 Open Journal of Respiratory Diseases, 2013, 3, 25-30 http://dx.doi.org/10.4236/ojrd.2013.31005 Published Online February 2013 (http://www.scirp.org/journal/ojrd) Leg Atherosclerosis in Japanese COPD Patients: Prevalence of Undiagnosed Peripheral Artery Disease and Association between Leg Atherosclerosis and Clinical Indices Hirofumi Matsuoka, Yusuke Matsumoto, Kengo Kimura, Midori Koyama, Towa Uzu, Yasuko Koma, Kensuke Fukumitsu, Yoshitaka Kasai, Nariyasu Nakashima, Daiki Masuya, Harukazu Yoshimatsu, Yujiro Suzuki Department of Respiratory Medicine, Shinko Hospital, Kobe, Japan Email: h-matsuoka@shinkohp.or.jp Received September 25, 2012; revised October 28, 2012; accepted November 7, 2012 ABSTRACT Introduction: Several studies have suggested that decreased FEV1 is associated with cardiovascular risk in COPD pa- tients. Objective: To identify the prevalence of undiagnosed peripheral artery disease (PAD) and the relationship be- tween leg atherosclerosis and clinical indices, which predict COPD mortality in Japanese COPD patients. Methods: We performed a cross-sectional study in 51 COPD patients and 51 age-matched, healthy control smokers. We measured ankle-brachial index (ABI) as a marker of atherosclerosis of the legs, pulmonary function, body mass index, modified Medical Research Council (MMRC) dyspnea scale, and smoking pack-years. We also calculated the ADO index (Age, Dyspnea, and Obstruction), an established predictor of mortality in COPD patients. Co-morbidities including diabetes mellitus, hypertension, and hypercholesterolemia were identified from blood laboratory tests and medical records. Re- sults: Five subjects (9.8%) had an ABI < 0.9. ABI was significantly lower in the COPD patients than in the healthy control smokers (p < 0.05). The prevalence of PAD was marginally higher in COPD patients than in control smokers (p = 0.09), with the prevalence of ABI < 1.0 being significantly higher in COPD patients than in control smokers (p = 0.04). In the COPD patients, ABI showed significant correlations with age (p = 0.006), FEV1 (p = 0.004), smoking pack-years (p = 0.047), MMRC dyspnea scale (p = 0.0005), SaO2 (p = 0.001), and ADO index (p < 0.001). Multiple linear regression modeling showed the factors associated independently with ABI were age, FEV1, smoking pack-years, MMRC dyspnea scale, and SaO2. Conclusion: The risk of leg atherosclerosis in Japanese COPD patients is higher than in smokers without COPD. Leg atherosclerosis in COPD patients is associated with clinical indices that predict COPD mortality. Keywords: COPD; Peripheral Artery Disease; Leg Atherosclerosis; Ankle-Brachial Index 1. Introduction Tobacco smoking is the most important risk factor for both the development and progression of COPD. Chronic obstructive pulmonary disease (COPD) is the fourth lead- ing cause of death worldwide [1]. Recently, COPD has been recognized as a systemic disease [2,3], and in par- ticular, is associated with a markedly increased risk of cardiovascular disease [4], which accounts for approxi- mately 25% to 40% of mortality in COPD patients [5,6]. OPD is characterized by chronic airflow limitation re- sulting from an excessive inflammatory response of the lungs to cigarette smoking [7], an established risk factor for cardiovascular disease. However, recent studies have also shown that COPD is associated with cardiovascular risk independent of classical risk factors [8-10]. Further- more, several studies have demonstrated that atheroscle- rosis is associated with FEV1 [11-14]. These findings in- dicate that the severity of COPD is associated with athe- rosclerosis. Peripheral arterial