Paper Menu >>
Journal Menu >>
 Modeling and Numerical Simulation of Material Science, 2013, 3, 9-12 Published Online January 2013 (http://www.SciRP.org/journal/mnsms) Copyright © 2013 SciRes. MNSMS Yb-Decorated Carbon Nanotubes As a Potential Capacity Hydrogen Storage Medium* Hong-Wen Lei, Hong Zhang, Wei-Dong Wu Science and Technology on Plasma physics Laboratory, Research Center of Laser Fusion, China Academy of Engineering Physics, Mianyang 621900, China Institution of Atomic and Molecular Physics, Sichuan University, Chengdu 610065, China College of Physical Science and Technology, Sichuan University, Chengdu 610065, China Email: wuweidongding@163.com Received 2012 ABSTRACT We report a first -principles study, which demonstrates that a single Yb atom coated on a single-walled nanotube (SWNT), B atom doped CNT and N atom doped CNT binds up to six hydrogen molecules. At high Yb coverage we show that a SWNT can strongly adsorb up to 3.18 wt % hydrogen. Yb-4f electrons have no contribution on the adsorp- tion of hydrogen molecules in Yb doped CNT. The charge analysis results show that 4f electrons remain in Yb. These results promote our fundamental understanding of dissociative adsorption of hydrogen in RE atom doped carbon nano- struct ur es. Keywords: Hydrogen Adsorption; Carbon Nanotubes; Density of States 1. Introduction As one of the most abundant elements in the universe, hydrogen is receiving increasing attention as an friendly and clean energy for environment [1]. However, a wealth of fundamental and technical challenges on hydrogen transport and storage, such as high gravimetric and volumetric density, safety and low cost must be overcome before hydrogen fuel economy realized. The Department of Energy (DOE) of the US targets for the ideal hydrogen storage materials with the gravimetric storage capacity of hydrogen should reach 9wt% by 2015[2]. Traditional methods to store hydrogen include using compressed gaseousor liquid H2, which demands high pressure and/or low temperature, or using solids that adsorb H2. Simultaneously, this approach is too expensive. In this cause, to achieve economic feasibility, hydrogen storage materials with high volumetric and gravimetric densities must be developed [3]. Suggestions for hydrogen adsorption in doped carbon nanotubes (CNTs) have been made because of the possibility of reversibility, fast kinetics, and high capacity (large surface area) [4-7]. However, it has been found that hydrogen on pure CNT currently falls short of the DOE targets. This is attributed to the weak interaction between hydrogen molecules and the carbon materials by physisorption [8,9]. More recent theoretical studies have been devoted to finding and designing materials whi c h enhance the interactions to the desirable binding energy of 0.2–0.6eV [10-16]. K. Hirano et al’s study