Paper Menu >>
Journal Menu >>
 Engineering, 2012, 5, 8-10 doi:10.4236/eng.2012.410B003 Published Online October 2012 (http://www.SciRP.org/journal/eng) Copyright © 2012 SciRes. ENG Red Blood Cell Mechanical Stability Oguz K . Baskurt Koc University School of Medicine, Rumelifeneri Kampusu, Sariyer, Istanbul, Turkey Email: obaskurt@ku.edu.tr Received 2012 ABSTRACT It has been well documented that shear forces effective above a certain magnitude under flow conditions causes damage in blood cells. This damage ranges from slight morphological alteration to the destruction of red blood cells (RBC). The hemolytic trauma to RBC can easily be detected by measuring free hemoglobin level in plasma, however there are no standardized protocols to quantitate subh emolytic trau ma. Ektacyto metr y has been used to study the alt eration s in RBC mechan ical pr operties i nduced by the appl ication of shear stress at various levels. Additionally, a protocol for measuring the hemolytic threshold as an indicator of subhemolytic dam- age to RBC has been developed based on ektacytometry. These standardized protocols may find applications in the pre-clinical and clini cal evaluati on of artifici al organs and biomedical devices contacting with blood. Keywords: Artificial Organs; Blood D a mage; Su bhemolytic Damage; Ektacytometry 1. Introduction Red blo od cells (RBC) are exp osed to mechanical st resses dur- ing their “everyday life”, at various levels of the circulatory s ys t em. They respond to this effect by changing their shape and orientation to the flow conditions; but they are not overtly damaged by these forces, under physiological conditions. However, if they are exposed to non-physiological conditions, like in case of circulating through artificial organs or devices, they might be affected resulting in deteriorations at various degrees, ranging from slight morphological alterations to the rupture of their membrane resulting in hemolysis [9]. The most effective factors under such conditions include the extremely high shear forces to which RBC are exposed to dur- ing their passage through the artificial flow environments [5, 9]. Additionally, turbulence, cavitation, contact of blood cells with artificial su rfaces, and collisi on between cells or b etweens cells and various parts of the device may also damage the cellular components of blood [5, 9]. Shear forces in the human arterial system are in the range of 1-10 Pascal (Pa) [7], while in artifi- cial circulatory environments (e.g., ventricular assist devices, total artificial heart, extracorporeal circulation systems, heart valves, stents, hemodialysis systems) these forces may ap- proach to several hundred Pa [5]. 