disease (PAD) is a manifestation of systemic atherosclerosis, and is a common disorder asso- ciated with a very high risk of myocardial infarction, is- chemic stroke, and death [15]. The prognosis of patients with lower extremity PAD is characterized by an increa- sed risk for cardiovascular ischemic events due to con- comitant coronary artery disease and cerebrovascular disease [16,17]. There is evidence that these cardiovas- cular ischemic events are more frequent than ischemic limb events in cohorts of patients with lower extremity C opyright © 2013 SciRes. OJRD  H. MATSUOKA ET AL. 26 PAD [18]. Lower extremity PAD should therefore be con- sidered as a sign of potentially diffuse and significant ar- terial disease [15]. Several studies have reported that the rate of cardio- vascular death in the smoking population in Japan is lower than in other developed countries [19,20]. How- ever, there are no data comparing the prevalence of PAD in COPD patients and healthy smokers in the Japanese population, and only limited data on the relationship be- tween leg atherosclerosis and clinical indices in COPD patients. 2. Method 2.1. Subjects Studied Subjects with COPD with a smoking history and age- matched control smokers were recruited from an outpa- tient clinic at Shinko Hospital. The control smokers with- out COPD were recruited from individuals treated at our hospital for chronic bronchitis without lung function ab- normalities, or for health status check-ups. Control smok- ers were ex-smokers or current smokers without lung func- tion abnormalities. An age-matched (within 1 year) con- trol smoker was selected randomly for each subject with COPD. Subjects with a history of respiratory infection within the previous 4 weeks, asthma, or active malignan- cy were not included in the study. Cardiovascular comor- bidity was recorded carefully. Patients already diagnosed with PAD were excluded from the study. Body mass index (BMI) was calculated as weight (in kilograms) divided by height squared (in meters). Hyper- tension was defined as either a systolic blood pressure ≧ 140 mmHg, diastolic blood pressure ≧ 90 mmHg, or self-reported use of antihypertensive medication. Diabe- tes mellitus was defined as either a fasting glucose level ≧ 126 mg/dl, a non-fasting glucose level ≧ 200 mg/dl, a self-reported physician diagnosis, or pharmacologic hy- poglycemic treatment. Subjects with a low-density lipo- protein (LDL)-cholesterol level ≧ 140 mg/dl or using li- pid-lowering drugs were considered to have hypercholes- terolemia. The subjects also completed a medical history that included questions about their current smoking status and history. Spirometry was performed on all subjects using a com- puted spirometer (CHESTAC-8800, CHEST M. I., Inc., Tokyo, Japan). The protocol for the lung function mea- surements conformed to the recommendations of the Ame- rican Thoracic Society [21]. This study was approved by the Ethics Committee of Shinko Hospital, and informed consent was obtained from all subjects prior to enroll- ment. 2.2. Ankle-Brachial Index The ABI is calculated as the ratio of ankle to arm systolic blood pressure and is used commonly in clinical practice to assess lower extremity PAD [15]. In all cases, the sub- jects rested in the supine position for 5 min before meas- urement of ABI. Using appropriately sized blood pres- sure cuffs, systolic blood pressure was measured in both brachial arteries and both leg arteries using an automated device. All measurements were performed by staff in a blinded manner. We used the measurement from the leg with the lower ABI in the analyses. 