suggest that 15 rare earth elements reveal characteristics of the capacity of hydrogen absorption. It is found that RE such as Eu prefers to adsorb at the hollow site of the hexagonal ring on the outer surface of the CNT. One of the most important advantages of RE (such as Eu) doping on CNT is that the electronic characteristics originating from the unpaired 4f electrons could serve as electron donors and thus increase Ead for H2 on CNT[17]. When H2 molecules are attach on RE atoms, electron transfers from H2 to Eu, filling an acceptor-like state. To our best knowledge, Ytterbium (Yb), a member of the rare earth group metals with a filled f shell and a divalent electronic character, had not been studied for hydrogen adsorption when it attached to (8,0) SWCNT. As the electron configuration of Yb is [Xe] 4f76s2, there are a large number of filled 4f orbital, and thus more H2 molecules could be adsorbed. In particular, we focus on the role of transferred charges between Yb and SWCNT on the affinity of H2. To achieve our goal, we conduct a systematic search for high-capacity hydrogen storage media consisting of indvidual Yb atoms decorated on CNT, B-CNT, N-CNT. Our results show that the hydrogen storage ability of SWCNT can be enhanced by doping Yb a t oms and Yb-4f electrons have no contribution on the hydrogen adsorption but Yb-5d. * This work was supported by Ministry of National Science and Technology Major Instrumentation Special (Grant No. 2011YQ130018)  H. Lei ET AL. Copyright © 2013 SciRes. MNSMS 2. Computation Details Density functional theory (DFT) methods, implemented in the DMol3 package [18,19], are used to study the systems which have been widely utilized for carbon nanot ubl es doped with lanthanide series metal [17,20].Double Numerical plus polarization (DNP) atomic orbits were taken as basis sets[21]. Due to the crucial role that spin plays in this system, all our calculations have been performed in a spin unrestricted framework. All structural optimizations were obtained with symmetry constraints by using a convergence tolerance of energy of 1.0×10-5 hartree, a maximum force is 0.002 hartree/Å and a maximum displacement of 0.005 Å. The orbital cut off was set to be global with a value of 5.0Å, and smearing was 0.005Ha (1Ha=27.2114eV). In the self-consi ste nt-field calculations, the electronic-density convergence was set to 1.0×10-6e/Å3. In the present calculation, a hexagonal supercell was adopted, whose size was 25 × 25 × c Å3 with the length of c in the axial of z direction being two ti mes of the periodicity of the (8,0) nanotube. Supercell calculations wer e employed throughout where the carbon atoms on adjacent nanotubes are separated by over 10 Å. and the interaction between neighboring images could be neglected. The Brillouin zone of the supercell was sampled by 1×1×6 k-points