Paper Menu >>
Journal Menu >>
 Advances in Ma terials Physics and Che mist ry, 2012, 2, 146-149 doi:10.4236/ampc.2012.24B038 Published Online December 2012 (http://www.SciRP.org/journal/ampc) Copyright © 2012 SciRes. AMPC Jet Plasma-Chemical Reactor for the Conversion of Methane: the Use of Cl ust e ring A. E. Zarvin, N. G. Korobeishchikov, M. D. Khodakov, V. V. Kalyada Department of Applied Physics, Novosibirsk State University, Novosibirsk, Russia Email: zarvin@phys.nsu.ru Received 2012 ABSTRACT The research results of processes proceeding in supersonic jets of light hydrocarbons, activated by an electron beam are present ed. It is shown, that condensation suppressed at activation by electrons in the initial stage of condensation. The developed condensation conditions mode leads to increasing of a part of heavy corpuscles in activated stream and not only owing to stimulation of condensa- tion but because of formation of heavy hydrocarbonic molecules. Keywords: Conversion of Methane; GTL-Production Engineering; Clusters; a Supersonic Jet; P lasmache mical Reactions; Gas Phase Reactions; an Electron B eam; a Gas Di s charge 1. Introduction Associated petroleum gas (a mixture of methane - more than 70%, ethane, propane and butane) is traditionally burned during the oil production. Its processing is not provided in the existing technology of oil refining. As a result, approximately 10% of the hydrocarbons contained in the field, does not used in the economy only, but also contributes to further pollution of the earth's atmosphere. Processing technology of volatile hydrocarbons exists for a long time. It’s primarily based on the use of catalysts in Fisch- er-Tropsch process, focused on the steady-state production , and remote oil fields is almost irrelevant. It should also be borne in mind that any large-tonnage commercial product should be exported, and the only available high-performance transport of liquid products from the field - an oil pipeline. In this situation it is extremely attractive the develop ment of the direct technology conversion method, bypassing the stage of synthesis gas, of natural and petroleum gas to heavy hydro- carbons, which is implemented on a compact high-performance device. The resulting liquid should be suitable for pipeline transport, ie its vapor pressure must not exceed 0.5 excess air. Such a formulation of the problem points directly to the use- fulness of its solution methods and approaches based on plas- ma-chemical processes in the gas phase: the large interaction cross section of ionized and activated particles provide an ex- tremel y high rate of any collis ional processes. 