2. Mechanical Trauma to Blood It is well known for many decades that shear forces exceeding a critical level may cause hemolysis [1 2]. It has been demon- strated that both magnitude and exposure time are important determinants of the hemolytic effect of shear stress [6]. Hemo- lytic damage to RBC can easily be quantitated by measuring free hemoglobin in the suspending medium of blood (plasma), which is usually expressed as the ratio to the total hemoglobin content of whole blood. This hemolysis ratio or its normalized forms are widely used in expressing the extent of mechanical trauma to blood and used for the evaluation of artificial circu- latory environments [9]. However, it should be noted that hemolysis is the end-stage damage to RBC, corresponding to the destruction of the cell. It has been documented that the mechani cal impact to RBC under the hemolytic threshold also induce alterations in the cellular properties, at varying degrees determined by the magnitude of the effectiv e forces [9,15]. It has been argued that even physio- logical shear forces u nder normal circul atory conditions induce slight changes in RBC properties, which may accumulate dur- ing the life-span of RBC (~120 days for human RBC). This “non-lethal” trauma to RBC might be enhanced if they are ex- posed to the conditions in artificial circulatory environment [9]. Subhemolytic trauma might be manifest by shortened life span [3], increased trapping i n spleen [14], morphological alte- rations [13], or deterioration in mechanical properties of the cells [2] . Subhemolytic trauma is also determined by both the magnitude and duration of the effective shear stresses [2,9 ]. RBC membrane st abili ty is affected by subh emolytic trau ma, resulting in increased mechanical fragility indicated by higher rate of hemolysis during the subsequent exposure to extreme shear forces. Various tests to determine mechanical fragil ity has been developed and proposed to be used in testing the artificial circulatory environments [4 ,8 ]. Mechanical fragility is ex- pressed as percent free hemoglobin following a standardized mechanical stress to RBC suspensions [9]. However, the mag- nitude of the mechanical impact to which RBC are exposed cannot be standardized or even accurately estimated with the propos e d pr ot oc ol s [4,8 ]. 3. Detection of Subhemolytic Damage to Red Blood Cells Accurate quantification of subhemolytic damage to RBC is important in studies aiming to evaluate the blood damage in- duce by artificial organs and biomedical devices contacting with blood. Additionally, test protocols and models based on subhemolytic levels of mechanical trauma to RBC may help 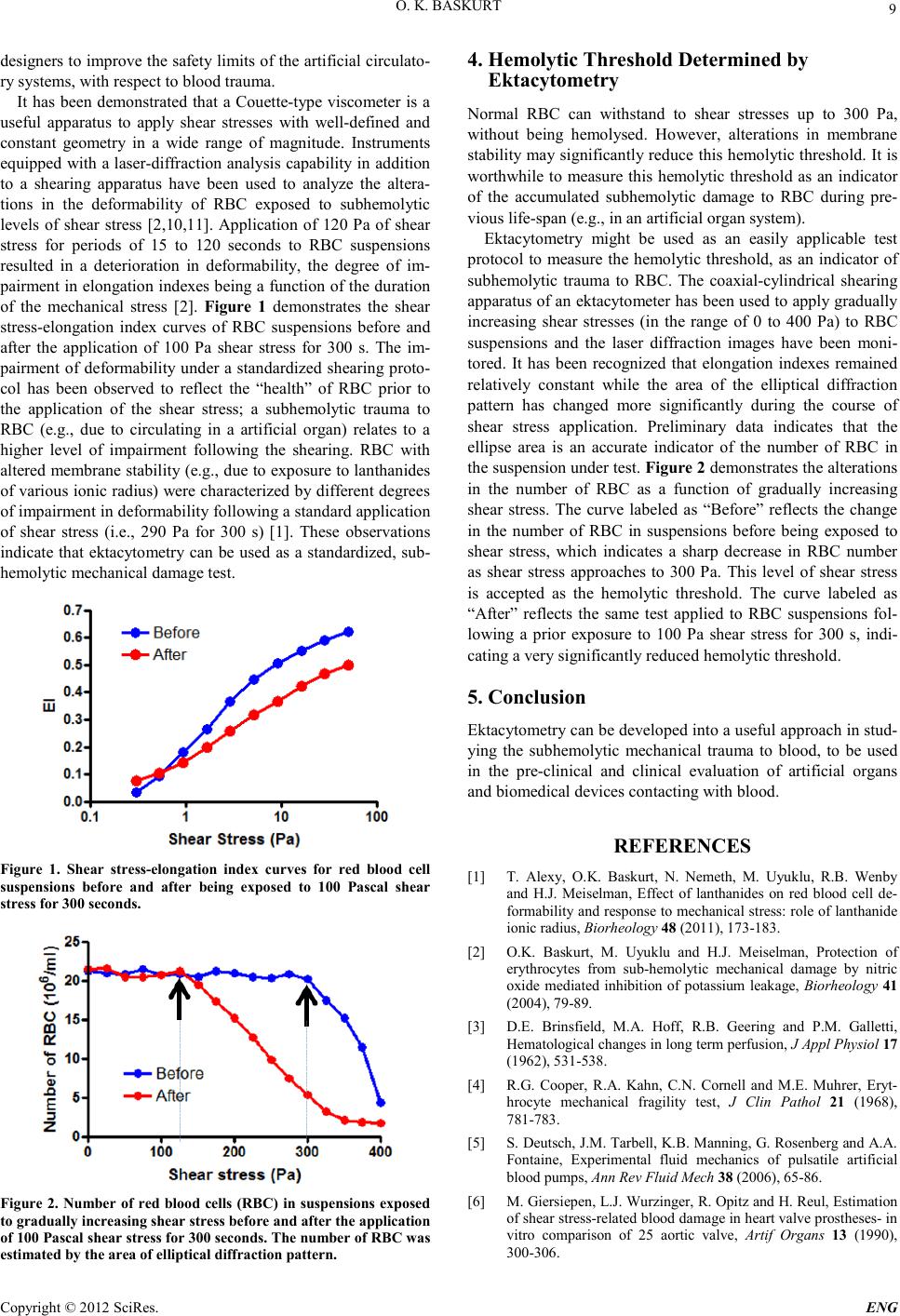 O. K. BASKURT Copyright © 2012 SciRes. E NG 9 designers to improve the safety limits of the artificial circulato- ry systems, with respect to blood trauma. It has been demonstrated that a Couette-type viscometer is a useful apparatus to apply shear stresses with well-defined and constant geometry in a wide range of magnitude. Instruments equipped with a laser-diffraction analysis capability in addition to a shearing apparatus have been used to analyze the altera- tions in the deformability of RBC exposed to subhemolytic levels of shear stress [2,10,11]. Application of 120 Pa of shear stress for periods of 15 to 120 seconds to RBC suspensions resulted in a deterioration in deformability, the degree of im- pairment in elongation indexes being a function of the duration of the mechanical stress [2]. Figure 1 