Paper Menu >>
Journal Menu >>
 Vol.2, No.10, 1156-1162 (2010) Health doi:10.4236/health.2010.210169 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ Slow movement resistance training in women Shunsuke Yamaji1*, Shinichi Demura2, Narihiro Watanabe3, Masanobu Uchiyama4 1Faculty of Medical Sciences, University of Fukui, Fukui, Japan; *Corresponding Author: yamaji@u-fukui.ac.jp 2Graduate School of Natural Science & Technology, Kanazawa University, Ishikawa, Japan 3hiroNARI, Fukui, Japan 4Research and Education Center for Comprehensive Science, Akita Prefectural University, Akita, Japan Received 6 July 2010; revised 13 July 2010; accepted 21 July 2010. ABSTRACT A resistance training protocol of low intensity and short duration allows for increased training frequency and improved compliance. This study aimed to examine the short-term (response of growth hormone (GH) and testosterone after one exercise session) and long-term (change of fitness level and body fat percentage after the exercise period) effects of slow movement resi- stance training using the individual’s body mass (hiroNARI style training) in adult women and to clarify their subjective sense of training contin- uity. Nineteen healthy adult women performed hiroNARI style training three times a week for 12 weeks. This protocol consisted of 12 types of exercise for 7 muscle groups. GH and testos- terone increased significantly after one exercise session (70% and 23.3%, p < 0.05, respectively). Height and the circumferences of the upper arm (flexed), chest, waist, hip, and thigh changed si- gnificantly. Except for the upper arm circumfe- rence, these parameters improved significantly after 6 weeks. There were significant improvem- ents in measurements of physical fitness after 6 weeks including one leg raise with eyes closed, side step, and repeated sit ups for 30 s. After 12 weeks, anteflexion from a long sitting position improved as well. Triglycerides and HDL and LDL cholesterol changed significantly after 12 weeks. In conclusion, resistance training may have po- sitive effects and is associated with high comp- liance. However, it will be necessary to reexam- ine the training protocol for increasing back and lower limb muscle strength and necessary vari- ations to prevent overtraining of certain muscle groups. Keywords: Resistance Training; Growth Hormone; Testosterone; Physical Fitness 1. INTRODUCTION Resistance training improves the musculoskeletal system, maintains various physical functions and prevents oste- oporosis and sarcopenia [1,2]. Recently, researchers rep- orted that resistance training may positively affect risk factors such as insulin resistance [3], resting metabolic rate [4], glucose metabolism [5], blood pressure [6], and body fat [7], which are associated with diabetes, heart disease [8], and cancer [9]. In order to gain the above effects, a basic resistance pr- otocol consisting of two or three sets of three to 15 repe- titions using 60 to 80% 1RM (repetition maximum) must be performed at a frequency of two to three times a week [10-12]. However, for inexperienced individuals or those with time constraints, it is difficult to maintain the above protocol. Because the effects of exercise and physical ac- tivity, including resistance training, can only occur after adherence to a protocol for a certain period of time, pri- ority should be given to the design of exercise protocols that are amenable to long-term compliance. The goals of resistance training for the majority of mi- ddle-aged people are to maintain and enhance good hea- lth and to improve body proportion rather than hypertr- ophy and improvement of muscle function. The above benefits can be acquired with a safe, low intensity train- ing protocol [10]. Furthermore, training frequency