Paper Menu >>
Journal Menu >>
 Engineering, 2012, 5, 84-87 doi:10.4236/eng.2012.410B022 Published Online October 2012 (http://www.SciRP.org/journal/eng) Copyright © 2012 SciRes. ENG Partial Reflection of Hyal uronan Molecul es Inside the Taylor-cone During Electrospray Zhikai Tan School of Biology, Hunan University, Changsha, Hunan, China Received 2012 ABSTRACT Electrospray (ES) is of great interest in research for its finely controlled delivery of molecules. This study investigated mechanisms involved in the electrospray of biological macromolecules which may cause spraying instabilities. Hyaluronan (HA) has been studied for its biological significance. A mixture of ethylene glycol and deionized water with volume ratio of 1:1 is used to dissolve HA mo- lecules. HA solutions with different concentrations and molecular sizes are investigated. Experimental results demonstrate that the molecular size and solution concentration of macro-molecules are critical factors affecting the spraying process. A concentration reduction of HA molecules happened during the ES process, and a hypothesis on partial reflection of HA molecules inside the Tay- lor-cone is pre sented i n the study. Keywords: Electro s pray; Macro-molecules; Reflection; Talor-cone 1. Introduction With the emergence of nanotechnology, researchers become more interested in the electrospray (ES) technique in recent years due to its versatility and potential for applications in di- verse fields [1,2]. By applying a suitable voltage to a conduct- ing liquid supplied into a capillary, the liquid meniscus will take a form of cone [3,4]. Further increasing the electric field on the surface of the liquid to overcome the surface tension, a controlled μm-sized jet will emerge from the tip of the liquid cone whi ch i s called the st abl e cone-jet mode [3]. Consequently, by manipulating the microscopic fluid jet and ejected charged droplets onto a desired place, an e-jet del ivery or d eposit ion can be achi eved. By using micro-sized fine nozzles, ES can oper ate at a low flow rate, which accordingly makes t he delivery of tiny volumes possible [5]. The electrospray of macromolecules has seen a tremendous increase in research and commercial attention over the past decade due to a number of applications such as in fabrication of enzyme elect ro des [ 6] , micro-cel ls for bi och emical reacti on s [7 , 8] and thin polymer films [9]. In this study, molecules of Hya- luronan (HA, i.e. hyaluronic acid) are chosen to be investigated due to its unique rheological properties and complete biocom- patibility. As a naturally occurring linear polysaccharide, HA is popularly found in connective tissues such as umbilical cord, synovial fluid and vitreous etc. Meanwhile, HA has been widely used in fields of drug delivery, cell encapsulation and tissue r egeneration [10]. The chemical stru cture of H A consists of alternating disaccharide units of D-glucuronic acid and N-acetyl-D-glucosamine. Molecular weight of HA can vary from 5 kDa to 2.1 MDa in vivo which will result in varied mo- lecular s izes [11]. Increasingly considerable attentions has been received nowadays for the electrospray of HA solutions into types of microfibers or membranes [10,12]. However, how to achieve stable electrospray of HA solutions still remains problematic due to uncertain reasons. Fluid motions within the Taylor-cone and the way that carries HA macro-molecules is of some rele- vance in understanding of HA spraying jet stability controlling difficulties. These motions during electrospray are driven by the tan gential electrical stress acting o n the liquid-gas int erface and the flow rate injected through the electrified emitt er [1 3]. Based on work of Hayati, Barrero and Sheldon for liquid motions inside the Taylor-cone containing both experimental results and theoretical modelling, there is a recirculating me- ridional motion during the ES process, liquid moves towards