demonstrates the shear stress-elongation index curves of RBC suspensions before and after the application of 100 Pa shear stress for 300 s. The im- pairment of deformability under a standardized shearing proto- col has been observed to reflect the “health” of RBC prior to the application of the shear stress; a subhemolytic trauma to RBC (e.g., due to circulating in a artificial organ) relates to a higher level of impairment following the shearing. RBC with altered membran e stabili ty (e.g., du e to exposure to lanthanides of variou s ionic r adius) were charact erized b y different degree s of impairment in deformability following a standard application of shear stress (i.e., 290 Pa for 300 s) [1]. These observations indicate that ektacytometry can be used as a standardized, sub- hemolytic mechan ical damage test. Figure 1. Shear stress-elongation index curves for red blood cell suspensions before and after being exposed to 100 Pascal shear stress for 300 seconds. Figure 2. Number of red blood cells (RBC) in suspensions exposed to gradually increasing shear stress before and after the application of 100 Pa scal s hear s tress for 300 s eco nds. T he number of RB C was estimated by the area of ellipti cal d iffraction pattern. 4. Hemolytic Threshold Determined by Ektacytom etry Normal RBC can withstand to shear stresses up to 300 Pa, without being hemolysed. However, alterations in membrane stability may significantly reduce this hemolytic threshold. It is worthwhile to measure this hemolytic threshold as an indicator of the accumulated subhemolytic damage to RBC during pre- vious life-span (e.g., i n an artificial organ syste m) . Ektacytometry might be used as an easily applicable test protocol to measure the hemolytic threshold, as an indicator of subhemolytic trauma to RBC. The coaxial-cylindrical shearing apparat us of an ektacytometer ha s been used to appl y gradu ally increasing shear stresses (in the range of 0 to 400 Pa) to RBC suspensions and the laser diffraction images have been moni- tored. It has been recognized that elongation indexes remained relatively constant while the area of the elliptical diffraction pattern has changed more significantly during the course of shear stress application. Preliminary data indicates that the ellipse area is an accurate indicator of the number of RBC in the suspension under test. Figure 2 demonstrat es th e alt erati on s in the number of RBC as a function of gradually increasing shear stress. The curve labeled as “Before” reflects the change in the number of RBC in suspensions before being exposed to shear stress, which indicates a sharp decrease in RBC number as shear stress approaches to 300 Pa. This level of shear stress is accepted as the hemolytic threshold. The curve labeled as “After” reflects the same test applied to RBC suspensions fol- lowing a prior exposure to 100 Pa shear stress for 300 s, indi- cating a very significantly reduced hemolytic threshold. 