Paper Menu >>
Journal Menu >>
 Advances in Ma terials Physics and Che mist ry, 2012, 2, 110-114 doi:10.4236/ampc.2012.24B030 Published Online December 2012 (http://www.SciRP.org/journal/ampc) Copyright © 2012 SciRes. AMPC Thermal Degradation Kinetics of iPP/Pd Nanocomposite Prepared by a Drying Process Y Jae-Young Lee1, H ong-Ki Lee1, Sung-Wan Hong 2, Il-Yub Choi3 1Hydrogen Fuel Cell Parts and Applied Technology RIC Woosuk Univ., Wanju, Korea 2Dept. of Cosmetics, Woosuk Univ. Samnye, K o r ea 3Dept. of En viro. Eng., Univ. of S eoul, Seoul, Korea Email: hongkil@woosuk.ac.kr Received 2012 ABSTRACT Palladium (Pd) nanoparticles were incorporated into isotactic polypropylene (iPP) film by a one-step dry process. iPP film was ex- posed to the sublimed Pd (acac)2 vapor i n a glass vessel at 1 80oC. The Pd nanoparticles were observed by transmission electron mi- croscope (TEM), and it was found that metallic nanoparticles were selectively loaded on the amorphous regions between the lamellae in iPP. Thermal degrad atio n kinetics w as investi gated b y intro ducing the d ata of thermogra vimetric analysis ( TGA) to Fl ynn & Wall equation. TGA data showed that thermal degradation temperature (Td) of the neat iPP was improved about 35oC by loading 0.27 wt% Pd nanoparticles. Thermal degradation activation energy (Ed) for iPP/Pd nanocomposite was 227.85 kJ/mol while that of neat iPP was 220 .57 kJ/mol. Th ese result s meant th at the Pd nan oparticl es acted as a retard ant in t he thermal d egradatio n of neat iPP polymer chain . Keywords: Pd Nanoparticles; Polymer Nanocomposite; Isotactic Polypropylene; Thermal Degradation; Flynn & Wall Equation 1. Introduction Uniformly dispersed metallic nanoparticles into a polymer ma- trix can offer new functional materials in the various applica- tions such as catalysts, optics, senses, magnetics and electrics. Therefore many researchers have investigated to develop new methods for the preparation of polymer/metal nanocomposites avoiding their easy oxidation, contamination, and aggregation problems, and these methods are mainly classified into five: (1) a metallic precursor solution and a polymer solution are mixed in a reacto r, and then the metal lic p recu rso r is redu ced to the metallic nanoparticles during stirring, heating and evapo- rating the solvent [1-3]; (2) a metallic precursor is dissolved in a monomer and then is thermally reduced during the polymeri- zation at high reaction temperature [4,5]; (3) a colloidal metal- lic n anopar ticles p re-prep ared by ot her methods are mixed with a polymer solution or monomer and then it was evaporated or polymerized [6,7]; (4) a metallic precursor is dissolved in a solvent and the solution impregnates into a polymer matrix, and then metallic nanoparticles are generated by treating with re- duction agents or thermolysis [8,9]; and (5) sublimed metallic precursor molecules penetrate into a polymer matrix and are reduced to self-assembled metallic nanoparticles [10,11]. The methods 1~4 are wet process, while the last method 5 is a dry proces s. Crystalline polypropylenes have strong mechanical proper- ties and thei r numerous and versatile applications can be easily achieved by wide range of PP homopolymers and copolymers and by easy processability. Therefore they are widely used as bumpers and dashboards in automobiles, OPP films, fibers for membranes, pipes, etc., and the crystallinity is one of the most important factors for the determination of the properties [12, 13]. The explanation of the crystal structure in PP is started by stereo-isomerism. When PP monomers are polymerized, all the neighboring methyl groups in the PP chain can have two ste- reo-isomeric positions. If all the methyl groups arrange on the same side o f the zigzag plain , it is defined as isotactic p olypro- pylene (iPP), and if they arrange alternatively, it is called as syndiotactic PP (sPP), and each has polymorphism of crystal structure resulting from various thermal and solvent conditions [12,14]. It was reported that the onset temperature of the thermal degradation, Td of alkyl polymer chains was improved by the incorporation of very small amount of palladium (Pd) nanopar- ticl es [ 10,11] . E speciall y th e Td values for crystalli ne polymers were remarkably increased, that is, Td of syndiotactic polysty- rene was improved about 50oC through the incorporation of 1.5 wt% of Pd nanoparticles. It was maybe due that the dry process, the above method 5 could incorporate metallic nanoparticles into the polymer films without the destruction of bulk shape and even without the change of crystallinity, because metallic nanoparticles were generated and positioned on the amorphous regions in sPS. However that of atatic polystyrene was im- proved only about 18oC by 1.68 wt% Pd nanoparticles, while that of nylon 6 even decreased about 52.8oC by 15.6 wt% Pd nanop articles. In this study, Pd nanoparticles were incorporated into iPP via the above method 5 and the thermal degradation kinetics was studied by Flynn & Wall equation [15,16] as follows: 1 (log ) 0.457 () d Rd EdT β − − = where, Ed was acti vation en ergy for th e thermal degradation, R was the universal gas constant, 8.314 kJ/mol, β was heating rate, 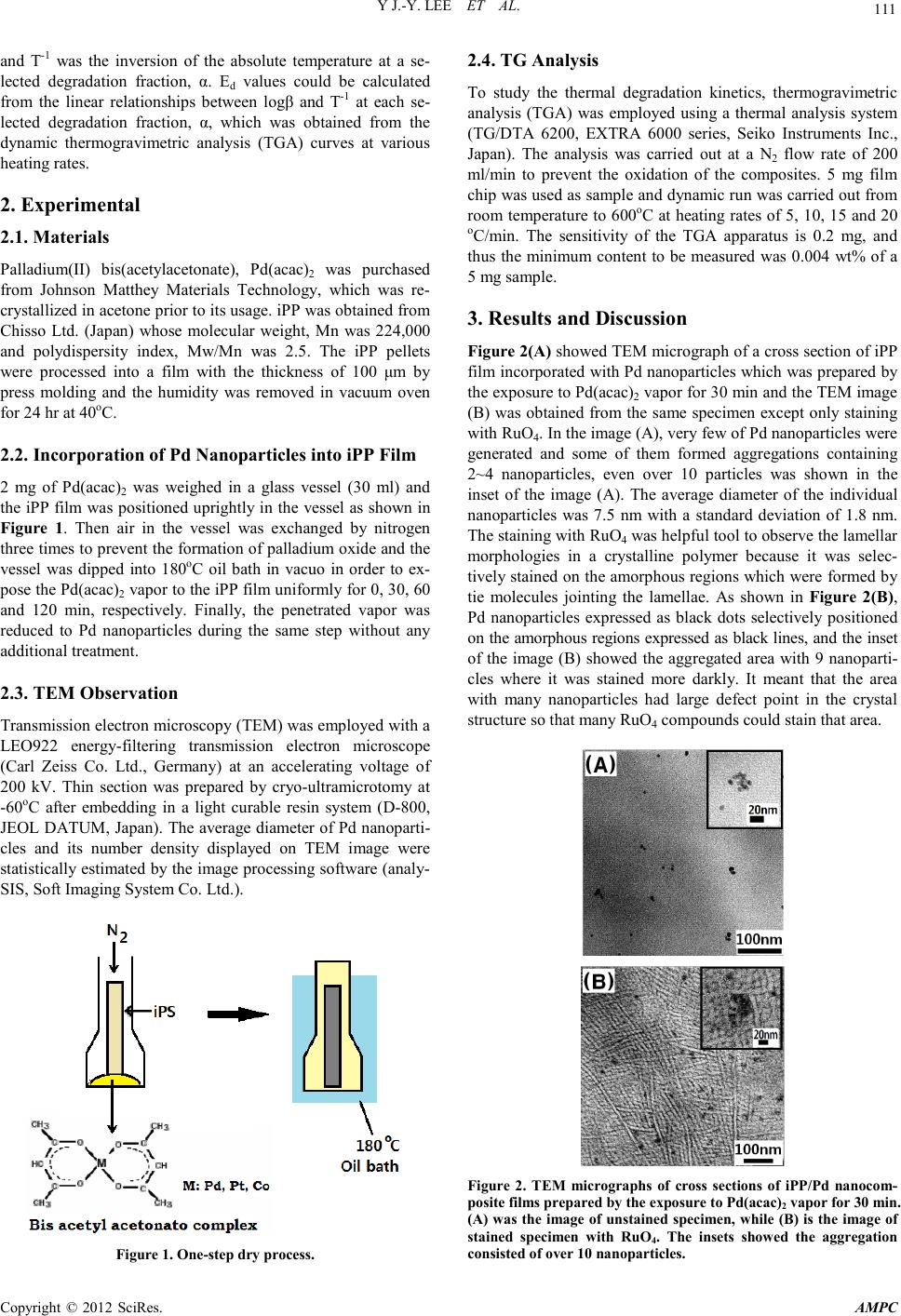 Y J.-Y. LEE ET AL. Copyright © 2012 SciRes. AMPC 111 and T-1 was the inversion of the absolute temperature at a se- lected degradation fraction, α. Ed values could be calculated from the linear relationships between logβ and T-1 at each se- lected degradation fraction, α, which was obtained from the dynamic thermogravimetric analysis (TGA) curves at various heating rates. 