Paper Menu >>
Journal Menu >>
 Advances in Ma terials Physics and Che mist ry, 2012, 2, 150-153 doi:10.4236/ampc.2012.24B039 Published Online December 2012 (htt p://www.SciRP.org/journal/ampc) Copyright © 2012 SciRes. AMPC Micromixing of a Two Phase System in a St irr ed Tank with Multiple Impellers Lei Yang, Jingcai Cheng, Ping Fan, Chao Yang Key Laboratory of Green P r ocess and Engineering, Institu te of P r o cess Engi neering Ch inese Academy of Sciences, Beijing 100190, Chi na Email: jccheng@mail.ipe.ac.cn, chaoyang@home.ipe.ac.cn Received 2012 ABSTRACT The co mpeti tive iod ide/ iodate react ion s cheme was used to ascert ain the micromixing in the stirred solid-liquid systems. Two differ- ent glass b eads from 450 to 1250 μm were tested. The effect of solid particles on reaction selectivity with multiple impellers at dif- ferent fe ed poin ts has been i nvestigated. It was confir med that glass beads as a second phase were s uitab le for the stu dy. The segre- gation index has ch anged significantly only for the medium-sized particles at relatively high solid holdups. The cloud formation was clearl y observed for th e mediu m-sized parti cles at a concen trat ion of 12 .12 wt. %. When feed in g into the clear l iqu id abo ve the cl oud, the value of the segregation index increased significantly. However, in the presence of particles of 1-1.25 mm, the influence on the selectivity was negl igible when the agitation speed was increased . Keywords: Micromixing; Parallel Reactions; Two Phases; Multiple Impellers 1. Introduction Micromixing could have an effect on the selectivity, yield and quality of the desired products in many industrial processes including precipitation, mineral processing, crystallization and biochemical processes. Poor micromixing may reduce the productivity of the desired products and also leads to higher purification costs. Therefore, many physical and chemical me- thods easy to be imp l eme n ted have been developed to charac- terize micromixing. A large amount of work has been done to improve the micromixing in single-phase systems, while rela- tively little work has been conducted in the two-phase systems especially with multiple impellers [1,2]. The dissipation rate of local energy determines the local micromixing. Techniques such as laser Doppler velocimetry (LDV) and particle image veloc imetry (PIV) can be used to give reliable values of local energy dissipation in single-phase systems. However, it is much difficult for them to be applied to dense slurries which are commonly opaque. So more experiments n eed to be undert aken to provide data for multiphase systems. Furthermore, the mod- els used by computational fluid dynamics (CFD) like turbu- lence model are not so mature. The data are crucial for suc- cessful turbulence modelling of multi-phase systems especially for high solids concentration systems. The influence of suspended solids on the selectivity of fast reactions has been investigated for a few years, but the results are a little contradictory. Villermaux et al. (1994) [1] used the iodide/iodate method to study the effect of suspended solids (0< dp < 40 μm) on the micromixing in stirred reactors. They found that the micromixing efficiency was enhanced by the presence of solids without significantly change in the power consump- tio n. However, aft er Guichardo n et al. (1995 ) [2] calculat ed the loss of iodine during filtration, the glass beads (20 μm < dp < 1300 μm) had a negligible effect on micromixing up to 5 wt. %. According to Barresi (1997) [3], si gnifican t ch an ges in s electiv- ity were observed only at relatively