2. Plasma Chemical Technologies First of all, it was started investigation of the possibilities for application of discharge plasma to initiate gas-phase reactions [1-4]. In the discharge can be easily obtained active particles that provide high-speed reactions. As a result, engineers have created several versions of devices to produce hydrogen from hydrocarbons using microwave discharge plasma torch, a com- bination of discharge with distributed finely dispersed catalyst [5], etc. In the 2000 method of conversion of hydrocarbons in a barrier discharge, where gas diffuses through the porous dielec- tric layer containing catalyst particles, was proposed [6]. A limitation of this method is the low rate of diffusion of gas as well as the n eed for high pressure. There are a large nu mber of patents on the use of a glow, corona, streamer and other types of discharge, electric arc, plasma torch and so on [7-9]. How- ever, no one has to initiate a one-step synthesis of heavy hy- drocar bons on an i ndustrial s cale yet/ Using th e disch arge faces a number o f difficul ties associated with the influence of the electrodes, the background gas and the flow in a quiescent gas, or the reverse reactions in a subsonic flow, limiting the equilibrium concentration of the reaction products. These negative factors can be excluded if use the supersonic reagent jet to provide gas into reaction zone. Shock waves which are formed at the boundaries of the jet, are iso- lated the jet core from the influence of background gas. Freez- ing of the reverse reactions is provided by a sharp drop in den- sity in a supersonic jet. It is much harder to get rid of the bad handling of the discharge, because its charact eristics dep end on the parameters of the gas in the area of activation, and to cope with the fact that a significant portion of the discharge of elec- trons is not involved in the activation process since energy of such el ecnrons l ie below the threshold for dissociation. Thus, a technology conversion of light hydrocarbons in the gasoline fraction, based on plasma-chemical approach has not succeeded. The main reason, apparently, is the apparent "equi- librium" of processes in the flow of weakly ionized methane plasma. Activ ati on of any kind of disch arge lead s to an increase of gas temperature in the flow, and heavy hydrocarbons which are produced with the participation of radicals formed in the discharge, will inevitably disintegrate soon after birth. This pessimistic conclusion was confirmed with both numerous experiments and calculations. 3. Gas-Jet Plasma Chemictry 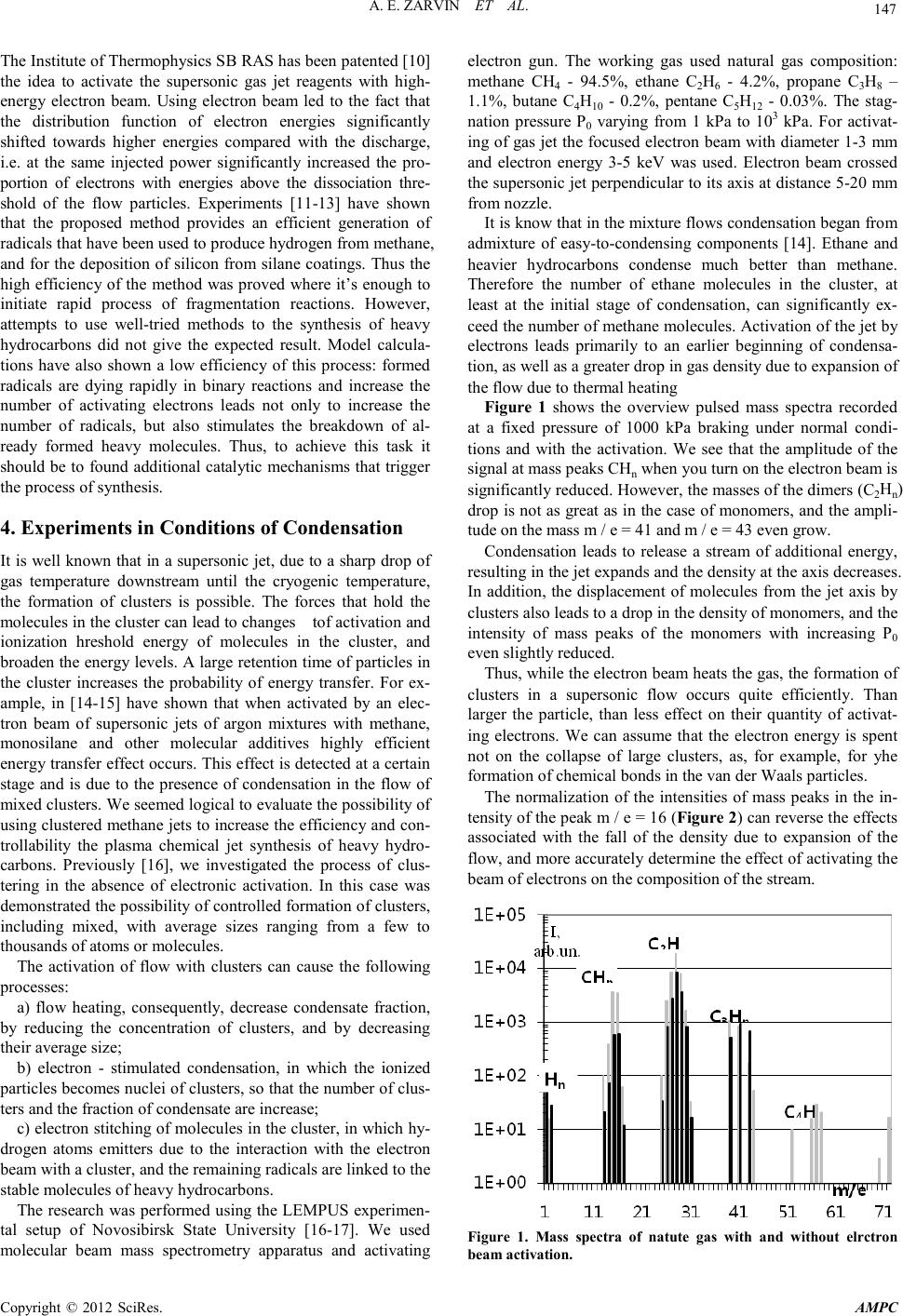 A. E. ZARVIN ET AL. Copyright © 2012 SciRes. AMPC 147 The Institute of Thermophysics SB RAS has been patented [10] the idea to activate the supersonic gas jet reagents with high- energy electron beam. Using electron beam led to the fact that the distribution function of electron energies significantly shifted towards higher energies compared with the discharge, i.e. at the same injected power significantly increased the pro- portion of electrons with energies above the dissociation thre- shold of the flow particles. Experiments [11-13] have shown that the proposed method provides an efficient generation of radicals that have been used to produce hydrogen from methane, and for the deposition of silicon from silane coatings. Thus the high efficiency of the method was proved where it’s enough to initiate rapid process of fragmentation reactions. However, attempts to use well-tried methods to the synthesis of heavy hydrocarbons did not give the expected result. Model calcula- tions have also shown a low efficiency of this process: formed radicals are dying rapidly in binary reactions and increase the number of activating electrons leads not only to increase the number of radicals, but also stimulates the breakdown of al- ready formed heavy molecules. Thus, to achieve this task it should be to found additional catalytic mechanisms that trigger the process of synthesis. 4. Experiments in Conditions of Condensation It is well known that in a supersonic jet, due to a sharp drop of gas temperature downstream until the cryogenic temperature, the formation of clusters is possible. The forces that hold the molecules i n t he cl ust er can lead to chan ges tof activat io n an d ionization hreshold energy of molecules in the cluster, and broad en the energy levels. A larg e retention time of particles i n the cluster increases the probability of energy transfer. For ex- ample, in [14-15] have shown that when activated by an elec- tron beam of supersonic jets of argon mixtures with methane, monosilane and other molecular additives highly efficient energy trans fer e ffect occurs . Thi s effect is d etect ed at a cert ain stage and is due to the presence of condensation in the flow of mixed clusters. We seemed logical to evaluate the possibility of usin g clustered methan e jets to increase t he efficienc y and con- trollability the plasma chemical jet synthesis of heavy hydro- carbons. Previously [16], we investigated the process of clus- tering in the absence of electronic activation. In this case was demonstrated the possibility of controlled formation of clusters, including mixed, with average sizes ranging from a few to thousands of atoms or molecules. The activation of flow with clusters can cause the following processes: a) flow heating, consequently, decrease condensate fraction, by reducing the concentration of clusters, and by decreasing their aver age si ze; b) electron - stimulated condensation, in which the ionized particl es becomes nu clei o f clusters, so that the nu mber of clus- ters and the fraction of condensate are increase; c) electron stitching of molecules in the cluster, in which hy- drogen atoms emitters due to the interaction with the electron beam with a cluster, and the r emaining radicals are li nked to the stable mol ecules of h eavy h ydrocarbons. The research was performed usi ng th e LEMP US experimen- tal setup of Novosibirsk State University [16-17]. We used molecular