Paper Menu >>
Journal Menu >>
 Engineering, 2012, 5, 27-29 doi:10.4236/eng.2012.410B007 Published Online October 2012 (http://www.SciRP.org/journal/eng) Copyright © 2012 SciRes. ENG Efficacy and Safety of Nasal Administration of “Na- no-L-DOPA” Based on PLGA Nanoparticles I. G. Kondr asheva1, T. A. Antipova1, G. G. Barsegyan1, P. Ye. Gambaryan2, A. A. Guseva2, A. A. Kamensky2 1Research Center of Molecular Diagnostics & Therapy, Moscow, Russia 2Moscow State University, Moscow, Russia Email: kondrashevl@list.ru Received 2012 ABSTRACT Inclusion of L-DOPA , th e standard Parkinson’s disease medication, into polymeric particles (PLGA) results in optimization the drug metabol ism and increas ing its bioavailability, significantly increases of physical endurance, better coordination and lower anxiety in Wistar rats, when chronical ly administ er ed nasal ly. Keywords: Nanopaticls; L-DOPA; Nasal Administration; Parki nson’s Disease 1. Introduction Nasal administration of medications is a well known and cur- rently widely applicable method of administration of a number of broad-spectrum drugs, both locally and systemically acting, the interest in which, as a non-invasive method of administra- tion drugs, deepens on [1]. Due to vascular plexuses in nasal cavity and a large absorption area, nasal administration of me- dications becomes a prospective method of delivering drugs directly into the blood stream, promoting fast achievement of the effective drug level in blood. Additional benefits of nasal administration are ease of performing, usability for patients, and a relatively low cost. Moreover, drugs can be transported from nasal cavity into the central nervous system (CNS) with- out involving the blood circulatory system of nasal-cavity lin- ing mucosa, b y extra cell ul ar way – alon g th e tri facial ner ve an d the olfactory nerve. Olfactory nerve sheathes do not have blood-brain barriers, and the drug can directly enter the brain [2-5]. This fact is the focus of attention, since it provides new opportunities in treating CNS diseases, particularly neurodege- nerative brain diseases, such as Parkinson’s disease or Alzhei- mer’s disease [6]. The “nose-brain” way leads to practically immediate delivery of some drugs into the cerebrospinal fluid, bypassing blood and the blood-brain barrier (BBB) [7-10]. L-3,4-dihydroxyphenylalanine (L-DOPA) is the precursor of dopamine that is responsible for the central mechanism of mo- tion control and coordination. Applying L-DOPA in the treat- ment of Parkinson’s disease has a number of disadvantages. Optimizing the drug metabolism and increasing its bioavaila- bility and potency are possible, if it is included into nanopar- ticles based on poly(lactic-co-glycolic acid) (PLGA), a biode- gradable and non-toxic polymer. Including a drug into the par- ticles allows modulating the biopharmaceutical properties of the medications administered, their bioavailability and, as a consequence, their potency [11]. Moreover, nanocontainers protect the drug components against chemical/enzymatic de- gradation, provide durable release, decrease the drug toxicity for peripheral organs, and represent drug containers suitable for treating the CNS diseases [12]. The purpose of this research is the estimation of efficacy and safety of the nasal administration of L-DOPA, the precur- sor of the dopamine neurotransmitter, in form of a nanoparticle medication (“Nano-L-DOPA”) developed on the basis of poly(lactic-co-glycolic acid) (PLGA) for modulating (increas- ing) the exercise performance o f rats Wistar, as compared to the efficacy and safet y of L-DOPA, the initial standard medication. 