high particle loadings (>10 vol. % for th e glass sph eres (dp = 100-177 μm and 425-500 μm) and the selectivity was not affected with larger cylindrical PET beads ( equ ivalen t d iameter = 3 m m) . In 1999, Brilman et al. [4] investigated the effe ct of particles (70-700 μm) on product dis- tribution using the diazo-coupling reaction. They found that the segregation index increased while the holdup was enhanced except the particles of 290μm. Barresi (2000) [5] reported the effect of particles on reaction selectivity and related it to the changes in the power input and hydrodynamics of the suspen- sion. With the Rushton turbine, a lower selectivity was ob- served in the slurry. On the contrary, selectivities were higher in the slurry than those in the single phase when the pitched blad e was used. Recent ly, micromixing was u naffected near the impeller and near the surface with glass beads of 500 μm at concentrations up to 2.5wt.% by Hofinger et al. [6]. Cloud for- mation was observed, and when feeding into the clear liquid above the cloud, the segregation index increased significantly. From the literature review above, it can be concluded that there st ill exist s disagree ment between researcher s on t he effect of solid particles. The aim of this paper is to investigate the effect of solid particles on micromixing and give data on mean energy dissipation with multiple stirrers in the solid-liquid sys- tems. 2. Experimental Set-Up and Methods 2.1. Experimental Setup and Agitation Condition Experiments have been carried out at room temperature in a cylindrical Perspex vessel (diameter, T = 0.384 m) with a dis- hed bottom. A schematic of the experimental setup is given in Figure 1. The tan k is equipped with four baffles of width T/10 perpendicularly. In this work, a Rushton turbine (blade: height 21 mm, width 32 mm, thickness 1 mm) and a 45o down pump-  L. YANG ET AL. Copyright © 2012 SciRes. AMPC 151 ing 6-blade pitched blade turbine (blade: height 25 mm, width 47mm, thickness 1 mm), all of which have a diameter D= T/3, were used. In order to avoid surface aeration, H/T is 1.6. The impeller off-bottom clearance is T/3. The power drawn by the impeller was determined via the torque from a strain gauge attached to the shaft. A steel pipe with an inner diameter of 2 mm was used at each position. The geometrical details of the feed pipe tip are given in Table 1. Different s izes o f glass bead s were tested: 1) medium size: dp = 600-425 μm (ρs = 2.431 g/L); 2) large size:dp = 1.25 mm > d >1 mm (ρs = 2. 431 g/L). 2.2. Chemical Test Reactio n Micromixing experiments were carried out in the semi-batch mode using the reaction developed by Fournier et al. [7]. Ac- cording to the following steps: -+ 23 33 HBO + H HBO→ (i) -- + 3 22 5 I+ IO + 6 H 3 I + 3 HO↔ (ii) -- 23 I + I I↔ (3) The equilibrium constant KB of(iii)is well known as a function of the temperature. [8] 10 10 log= 555/+ 7.355 - 2.575log, B KT T KB in M-1 (1) The experiment procedure consists of injecting 0.03 L of sulphuric acid ([H+] = 1. 0 M) to the solution containing iodide, iodate and borate ions, whose concentrations follow that of Guichardon and Falk [9]. It has been confirmed that XQ, the selectivity of iodine formation, whose valu e lies between 0 and 1, is a measur ement of micromixin g efficien c y. XQ is defined as : XQ = Y/YST, where, 1 2 3 T D r H CC Figure 1. Schematic of the tank and feed points. Tabl e 1. Geometrical details of the feed pipe tip. Feed position Geome trical detail s Position <1> Position <2> Position <3> 2r/D z/H 2.2 0.21 2.2 0.31 2.2 0.84 23 0 II tank 23 injection 0 H 2(+) 2([I]+[I]) =[H ] nn V YnV − + − + = (2) and 30 ST 302 30 6[IO ] 6[IO] [HBO] Y − −− = + (3) Based on mass balances on iodine atoms, the following ex- pression is produ ced: --- - 0 233 [I]= [I]- 5/3( [I] + [I] ) - [I] (4) The value of XQ is calculated by combining (1) and (4). 