2.3. Clinical Evaluation Patients completed the modified Medical Research Coun- cil dyspnea scale (MMRC) questionnaire [22]. The ADO index is calculated using age, MMRC, and FEV1, and is a better predictor of mortality from COPD than the tradi- tional BODE index [23]. The ADO score ranges from 0 to 10 points, with higher scores indicating higher morta- lity. 2.4. Statistical Analysis JMP software (SAS Institute Inc., Cary, NC, USA) was used for the analyses. The results are presented as mean (SEM) or number (percentage). Differences between the COPD patients and control smokers were compared us- ing unpaired Student’s t-tests for continuous variables and χ2-tests for categorical data. Spearman’s rank test was used to examine correlations between the variables. Multivariate linear regression was performed using each parameter as a dependent variable in order to determine the independent predictors of ABI. Due to the strong as- sociation between age and SaO2, these variables were in- cluded in separate models as candidate variables. P-va- lues < 0.05 were considered statistically significant. 3. Results 3.1. Subject Characteristics The characteristics of the subjects are shown in Table 1. The mean age of the COPD subjects was 72.4 years. The prevalence of ABI < 0.9 was marginally higher in the COPD group than in the control group (9.8% vs 2.0%, p = 0.092). The prevalence of ABI < 1.0 was significantly higher in the COPD group than in the control group (19.6% vs 5.9%, p = 0.037). FEV1, BMI, and ABI were significantly lower in the COPD subjects compared to the control smokers (all p < 0.05). Age, gender, smoking status and pack-year histories, and prevalence of comor- bidities were similar between the two groups. The association of ABI with cardiovascular risk factors and clinical indices In the COPD patients, ABI correlated significantly with age (r = −0.37, p = 0.006), FEV1 (r = 0.28, p = 0.004), smoking pack-years (r = −0.28, p = 0.047), Copyright © 2013 SciRes. OJRD 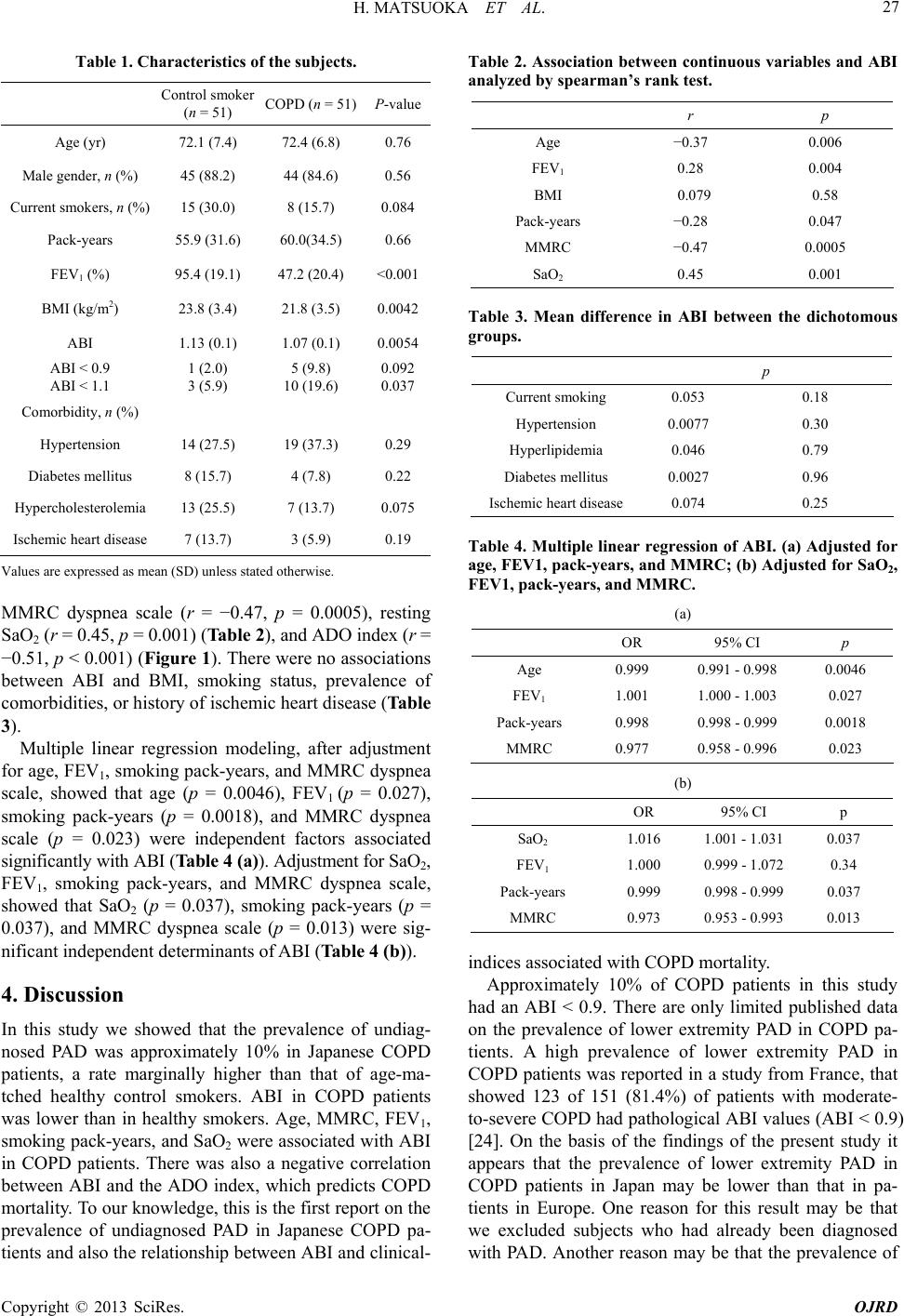 H. MATSUOKA ET AL. 27 Table 1. Characteristics of the subjects. Control smoker (n = 51) COPD (n = 51)P-value Age (yr) 72.1 (7.4) 72.4 (6.8) 0.76 Male gender, n (%) 45 (88.2) 44 (84.6) 0.56 Current smokers, n (%) 15 (30.0) 8 (15.7) 0.084 Pack-years 55.9 (31.6) 60.0(34.5) 0.66 FEV1 (%) 95.4 (19.1) 47.2 (20.4) <0.001 BMI (kg/m2) 23.8 (3.4) 21.8 (3.5) 0.0042 ABI 1.13 (0.1) 1.07 (0.1) 0.0054 ABI < 0.9 ABI < 1.1 1 (2.0) 3 (5.9) 5 (9.8) 10 (19.6) 0.092 0.037 Comorbidity, n (%) Hypertension 14 (27.5) 19 (37.3) 0.29 Diabetes mellitus 8 (15.7) 4 (7.8) 0.22 Hypercholesterolemia 13 (25.5) 7 (13.7) 0.075 Ischemic heart disease 7 (13.7) 3 (5.9) 0.19 Values are expressed as mean (SD) unless stated otherwise. MMRC dyspnea scale (r = −0.47, p = 0.0005), resting SaO2 (r = 0.45, p = 0.001) (Table 2), and ADO index (r = −0.51, p < 0.001) (Figure 1). There were no associations between ABI and BMI, smoking status, prevalence of comorbidities, or history of ischemic heart disease (Table 3). Multiple linear regression modeling, after adjustment for age, FEV1, smoking pack-years, and MMRC dyspnea scale, showed that age (p = 0.0046), FEV1 (p = 0.027), smoking pack-years (p = 0.0018), and MMRC dyspnea scale (p = 0.023) were independent factors associated significantly with ABI (Table 4 (a)). Adjustment for SaO2, FEV1, smoking pack-years, and MMRC dyspnea scale, showed that SaO2 (p = 0.037), smoking pack-years (p = 0.037), and MMRC dyspnea scale (p = 0.013) were sig- nificant independent determinants of ABI (Table 4 (b)). 