2. Materials and Methods In this paper, L-DOPA (Sigma, China), 1 mg/kg, and “Nano-L- DOPA” (Resear ch Cen ter of Molecul ar Diagn o stics & Th erapy, Russia), 1 mg/kg, 5 and 10 mg/kg (by L-DOPA), were re- searched. Particle sizes of the latter one varied from 250 through 400 nm, while the L-DOPA substance content within the “Nano-L-DOPA” composition ranged from 9.5 through 12.0% of the mass. The research in the dynamics of the drug release from the nanocomposition in a model experiment in vitro testifies that up to 70% of L-DOPA contained in the par- ticles are released within the first 30 minutes, while the resting 30% o f the active substance are r eleased withi n the subsequent 10 hours. The solutions of L-DOPA were prepared i mmediately prior to the start of the experiment. Normal saline solution was used as a dissolution medium, ascorbic acid (1%, pH 2.6) being added for bettering the dissolubility and preventing the drug from oxidation on dissolving. Animals in the Baseline were administered with blank particles in the amount corresponding with the maximum dose of the nano-medicatio n. In the resear ch, male Wistar rats weighing 200-250 g and BALB/c mice (20 g) were used. The drug influence upon the exercise performance, the mo- tion activity and the behavior of the laboratory animals in the long-term experiment was researched comprehensively using a number of tests. 1) In the dynamic load conditions, in test “Forced Swim- ming” up to complete fatigue with weights (15% of their body masses), the drug influence upon the exercise performance of 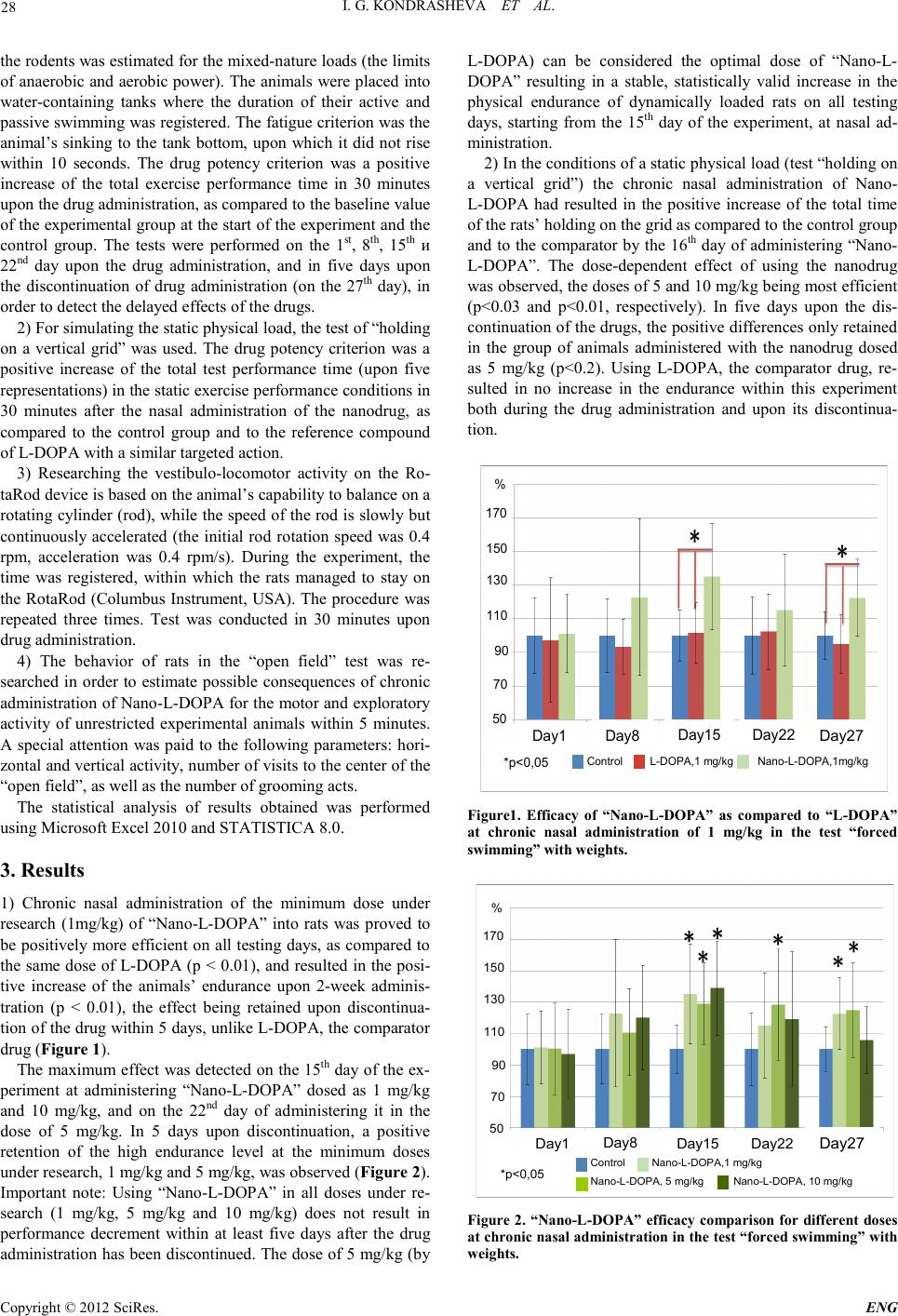 I. G. KONDRASHEVA ET AL. Copyright © 2012 SciRes. ENG 28 the rod ents was estimated fo r the mixed-nature loads (the limits of anaerobic and aerobic power). Th e animals were placed in to water-containing tanks where the duration of their active and passive s wimming was register ed. The fatigu e criterio n was th e animal’s sinking to the tank bottom, upon which it did not rise within 10 seconds. The drug potency criterion was a positive increase of the total exercise performance time in 30 minutes upon the drug administration, as compared to the baseline value of the experimental group at the start of the experiment and the control group. The tests were performed on the 1st, 8th, 15th и 22nd day upon the drug administration, and in five days upon the discontinuation of drug administration (on the 27th day), in order to detect th e delayed effects of th e drugs. 2) For simulating the static physical load, the test of “holding on a vertical grid” was used. The drug potency criterion was a positive increase of the total test performance time (upon five representatio ns) in the stati c exercise performance co nd itions in 30 minutes after the nasal administration of the nanodrug, as compared to the control group and to the reference compound of L-DOP A with a similar targeted action. 3) Researching the vestibulo-locomotor activity on the Ro- taRod device is based on the animal’s capab il ity to bal ance on a rotating cylinder (rod), while the speed of the rod is slowly but continuously accelerated (the initial rod rotation speed was 0.4 rpm, acceleration was 0.4 rpm/s). During the experiment, the time was registered, within which the rats managed to stay on the RotaRod (Columbus Instrument, USA). The procedure was repeated three times. Test was conducted in 30 minutes upon drug administration. 