3. Results and Discussion 3.1. Determination of Molar E xtinction Coefficient The molar extinction coefficient, ε, of triiodide ion was deter- mined by measuring the optical density of the solutions con- taining potassium iodide and iodine with a ratio of 2:1. The results are depicted in Fig ure 2. In this paper, th e analysis was conducted at 353 nm with a single beam where the int erference with iodide ion and solvent absorption could be reduced. At 353 nm, we found that ε was 2624 mol-1m2, which is in good agreement wit h the l iterature [7,10] . 3.2. The Influence of Feed Time With the purpose of finding out the conditions in which micro- mixing were free from macromixing, we changed the feed rate both in the single phase and the slurry system. From Figure 3, it can be seen that the segregation indexes come to asymptotic values when the feed time is larger than 2145 s, no matter what the system is. Thus, all our experiments were carried out with feed times larger th an 2145 s. 0.00 0.01 0.02 0.03 0.04 0.0 0.4 0.8 1.2 D C(l/L) Figure 2. The molar extinction coefficient. 01000 2000 3000 4000 5000 0.3 0.4 0.5 0.6 X Q t (s) m edium size, wt.% = 5, N = 7.2 s -1 single phase, N = 4.2 s -1 Figure 3. In fluence of feed time on segregat i on i n dex , f eed point 3. 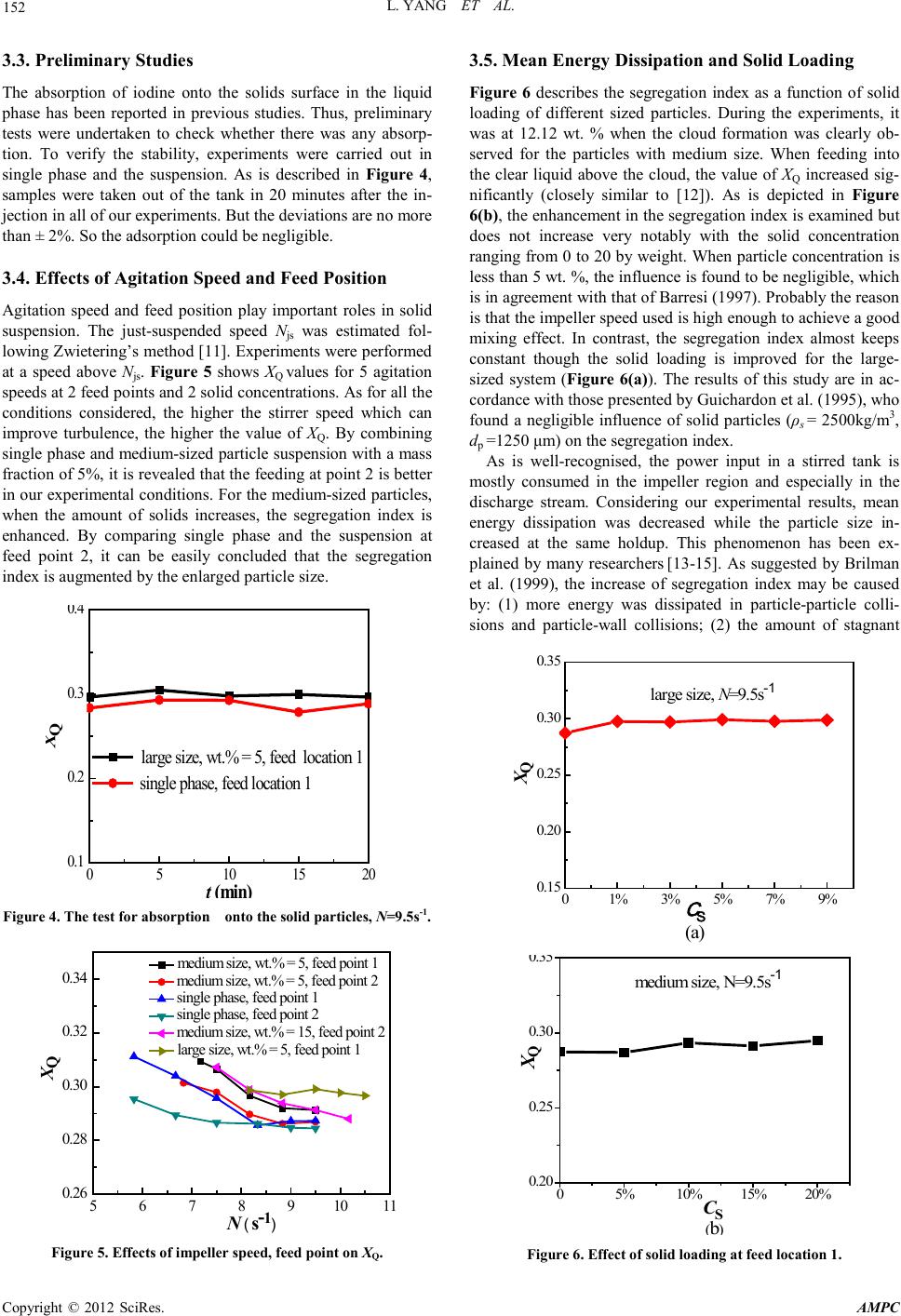 L. YANG ET AL. Copyright © 2012 SciRes. AMPC 152 3.3. Preliminary Studies The absorption of iodine onto the solids surface in the liquid phase has been reported in previous studies. Thus, preliminary tests were undertaken to check whether there was any absorp- tion. To verify the stability, experiments were carried out in single phase and the suspension. As is described in Figure 4, samples were taken out of the tank in 20 minutes after the in- jection in all of our experiments. But the deviations are no more than ± 2%. So the adsorption could be ne gligibl e . 3.4. Effects of Agit at ion Speed and Feed Position Agitation speed and feed position play important roles in solid suspension. The just-suspended speed Njs was estimated fol- lowing Zwietering’s meth od [ 11]. Experi ments were p erformed at a speed above Njs. Figure 5 shows XQ values for 5 agitation speeds at 2 feed points and 2 solid concentrations. As for all the conditions considered, the higher the stirrer speed which can improve turbulence, the higher the value of XQ. By combining single phase and medium-sized particl e suspens ion with a mass fraction of 5%, it is revealed that the feeding at point 2 is better in our experimental conditions. For the medium-sized p articles, when the amount of solids increases, the segregation index is enhanced. By comparing single phase and the suspension at feed point 2, it can be easily concluded that the segregation ind ex i s augmented by the enlarged p ar ticle size. 05 10 15 20 0.1 0.2 0.3 0.4 X Q t (min) large size, w t.% = 5, feed location 1 single phase, feed location 1 Figure 4. The test for absorptio n onto the solid particles, N=9.5s-1. 5678910 11 0.26 0.28 0.30 0.32 0.34 X Q N ( s- 1) medium size, wt.% = 5, feed point 1 m edium size, wt.% = 5, feed point 2 single phase, feed point 1 single phase, feed point 2 m edium size, wt.% = 15, feed point 2 large size, wt.