may be a more important factor than intensity and repetition of exercise [13]. Training for only one day each week produces slight improvement of strength and hypertro- phy outcomes, but its benefit on other risk factors related to the health-related disease and obesity remains unclear [2,13]. An adequate resistance training protocol of limited du- ration contributes to increased training frequency and improved compliance [10]. The total duration of aerobic exercise is determined by perceived exertion (RPE) or heart rate. However, that of resistance training can not be easily determined because it depends on a combina- tion of repetition, number of sets, interval time, and trai-  S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 115 1157 ning region, using the specific rate of one repetition ma- ximum (1RM) as intensity [14]. Moreover, the training protocol (intensity, repetition, and number of sets) must be carefully designed because delayed onset muscle sor- eness caused by resistance training hinders exercise con- tinuity. Light training of low intensity decreases physical distress but does not yield dramatic results. Tanimoto and Ishii [15] reported that low intensity, less than 50% 1RM, exercise with slow movement (3-4 sec in concentric and eccentric contraction) resulted in si- milar improvements in muscle mass and strength as high intensity (80% 1RM) training with normal speed move- ments. This suggested that slow movements produce an ischemia caused by increased intramuscular pressure and accelerated secretion of growth hormone (GH) and insu- lin like growth factor I (IGF-I). On the other hand, stabi- lization exercises, which are commonly used for patients with low back pain in rehabilitation settings, consist of slow concentric and eccentric contractions using the ind- ividual’s body weight or manual loads for 3-4 sec and is- ometric contraction for 5 sec (posing phase) [16]. Stev- ens et al. [17] reported that performance of the above ex- ercises for 8 weeks increased muscle activation volume and muscle fiber recruitment. Short duration resistance training with slow movements using the individual’s bo- dy weight as a load provides physical benefits and is as- sociated with improved compliance. This study aimed to examine the short-term (response of GH and testosterone after one exercise session) and long-term effects (change of anthropometric characteris- tics, fitness level and serum chemistry value after the ex- ercise period) of resistance training with slow movem- ents using the individual’s body weight (hiroNARI style training) in adult women. It also sought to clarify their subjective sense of training continuity. 2. METHOD 2.1. Subjects Subjects included 19 healthy adult women who did not have emmeniopathy or regular participation in any form of resistance training and exercise (Age: 33.4 ± 10.8 yr, range = 20 – 48 yr). All subjects completed a health que- stionnaire. All subjects rated their health as “good” or “fairly good” except for one person (“not too good”). Four subjects rated their physical strength as “average” and fifteen answered “inferior”. After explaining the aim and method of this study, all subjects provided informed consent. The Ethics and Research Committee of the Fa- culty of Medical Sciences, University of Fukui, ap- proved this study. 2.2. Design The objective of this study was to examine the short-te- rm effects of slow movement training (hiroNARI style training) on GH and testosterone after one exercise ses- sion, and the long-term effects on anthropometric char- acteristics, physical fitness and serum chemistry after 12 weeks. For these reasons, healthy adult women perfor- med hiroNARI style training three times a week for 12 weeks. The above parameters were measured at the be- ginning of the training program and after 6 and 12 weeks of training. 