4) The behavior of rats in the “open field” test was re- searched in order to estimate possible consequences of chronic administration of Nano-L-DOPA for the motor and exploratory activity of unrestricted experimental animals within 5 minutes. A special attention was paid to the following parameters: hori- zontal and vertical activity, number of visits to the center of the “open field”, as well as the number of grooming acts. The statistical analysis of results obtained was performed using Microsoft Excel 2010 and STATISTICA 8.0. 3. Results 1) Chronic nasal administration of the minimum dose under research (1mg/kg) of “Nano-L-DOPA” into rats was proved to be positively more effi cient on all test ing days, as co mpared to the same dose of L-DOPA (p < 0.01), and resulted in the posi- tive increase of the animals’ endurance upon 2-week adminis- tration (p < 0.01), the effect being retained upon discontinua- tion of the drug within 5 days, u nlike L-DOP A, the comparato r drug (Figure 1). The maxi mum effect was detecte d on the 15th day of the ex- periment at administering “Nano-L-DOPA” dosed as 1 mg/kg and 10 mg/kg, and on the 22nd day of administering it in the dose of 5 mg/kg. In 5 days upon discontinuation, a positive retention of the high endurance level at the minimum doses under research, 1 mg/kg and 5 mg/kg, was observed (Figure 2). Important note: Using “Nano-L-DOPA” in all doses under re- search (1 mg/kg, 5 mg/kg and 10 mg/kg) does not result in performance decrement within at least five days after the drug administration has been discontinued. The dose of 5 mg/kg (by L-DOPA) can be considered the optimal dose of “Nano-L- DOPA” resulting in a stable, statistically valid increase in the physical endurance of dynamically loaded rats on all testing days, starting from the 15th day of the experiment, at nasal ad- ministration. 2) In the conditions of a static physical load (test “holding on a vertical grid”) the chronic nasal administration of Nano- L-DOPA had resulted in the positive increase of the total time of the rats’ holding on the grid as compared to the control group and to the comparator by the 16th day of administering “Nano- L-DOPA”. The dose-dependent effect of using the nanodrug was observed, the doses of 5 and 10 mg/kg being most efficient (p<0.03 and p<0.01, respectively). In five days upon the dis- continuation of the drugs, the positive differences only retained in the group of animals administered with the nanodrug dosed as 5 mg/kg (p<0.2). Using L-DOPA, the comparator drug, re- sulted in no increase in the endurance within this experiment both during the drug administration and upon its discontinua- tion. Day1 Day8 Day15 Day22 Effect Control L-DOPA,1 mg/kg Nano-L-DOPA,1mg/kg 170 150 130 110 90 70 50 % *p<0,05 Day1 Day8 Day15 Day22 Effect Control L-DOPA,1 mg/kg Nano-L-DOPA,1mg/kg 170 150 130 110 90 70 50 % 170 150 130 110 90 70 50 % *p<0,05 Day27 Day1 Day8 Day15 Day22 Effect Control L-DOPA,1 mg/kg Nano-L-DOPA,1mg/kg 170 150 130 110 90 70 50 % *p<0,05 Day1 Day8 Day15 Day22 Effect Control L-DOPA,1 mg/kg Nano-L-DOPA,1mg/kg 170 150 130 110 90 70 50 % 170 150 130 110 90 70 50 % *p<0,05 Day27 Figure1. Efficacy of “Nano-L-DOPA” as compared to “L-DOPA” at chronic nasal administration of 1 mg/kg in the test “forced swimming” with weights. 