” American Journal of Psychiatry, Vol. 136, 1973, pp. 129-132. [56] R. M. Freitas, S. M. M. Vasconcelos, F. C. F. Souza, G. S. B. Viana and M. M. F. Fonteles, “Monoamine Levels af- ter Pilocarpine-Induced Status Epilepticus in Hippocam- pus and Frontal Cortex of Wistar Rats,” Neuroscience Letters, Vol. 370, No. 2-3, 2004, pp. 196-200. doi:10.1016/j.neulet.2004.08.024 [57] V. S. Nascimento, A. A. Oliveira, R. M. Freitas, F. C. Sousa, S. M. M. Vasconcelos, G. S. B. Viana and M. M. F. Fonteles, “Pilocarpine-Induced Status Epilepticus: Mo- noamine Level, Muscarinic and Dopaminergic Receptors Alterations in Striatum of Young Rats,” Neuroscience Letters, Vol. 383, No. 1-2, 2005, pp. 165-170. doi:10.1016/j.neulet.2005.04.006 [58] V. D. Petkov, S, Belcheva, E. Konstantinova and R. Ke- hayov, “Participation of Different 5-HT Receptors in the Memory Process in Rats and Its Modulation by the Sero- tonin Depletor p-Chlorophenylalanine,” Acta Neurobio- logiae Experimentalis (Wars), Vol. 55, 1995, pp. 243- 252. [59] G. M. Abu-Taweel, J. S. Ajarem and M. Ahmad, “Neuro- behavioral Toxic Effects of Perinatal Oral Exposure to Aluminum on the Developmental Motor Reflexes, Learn- ing, Memory and Brain Neurotransmitters of Mice Off- spring,” Pharmacology Biochemistry and Behavior, Vol. 101, No. 1, 2012, pp. 49-56. doi:10.1016/j.pbb.2011.11.003 [60] G. Richter-Levin and M. Segal, “The Effects of Serotonin Depletion and Raphe Grafts on Hippocampal Electrophy- siology and Behavior,” The Journal of Neuroscience, Vol. 11, No. 6, 1991, pp. 1585-1596. [61] I. S. Jamall and J. J. Sprowls, “Effects of Cadmium and Dietary Selenium on Cytoplasmic and Mitochondrial An- tioxidant Defense Systems in the Heart of Rats Fed High Dietary Copper,” Toxicology and Applied Pharmacology, Vol. 87, No. 1, 1987, pp. 102-110. doi:10.1016/0041-008X(87)90088-3 [62] S. Sarkar, P. Yadav and D. Bhatnagar, “Lipid Peroxida- tive Damage on Cadmium Exposure and Alterations in Antioxidant System in Rat Erythrocytes: A Study with Relation to Time,” Biometals, Vol. 11, No. 2, 1998, pp. 153-157. doi:10.1023/A:1009286130324 [63] E. Casalino, G. Valzaretti, C. Sblano, V. Landriscina, M. Felice Tesse and C. Landriscina, “Antioxidant Effect of Hydroxytyrosol (DPE) and Mn2+ in Liver of Cadmium- Intoxicated Rats,” Comparative Biochemistry and Physi- ology, Vol. C133, No. 4, 2002, pp. 625-632. [64] B. Ognjanovic, S. Z. Pavlovic, S. D. Maletic, R. V. Žikic, A. Štajn, R. M. Radojicic, Z. S. Saicic and V. M. Petrovic, “Protective Influence of Vitamin E on Antioxidant De- fense System in the Blood of Rats Treated with Cad- mium,” Physiological Research, Vol. 52, No. 5, 2003, pp. 563-570. [65] Z. A. Shaikh, T. T. Vu and K. Zaman, “Oxidative Stress as a Mechanism of Chronic Cadmium-Induced Hepato- toxicity and Renal Toxicity and Protection by Antioxi- dants,” Toxicology and Applied Pharmacology, Vol. 154, No. 3, 1999, pp. 256-263. doi:10.1006/taap.1998.8586 [66] M. Waisberg, P. Joseph, B. Hale and D. Beyersmann, “Molecular and Cellular Mechanisms of Cadmium Car- cinogenesis: A Review,” Toxicology, Vol. 192, No. 2-3, 2003, pp. 95-117. doi:10.1016/S0300-483X(03)00305-6 [67] S. J. Stohs, D. Bagchi, E. Hassoun and M. Bagchi, “Oxi- dative Mechanisms in the Toxicity of Chromium and Cad- mium Ions,” Journal of Environmental Pathology, Toxi- cology and Oncology, Vol. 20, No. 2, 2001, pp. 77-88. doi:10.1615/JEnvironPatholToxicolOncol.v20.i2.10 [68] V. Calabrese, D. A. Butterfield and A. M. Stella, “Nutri- tional Antioxidants and the Heme Oxygenase Pathway of Stress Tolerance: Novel Targets for Neuroprotection in Alzheimer’s Disease,” Italian Journal of Biochemistry, Vol. 52, No. 4, 2003, pp. 177-181. [69] K. Kitani, T. Yokozawa and T. Osawa, “Interventions in Aging and Age-Associated Pathologies by Means of Nu- tritional Approaches,” Annals of New York Academy of Sciences, Vol. 1019, No. 1, 2004, pp. 424-426. doi:10.1196/annals.1297.075 [70] R. Motterlini, R. Foresti, R. Bassi