within the Monkhorst-Pack sche me[22]. For comparison, the identical simulation parameters were employed in all simulations. Because the interaction between H2 and Yb usually renders significantly smaller or even repulsive interactions using GGA, however, the local density approximation (LDA) generally overestimates the H2 binding strength. So both LDA and GGA are described the binding energy of H2 dispersed CNT B-CNT, N-CNT, respectively [17, 23-25]. 3. Results and Discussion (a) (b) Figure 1. Coronene (C24H12) planar sheet(a) and the hydrogen binding energy(b).Red, gray, green spheres represent hydrogen atoms, carbon atoms and Yb atoms, respectively Coro nene -like (C24H12) planar sheet used as clus- ter-modeled carbon and hydrogen atoms. It is well know that carbon nanotubes curled from grap he me. One Yb atom doped C24H12 can attach five hydrogen molecules and average distanc e between Yb atom and the center of hydrogen molecules is 2.24 Å. The purpose of the hy- drogen atoms is to passivate the dangling bonds of the polycyclic sp2 hybridized carbon structure . Binding energy and optimized structure are shown in Fi g.1 . To search for hydrogen-storage nanostructures consisting of combination of carbon nanotubes and Yb, we consider a pristine (8,0) CNT and the case of boron substitutional doping, nitrogen substitutional doping CNT to examine the local structure of the Yb attachment. A single Yb atom is attached to each of the above three struc ture s. We first evaluated the binding energy of a Yb atom (persu- percell) to the pristine (8,0) CNT. The Yb atom favors to locate at the hollow site of the hexagonal ring on the out- er surface, which is consistent with other RE atoms [17]. The binding energy of a single Yb atom at hexagonal sites is defined as EB(Yb)=E(CNT)+E(Ti)-E(CNT+Ti). The calculated binding energy of the Yb atom to the (8,0)CNT is 1.74 eV and the Yb-C bonds exist with bond length 2.39 and 2.67Å. Mulliken population analysis indicates that 0.45 e electrons are transferred from the Yb atom to CNT, resulting in partially cationic Yb atom. When the first hydrogen molecule is adsorbed on the Yb/CNT, H-H bond lengthn is 0.81 Å. Our calculations show that each Yb atom can hold up to six H2 molecules are attached on the Yb/CNT both LDA and GGA. The resulting system, which is shown in Fig. 2, is denoted by CNT/Yb/(H2)6. The final configuration is very symmetric, and all the hydrogen molecules benifit equally from the bonding with the Yb atom. The average binding energy per H2 is about 0.46eV(LDA) , i.e., slightly smaller than that obtained for the first adsorption. We have also cal- culated the binding energy of H2 molecules on Yb in these three struc tur es as a function of the number of hy- drogen molecules adsorbed. The binding energy of H2 for an isolated Yb atom decorated on a pristine (8,0) CNT, a nitro ge n-dopant, a boron-do pant are shown in table 1. The distance between Yb and the center of H2 and H-H bond length on average are 2.64 and 0.76 Å for CNT/Yb/(H2)6. (The bond length of isolated H2 is calcu- lated to be about 0.75 Å with GGA). Since H2 physisorp- tion on CNT is not reproduced at all in GGA [26,27], this observation implies that H2 adsorption on Yb is not in the physisorption regime. The binding energy of hydrogen molecules for LDA is excess the twice for GGA. Figure 4 shows the fully optimized structure for a maxi mu m hydrogen-storage capacity in (8,0) CNT co r- responding to 3.18 wt.%. We also computed the hydro- gen adsorption of B-CNT and N-CNT , the results ind i- cate that six H2 per Yb atom can be adsorbed onto B-CNT and N-CNT system where the molecular formula may be expressed as(C60N4·4Yb·24H2)n and (C60B4·4Yb·24H2)n (n is an integer). Eu3/SWCNT can 10 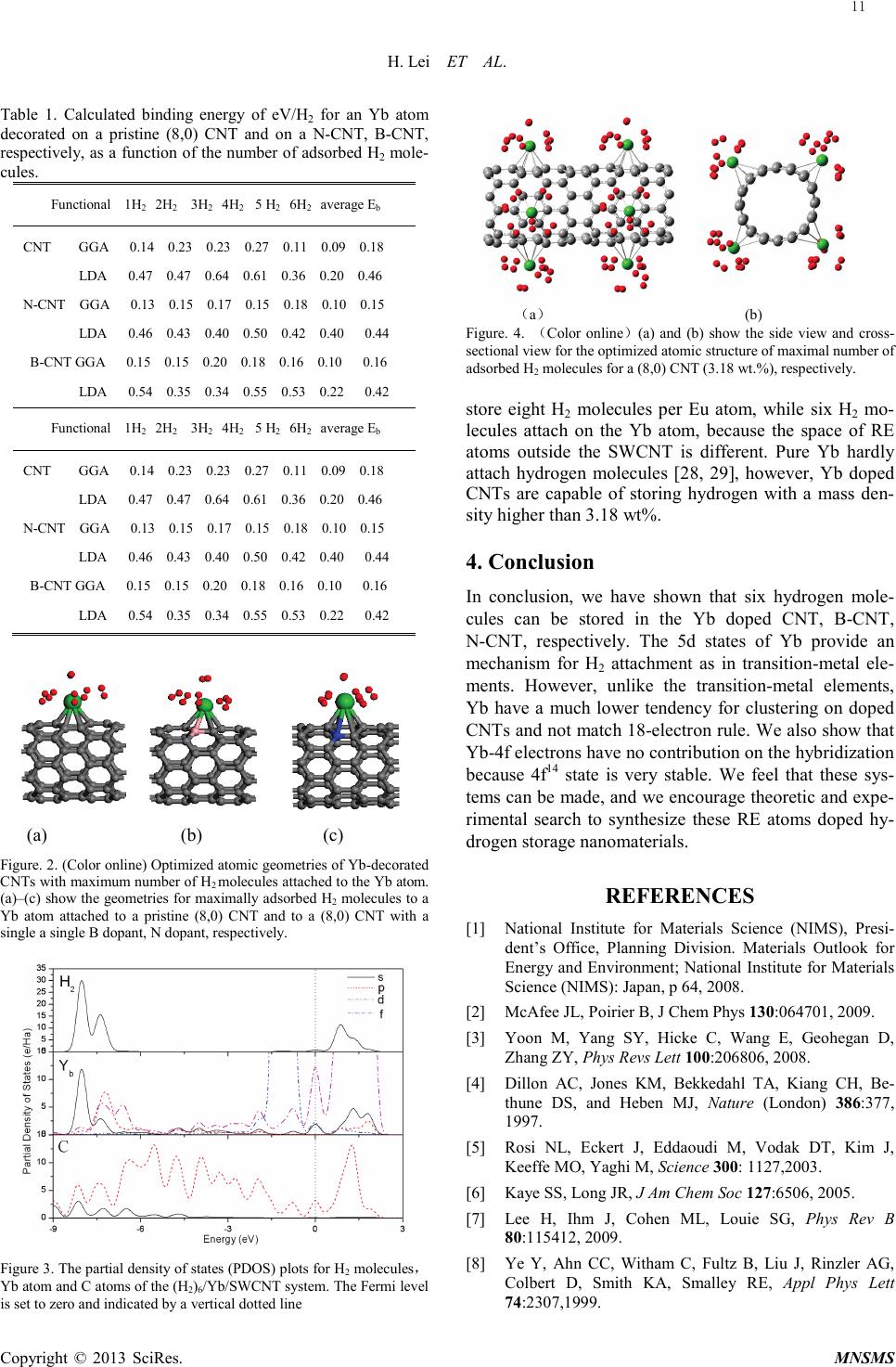 H. Lei ET AL. Copyright © 2013 SciRes. MNSMS Table 1. Calculated binding energy of eV/H2 for an Yb atom decorated on a pristine (8,0) CNT and on a N-CNT, B-CNT, respectively, as a function of the number of adsorbed H2 mole- cules. Functional 1H2 2H2 3H2 4H2 5 H2 6H2 average Eb CNT GGA 0.14 0.23 0.23 0.27 0.11 0.09 0.18 LDA 0.47 0.47 0.64 0.61 0.36 0.20 0.46 N-CNT GGA 0.13 0.15 0.17 0.15 0.18 0.10 0.15 LDA 0.46 0.43 0.40 0.50 0.42 0.40 0 .44 B-CNT GGA 0.15 0.15 0.20 0.18 0.16 0.10 0 .16 LDA 0.54 0.35 0.34 0.55 0.53 0.22 0.42 Functional 1H2 2H2 3H2 4H2 5 H2 6H2 average Eb CNT GGA 0.14 0.23 0.23 0.27 0.11 0.09 0.18 LDA 0.47 0.47 0.64 0.61 0.36 0.20 0.46 N-CNT GGA 0.13 0.15 0.17 0.15 0.18 0.10 0.15 LDA 0.46 0.43 0.40 0.50 0.42 0.40 0 .44 B-CNT GGA 0.15 0.15 0.20 0.18 0.16 0.10 0 .16 LDA 0.54 0.35 0.34 0.55 0.53 0.22 0 .42 (a) (b) (c) Figure. 2. (Color online) Optimized atomic geometries of Yb -decorated CNTs with maximum number of H2 molecules attached to the Yb atom. (a)–(c) show the geometries for maximally adsorbed H2 molecules to a Yb atom attached to a pristine (8,0) CNT and to a (8,0) CNT with a single a single B dopant, N dopant, respectively. Figure 3. The partial density of states (PDOS) plots for H2 molecules , Yb atom a nd C atoms of the (H2)6/Yb/SWCNT system. The Fermi level is set to zero and indicated by a vertical dotted line (a) (b) Figure. 4. (Color online)(a) and (b) show the side view and cross- sectional view for the optimized atomic structure of maximal number of adsorbed H2 molecules for a (8,0) CNT (3.18 wt . %), respectively. store eight H2 molecules per Eu atom, while six H2 mo- lecules attach on the Yb atom, because the space of RE atoms outside the SWCNT is different. Pure Yb hardly attach hydrogen molecules [28, 29], however, Yb doped CNTs are capable of storing hydrogen with a mass den- sity higher than 3.18 wt%. 