4. Discussion In this study we showed that the prevalence of undiag- nosed PAD was approximately 10% in Japanese COPD patients, a rate marginally higher than that of age-ma- tched healthy control smokers. ABI in COPD patients was lower than in healthy smokers. Age, MMRC, FEV1, smoking pack-years, and SaO2 were associated with ABI in COPD patients. There was also a negative correlation between ABI and the ADO index, which predicts COPD mortality. To our knowledge, this is the first report on the prevalence of undiagnosed PAD in Japanese COPD pa- tients and also the relationship between ABI and clinical- Table 2. Association between continuous variables and ABI analyzed by spearman’s rank test. r p Age −0.37 0.006 FEV1 0.28 0.004 BMI 0.079 0.58 Pack-years −0.28 0.047 MMRC −0.47 0.0005 SaO2 0.45 0.001 Table 3. Mean difference in ABI between the dichotomous groups. p Current smoking 0.053 0.18 Hypertension 0.0077 0.30 Hyperlipidemia 0.046 0.79 Diabetes mellitus 0.0027 0.96 Ischemic heart disease 0.074 0.25 Table 4. Multiple linear regression of ABI. (a) Adjusted for age, FEV1, pack-years, and MMRC; (b) Adjusted for SaO2, FEV1, pack-years, and MMRC. (a) OR 95% CI p Age 0.999 0.991 - 0.998 0.0046 FEV1 1.001 1.000 - 1.003 0.027 Pack-years 0.998 0.998 - 0.999 0.0018 MMRC 0.977 0.958 - 0.996 0.023 (b) OR 95% CI p SaO2 1.016 1.001 - 1.031 0.037 FEV1 1.000 0.999 - 1.072 0.34 Pack-years 0.999 0.998 - 0.999 0.037 MMRC 0.973 0.953 - 0.993 0.013 indices associated with COPD mortality. Approximately 10% of COPD patients in this study had an ABI < 0.9. There are only limited published data on the prevalence of lower extremity PAD in COPD pa- tients. A high prevalence of lower extremity PAD in COPD patients was reported in a study from France, that showed 123 of 151 (81.4%) of patients with moderate- to-severe COPD had pathological ABI values (ABI < 0.9) [24]. On the basis of the findings of the present study it appears that the prevalence of lower extremity PAD in COPD patients in Japan may be lower than that in pa- tients in Europe. One reason for this result may be that we excluded subjects who had already been diagnosed with PAD. Another reason may be that the prevalence of Copyright © 2013 SciRes. OJRD 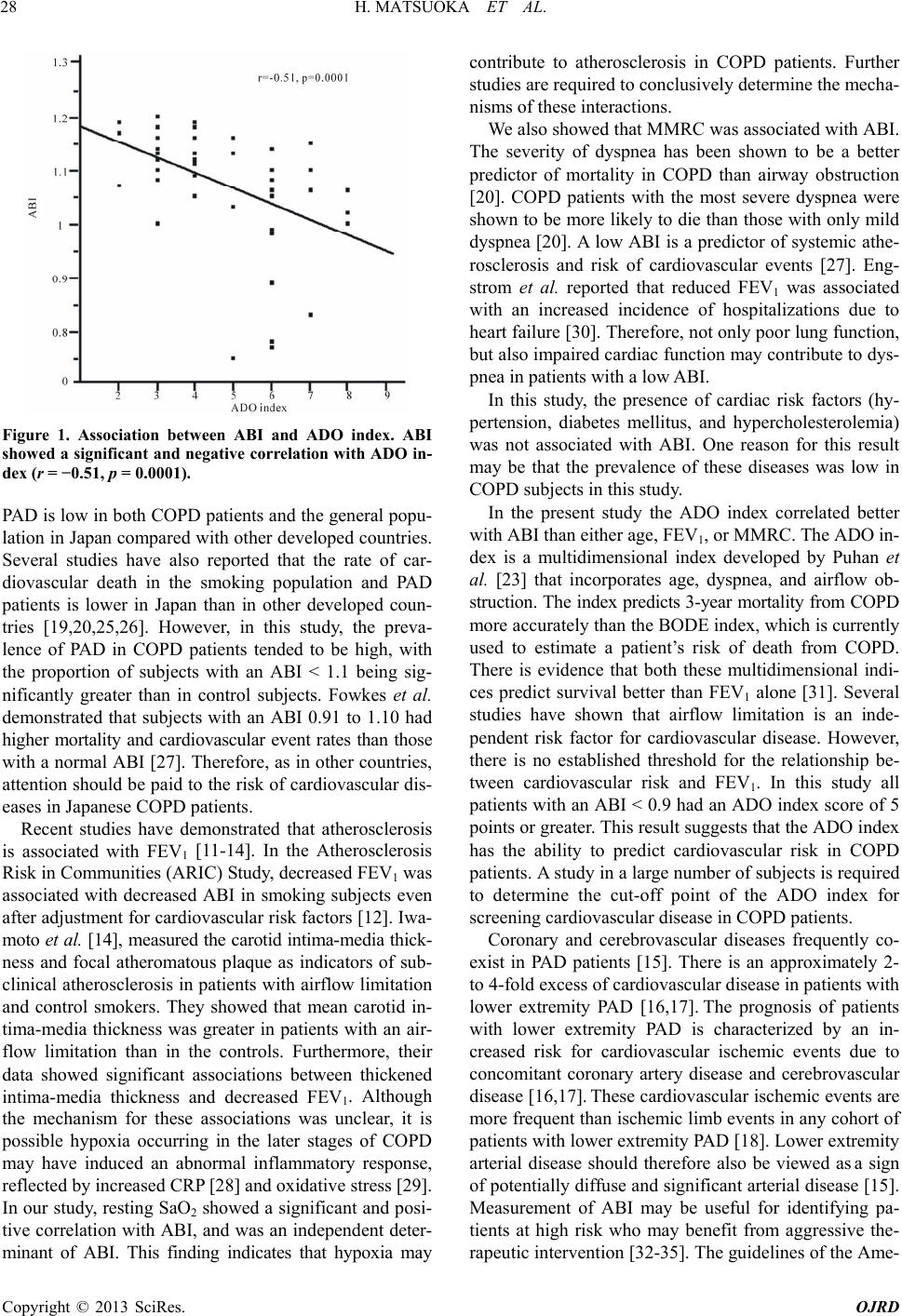 H. MATSUOKA ET AL. 28 Figure 1. Association between ABI and ADO index. ABI showed a significant and negative correlation with ADO in- dex (r = −0.51, p = 0.0001). PAD is low in both COPD patients and the general popu- lation in Japan compared with other developed countries. Several studies have also reported that the rate of car- diovascular death in the smoking population and PAD patients is lower in Japan than in other developed coun- tries [19,20,25,26]. However, in this study, the preva- lence of PAD in COPD patients tended to be high, with the proportion of subjects with an ABI < 1.1 being sig- nificantly greater than in control subjects. Fowkes et al. demonstrated that subjects with an ABI 0.91 to 1.10 had higher mortality and cardiovascular event rates than those with a normal ABI [27]. Therefore, as in other countries, attention should be paid to the risk of cardiovascular dis- eases in Japanese COPD patients. Recent studies have demonstrated that atherosclerosis is associated with FEV1 [11-14]. In the Atherosclerosis Risk in Communities (ARIC) Study, decreased FEV1 was associated with decreased ABI in smoking subjects even after adjustment for cardiovascular risk factors [12]. Iwa- moto et al. [14], measured the carotid intima-media thick- ness and focal atheromatous plaque as indicators of sub- clinical atherosclerosis in patients with airflow limitation and control smokers. They showed that mean carotid in- tima-media thickness was greater