the ap ex along th e generatr ix and away fro m it alon g th e axis as shown in Figure 1 [13-16],. Their results show that only fluid particles lying close to the surface are ejected through the jet while th e rest r ecircu lates to ward s th e apex al on g th e generat rix and away from it along the axis [13,17]. When solutions of HA macro-molecules are sprayed under stable cone-jet conditions, the anticipated molecules movement could be as shown in Fig- ure 1. HA molecules move following liquid towards the apex and away from it along the axis [15,16]. Figure 1. Streamlines and movements of HA molecules within the Taylor-cone during the electrospray: Meridional circulation of liquid with molecules towards the apex along the generatrix and away fr om th e apex along the axis .  Z. K. TAN Copyright © 2012 SciRes. E NG 85 During the process, only that fraction of HA molecules lying close to the surface are ejected through the jet while the rest recirculated towards the apex away from it along the axis [13, 17]. This recirculation may cause a back diffusion movement of HA molecules, and with a higher HA concentration nears the tip which would result in partial reflection of HA molecules durin g the electr ospray. It is possible then that this process may cause in stabilities t o the ES process and a con centration reduc- tion of HA molecules after t he electrospray process. 2. Materials and Methods Hyaluronan powders (sodium salt, molecular weight: 2.1 MDa) were purchased from Genzemy Co. (USA) and diluted in eth- ylene glycol and water (EG/H2O, volume ratio=1) with differ- ent concentrations (0.1, 0.2 and 0.4 mg/ml). A sonication method was used to r educe the mo lecular si ze and chain length of H A [ 18]. After so nicati on, average molecul ar weights o f H A were measured (200 kDa, 390 kDa and 1000 kDa) by size- exclusion high performance liquid chromatography (HPLC). In order to examine the partial reflection of HA molecules, experiments were undertaken to investigate whether the elec- trospray process was acting as a filter of HA molecules or not. The hypothesis is that if the partial reflection takes place then downstream of the Taylor cone, liquid collect from the jet end may have a lower concentration of HA molecules than in the initial fluid solution. Figure 2 shows the experimental sche- matic of the ES apparatus for the study. The main components consist of fluid supply system, spraying nozzle, high voltage power supply, current measuring unit and an imaging system to monitor the spraying cone and jet. Nozzles were made from glass tubes. A micropipette puller P-97 (Sutter Instrument Co.) was used to produce tips of noz- zles down to 30 μm. Liquid contained in a small reservoir was forced through a narrow silica tube into the nozzle by pressur- ized nitrogen gas. All experiments were completed at atmos- pheric conditions at the emitter tip, with the same initial fixed pressure. The change in pressure as the reservoir drained was insignificant, as each spray used less than 0.5 ml, which re- sulted in a change in heigh t less than 1 mm. The power supply (F.u.G. Electronik) can provide a voltage up to 6 kV. The spray distance between the nozzle tip and the Figure 2. Expe rimen t al schemat ic for el ect rospr a y of HA s olutions. substrate was set to 3 mm. A voltage ranged in 2.3 ~ 2.5 kV was applied to produce a stable co ne-jet electrospray. The slight adjustment of the applied voltage was made to maintain a spraying due to different physical properties of solutions. The current was determined by measuring the voltage drop across a 100MΩ resistor. An ISO-TECH IDM 207 voltmeter logged the voltage to a PC at a freq uency of 2 Hz. The jet during an electrospray process was monitored by an optical system based on a microscope. Images were recorded and captured by a computer. The system consists of a combined zoom lens and a CCD camera (UEYE). An infinity corrected objective lens from Mitatoyo and a variable zoom from Thales Optem were used. The optical resolution can reach to 1μm. Additionally, a cold light source was used for illumination. 