2.3. Methodology Table 1 shows a schema of slow movement resistance using the subject’s own body mass (hiroNARI’s method). Subjects performed 12 exercises for the following 7 mu- scle groups: abdominal (three forms), femoral and glu- teal (two forms), back (one form), pectoral (one form), brachial (two forms), shoulder (two forms), and lower th- igh (one form). Oblique curls and lateral leg raises were performed bilaterally. The exercise session consisted of one set of 6 repetitions of each exercise and was per- formed three times a week for 12 weeks. The subjects performed a slow movement consisting of tonic force generation without relaxation as follows: 3 s concentric action, 6 s pause, 2 s concentric action, and 3 s eccentric action (Figure 1). The inter-rest period between each exercise and the total exercise time were 15s and approximately 23 min, respectively. The subjects perfor- med the exercise according to the instructor’s perform- ance which was projected by DVD on a screen. The short term effects of the exercise were examined by the levels of GH and testosterone in serum samples after one exercise session. Blood (6 ml) was drawn from the antecubital vein before exercise and at 15 min after exercise. Subjects ate nothing for 4 h and refrained from ingesting alcohol and caffeine for 24 h before sampling. Table 1. Slow motion resistance training. (hiroNARI’s method) Repetition 6 times Set 1 set Interval 15 s Training formsAbdominal Curls Oblique Curls Dual Torso Curls Modified Squats Lateral Leg Raises Hip Adduction Back Extension Modified Push Ups Back Row Shoulder Press Bicep Flexion Tricep Extension Calf Raises and Face 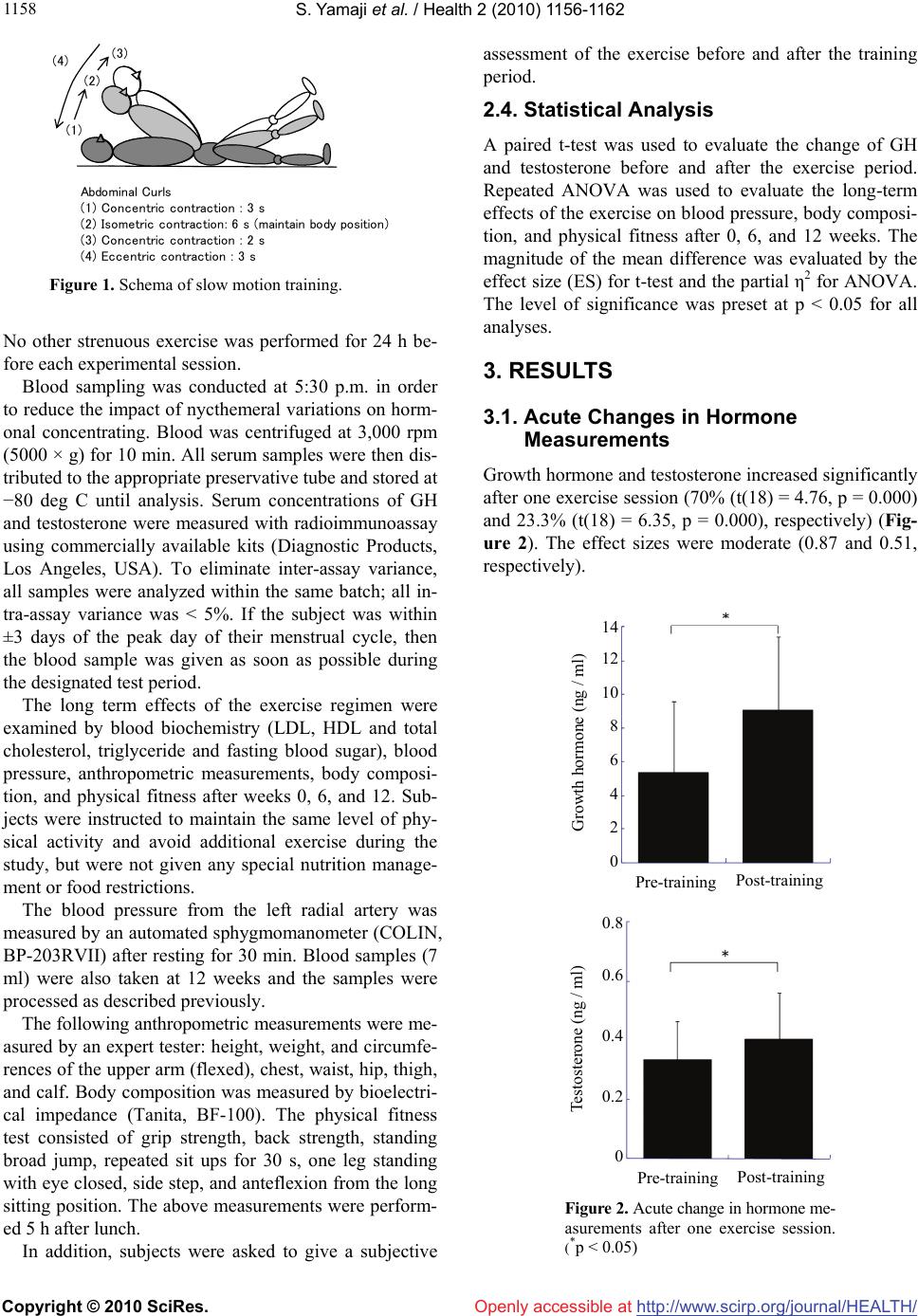 S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1158 Abdominal Curls (1) Concentric contraction : 3 s (2) Isometric contraction: 6 s (maintain body position) (3) Concentric contraction : 2 s (4) Eccentric contraction : 3 s (1) (2) (3) (4) Figure 1. Schema of slow motion training. No other strenuous exercise was performed for 24 h be- fore each experimental session. Blood sampling was conducted at 5:30 p.m. in order to reduce the impact of nycthemeral variations on horm- onal concentrating. Blood was centrifuged at 3,000 rpm (5000 × g) for 10 min. All serum samples were then dis- tributed to the appropriate preservative tube and stored at −80 deg C until analysis. Serum concentrations of GH and testosterone were measured with radioimmunoassay using commercially available kits (Diagnostic Products, Los Angeles, USA). To eliminate inter-assay variance, all samples were analyzed within the same batch; all in- tra-assay variance was < 5%. If the subject was within ±3 days of the peak day of their menstrual cycle, then the blood sample was given as soon as possible during the designated test period. The long term effects of the exercise regimen were examined by blood biochemistry (LDL, HDL and total cholesterol, triglyceride and fasting blood sugar), blood pressure, anthropometric measurements, body composi- tion, and physical fitness after weeks 0, 6, and 12. Sub- jects were instructed to maintain the same level of phy- sical activity and avoid additional exercise during the study, but were not given any special nutrition manage- ment or food restrictions. The blood pressure from the left radial artery was measured by an automated sphygmomanometer (COLIN, BP-203RVII) after resting for 30 min. Blood samples (7 ml) were also taken at 12 weeks and the samples were processed as described previously. The following anthropometric measurements were me- asured by an expert tester: height, weight, and circumfe- rences of the upper arm (flexed), chest, waist, hip, thigh, and calf. Body composition was measured by bioelectri- cal impedance (Tanita, BF-100). The physical fitness test consisted of grip strength, back strength, standing broad jump, repeated sit ups for 30 s, one leg standing with eye closed, side step, and anteflexion from the long sitting position. The above measurements were perform- ed 5 h after lunch. In addition, subjects were asked to give a subjective assessment of the exercise before and after the training period. 