% = 5, feed point 1 Figure 5. Effects of impeller speed, feed point on XQ. 3.5. Mean Energy Dissipation and Solid Loading Figure 6 describes the segregation index as a function of solid loading of different sized particles. During the experiments, it was at 12.12 wt. % when the cloud formation was clearly ob- served for the particles with medium size. When feeding into the clear liquid above the cloud, the value of XQ increased sig- nificantly (closely similar to [12]). As is depicted in Figure 6(b), th e enhan cement in t he segregat ion index is examined but does not increase very notably with the solid concentration ranging from 0 to 20 by weight. When particle concentration is less than 5 wt. %, the influence is found to be negligible, which is in agree ment wit h t hat of Barresi (1997). Probably the reason is that the impeller speed used is high enough to achieve a good mixing effect. In contrast, the segregation index almost keeps constant though the solid loading is improved for the large- sized system (Figure 6(a)). The results of this study are in ac- cordance with those presented by Guichardon et al. (1995), who found a negligible influence of solid particles (ρs = 2500kg/m3, dp =1250 μm) on the segregation index. As is well-recognised, the power input in a stirred tank is mostly consumed in the impeller region and especially in the discharge stream. Considering our experimental results, mean energy dissipation was decreased while the particle size in- creased at the same holdup. This phenomenon has been ex- plained by many researchers [13-15]. As suggested by Brilman et al. (1999), the increase of segregation index may be caused by: (1) more energy was dissipated in particle-particle colli- sions and particle-wall collisions; (2) the amount of stagnant 01% 3% 5% 7% 9% 0.15 0.20 0.25 0.30 0.35 X Q C S (a) large size, N=9.5s -1 05%10% 15% 20% 0.20 0.25 0.30 0.35 XQ CS (b) m edium size, N=9.5s -1 Figure 6. Effect of solid loading at f eed lo cation 1.  L. YANG ET AL. Copyright © 2012 SciRes. AMPC 153 0.5 1.01.5 2.0 2.5 3.0 3.5 0.26 0.28 0.30 0.32 X Q average energy dissipation (W/kg) single qhase, feed point 2 m edium size, w t.% = 5, feed point 1 large size, wt.% = 5 feed point 1 Figure 7. Comparison of mean specific energy dissipatio ns. liquid moving with the particles was increased. Figure 7 de- scribes the correlation between the mean en erg y dissip ati on and the segrega tion index. At higher particle sizes, there is an in- crease in segregation index due to more energy distributed to the particles. The results accord with those of Barresi (2000) and Brilman et a l. (1999). 4. Conclusions The effect of solid particles on reaction selectivity in a dished- bottom stirred tank with multiple impellers at different feed points has been investigated. It has been confirmed that glass beads as a second phase were suitable for the study. Large changes in the segregation index were obtained only for the medium-sized particles at relatively high solid holdups. During the experiments, the cloud formation was clearly observed for the medium-sized particles at the hold of 12.12 wt. %. When feeding into the clear liquid above the cloud, the value of XQ increased significantly and was up to 0.56. In the presence of the glass particl es of 1-1.25 mm, the influence on the selectivity was negligible though the solid concentration was enhanced when feeding at the tip of the pitched blade turbine. From our experimental results, it can be concluded that the segregation index improved but not apparently in the feed points studied when the agitation speeds increased. In order to reduce the selectivity towards undesired product, it is indis- pensable to improve the mixing, such as by increasing the cir- culation. However, it may consume more power. As suggested by Bourne and Hilber [16], an alternative could be the use of multiple feeds. Further work has been intended to find out the maximum energy dissipation rate of the pitched blade turbine and then carry out multiple feeds. 