3. Results and Discussion Figure 3 shows optical micrographs of spraying pure EG/H2O solvent and HA solutions (molecular weight: 1000 kDa) with concentrations of 0.1, 0.2 and 0.4 mg/ml. Respectively, stable spraying jets were observed in HA solutions with concentra- tions of 0, 0.1, and 0.2 mg/ml. However, further double the concentration up to 0.4 mg/ml the stable jet was not observed, which clearl y indicated that the stable electrospray process can be hampered by adding HA macromolecules. Varied concentrated HA solutions (Cin) with different mo- lecular sizes were electrosprayed at stable cone-jet mode for further investigations. Solutions of HA 1000 kDa molecules were only be cone-jet electrosprayed when Cin < 0.4 mg/ml while other smaller HA molecules (200 kDa and 390 kDa) could reach the stable cone-jet electrospray at the highest con- centrati on. Molecular si ze and concentration do affect th e elec- trospray stability of HA solutions. Samples of liquid from downstream spraying jet were col- lected every 20 minutes from the plate electrode after cone-jet electrospray have been reached, and then concentrations of samples (Cout) were measured. Concentration change R was calculat ed as following: Figure 3. Microphotography images showing the electrospray of solutions: a) electrospray of pure EG/H2O solvent. b)-d) electros- pray of HA (1000 kDa) solutions with a concentration of 0.1, 0.2 and 0.4 mg/ml, respectively. 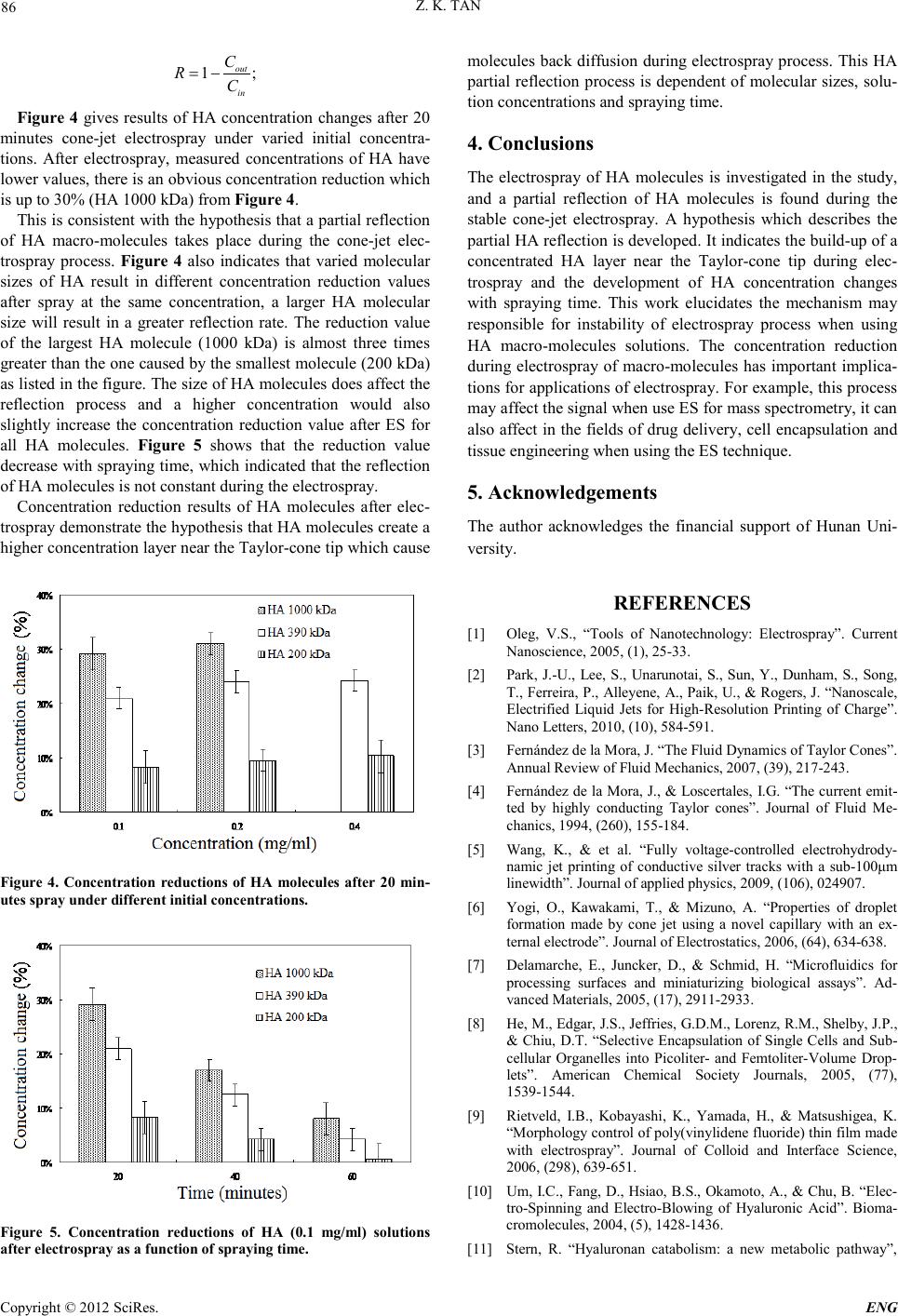 Z. K. TAN Copyright © 2012 SciRes. ENG 86 1; out in C RC = − Figure 4 gives results of HA concentration changes after 20 minutes cone-jet electrospray under varied initial concentra- tions. After electrospray, measured concentrations of HA have lower values, there is an obvious concentration reduction which is up to 30% (HA 1000 kDa) from Figure 4. This is consistent with the hypothesis that a partial reflection of HA macro-molecules takes place during the cone-jet elec- trospray process. Figure 4 also indicates that varied molecular sizes of HA result in different concentration reduction values after spray at the same concentration, a larger HA molecular size will result in a greater reflection rate. The reduction value of the largest HA molecule (1000 kDa) is almost three times greater th an the o ne cau sed by the smallest molecu le (20 0 kDa) as listed in the figure. The size o f HA molecules do es affect the reflection process and a higher concentration would also slightly increase the concentration reduction value after ES for all HA molecules. Figure 5 shows that the reduction value decrease with spraying ti me, which indi cated that the reflect ion of HA molecules is not constant during the electrospray. Concentration reduction results of HA molecules after elec- trosp ray demonstrat e the hypo thesis th at HA molecules create a higher con cen tratio n layer near th e Tayl or-cone tip which cause Figure 4. Concentration reductions of HA molecules after 20 min- utes spray under differe nt initial concentrations. Figure 5. Concentration reductions of HA (0.1 mg/ml) solutions after elect r o sp ra y as a fu n ct ion o f s p r a ying time. molecules back diffusion during electrospray process. This HA partial reflection pro cess is depend ent of molecular sizes, so lu- tion concent rations and spraying time. 4. Conclusions The electrospray of HA molecules is investigated in the study, and a partial reflection of HA molecules is found during the stable cone-jet electrospray. A hypothesis which describes the partial HA reflectio n is develo ped. It indicates the build-up of a concentrated HA layer near the Taylor-cone tip during elec- trospray and the development of HA concentration changes with spraying time. This work elucidates the mechanism may responsible for instability of electrospray process when using HA macro-molecules solutions. The concentration reduction during electrospray of macro-molecules has important implica- tions for applications of electrospray. For example, this process may affect t he signal when use E S for mass spectrometry, it can also affect in the fields of drug delivery, cell en capsulation and tissue engineering when using the ES technique. 