beam mass spectrometry apparatus and activating electron gun. The working gas used natural gas composition: methane CH4 - 94.5%, ethane C2H6 - 4.2%, propane C3H8 – 1.1%, butane C4H10 - 0.2%, pentane C5H12 - 0.03%. The stag- nation pressure P0 varying from 1 kPa to 103 kPa. For activat- ing of gas jet t he focused electron b eam with diameter 1-3 mm and electron energy 3-5 keV was used. Electron beam crossed the su personic jet perpendicu lar to its axis at distance 5 -20 mm from nozzle. It is know that in the mixture flows condensation began from admixture of easy-to-condensing components [14]. Ethane and heavier hydrocarbons condense much better than methane. Therefore the number of ethane molecules in the cluster, at least at the initial stage of condensation, can significantly ex- ceed the number of methane molecules. Activation of the jet by electrons leads primarily to an earlier beginning of condensa- tion, as well as a greater drop in gas density due to expansion of the flow due to thermal heating Figure 1 shows the overview pulsed mass spectra recorded at a fixed pressure of 1000 kPa braking under normal condi- tions and with the activation. We see that the amplitude of the signal at mass peaks CHn when you turn on the electron beam is significant l y reduced . Ho wever, th e masses o f the d imers ( C2Hn) drop is not as great as in the case of monomers, and the ampli- tude on the mass m / e = 41 and m / e = 43 even grow. Condensation leads to release a stream of additional energy, result in g in th e jet exp and s an d the densi ty at the a xis decre ases . In addition, the displacement of molecules from the jet axis by clusters also leads to a drop in the density of monomers, and the intensity of mass peaks of the monomers with increasing P0 even sligh tly reduced. Thus, while the electron beam heats the gas, the formation of clusters in a supersonic flow occurs quite efficiently. Than larger the particle, than less effect on their quantity of activat- ing electrons. We can assume that the electron energy is spent not on the collapse of large clusters, as, for example, for yhe formation of chemical bonds in the van der Waals part icles. The normalization of the intensities of mass peaks in the in- tensit y of the peak m / e = 16 (F igure 2) can reverse t he effects associated with the fall of the density due to expansion of the flow, and more accu ratel y determ ine t he effect of acti vating th e beam of electrons on the composition of the stream. Figure 1. Mass spectra of natute gas with and without elrctron beam activation. 