170 150 130 110 90 70 50 % Day1 Day8 Day15 Day22 Effect Control Nano-L-DOPA,1 mg/kg Nano-L-DOPA, 5 mg/kg Nano-L-DOPA, 10 mg/kg *p<0,05 Day27 170 150 130 110 90 70 50 % Day1 Day8 Day15 Day22 Effect Control Nano-L-DOPA,1 mg/kg Nano-L-DOPA, 5 mg/kg Nano-L-DOPA, 10 mg/kg *p<0,05 Day27 Figure 2. “Nano-L-DOPA” efficacy comparison for different doses at chronic nasal administration in the test “forced swimming” with weig h ts.  I. G. KONDRASHEVA ET AL. Copyright © 2012 SciRes. E NG 29 3) The research in the vestib ulo-locomotor activity of Wistar rats using RotaRod in the conditions of chronic nasal adminis- tration of “L-DOPA” compounds within 20 days, in the minim- al daily doses of 1, 5 or 10 mg/kg, did not show any distur- bances in locomotor activity or any spatial disorientation in the animals. Administering “Nano-L-DOPA” in all doses under research promoted the positive improvement of the animals’ rotarod performance and sp atial orientation ability. The largest values o f RotaRod p erformance had reached sta- tistically valid differences in all group s of animals admin istered with the nanodrug, as compared to the control group (p>0.1) and to the group administered with standard medication L-DOPA (p>0.2), by the end of the experiment (the 20th day o f nasal administration). The effect observed was dose-indepen- dent and had practically identical representation for all nano- drug doses used. The effect above retained even 6 days upon the nanodrug discontinuation in the groups of animals ad mi ni s- tered with the doses of 1 or 5 mg/kg. Thus, the identified bet- tering the coordination of spatial motions and of the animals’ spatial orientation ability is the result of using “L-DOPA” in a prolonged form. 4) When studying the drugs influence upon the animals’ motor performance and their exploratory activities in the “Open Field” test, the chronic nasal administration of L-DOPA com- pounds in all doses used was not detected to influence on test results, such as total distance, number of postures and visits to the center of the field. At th e same time, it is important to note the disappearance of grooming acts by the end of experiment for all drugs under research, but with the high degree of confi- dence – in the case of “Nano-L-DOPA” only. This fact may testify the decrease in emotional reactivity/anxiety, as well as the increase in the decision making processes in experimental animals upon the chronic nasal administration of “Nano-L- DOPA” and, to a smaller extent, L-DOPA. Thus, the comprehensive preclinical efficacy of the nasal administration of “Nano-L-DOPA” showed that, in the condi- tions of a chronic experiment, in the background of the pro- longed action of “Nano-L-DOPA”, a long-term increase in the animals’ endurance under the dynamic and static loads had been d etected. Of th e doses resea rched, t he optimal do ses are 1 mg/kg and 5 mg/kg (by L-DOPA), where the performance in- creases by maximum (up to 35% of the background values), the positively significant effect being retained even after the dis- continuation of the administration. The results obtained allow us to consider “Nano-L-DOPA” a prospective compound for nasal administration as a drug influencing the performance. Comparative studies of general toxicity (acute, subacute) at the intranasal administration of the “Nano-L-DOPA” composi- tion, conducted on 2 animal species of both genders (BALB/c mice and Wistar rats) within the doses experimented did not show any toxic actions of “Nano-L-DOPA”. In acute toxicity studies, the intranasal (repeated, at short intervals) administra- tion of “Nano-L-DOPA” in a maximum species-relevant