2. Experimental 2.1. Materials Palladium(II) bis(acetylacetonate), Pd(acac)2 was purchased from Johnson Matthey Materials Technology, which was re- crystalli zed i n acet on e pr io r to it s usage. iP P was ob tain ed fro m Chisso Ltd. (Japan) whose molecular weight, Mn was 224,000 and polydispersity index, Mw/Mn was 2.5. The iPP pellets were processed into a film with the thickness of 100 μm by press molding and the humidity was removed in vacuum oven for 24 hr at 40oC. 2.2. Incorporation of Pd Nanoparticles into iPP Film 2 mg of Pd(acac)2 was weighed in a glass vessel (30 ml) and the iPP film was positioned uprightly in the vessel as shown in Figure 1. Then air in the vessel was exchanged by nitrogen three times to prevent the formation of palladium oxide and the vessel was dipped into 180oC oil bath in vacuo in order to ex- pose the P d( acac)2 vapor to the iPP film uniformly for 0, 30, 60 and 120 min, respectively. Finally, the penetrated vapor was reduced to Pd nanoparticles during the same step without any additional treatment. 2.3. TEM Observation Transmission electron microscopy (TEM) was employed with a LEO922 energy-filtering transmission electron microscope (Carl Zeiss Co. Ltd., Germany) at an accelerating voltage of 200 kV. Thin section was prepared by cryo-ultramicrotomy at -60oC after embedding in a light curable resin system (D-800, JEOL DATUM, Japan). The average diamet er of Pd nanoparti- cles and its number density displayed on TEM image were statist ically estimat ed by the i mage processin g software (an aly- SIS, Soft Imaging System Co. Lt d.). Figure 1. One-step dry process. 2.4. TG Analysis To study the thermal degradation kinetics, thermogravimetric analysis (TGA) was employed using a thermal analysis system (TG/DTA 6200, EXTRA 6000 series, Seiko Instruments Inc., Japan). The analysis was carried out at a N2 flow rate of 200 ml/min to prevent the oxidation of the composites. 5 mg film chip was used as sample and dynamic run was carried out from room temperature to 600oC at heating rates of 5, 10, 15 and 20 oC/min. The sensitivity of the TGA apparatus is 0.2 mg, and thus the minimum content to be measured was 0.004 wt% of a 5 mg sample. 3. Results and Discussion Figure 2(A) showed TEM micrograph of a cross section of iPP film incorporated with Pd nanoparticles which was prepared by the expo sure to Pd(acac)2 vapor for 30 min and the TEM image (B) was obtained from the same specimen except only staining with RuO4. In the image (A), very few of Pd nanop arti cles were generated and some of them formed aggregations containing 2~4 nanoparticles, even over 10 particles was shown in the inset of the image (A). The average diameter of the individual nanoparticles was 7.5 nm with a standard deviation of 1.8 nm. The staining with RuO4 was helpful tool to ob serve th e lamellar morphologies in a crystalline polymer because it was selec- tively stained on the amorphous regions which were formed by tie molecules jointing the lamellae. As shown in Figure 2(B), Pd nanoparticles expressed as black dots selectively positioned on the amorphous regions expressed as black lines, and the inset of the image (B) showed the aggregated area with 9 nanoparti- cles where it was stained more darkly. It meant that the area with many nanoparticles had large defect point in the crystal structure so that many RuO4 compounds could stain that area. Figure 2. TEM micrographs of cross sections of iPP/Pd nanocom- posite films prepared by the exposure to Pd(acac)2 vapor f or 30 min. (A) was the image of unstaine d specimen, w hile (B) is the image of stained specimen with RuO4. The insets showed the aggregation consist ed of ov er 1 0 n anoparticles .  