in patients with an air- flow limitation than in the controls. Furthermore, their data showed significant associations between thickened intima-media thickness and decreased FEV1. Although the mechanism for these associations was unclear, it is possible hypoxia occurring in the later stages of COPD may have induced an abnormal inflammatory response, reflected by increased CRP [28] and oxidative stress [29]. In our study, resting SaO2 showed a significant and posi- tive correlation with ABI, and was an independent deter- minant of ABI. This finding indicates that hypoxia may contribute to atherosclerosis in COPD patients. Further studies are required to conclusively determine the mecha- nisms of these interactions. We also showed that MMRC was associated with ABI. The severity of dyspnea has been shown to be a better predictor of mortality in COPD than airway obstruction [20]. COPD patients with the most severe dyspnea were shown to be more likely to die than those with only mild dyspnea [20]. A low ABI is a predictor of systemic athe- rosclerosis and risk of cardiovascular events [27]. Eng- strom et al. reported that reduced FEV1 was associated with an increased incidence of hospitalizations due to heart failure [30]. Therefore, not only poor lung function, but also impaired cardiac function may contribute to dys- pnea in patients with a low ABI. In this study, the presence of cardiac risk factors (hy- pertension, diabetes mellitus, and hypercholesterolemia) was not associated with ABI. One reason for this result may be that the prevalence of these diseases was low in COPD subjects in this study. In the present study the ADO index correlated better with ABI than either age, FEV1, or MMRC. The ADO in- dex is a multidimensional index developed by Puhan et al. [23] that incorporates age, dyspnea, and airflow ob- struction. The index predicts 3-year mortality from COPD more accurately than the BODE index, which is currently used to estimate a patient’s risk of death from COPD. There is evidence that both these multidimensional indi- ces predict survival better than FEV1 alone [31]. Several studies have shown that airflow limitation is an inde- pendent risk factor for cardiovascular disease. However, there is no established threshold for the relationship be- tween cardiovascular risk and FEV1. In this study all patients with an ABI < 0.9 had an ADO index score of 5 points or greater. This result suggests that the ADO index has the ability to predict cardiovascular risk in COPD patients. A study in a large number of subjects is required to determine the cut-off point of the ADO index for screening cardiovascular disease in COPD patients. Coronary and cerebrovascular diseases frequently co- exist in PAD patients [15]. There is an approximately 2- to 4-fold excess of cardiovascular disease in patients with lower extremity PAD [16,17]. The prognosis of patients with lower extremity PAD is characterized by an in- creased risk for cardiovascular ischemic events due to concomitant coronary artery disease and cerebrovascular disease [16,17]. These cardiovascular ischemic events are more frequent than ischemic limb events in any cohort of patients with lower extremity PAD [18]. Lower extremity arterial disease should therefore also be viewed as a sign of potentially diffuse and significant arterial disease [15]. Measurement of ABI may be useful for identifying pa- tients at high risk who may benefit from aggressive the- rapeutic intervention [32-35]. The guidelines of the Ame- Copyright © 2013 SciRes. OJRD  H. MATSUOKA ET AL. 29 rican College of Cardiology (ACC) and American Heart Association (AHA) for the management of patients with PAD recommends that ABI should be considered as a routine test for all patients who are 49 years of age and younger with a history of diabetes and 1 other risk factor, those 50 to 69 years of age with a history of smoking or diabetes, and those aged 70 years or older [15]. In accor- dance with these guidelines the majority of COPD pa- tients should have ABI measured. There were some limitations in this study. The number of subjects was small and therefore a study on a larger number of subjects is needed to conclusively establish the prevalence of PAD in COPD subjects. Although some studies have reported an association between atheroscle- rosis and nocturnal hypoxia [36], the current study did not evaluate this relationship. 5. Conclusion In this study we showed that the rate of atherosclerosis in COPD patients in Japan was lower than in similar pa- tients in other developed countries. However, we showed the rate of atherosclerosis in COPD patients was higher than in healthy smokers, with this finding being consis- tent to data of other countries. Leg atherosclerosis was also shown to be associated with clinical indices related to COPD mortality. It is therefore important that more attention is paid to leg atherosclerosis in Japanese COPD patients. 6. Acknowledgements We thank Masahiro Motoki, Takanori Matsutani, Taka- hiko Ando, Kaori Tai, Megumi Sakano, Nanae Kiyokawa, and Kanako Ichimaru of the clinical laboratory of Shinko Hospital for carrying out the pulmonary function tests and measuring ABI. REFERENCES [1] C. J. Murray and A. D. Lopez, “Global Mortality, Dis- ability, and the Contribution of Risk Factors: Global Bur- den of Disease Study,” Lancet, Vol. 349, No. 9063, 1997, pp. 1436-1442. doi:10.1016/S0140-6736(96)07495-8 [2] L. M. Fabbri, F. Luppi, B. Beghe, et al., “Complex Chro- nic Comorbidities of COPD,” European Respiratory Jour- nal, Vol. 31, No. 1, 2008, pp. 204-212. doi:10.1183/09031936.00114307 [3] J. D. Maclay, D. A. McAllister and W. Macnee, “Cardio- vascular Risk in Chronic Obstructive Pulmonary Disea- se,” Respirology, Vol. 12, No. 5, 2007, pp. 634-641. doi:10.1111/j.1440-1843.2007.01136.x [4] D. D. Sin and S. F. Man, “Chronic Obstructive Pulmona- ry Disease as a Risk Factor for Cardiovascular Morbidity and Mortality,” Proceedings of the American Thoracic Society, Vol. 2, No. 1, 2005, pp. 8-11. doi:10.1513/pats.200404-032MS [5] P. M. Calverley, J. A. Anderson, B. Celli, et al., “Salme- terol and Fluticasone Propionate and Survival in Chronic Obstructive Pulmonary Disease,” The New England Jour- nal of Medicine, Vol. 356, No. 8, 2007, pp. 775-789. doi:10.1056/NEJMoa063070 [6] R. A. Pauwels, C. G. Lofdahl, L. A. Laitinen, et al., “Long- Term Treatment with Inhaled Budesonide in Persons with Mild Chronic Obstructive Pulmonary Disease Who Con- tinue Smoking,” The New England Journal of Medicine, Vol. 340, No. 25, 1999, pp. 1948-1953. doi:10.1056/NEJM199906243402503 [7] W. MacNee, “Pathogenesis of Chronic Obstructive Pul- monary Disease,” Proceedings of the American Thoracic Society, Vol. 2, No. 4, 2005, pp. 258-266. doi:10.1513/pats.200504-045SR [8] H. J. Schunemann, J. Dorn, B. J. Grant, et al., “Pulmo- nary Function Is a Long-Term Predictor of Mortality in the General Population: 29-Year Follow-Up of the Buf- falo Health Study,” Chest, Vol. 118, No. 3, 2000, pp. 656-664. doi:10.1378/chest.118.3.656 [9] D. D. Sin, L. Wu and S. F. Man, “The Relationship Be- tween Reduced Lung Function and Cardiovascular Mor- tality: A Population-Based Study and a Systematic Re- view of the Literature,” Chest, Vol. 127, No. 6, 2005, pp. 