2.4. Statistical Analysis A paired t-test was used to evaluate the change of GH and testosterone before and after the exercise period. Repeated ANOVA was used to evaluate the long-term effects of the exercise on blood pressure, body composi- tion, and physical fitness after 0, 6, and 12 weeks. The magnitude of the mean difference was evaluated by the effect size (ES) for t-test and the partial η2 for ANOVA. The level of significance was preset at p < 0.05 for all analyses. 3. RESULTS 3.1. Acute Changes in Hormone Measurements Growth hormone and testosterone increased significantly after one exercise session (70% (t(18) = 4.76, p = 0.000) and 23.3% (t(18) = 6.35, p = 0.000), respectively) (Fig- ure 2). The effect sizes were moderate (0.87 and 0.51, respectively). 14 12 10 8 6 4 2 0 Growth hormone (ng / ml) Testosterone (ng / ml) Pre-training Post-training 0.8 0.6 0.4 0.2 0 Pre-trainin g Post-training Figure 2. Acute change in hormone me- asurements after one exercise session. (*p < 0.05) 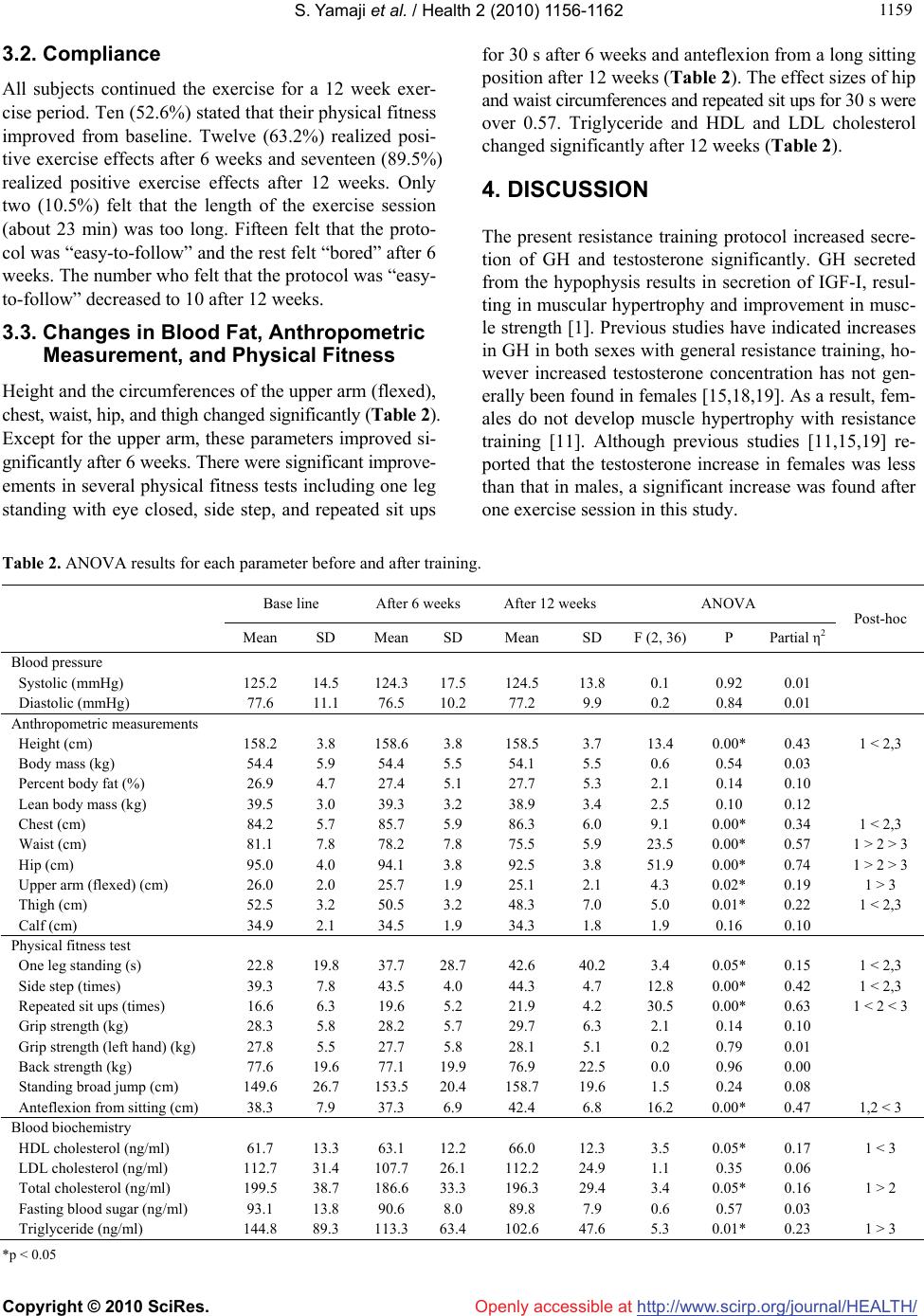 S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 115 1159 3.2. Compliance All subjects continued the exercise for a 12 week exer- cise period. Ten (52.6%) stated that their physical fitness improved from baseline. Twelve (63.2%) realized posi- tive exercise effects after 6 weeks and seventeen (89.5%) realized positive exercise effects after 12 weeks. Only two (10.5%) felt that the length of the exercise session (about 23 min) was too long. Fifteen felt that the proto- col was “easy-to-follow” and the rest felt “bored” after 6 weeks. The number who felt that the protocol was “easy- to-follow” decreased to 10 after 12 weeks. 3.3. Changes in Blood Fat, Anthropometric Measurement, and Physical Fitness Height and the circumferences of the upper arm (flexed), chest, waist, hip, and thigh changed significantly (Table 2). Except for the upper arm, these parameters improved si- gnificantly after 6 weeks. There were significant improve- ements in several physical fitness tests including one leg standing with eye closed, side step, and repeated sit ups for 30 s after 6 weeks and anteflexion from a long sitting position after 12 weeks (Table 2). The effect sizes of hip and waist circumferences and repeated sit ups for 30 s were over 0.57. Triglyceride and HDL and LDL cholesterol changed significantly after 12 weeks (Table 2). 4. DISCUSSION The present resistance training protocol increased secre- tion of GH and testosterone significantly. GH secreted from the hypophysis results in secretion of IGF-I, resul- ting in muscular hypertrophy and improvement in musc- le strength [1]. Previous studies have indicated increases in GH in both sexes with general resistance training, ho- wever increased testosterone concentration has not gen- erally been found in females [15,18,19]. As a result, fem- ales do not develop muscle hypertrophy with resistance training [11]. Although previous studies [11,15,19] re- ported that the testosterone increase in females was less than that in males, a significant increase was found after one exercise session in this study. Table 2. ANOVA results for each parameter before and after training. Base line After 6 weeks After 12 weeks ANOVA Mean SD Mean SD Mean SD F (2, 36)P Partial η2 Post-hoc Blood pressure Systolic (mmHg) 125.2 14.5 124.3 