Y J.-Y. LEE ET AL. Copyright © 2012 SciRes. AMPC 112 Figure 3 showed the dynamic TGA and DTG curves for iPP/ Pd nanocomposites with various amounts of the Pd nanoparti- cles. The h eating rat e was at 10 oC/min in nitrogen atmosphere. It was clearly shown that the incorporation of the Pd nanoparti- cles shifted the TGA curves to higher temperature. It implied that the thermal stability was remarkably improved by the in- corporation of Pd nanoparticles. In the TG curve of neat iPP, there was almost no weight loss before 372oC and it decreased very slowly un til 404oC, but it was very q ui ckl y decreased a fter that temperature and almost all polymer chains were degraded abruptly between 404~482oC. However, the TGA curve of the iPP/Pd nanocomposite prepared by the exposure to Pd(acac)2 vapor for 30 min was very different from that of neat iPP. It showed no weight loss before 409oC, slow decrement until 439oC, and abrupt degradation between 439~483oC, where the Pd incorporation weight of the sample was 0.27 wt%. It was an amazing result that the thermal stability of the neat iPP was improved by 35oC via the incorporation of such very small amount of the Pd nanoparticles. In here, the incorporation weight of Pd nanoparticles was obtained from the prolysis at 1,000oC nitrogen atmosphere in an electric furnace for 3 hr. In the DTG curves, compared to that of iPP/Pd nanocomposite, the steeper slope in the former part and the similar slope in the latter part of the neat iPP maybe said that the Pd nanoparticles mainly contributed to the improvement in the initial degrada- tion of neat iPP, and it was maybe due that nanoparticles posi- tioned on the defects of crystal structure held the polymer chains tightly until higher temperature. Figure 3 also showed that the increasing Pd content from 0.27 wt% to 0.57 wt% coul d no t bring the better res ults as was expected. - 1.0 - 0.8 - 0.6 - 0.4 - 0.2 0.0 TG 350 400 450 500 0.0 0.1 0.2 0.3 0.4 Pd loading time (loading w eight) 0min (0 wt%) 30min (0.27wt%) 60min (0.39wt%) 120min (0.57 wt%) DTG (min-1) Temperature (o C ) Figure 3. TGA and DTG curves for iPP/Pd nanocomposites pre- pared at different Pd(acac)2 exposure time. In order to estimate the thermal degradation rate, the TGA data at various heating rates were introduced to Flynn & Wall equation. Figure 4 showed the TGA curves for the iPP/Pd (0.27 wt%) nanocomposite at the heating rates of 5, 10, 15 and 20 oC/min. The temperature values at α = 0.05 for each heating rate were 428.30, 438.16, 447.26, and 452.95oC, respectively. To get Ed value at each α, these