vo- lumes and doses was established not to be accompanied by lethal outcomes for the animals under test. The highest sum- mary dose of “Nano-L-DOPA” reached at repeated intranasal administration made 540 mg/kg in BALB/c mice (by 10 mcl in each nostril, every 5 minutes) and 150 mg/kg in Wistar rats (by 20 mcl in each nostr il, every 5 min ut es), resp ectivel y. The sa me procedure of L-DOPA intranasal administration did not result in any mortality of the animals under test, either. However, transient toxic manifestations were detected at positively smaller doses and were more evident than in administering “Nano-L-DOPA”. In subacute toxicity studies, the 7-day intranasal “Nano-L- DOPA” administration in doses under research (rats 20 mg/kg, 50 mg/kg) were not accompanied by any lethal outcomes or side effects during the administration period and within 14-day observations after the last administration. The results of hema- tologic and morphometric studies, pathomorphological and pathohistological examinations did not detect any essential differences in organs and tissues of the animals under test, as compared to those of control animals. The results obtained in the studies allowed us to conclude that the intranasal adminis- tration of “Nano-L-DOPA” was a relatively safe way of admi- nistering the drug under development. The results obtained allow us t o forecast t hat th e intran asal way of ad ministering the “Nano-L-DOPA” will be prospective for further clinical man- agement thereo f. REFERENCES [1] A. Pires, A. Fortuna, G. Alves, A. Falcao Intranasal Drug De- livery: Ho w, Why and Wha t for? J Pharm Pharmaceut Sci, 2009, vol. 12(3) pp. 288 - 311 [2] Graff CL, Pollack GM. Nasal drug administration: Potential for ta rgeted cent ral nervou s s ystem d eliv er y. J Ph arm Sci, 2005, vol. 94, pp. 1187–1195. [3] Illum L. Transport of drugs from the nasal cavity to the central nervous system. Eur J Pha rm Sci, 2000, vol. 11, pp 1–18. [4] Illum L. Is nose-to-brain transport of drugs in man a reality. J Pharm Ph armacol, 2004, vol. 56, pp. 3–17. [5] Dah lin M, Jan sson B , Bj ork E. . Level s of d op amine in blood and brain following nasal administration to rats. Eur J Pharm Sci, 2001, vol. 14. pp. 75–80. [6] M.Fazil, Shadab, S. Baboota, JK Sahni, J Ali Nanotherapeutics for Al zh ei mer' s d i s eas e (A D): p a s t, pr esen t and future, Journal of Drug Targeting, 2012, vol. 20 (2), pp. 97-113. [7] Henry, R.J., Ruano N, Casto D, Wolf RH, A pharmacokinetic study of midazolam in dogs: nasal drop vs. atomizer administration. Pediatr Dent, 1998, vol. 20(5) , p p. 3 21-326. [8] Sakane, T., Akizuki M, Yoshida M, Yamashita S, Nadai T, Hashida M, Sezaki H. Transport of cephalexin to the cerebrospinal fluid directly from the nasal cavity. J Pharm Pharm acol, 1991, vol. 43(6), pp. 449 -451. [9] Banks, W.A., M.J. During, and M.L. Niehoff Brain uptake of the glucagon-like peptide-1 antagonist exendin (9-39) after intranasal administration. J Pharmacol Exp Ther, 2004, vol. 309(2), pp . 469-475. [10] Westin, Bost rom E, Gr as j o J , Hammarlund-Uden a es M , Bj örk E. Direct nose-to-brain transfer of morphine after nasal admin istrat ion to rats. Pharm Res, 2006, vol. 23(3), pp. 565-572. [11] Kreuter J. Nanoparticulate system for brain delivery of drags. Adv Drug Deliv R ev, 20 01, vol. 47, pp. 65-81. [12] Hall JB, Dobrovolskaia MA, Patri AK, McNeil SE. Characteri- zation of nanoparticles for therapeutics. Nanomedicine (Lond). 2007, Dec, vol. 2( 6), pp. 789 -803 |