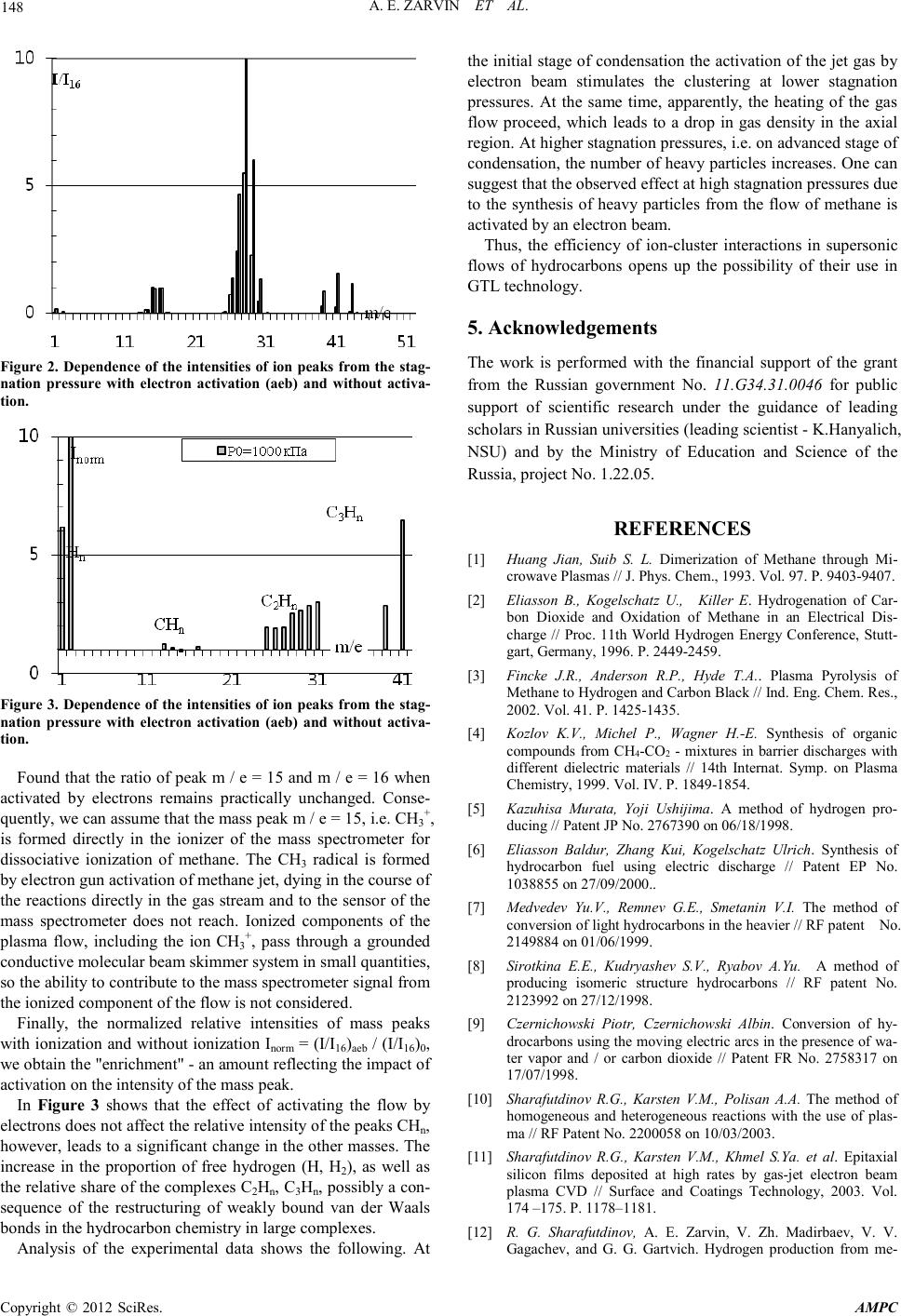 A. E. ZARVIN ET AL. Copyright © 2012 SciRes. AMPC 148 Figure 2. Dependence of the intensities of ion peaks from the stag- nation pressure with electron activation (aeb) and without activa- tion. Figure 3. Dependence of the intensities of ion peaks from the stag- nation pressure with electron activation (aeb) and without activa- tion. Found that the ratio of peak m / e = 15 and m / e = 16 when activated by electrons remains practically unchanged. Conse- quently, we can assu me that t he mass peak m / e = 15 , i.e. CH3+, is formed directly in the ionizer of the mass spectrometer for dissociative ionization of methane. The CH3 radical is formed by electron gun activation of methane jet, dying in the course of the reactions directly in the gas stream and to the sensor of the mass spectrometer does not reach. Ionized components of the plasma flow, including the ion CH3+, pass through a grounded condu ctive molecular beam skimmer system in small quan tities, so the ability to co nt ribu te to the mass spect ro meter sign al fro m the ionized component of the flow is not considered. Finally, the normalized relative intensities of mass peaks with ionization and without ionization Inorm = (I/I16)aeb / (I/I16)0, we obtain the "enrichment" - an amoun t reflecti ng t he impact o f activation on t he inten s ity of the mas s peak. In Figure 3 shows that the effect of activating the flow by electron s d o es no t affect t he r elati ve i nt ensi ty o f the p eaks C Hn, however, le ads to a si gnificant ch ange in the other masses. The increase in the proportion of free hydrogen (H, H2), as well as the relat ive share of the complexes C2Hn, C3Hn, possibly a con- sequence of the restructuring of weakly bound van der Waals bonds in the hydrocarbon chemistry in large complexes. Analysis of the experimental data shows the following. At the initial stage of condensation the activation of the jet gas by electron beam stimulates the clustering at lower stagnation pressures. At the same time, apparently, the heating of the gas flow proceed, which leads to a drop in gas density in the axial region . At hi gher stagn atio n pr essures, i .e. on ad vanced st age of condensation, the number of heavy particles increases. One can suggest that the observed effect at high stagnation pressures due to the synthesis of heavy particles from the flow of methane is activated by an electro n beam. Thus, the efficiency of ion-cluster interactions in supersonic flows of hydrocarbons opens up the possibility of their use in GTL technology. 5. Acknowledgements The work is performed with the financial support of the grant from the Russian government No. 11.G34.31.0046 for public support of scientific research under the guidance of leading scholars in Russian universities (leading scientist - K.Hanyalich, NSU) and by the Ministry of Education and Science of the Russia, project No. 1.22.05. REFERENCES [1] Huang Jian, Suib S. L. Dimerization of Methane through Mi- crowave Plasmas // J. Phys. Chem., 1993. Vol. 97. P. 9403-9407. [2] Eliasson B., Kogelschatz U., Killer E. Hydrogenation of Car- bon Dioxide and Oxidation of Methane in an Electrical Dis- charge // Proc. 11th World Hydrogen Energy Conference, Stutt- gart, Germany, 1996. P. 244 9-2459. [3] Fincke J.R., Anderson R.P., Hyde T.A.. Plasma Pyrolysis of Metha ne to Hydrogen a nd Ca rb on B lac k // Ind . En g. Ch em. R es ., 2002. Vol. 41. P. 1425-1435. [4] Kozlov K.V., Michel P., Wagner H.