17.5124.5 13.8 0.1 0.92 0.01 Diastolic (mmHg) 77.6 11.1 76.5 10.277.2 9.9 0.2 0.84 0.01 Anthropometric measurements Height (cm) 158.2 3.8 158.6 3.8 158.5 3.7 13.4 0.00* 0.43 1 < 2,3 Body mass (kg) 54.4 5.9 54.4 5.5 54.1 5.5 0.6 0.54 0.03 Percent body fat (%) 26.9 4.7 27.4 5.1 27.7 5.3 2.1 0.14 0.10 Lean body mass (kg) 39.5 3.0 39.3 3.2 38.9 3.4 2.5 0.10 0.12 Chest (cm) 84.2 5.7 85.7 5.9 86.3 6.0 9.1 0.00* 0.34 1 < 2,3 Waist (cm) 81.1 7.8 78.2 7.8 75.5 5.9 23.5 0.00* 0.57 1 > 2 > 3 Hip (cm) 95.0 4.0 94.1 3.8 92.5 3.8 51.9 0.00* 0.74 1 > 2 > 3 Upper arm (flexed) (cm) 26.0 2.0 25.7 1.9 25.1 2.1 4.3 0.02* 0.19 1 > 3 Thigh (cm) 52.5 3.2 50.5 3.2 48.3 7.0 5.0 0.01* 0.22 1 < 2,3 Calf (cm) 34.9 2.1 34.5 1.9 34.3 1.8 1.9 0.16 0.10 Physical fitness test One leg standing (s) 22.8 19.8 37.7 28.742.6 40.2 3.4 0.05* 0.15 1 < 2,3 Side step (times) 39.3 7.8 43.5 4.0 44.3 4.7 12.8 0.00* 0.42 1 < 2,3 Repeated sit ups (times) 16.6 6.3 19.6 5.2 21.9 4.2 30.5 0.00* 0.63 1 < 2 < 3 Grip strength (kg) 28.3 5.8 28.2 5.7 29.7 6.3 2.1 0.14 0.10 Grip strength (left hand) (kg) 27.8 5.5 27.7 5.8 28.1 5.1 0.2 0.79 0.01 Back strength (kg) 77.6 19.6 77.1 19.976.9 22.5 0.0 0.96 0.00 Standing broad jump (cm) 149.6 26.7 153.5 20.4158.7 19.6 1.5 0.24 0.08 Anteflexion from sitting (cm) 38.3 7.9 37.3 6.9 42.4 6.8 16.2 0.00* 0.47 1,2 < 3 Blood biochemistry HDL cholesterol (ng/ml) 61.7 13.3 63.1 12.266.0 12.3 3.5 0.05* 0.17 1 < 3 LDL cholesterol (ng/ml) 112.7 31.4 107.7 26.1112.2 24.9 1.1 0.35 0.06 Total cholesterol (ng/ml) 199.5 38.7 186.6 33.3196.3 29.4 3.4 0.05* 0.16 1 > 2 Fasting blood sugar (ng/ml) 93.1 13.8 90.6 8.0 89.8 7.9 0.6 0.57 0.03 Triglyceride (ng/ml) 144.8 89.3 113.3 63.4102.6 47.6 5.3 0.01* 0.23 1 > 3 *p < 0.05  S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1160 The degree to which hormone response contributes to muscle hypertrophy and muscle strength remains unclear. Hence, the increased hormone level in this study must be compared to the secretory volume before and after gen- eral resistance training [1,2,20,21]. However, these hor- mones are largely affected by individual differences inc- luding age and trainability [15], which tended to be large in this study. Furthermore, using the individual’s weight for intensity may have also affected the above. Further study with an electromyogram will be necessary to dete- rmine whether the present training intensity corresponds to a range of relative intensity level (% 1RM). The effects of resistance training on weight and body fat are unclear. The consumption energy for resistance training was reported to be 4-10 kcal/min even if the protocol involved use of major muscle groups without rests between each training item [14]. This consumption energy is less than that of aerobic exercise with moder- ate intensity [22-24]. Body weight and fat decreased sli- ghtly after 12 weeks. Previous studies reported that res- istance training increases basal metabolism by increasing muscle volume, thereby decreasing body weight and fat [14]. However, the 12 week training did not affect these measurements. Height significantly increased at 6 weeks, even thou- ght the subjects were adult women. Although height has a circadian variation of ±2 cm [25,26], this was not felt to play a role in these findings as height was measured at the same time of day (17:30). The observed height incre- ase may be the result of improved standing posture and antigravity muscle strength (i.e., abdominal and back mu- scles) and decreased spine shrinkage. As stated above, the waist, hip, flexed upper arm, and femur circumferences significantly decreased despite ma- intenance of body weight and fat. Particularly, the de- crease in waist and hip circumference tended to be large (mean decrease in volume: 5.6 cm and 2.5 cm, ES: 0.57 and 