5. Acknowledgements The author acknowledges the financial support of Hunan Uni- versity. REFERENCES [1] Oleg, V.S., “Tools of Nanotechnology: Electrospray”. Current Nanosc ie nce , 20 0 5, ( 1), 2 5-33. [2] Park, J.-U., Lee, S., Unarunotai, S., Sun, Y., Dunham, S., Song, T., Ferreira, P., Alleyene, A., Paik, U., & Rogers, J. “Nanoscale, Electrified Liquid Jets for High-Resolution Printing of Charge”. Nano Letters, 2010, (10), 584-591. [3] Fernández de la Mora, J. “The Fluid Dynamics of Taylor Cones”. Annu al R e view of Fluid Me c h anics, 20 0 7, (39) , 2 17-243. [4] Fer nández d e la Mora, J. , & Loscert ales, I.G. “The c urrent emit- ted by highly conducting Taylor cones”. Journal of Fluid Me- chani cs, 1994, (260), 155-184. [5] Wang, K., & et al. “Fully voltage-controlled electrohydrody- namic jet printing of conductive silver tracks with a sub-100μm linewid th”. Journal of applied physics, 2009, (106 ), 024907. [6] Yogi, O., Kawakami, T., & Mizuno, A. “Properties of droplet formation made by cone jet using a novel capillary with an ex- ternal electrode”. Journal of Electrostatics, 2006, (64), 634-638. [7] Delamarche, E., Juncker, D., & Schmid, H. “Microfluidics for processing surfaces and miniaturizing biological assays”. Ad- vanced Materials, 2005, (17), 2911-2933. [8] He, M., Edgar, J.S., Jeffries, G.D.M., Lorenz, R.M., Shelby, J.P., & Chiu , D.T. “Selective Encap sulation of Single Cells and Sub- cellular Organelles into Picoliter- and Femtoliter-Volume Drop- lets”. American Chemical Society Journals, 2005, (77), 1539-1544. [9] Rietveld, I.B., Kobayashi, K., Yamada, H., & Matsushigea, K. “Morphology control of poly(vinylidene fluoride) thin film made with electrospray”. Journal of Colloid and Interface Science, 2006, (298), 639 -651. [10] Um, I.C., Fang, D., Hsiao, B.S., Okamoto, A., & Chu, B. “Elec- tro-Spinning and Electro-Blowing of Hyaluronic Acid”. Bioma- cromolecules, 2004, (5), 1428-1436. [11] Stern, R. “Hyaluronan catabolism: a new metabolic pathway”,  Z. K. TAN Copyright © 2012 SciRes. E NG 87 European Journal of Cell Biolog y, 2004, ( 83), 317-325. [12] Ji, Y., Ghosh, K., Shu, X.Z., Li, B., Sokolov, J.C., Prestwich, G.D., Clark, R.A.F., & Rafailovich, M.H. “Electrospun three-dimensional hyaluronic acid nanofibrous scaffolds”. Bio- materials, 2006, (27), 3782-3792. [13] Barrero, A., Gañán-Calvo, A.M., Dávila, J., Palacio, A., & Gómez-González, E. “Low and high Reynolds number flows in- side Taylor cones”. Physical Review E, 1998, (58), 7309. [14] Hayati, I., Bailey, A.I., & Tadros, T.F. “Mechanism of stable jet formation in electrohydrodynamic atomization”. Nature, 1986, (319), 41-43. [15] Hayati, I., Bailey, A.I., & Tadros, T.F. “Investigations into the mechanisms of electrohydrodynamic spraying of liquids : I. Ef- fect of electric field and the environment on pendant drops and factors affecting the formation of stable jets and atomization”. Journ al of Colloid and Interface Sci enc e, 1987, (117), 205-221. [16] Sheldon, A.M. “Visualization of fluid motions in Taylor Cones using dye tracer”. Queen Mary, University of London, London. 2009 [17] Barrero, A., Ganan-Calvo, A.M., & Fernandez-Feria, R. “he role of liquid viscosity and electrical conductivity on the motions in- side Taylor cones in E.H.D. spraying of liquids”. Journal of Aerosol Scienc e, 1996, (27), S175 -S176. [18] Coleman, P.J., Scott, D., Mason, R.M., & Levick, J.R. “Role of hyaluronan chain length in buffering interstitial flow across syn- ovium in rabbits”. Journal of Physiology, 2000, (526), 425-434. |