values were converted to T-1 and introduced to the Flynn & Wall equation together with logβ listed in Table 1. Usi ng the same procedure, the temperatures at different α for each heating rate were obtained from Figure 4, and the T-1 values were also listed in Table 1. Then a straight line for each α was plotted by the relationship between log β and T-1 data for F lynn & Wall equation, as displayed in Figure 5. The linear correlation for α = 0.05 was expressed as log β = -12.25×103 · T-1 + 18.19. So, the Ed value was calculated from the slop e, -12.25×103 = -0.457·Ed/R, so that the Ed value at α = 0.05 was 222.86 kJ/mol. The Ed values for various α were also obtained through the same procedure and shown in Figure 6. The average Ed value for iPP/Pd (0.27 wt%) nanocomposite was 227.85 kJ/mol, which was very similar value, 228 kJ/mol calculated from Kissinger equation [11]. The Ed value for neat iPP was also calculat ed by the same proced ure and we obt ained the average value of 220.57 kJ/mol, which was also displayed in Figure 6 and was 7.28 kJ/mol lower. These results meant that the Pd nanopart icles could act as a retardant in the ther mal degradation of neat iPP polymer chain. 350 400 450 500 - 1.0 - 0.8 - 0.6 - 0.4 - 0.2 0. 0 dc b Temperature ( o C) TG a Heating rate a : 5 o C/min b :10 o C/min c :15 o C/min d :20 o C/min Figure 4. TGA curves for iPP/Pd (0.27 wt%) nanocomposite at different heating rates. Table 1. Relationships between logβ and T-1 for the iPP/Pd (0.27 wt%) Nanocomposite. α logβ T-1 x 103 (K-1) 0.05 0.10 0.15 0 .2 0 0.25 0.30 0.35 0 .4 0 0.45 0.50 0.70 1.00 1.18 1.30 1.425 1.406 1.388 1.377 1.413 1.394 1.375 1.366 1.406 1.388 1.368 1.360 1.398 1.379 1.340 1.352 1.397 1.378 1.359 1.351 1.394 1.375 1.356 1.348 1.391 1.372 1.353 1.345 1.388 1.369 1.351 1.342 1.387 1.366 1.348 1.340 1.384 1.364 1.346 1.337 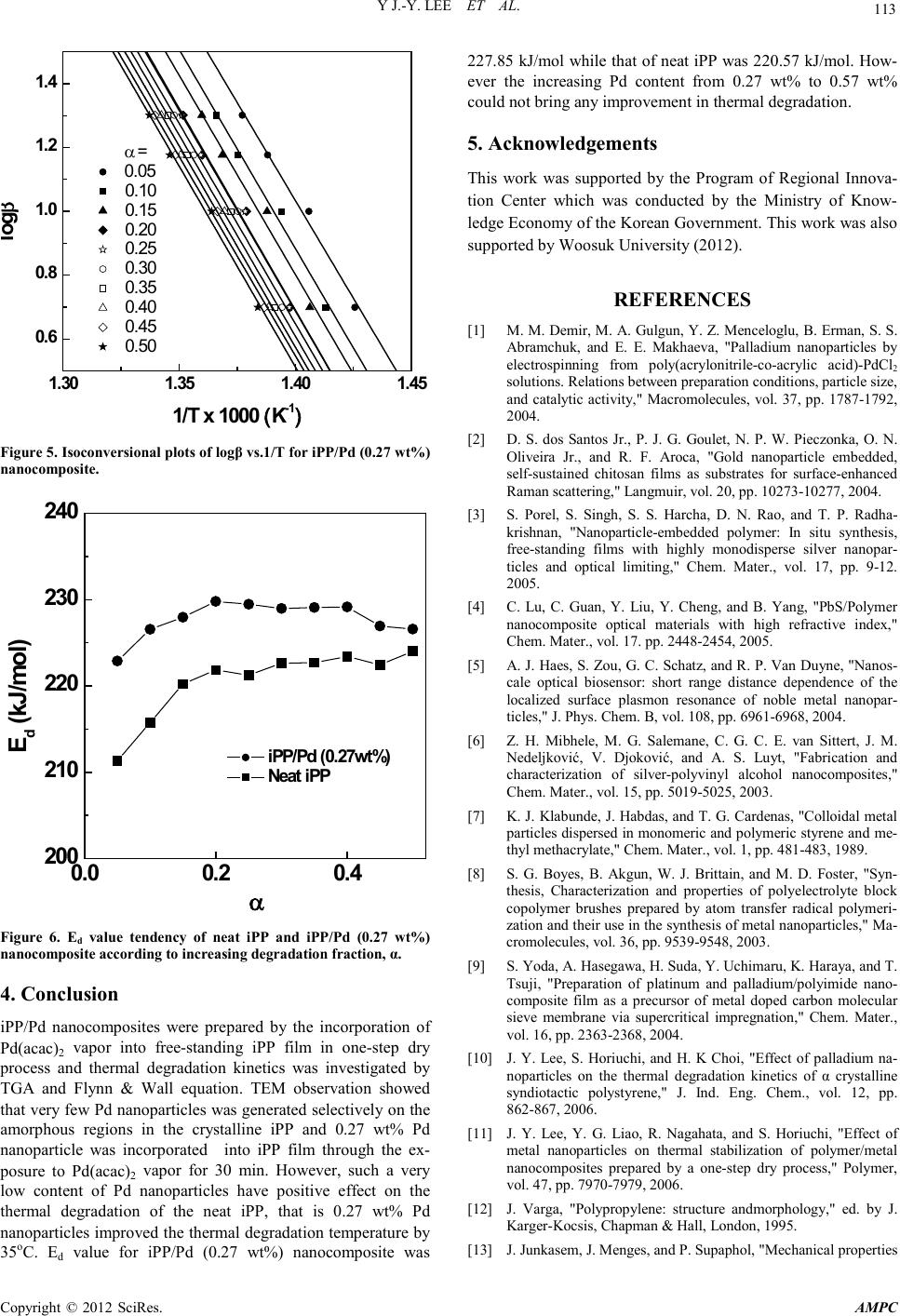 Y J.-Y. LEE ET AL. Copyright © 2012 SciRes. AMPC 113 1.30 1.351.40 1.45 0.6 0.8 1.0 1.2 1.4 α = 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0.50 l ogβ 1/T x 1000 (K -1 ) Figure 5. Isoconversional plots of logβ vs.1/T for iPP/Pd (0.2 7 wt%) nanocomposite. 0.0 0.2 0.4 200 210 220 230 240 iPP/Pd (0.27wt%) Neat iP P E d (kJ/m ol) α Figure 6. Ed value tendency of neat iPP and iPP/Pd (0.27 wt%) nanocomposite according to increasing degradation fraction, α. 4. Conclusion iPP/Pd nanocomposites were prepared by the incorporation of Pd(acac) 2 vapor into free-standing iPP film in one-step dry process and thermal degradation kinetics was investigated by TGA and Flynn & Wall equation. TEM observation showed that very few Pd nanoparticl es was generated selecti vely on the amorphous re gions in the crystalline iPP and 0.27 wt% Pd nanoparticle was incorporated into iPP film through the ex- posure to Pd(acac)2 vapor for 30 min. However, such a very low content of Pd nanoparticles have positive effect on the thermal degradation of the neat iPP, that is 0.27 wt% Pd nanoparticles improved the thermal degrad ation temperatu re by 35oC. Ed value for iPP/Pd (0.27 wt%) nanocomposite was 227.85 kJ/mol while that of neat iPP was 220.57 kJ/mol. How- ever the increasing Pd content from 0.27 wt% to 0.57 wt% could not br i ng any improvement in thermal degradation. 