0.74, respectively). Waist circumference is deter- mined mainly by the entrails, visceral and subcutaneous fat, and muscles. As body fat was unchanged by training, the decrease of waist circumference may be explained by an increase of abdominal pressure based on the im- provement of abdominal muscles. On the other hand, the decrease of hip circumference may be explained by improvement in the subcutaneous fat near the gluteal muscles. However, this is speculation as this study did not measure changes in visceral and su- bcutaneous fat. In general, there have been reports that exercise causes a selective decrease in subcutaneous fat of the region stimulated by exercise [27]. Hence, it may be valid to interpret the present results as improvements of body proportion, rather than decreases of body cir- cumference. The performances on the physical fitness test at base- line were below same age standard values. All measure- ements except for grip and back strength and standing broad jump improved significantly after the exercise period. In particular, the improvement of sit ups for 30 s was the largest (partial η2 = 0.63). This test requires mu- scle endurance and strength. This observation is likely the result of the greater proportion of abdominal exer- cises in the exercise protocol (3 of 12 forms). On the other hand, back strength did not improve sig- nificantly. This may indicate that the training protocol did not focus on the back muscles adequately. In short, back muscle exercises consist of a small range of motion, involving repeated deflection from lying flat on the sto- mach. Thus when compared to abdominal muscle exer- cises, the exercise stimulus may not be sufficient to cre- ate satisfactory results. These back muscle exercises will need to be revised and reexamined in the future. Lower limb exercises included modified squat, lateral leg raise hip adduction, and calf raise. Performance of side steps and one-leg standing with closed eyes impro- ved significantly after the exercise period, but the stand- ing broad jump did not. These results may be the result of differences in muscle exertion. The former exercises require continuous and repeated exertion, but the latter requires maximal explosive exertion. Westcott et al. [28] reported that slow repetitions of low intensity (about 50% 1RM) may result in greater strength outcomes than faster repetitions. Tanimoto and Ishii [15] reported that slow movements (3-4 sec in concentric and eccentric co- ntractions with low intensity less than 50% 1RM) resul- ted in the same improvements in muscle hypertrophy and muscle strength as high intensity exercise (80% 1RM). However, the improvement of maximal strength and leg power observed with slow movement resistance training may be lower than that in previous studies. The effect of exercise intensity on leg muscle groups should be reex- amined in the future. Cholesterol and triglyceride levels improved significa- ntly after the exercise period. Thus, the present exercise protocol will contribute to the prevention of life-related disease such as arteriosclerosis. In particular, subjects who were initially diagnosed with metabolic syndrome (1 subject) and its reserve (8 subjects) no longer met the diagnostic criteria at 12 weeks. In addition, in spite of lack of a statistical change in blood pressure, all cases of hypertension (systolic BP > 130 mmHg or diastolic BP > 85 mmHg) decreased to normal levels after 12 weeks. Previous studies have consistently reported that resis- tance training significantly reduces (~ 3 mmHg, systolic and diastolic) blood pressure [8]. Further studies may be needed that focus on hypertensive populations. The improvement in cholesterol and triglyceride lev-  S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 