1952-1959. doi:10.1378/chest.127.6.1952 [10] S. M. Curkendall, C. DeLuise, J. K. Jones, et al., “Car- diovascular Disease in Patients with Chronic Obstructive Pulmonary Disease, Saskatchewan Canada Cardiovascu- lar Disease in COPD Patients,” Annals of Epidemiology, Vol. 16, No. 1, 2006, pp. 63-70. doi:10.1016/j.annepidem.2005.04.008 [11] M. Zureik, A. Benetos, C. Neukirch, et al., “Reduced Pul- monary Function Is Associated with Central Arterial Stiff- ness in Men,” American Journal of Respiratory and Criti- cal Care Medicine, Vol. 164, No. 12, 2001, pp. 2181-2185. [12] E. B. Schroeder, V. L. Welch, G. W. Evans, et al., “Im- paired Lung Function and Subclinical Atherosclerosis. The ARIC Study,” Atherosclerosis, Vol. 180, No. 2, 2005, pp. 367-373. doi:10.1016/j.atherosclerosis.2004.12.012 [13] D. A. McAllister, J. D. Maclay, N. L. Mills, et al., “Arte- rial Stiffness Is Independently Associated with Emphy- sema Severity in Patients with Chronic Obstructive Pul- monary Disease,” American Journal of Respiratory and Critical Care Medicine, Vol. 176, No. 12, 2007, pp. 1208-1214. doi:10.1164/rccm.200707-1080OC [14] H. Iwamoto, A. Yokoyama, Y. Kitahara, et al., “Airflow Limitation in Smokers Is Associated with Subclinical Atherosclerosis,” American Journal of Respiratory and Critical Care Medicine, Vol. 179, No. 1, 2009, pp. 35-40. doi:10.1164/rccm.200804-560OC [15] A. T. Hirsch, Z. J. Haskal, N. R. Hertzer, et al., “ACC/ AHA 2005 Practice Guidelines for the Management of Patients with Peripheral Arterial Disease (Lower Extremi- ty, Renal, Mesenteric, and Abdominal Aortic): A Col- laborative Report from the American Association for Va- scular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interven- Copyright © 2013 SciRes. OJRD  H. MATSUOKA ET AL. Copyright © 2013 SciRes. OJRD 30 tional Radiology, and the ACC/AHA Task Force on Prac- tice Guidelines (Writing Committee to Develop,” Circu- lation, Vol. 113, No. 11, 2006, pp. e463-e654. [16] M. H. Criqui, J. O. Denenberg, R. D. Langer, et al., “The Epidemiology of Peripheral Arterial Disease: Importance of Identifying the Population at Risk,” Vascular Medicine, Vol. 2, No. 3, 1997, pp. 221-226. [17] J. Ness, W. S. Aronow, “Prevalence of Coexistence of Coronary Artery Disease, Ischemic Stroke, and Peripheral Arterial Disease in Older Persons, Mean Age 80 Years, in an Academic Hospital-Based Geriatrics Practice,” Jour- nal of the American Geriatrics Society, Vol. 47, No. 10, 1999, pp. 1255-1256. [18] J. I. Weitz, J. Byrne, G. P. Clagett, et al., “Diagnosis and Treatment of Chronic Arterial Insufficiency of the Lower Extremities: A Critical Review,” Circulation, Vol. 94, No. 11, 1996, pp. 3026-3049. doi:10.1161/01.CIR.94.11.3026 [19] D. R. Jacobs Jr., H. Adachi, I. Mulder, et al., “Cigarette Smoking and Mortality Risk: Twenty-Five-Year Follow- Up of the Seven Countries Study,” The Archives of In- ternal Medicine, Vol. 159, No. 7, 1999, pp. 733-740. doi:10.1001/archinte.159.7.733 [20] K. Nishimura, T. Izumi, M. Tsukino, et al., “Dyspnea Is a Better Predictor of 5-Year Survival than Airway Obstruc- tion in Patients with COPD,” Chest, Vol. 121, No. 5, 2002, pp. 1434-1440. doi:10.1378/chest.121.5.1434 [21] American Thoracic Society, “Standardization of Spi- rometry,” American Journal of Respiratory and Critical Care Medicine, Vol. 152, No. 3, 1995, pp. 1107-1136. [22] D. A. Mahler, C. K. Wells, Evaluation of Clinical Meth- ods for Rating Dyspnea,” Chest, Vol. 93, No. 3, 1988, pp. 580-586. doi:10.1378/chest.93.3.580 [23] M. A. Puhan, J. Garcia-Aymerich, M. Frey, et al., “Ex- pansion of the Prognostic Assessment of Patients with Chro- nic Obstructive