and C. J. Green, “Cur- cumin, an Antioxidant and Anti-Inflammatory Agent, In- Copyright © 2013 SciRes. JBBS  G. M. ABU-TAWEEL ET AL. Copyright © 2013 SciRes. JBBS 84 duces Heme Oxygenase-1 and Protects Endothelial Cells against Oxidative Stress,” Free Radical Biology and Me- dicine, Vol. 28, 2000, pp. 1303-1312. doi:10.1016/S0891-5849(00)00294-X [71] A. Ray, “Cancer Preventive Role of Selected Dietary Fac- tors,” Indian Journal of Cancer, Vol. 42, No. 1, 2005, pp. 11-20. doi:10.4103/0019-509X.15095 [72] A. I. Ghoneim, A. B. Abdel-Naim, A. E. Khalifa and E. S. El-Denshary, “Protective Effects of Curcumin against Is- chaemia/Reperfusion Insult in Rat Forebrain,” Pharma- cological Research, Vol. 46, No. 3, 2002, pp. 273-279. doi:10.1016/S1043-6618(02)00123-8 [73] M. Thiyagarajan and S. S. Sharma, “Neuroprotective Ef- fect of Curcumin in Middle Cerebral Artery Occlusion Induced Focal Cerebral Ischemia in Rats,” Life Sciences, Vol. 74, No. 8, 2004, pp. 969-985. doi:10.1016/j.lfs.2003.06.042 [74] V. Rajakrishnan, P. Viswanathan, K. N. Rajasekharan and V. P. Menon, “Neuroprotective Role of Curcumin from Curcuma Longa on Ethanolinduced Brain Damage,” Phy- totherapy Research, Vol. 13, No. 7, 1999, pp. 571-574. doi:10.1002/(SICI)1099-1573(199911)13:7<571::AID-PT R494>3.0.CO;2-7 [75] G. P. Lim, T. Chu, F. Yang, W. Beech, S. A. Frautschy and G. M. Cole, “The Curry Spice Curcumin Reduces Oxi- dative Damage and Amyloid Pathology in an Alzheimer Transgenic Mouse,” Journal of Neuroscience, Vol. 21, No. 21, 2001, pp. 8370-8377. [76] S. J. Stohs and D. Bagchi, “Oxidative Mechanisms in the Toxicity of Metal Ions,” Free Radical Biology and Medi- cine, Vol. 18, No. 2, 1995, pp. 321-336. doi:10.1016/0891-5849(94)00159-H [77] R. Anane and E. E. Creppy, “Lipid Peroxidation as Path- way of Aluminium Cytotoxicity in Human Skin Fibro- blast Cultures: Prevention by Superoxide Dismutase and Catalase and Vitamins E and C,” Human Experimental Toxicology, Vol. 20, No. 9, 2001, pp. 477-481. doi:10.1191/096032701682693053 [78] P. O’brien and H. J. Salasinski, “Evidence That the Reac- tions of Cadmium in the Presence of Metallothionein Can Produce Hydroxyl Radicals,” Archives of Toxicology, Vol. 72, No. 11, 1998, pp. 690-700. doi:10.1007/s002040050562 [79] T. Koizumi, G. Shirakura, H. Kumagai, H. Tatsumoto and K. T. Suzuki, “Mechanism of Cadmium-Induced Cytotox- icity in Rat Hepatocytes: Cadmium-Induced Active Oxy- gen-Related Permeability Changes of the Plasma Mem- brane,” Toxicology, Vol. 114, 1996, pp. 124-134. doi:10.1016/S0300-483X(96)03477-4 [80] S. K. Tandon, S. Singh, S. Prasad, K. Khandekar, V. K. Dwivedi, M. Chatterjee and N. Mathur, “Reversal of Cad- mium Induced Oxidative Stress by Chelating Agent, An- tioxidant or Their Combination in Rat,” Toxicology Let- ters, Vol. 145, No. 3, 2003, pp. 211-217. doi:10.1016/S0378-4274(03)00265-0 [81] B. Ognjanovic, S. D. Markovic, S. Z. Pavlovic, R. V. Žikic, A. Štajn and Z. S. Saicic, “Combined Effects of Coenzyme Q10 and Vitamin E in Cadmium Induced Al- terations of Antioxidant Defense System in the Rat Heart,” Environmental Toxicology and Pharmacology, Vol. 22, 2006, pp. 219-224. doi:10.1016/j.etap.2006.03.008 [82] B. Halliwell and J. M. C. Gutteridge, “Free Radicals in Biology and Medicine,” 3rd Edition, Oxford University Press, New York, 1999. [83] M. Mates, “Effects of Antioxidant Enzymes in the Mo- lecular Control of Reactive Oxygen Species Toxicology,” Toxicology, Vol. 153, No. 1, 2000, pp. 83-104. doi:10.1016/S0300-483X(00)00306-1 [84] O. W. Griffith, “Biological and Pharmacologic Regula- tion of Mammalian Glutathione Synthesis,” Free Radical Biology and Medicine, Vol. 27, No. 9-10, 1999, pp. 922- 935. doi:10.1016/S0891-5849(99)00176-8 [85] R. E. Beyer, “The Role of Ascorbate in Antioxidant Pro- tection of Biomolecules: Interaction with Vitamin E and Coenzyme Q,” Journal of Bioenergy and Biomembrane, Vol. 26, No. 4, 1994, pp. 349-358. doi:10.1007/BF00762775
|