5. Conclusion Ektacyto metry can b e develop ed into a useful app roach in stud- ying the subhemolytic mechanical trauma to blood, to be used in the pre-clinical and clinical evaluation of artificial organs and biomedical devices contacting with blood. REFERENCES [1] T. Alexy, O.K. Baskurt, N. Nemeth, M. Uyuklu, R.B. Wenby and H.J. Meiselman, Effect of lanthanides on red blood cell de- formability and respon se to mec hanica l stres s: rol e of la nth anide ionic radius, Biorheology 48 (2011), 173-183. [2] O.K. Baskurt, M. Uyuklu and H.J. Meiselman, Protection of erythrocytes from sub-hemolytic mechanical damage by nitric oxide mediated inhibition of potassium leakage, Biorheology 41 (2004), 79-89. [3] D.E. Brinsfield, M.A. Hoff, R.B. Geering and P.M. Galletti, Hematologi ca l ch an ges in lon g term perfus ion , J Ap pl Phys iol 17 (1962), 53 1-538. [4] R.G. Cooper, R.A. Kahn, C.N. Cornell and M.E. Muhrer, Eryt- hrocyte mechanical fragility test, J Clin Pathol 21 (1968), 781-783. [5] S. Deutsch, J.M. Tarbell, K.B. Manning, G. Rosenberg and A.A. Fontaine, Experimental fluid mechanics of pulsatile artificial blood pumps, Ann Rev Fluid Mech 38 (2006), 65-86. [6] M . Giersi epen , L.J . Wurzi ng er, R. Opitz and H. Reul, Estimation of shear stress -rela ted blood da mage in h eart va lve prost h eses- in vitro comparison of 25 aortic valve, Artif Organs 13 (1990), 300-306.  O. K. BASKURT Copyright © 2012 SciRes. ENG 10 [7] J.M. Greve, A.S. Les, B.T. Tang, M.T. Draney Blomme, N.M. Wilson, R.L. Dalman, N.J. Pelc and C.A. Taylor, Allometric scaling of wall shear stress from mice to humans: quantification using cine phase-contrast MRI and computational fluid dynamics, Am J Physiol Heart Circ Physiol 291 (2006), H1700-H1708. [8] L. Gu, W.A. Smith and G.P. Chatzimavroudis, Mechanical fra- gility calibration of red blood cells, Asaio J 51 (2005), 194-201. [9] M.V. Kameneva and J.F. Antaki, Mechanical trauma to blood, in: O.K.Baskurt, M.R.Hardeman, M.W.Rampling and H.J.Meiselman (Eds.), Handbook of Hemorheology and Hemo- dynamics, Amsterdam, Berlin, Oxford, Tokyo, Washington DC, 2007, pp. 206-227. [10] S.S. Lee, K.H. Ahn, S.J. Lee, K. Sun, P.T. Goedhart and M.R. Hardeman, Shear induced damage of red blood cells monitored by th e d ec rea se o f t h ei r d efor ma b i li t y, Korea-Australia Rheology Journal 16 (20 04), 141-146. [11] P.J. Marascalco, S.P. Ritchie, T.A. Snyder and M.V. Kameneva, Devel opmen t of stand ard tes ts to exami ne visc oelast ic prop erties of blood of experimental animals for pediatric mechanical sup- port dev ice evaluation, Asaio J 52 (2006), 567-574. [12] C.G. Nevaril, E.C. Lynch, C.P. frey Jr and J.D. Hellums, Eryt- hrocyte damage and destruction induced by shear stress, J Lab Clin Med 71 (1968), 784-790. [13] E.A. Orear, M.M. Udden, J.A. Farmer, L.V. McIntire and E.C. Lynch, Increased Intracellular C alcium and Decrea sed Deforma- bility of Erythrocytes from Prosthetic Heart-Valve Patients, Clinical Hemorheology 4 (1984), 461-471. [14] J.G. Sandza, R.E. Clark, C.S. Weldon and S.P. Sutera, Subhe- molytic trauma of erythrocytes; recognition and sequestration by spleen as a function of s hear , ASAIO Trans 2 (1974), 457-462. [15] S.P. Sutera, Flow-induced trauma to blood cells, Circ Res 41 (1977), 2-8. |