116 1161 els appeared after 12 weeks, after the neuromuscular be- nefits. Due to their close relationship with visceral fat volumes, these levels, and body fat, may decrease with further training. All subjects realized an increase in subjective physical fitness and a positive training effect. When a person with inexperienced training begins continual exercise, drop- out commonly appears at approximately three to 4 weeks [29]. Fifteen subjects felt that this protocol was “easy-to- follow” after 6 weeks, but this number decreased to ten after 12 weeks. As the remaining respondents noted that they were “bored” by the protocol, it may need to be varied to prevent weariness. 5. CONCLUSIONS Slow movement resistance training using the individu- al’s body mass increased the concentration of growth ho- rmone and testosterone after one exercise session. This training also improved physical fitness and body circu- mference when continued three times a week for 12 we- eks. The exercise protocol may have high levels of com- pliance as compared with the typical resistance protocol. However, the training protocol should be reexamined to increase back and lower limb muscle strength and pre- vent boredom from lack of variation. REFERENCES [1] Nelson, M.E., Fiatarone, M.A., Morganti, C.M., Trice, I., Greenberg, R.A. and Evans, W. (1994) Effects of high- intensity strength training on multiple risk factors for os- teoporotic fractures. A randomized controlled trial. Journal of the American Medical Association, 272(24), 1909-1914. [2] Winett, R.A. and Carpinelli, R.N. (2001) Potential health-related benefits of resistance training. Preventive Medicine, 33(5), 503-513. [3] Hurley, B.F. and Roth, S.M. (2000) Strength training in the elderly: Effects on risk factors for age-related dis- eases. Sports Medicine, 30(4), 249-268. [4] Ryan, A.S., Pratley, R.E., Elahi, D. and Goldberg, A.P. (1995) Resistive training increases fat-free mass and maintains RMR despite weight loss in postmenopausal women. Journal of Applied Physiology, 79(3), 818-823. [5] Boulé, N.G., Haddad, E., Kenny, G.P., Wells, G.A. and Sigal, R.J. (2001) Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: A meta- anal- ysis of controlled clinical trials. Journal of the American Medical Association, 286(10), 1218-1227. [6] Kelley, G.A. and Kelley, K.S. (2000) Progressive resis- tance exercise and resting blood pressure: A meta-analy- sis of randomized controlled trials. Hypertension, 35(3), 838-843. [7] Hurley, B.F., Hagberg, J.M., Goldberg, A.P., Seals, D.R., Ehsani, A.A., Brennan, R.E. and Holloszy, J.O. (1988) Resistive training can reduce coronary risk factors with- out altering VO2max or percent body fat. Medicine and Science in Sports and Exercise, 20(2), 150-154. [8] Martel, G.F., Hurlbut, D.E., Lott, M.E., Lemmer, J.T., Ivey, F.M., Roth, S.M., Rogers, M.A., Fleg, J.L. and Hurley, B.F. (1999) Strength training normalizes blood pressure in 65- to 73-year-old men and women with high normal blood pressure. Journal of the American Geriat- rics Society, 478(10), 1215-1221. [9] Koffler, K.H., Menkes, A., Redmand, R.A., Whitehead, W.E., Pratley, R.E. and Hurley, B.F. (1992) Strength training accelerates gastrointestinal transit in middle- aged and older men. Medicine and Science in Sports and Exercise, 24(2), 415-419. [10] Feigenbaum, M.S. and Pollock, M.L. (1999) Prescription of resistance training for health and disease. Medicine and Science in Sports and Exercise, 31(1), 38-45. [11] Kraemer, W.J. and Ratamess, N.A. (2005) Hormonal responses and adaptations to resistance exercise and training. Sports Medicine, 35(4), 339-361. [12] McDonagh, M.J. and Davies, C.T. (1984) Adaptive re- sponse of mammalian skeletal muscle to exercise with high loads. European Journal of Applied Physiology and Occupational Physiology, 52(2), 139-155. [13] Hass, C.J., Feigenbaum M.S. and Franklin, B.A. (2001) Prescription of resistance training for healthy populations. Sports Medicine, 31(14), 953-964. [14] American College of Sports Medicine, (2006) ACSM’s guidelines for exercise testing and prescription. 