-E. Synthesis of organic compounds from CH4-CO2 - mixtures in barrier discharges with different dielectric materials // 14th Internat. Symp. on Plasma Chemistry, 1999. Vol. IV. P. 1849-1854. [5] Kazuhisa Murata, Yoji Ushijima. A method of hydrogen pro- ducing // Patent JP No. 2767390 on 06/18/1998. [6] Eliasson Baldur, Zhang Kui, Kogelschatz Ulrich. Synthesis of hydrocarbon fuel using electric discharge // Patent EP No. 1038855 on 27/09/2000.. [7] Medvedev Yu.V., Remnev G.E., Smetanin V.I. The method of conversi on of light hydroca rb on s in the heavi er // RF paten t No. 2149884 on 01/06/1999. [8] Sirotkina E.E., Kudryashev S.V., Ryabov A.Yu. A method of producing isomeric structure hydrocarbons // RF patent No. 2123992 on 27/12/1998. [9] Czernichowski Piotr, Czernichowski Albin. Conversion of hy- drocarbons using the moving electric arcs in the presence of wa- ter vapor and / or carbon dioxide // Patent FR No. 2758317 on 17/07/1998. [10] Sharafutdinov R.G., Karsten V.M., Polisan A.A. The method of homogeneous and heterogeneous reactions with the use of plas- ma // RF Patent No. 2200058 on 10/03/2003 . [11] Sharafutdinov R.G., Karsten V.M., Khmel S.Ya. et al. Epitaxial silicon films deposited at high rates by gas-jet electron beam plasma CVD // Surface and Coatings Technology, 2003. Vol. 174 –175. P. 1178–1181. [12] R. G. Sharafutdinov, A. E. Zarvin, V. Zh. Madirbaev, V. V. Gagachev, and G. G. Gartvich. Hydrogen production from me-  A. E. ZARVIN ET AL. Copyright © 2012 SciRes. AMPC 149 thane in electron-beam-generated plasma // Technical Physics Letters. 2005. Vol. 31. No. 8. P. 641-643. [13] V. A. Vinokurov, R. G. Sharafutdinov, Yu. I. Tychkov Plas- ma-chemical processing of natural gas // Chemistry and Tech- nology of Fuels and O ils. 2005 . Vol. 41. No. 2. P. 112 -115. [14] A. E. Zarvin, V. Zh. Madirbaev, N. G. Korobeishchikov, G. G. Gartvich, and R. G. Sharafutdinov. Effect of small methane an d monosilane additives on clustering in pulse supersonic argon jets // Tech. P hys. 2005. Vol. 50 . P. 1444-1450. [15] V. Zh. Madirbaev, A. E. Zarvin. Ion-cluster excitation of atomic argon levels in molecular gas mixtures // Vestnik Novosibirsk State University. Series: Ph ysics. 2007. Vol. 2. Issue 1. P. 36-43. [16] N. G. Korobeishchikov, A. E. Zarvin, V. Zh. Madirbaev and R. G. Sharafutdinov. Condensation of argon, monosilane and their mixture in a pulse free jet // Plasma Chem. Plasma Proc. 2005. Vol. 25. P. 319-349. [17] A. E. Zarvin, N. G. Korobeishchikov, V. V. Kalyada, V. Zh. Ma- dirbaev. Formation of mixed clusters in a pulsed helium - oxy- gen - isoprene supersonic jet // Eur. Phys. J. D. 2008. Vol. 49. P. 101-110. |