5. Acknowledgements The authors acknowledge the financial support from the 973 Program (2012CB224806), the National Natural Science Fund for Distinguished Young Scholars (21025627), the National Natural Science Foundation of China (21106154, 20990224), 863 Project (2012AA061503), Beijing Natural Science Foun- dation (2112038) and Jiangsu Province Project (BY2009133). REFERENCES [1] J. Villermaux, L. Falk, and M. C. Founier, “Potential use of a new par allel reaction system to characterize micromixing in stirred rea ct ors ,” AIChE Symp. Ser., vol. 90, pp. 50-54, 1994. [2] P. Guichardon, L. Falk, M. C. Founier, and J. Villermaux, “Study of micromixing in a liquid-solid suspension in a stirred tank,” AIChE Symp. Ser., vol. 91, pp. 123-130, 1995. [3] A. A. Barresi, “Experimental investigation of interation between turbulent liquid flow and solid particles and its effects on fast reactions,” Chem.En g. Sci., vol. 52, pp. 807-814, 1997. [4] D. W. F. Brilman, R. Antink, W. P. M. van Swaaij, and G. F. Versteeg, “Experimental study of the effect of bubbles, drops and particles on the product distribution for a mixing sensitive, pa- rallel-consecutive reaction system,” Chem. Eng. Sci., vol. 54, pp . 2325–2337, 1999. [5] A. A. Barresi, “Selectivity of mixing-sensiti ve reacti ons in slurry systems,” Chem.Eng. Sci., vol. 55, pp. 1929-1933, 2000. [6] J. Hofinger, R. W. Shape, W. Bujalski, S. Bakalis, M. Assirelli, A. Eaglesham, and A. W. Nienow, “Micromixing in two-phase (G-L and S-L)systems in a stirred vessel,” Can. J. Chem. Eng., vol. 89, pp. 1–11, 2011. [7] M. C. Fournier, L. Falk, and J. Villermaux, “A new parallel competin g react ion system for a ssessin g micromixing efficiency- —Experimental approach,” Chem.Eng. Sci., vol. 51, pp. 5053-5064, 1996. [8] D. A. Palmer, R. W. Ramette, and R. E. Mesmer, “Triiodide ion formation equilibrium and activity coefficients in aqueous solu- tion,” J. Soluti on Chem., vol. 13, pp. 673-683, 1984. [9] P. Guichardon and L. Falk, “Characterisation of micromixing efficiency by the iodide-iodate reaction system. Part I: experi- mental procedure,” Chem. Eng. Sci.,vol. 55, pp. 4233–4243, 2000. [10] Awtrey and Connick, “The absorption spectra of 2 I , - 3 I , - 3 IO , 2- 26 SO , 2- 23 SO . Heat of the Reaction - 3 I = 2 I +- I ,” J. Am. Chem. Soc, vol. 73, pp. 1844-1843, 1951. [11] Zwietering, Th. N, “Suspension of Solid Particles in Liquid by Agitators,” Chem. Eng. Sci ., vo l . 8, p p. 244-253, 1958. [12] M. Assirelli, W. Bujalski, A. Eaglesham, and A. W. Nienow, “Study of micromixing in a stirred tank using a Rushton Turbine: Compa ris on of feed positions and other mixing devices,” Chem. Eng. Res. Des., vol. 80, pp. 855–863, 2002. [13] Yu . A. B uyev ich, “Fluid dynamics of coarse dispersions,” Chem. Eng. Sci. vol. 49, pp. 1217-1228, 1994. [14] J. O. Hinze, “Turbulent fluid and particle interaction,” In Pro- ceedings of the International Symposium Two-Phase Systems, eds G. Hetsroni, S. Sid eman and J. P. Hartn ett Prog, Heat Mass Transfer, vol. 6, pp. 433-452. Perga m on Press, Oxford, 1972. [15] S. Elgobashi, “On predicting particle-laden turbulent flows,” Appl. Sci. Res., vol. 52, pp. 309-329, 1994. [16] J. R. Bourne and C. P. Hilber , “The produ ctivity of mic romixing controlled reactions: effect of feed distribution in stirred tanks,” Chem. Eng. Res. Des. vol. 68A, pp. 51-56, 1990. |