6th Edi- tion, Williams Wilkins, Lippincott. [15] Tanimoto, M. and Ishii, N. (2006) Effects of low-inten- sity resistance exercise with slow movement and tonic force generation on muscular function in young men. Journal of Applied Physiology, 100(4), 1150-1157. [16] Stevens, V.K., Vleeming, A., Bouche, K.G., Mahieu, N.M., Vanderstraeten, G.G. and Danneels, L.A. (2007) Electromyographic activity of trunk and hip muscles during stabilization exercises in four-point kneeling in healthy volunteers. European Spine Journal, 16(5), 711- 718. [17] Stevens, V.K., Coorevits, P.L., Bouche, K.G., Mahieu, N.N., Vanderstraeten, G.G. and Danneels, L.A. (2007) The influence of specific training on trunk muscle re- cruitment patterns in healthy subjects during stabilization exercises. Manual Therapy, 12(3), 271-279. [18] Kraemer, W.J., Gordon, S.E., Fleck, S.J., Marchitelli, L.J., Mello, R., Dziados, J.E., Friedl, K., Harman, E., Maresh, C. and Fry, A.C. (1991) Endogenous anabolic hormonal and growth factor responses to heavy resis- tance exercise in males and females. International Jour- nal of Sports Medicine, 12(2), 228-235. [19] Weiss, L.W., Cureton, K.J. and Thompson, F.N. (1983) Comparison of serum testosterone and androstenedione responses to weight lifting in men and women. European Journal of Applied Physiology and Occupational Physi- ology, 50(3), 413-419. [20] Aizawa, K., Akimoto, T., Inoue, H., Kimura, F., Joo, M., Murai, F. and Mesaki, N. (2003) Resting serum dehy- droepiandrosterone sulfate level increases after 8-week resistance training among young females. European Journal of Applied Physiology and Occupational Physi- ology, 90(5-6), 575-580. [21] Raastad, T., Bjøro, T. and Hallén, J. (2000) Hormonal  S. Yamaji et al. / Health 2 (2010) 1156-1162 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1162 responses to high- and moderate-intensity strength exer- cise. European Journal of Applied Physiology and Oc- cupational Physiology, 82(1-2), 121-128. [22] Beckham, S.G. and Earnest, C.P. (2000) Metabolic cost of free weight circuit weight training. The Journal of Sports Medicine and Physical Fitness, 40(2), 118-125. [23] Phillips, W.T. and Ziuraitis, J.R. (2003) Energy cost of the ACSM single-set resistance training protocol. Jour- nal of Strength and Conditioning Research, 17(2), 350- 355. [24] Wilmore, J.H., Parr, R.B., Ward, P., Vodak, P.A., Bar- stow, T.J., Pipes, T.V., Grimditch, G. and Leslie, P. (1978) Energy cost of circuit weight training. Medicine and Science in Sports, 10(2), 75-78. [25] Reilly, T., Tyrrell, A. and Troup, J.D. (1984) Circadian variation in human stature. Chronobiology International, 1(2), 121-126. [26] Tyrrell, A.R., Reilly, T. and Troup, J.D. (1985) Circadian variation in stature and the effects of spinal loading. Spine, 10(2), 161-164. [27] Tremblay, A. and Therrien, F. (2006) Physical activity and body functionality: implications for obesity preven- tion and treatment. Canadian Journal of Physiology and Pharmacology, 84(2), 149-156. [28] Westcott, W.L., Winett, R.A., Anderson, E.S., Wojcik, J.R., Loud, R.L., Cleggett, E. and Glover, S. (2001) Ef- fects of regular and slow speed resistance training on muscle strength. The Journal of Sports Medicine and Physical Fitness, 41(2), 154-158. [29] Sidney, K. and Jetté, M. (1992) Characteristics of women performing strength training: comparison of participants and dropouts. The Journal of Sports Medicine and Phys- ical Fitness, 32(1), 84-95. |