Pulmonary Disease: The Updated BODE Index and the ADO Index,” Lancet, Vol. 374, No. 9691, 2009, pp. 704-711. doi:10.1016/S0140-6736(09)61301-5 [24] O. Castagna, A. Boussuges, E. Nussbaum, et al., “Pe- ripheral Arterial Disease: An Underestimated Aetiology of Exercise Intolerance in Chronic Obstructive Pulmonary Disease Patients,” European Journal of Cardiovascular Prevention & Rehabilitation, Vol. 15, No. 3, 2008, pp. 270-277. doi:10.1097/HJR.0b013e3282f009a9 [25] T. Fujiwara, S. Saitoh, et al., “Prevalence of Asymptoma- tic Arteriosclerosis Obliterans and Its Relationship with Risk Factors in Inhabitants of Rural Communities in Ja- pan: Tanno-Sobetsu study,” Atheroscle rosis, Vol. 177, No. 1, 2004, pp. 83-88. [26] H. Ohnishi, Y. Sawayama, et al., “Risk Factors for and the Prevalence of Peripheral Arterial Disease and Its Re- lationship to Carotid Atherosclerosis: The Kyushu and Oki- nawa Population Study (KOPS),” Journal of Atheroscle- rosis and Thrombosis, Vol. 30, Vol. 17, 2010, pp. 751-758. [27] F. G. Fowkes, G. D. Murray, I. Butcher, et al., “Ankle Brachial Index Combined with Framingham Risk Score to Predict Cardiovascular Events and Mortality: A Meta- Analysis,” Journal of the American Medical Association, Vol. 300, No. 2, 2008, pp. 197-208. doi:10.1001/jama.300.2.197 [28] G. Hartmann, M. Tschop, R. Fischer, et al., “High Alti- tude Increases Circulating Interleukin-6, Interleukin-1 Re- ceptor Antagonist and C-Reactive Protein,” Cytoki ne, Vol. 12, No. 3, 2000, pp. 246-252. doi:10.1006/cyto.1999.0533 [29] V. Savransky, A. Nanayakkara, J. Li, et al., “Chronic Intermittent Hypoxia Induces Atherosclerosis,” American Journal of Respiratory and Critical Care Medicine, Vol. 175, No. 12, 2007, pp. 1290-1297. doi:10.1164/rccm.200612-1771OC [30] G. Engstrom, O. Melander, B. Hedblad, ‘Population-Ba- sed Study of Lung Function and Incidence of Heart Fai- lure Hospitalisations,” Thorax, Vol. 65, No. 7, 2010, pp. 633-638. doi:10.1136/thx.2010.135392 [31] B. R. Celli, C. G. Cote, J. M. Marin, et al., “The Body- Mass Index, Airflow Obstruction, Dyspnea, and Exercise Capacity Index in Chronic Obstructive Pulmonary Dis- ease,” The New England Journal of Medicine, Vol. 350, No. 10, 2004, pp. 1005-1012. doi:10.1056/NEJMoa021322 [32] M. McKenna, S. Wolfson and L. Kuller, “The Ratio of Ankle and Arm Arterial Pressure as an Independent Pre- dictor of Mortality,” Atherosclero sis, Vol. 87, No. 2, 1991, pp. 119-128. doi:10.1016/0021-9150(91)90014-T [33] M. T. Vogt, M. McKenna, S. J. Anderson, et al., “The Relationship between Ankle-Arm Index and Mortality in Older Men and Women,” Journal of the American Geri- atrics Society, Vol. 41, No. 5, 1993, pp. 523-530. [34] G. C. Leng, F. G. Fowkes, A. J. Lee, et al., “Use of Ankle Brachial Pressure Index to Predict Cardiovascular Events and Death: A Cohort Study,” British Medical Journal, Vol. 313, No. 7070, 1996, pp. 1440-1444. doi:10.1136/bmj.313.7070.1440 [35] H. E. Resnick, R. S. Lindsay, M. M. McDermott, et al., “Relationship of High and Low Ankle Brachial Index to All-Cause and Cardiovascular Disease Mortality: The Strong Heart Study,” Circulation, Vol. 109, No. 6, 2004, pp. 733-739. doi:10.1161/01.CIR.0000112642.63927.54 [36] R. Schulz, Seeger, et al., “Changes in Extracranial Arte- ries in Obstructive Sleep Apnoea,” European Respiratory Journal, Vol. 25, No. 1, 2005, pp. 69-74. Abbreviations COPD: Chronic Obstructive Pulmonary Disease FEV1: Forced Expiratory Volume in one second PAD: Peripheral Artery Disease A BI: Ankle-Brachial Index BMI: Body Mass Index MMRC: Modified Medical Research Council SaO2: Arterial Oxygen Saturation Ease of Use (Heading 2)
|