4. Conclusion In conclusion, we have shown that six hydrogen mole- cules can be stored in the Yb doped CNT, B-CNT, N-CNT, respectively. The 5d states of Yb provide an mechanism for H2 attachment as in transition-metal ele- ments. However, unlike the transition-metal elements, Yb have a much lower tendency for clustering on doped CNTs and not match 18-electron rule. We also show that Yb-4f electrons have no contribution on the hybridization because 4f14 state is very stable. We feel that these sys- tems can be made, and we encourage theoretic and expe- rimental search to synthesize these RE atoms doped hy- drogen storage nanomaterials. REFERENCES [1] National Institute for Materials Science (NIMS), Presi- dent’s Office, Planning Division. Materials Outlook for Energy and Environment; National Institute for Materials Science (NIMS): Japan, p 64, 2008. [2] M cAfee JL, Poirier B, J Chem Phys 130:064701, 2009. [3] Yoon M, Yang SY, Hicke C, Wang E, Geohegan D, Zhang ZY, Phys Revs Lett 100:206806, 2008. [4] Dillon AC, Jones KM, Bekkedahl TA, Kiang CH, Be- thune DS, and Heben MJ, Nature (London) 386:377, 1997. [5] Rosi NL, Eckert J, Eddaoudi M, Vodak DT, Kim J, Keeffe MO, Yaghi M, Science 300: 1127,2003. [6] K aye SS, Long JR, J Am Chem Soc 127:6506, 2005. [7] Lee H, Ihm J, Cohen ML, Louie SG, Phys Rev B 80:115412, 2009. [8] Ye Y, Ahn CC, Witham C, Fultz B, Liu J, Rinzler AG, Colbert D, Smith KA, Smalley RE, Appl Phys Lett 74:2307,1999. 11  H. Lei ET AL. Copyright © 2013 SciRes. MNSMS [9] Li u W, Zhao YH, Li Y, Jiang Q, Lavernia EG, J Phys Chem C 113:2028,2009. [10] Chandrakumar KRS, Ghosh SK, Nano Lett 8:13, 2008. [11] Sun Q, Jena P, Wang Q, Marguez M, J Am Chem Soc 128:9741, 2006. [12] Kim YH, Zhao Y, Williamson A, Heben MJ, Zhang SB, Phys Rev Lett 96:016102, 2006. [13] Park N, Hong S, Kim G, Jhi SH, J Am Chem Soc 129:8999, 2007. [14] Dag S, Ozturk Y, Ciraci S, Yildirim T, Phys Rev Lett 94:155504,2005. [15] Yildirim T, Ciraci S, Phys Rev Lett 94:175501, 2005. [16] Lee H, Choi WI, Ihm J, Ph ys Rev Lett. 97:056104, 2007. [17] Zhang ZW, Li JC, Jiang Q, J Phys Chem C 114:7733, 2010. [18] Delley B, J Chem Phys 92:508, 1990. [19] Delley B, J Chem Phys 113:7756, 2003. [20] Wan g XX, Li XY, Li HN, Phys Lett A 372:6677, 2008. [21] Perd ew JP, Burke K, Ernzerhof M, Phys Rev Lett 77:3865, 1996. [22] Monkhorst HJ, Pack JD, Phys Rev B 13:5188, 1976. [23] Gao Y, Zeng XC, J Phys: Condens Matter 19:386220, 2007. [24] Cobian M, Iniguez J, J Phys: Condens Matter 20: 285212, 2008. [25] Henwood D, Carey JD, Phys Rev B 75:245413, 2007. [26] Ferre-Vilaplana A, J Chem Ph y s 122:104709, 2005. [27] Wu XJ, Gao, Y, Z eng XC, J Phys Chem C 112: 8458, 2008. [28] Hiran o K, Kadono J, Yamamoto S, Tanabe T, Miyake H, J Alloy Compd 408-412:351, 2006. [29] Yoshihiro N, Kadono J, Nishiuchi S, Yamamoto S, Ta- nabe T, Miya ke H, J Alloy Compd 408–412:355, 2006 12 |