5. Acknowledgements This work was supported by the Program of Regional Innova- tion Center which was conducted by the Ministry of Know- ledge Economy of the Korean Government. This work was also supported by Woosuk University (2012). REFERENCES [1] M . M. Demir, M. A. Gulgun, Y . Z. Menceloglu, B. Erman, S. S. Abramchuk, and E. E. Makhaeva, "Palladium nanoparticles by electrospinning from poly(acrylonitrile-co-acrylic acid)-PdCl2 solutions. Relations between preparation conditions, particle size, and catalytic activity," Macromolecules, vol. 37, pp. 1787-1792, 2004. [2] D. S. dos Santos Jr., P. J. G. Goulet, N. P. W. Pieczonka, O. N. Oliveira Jr., and R. F. Aro ca , "Gold nanoparticle embedded, self-sustained chitosan films as substrates for surface-enhanced Raman scattering," Langmuir, vol. 20, pp. 10273-10277, 2004. [3] S. Porel, S. Singh, S. S. Harcha, D. N. Rao, and T. P. Radha- krishnan, "Nanoparticle-embedded polymer: In situ synthesis, free-standing films with highly monodisperse silver nanopar- ticles and optical limiting ," Chem. Mater., vol. 17, pp. 9-12. 2005. [4] C. Lu, C. Guan, Y. Liu, Y. Cheng, and B. Yang, "PbS/Polymer nanocomposite optical materials with high refractive index," Chem. Mater., vol. 17. pp. 2448-2454, 200 5. [5] A. J. Haes, S. Zou, G. C. Schatz, and R . P. Van Duyne, "Nanos- cale optical biosensor: short range distance dependence of the localized surface plasmon resonance of noble metal nanopar- ticles," J. Phys. Chem. B, vol. 108, pp. 6961-6968, 2004. [6] Z. H. Mibhele, M. G. Salemane, C. G. C. E. van Sittert, J. M. Nedeljković, V. Djokovi ć, and A. S. Luyt, "Fabrication and characterization of silver-polyvinyl alcohol nanocomposites," Chem. Mater., vol. 15, pp. 5019-5025, 200 3. [7] K. J. Klabunde, J. Habdas, and T. G. Cardenas, "Colloid al meta l pa rticles disp ersed in monomeri c and polymeric styrene and me- thyl methacrylate," Chem. Mater., vol. 1, pp. 481-483, 1989. [8] S. G. Boyes, B. Akgun, W. J. Brittain, and M. D. Foster, "Syn- thesis, Characterization and properties of polyelectrolyte block copolymer brushes prepared by atom transfer radical polymeri- zati on a nd their use i n t h e synthesis of metal nanoparticles," Ma- cromolecules, vol. 36, pp. 9539-9548, 2003. [9] S. Yoda, A. Hasegawa, H. Suda, Y. Uchimaru, K. Haraya, and T. Tsu ji, "Preparation of platinum and palladium/polyimide nano- composite film as a precursor of metal doped carbon molecular sieve membrane via supercritical impregnation," Chem. Mater., vol. 16, pp. 2363-2368, 2004. [10] J. Y. Lee, S. Horiuchi, and H. K Choi, "Effect of palladium na- noparticles on the thermal degradation kinetics of α crystalline syndiotactic polystyren e ," J. Ind. Eng. Chem., vol. 12, pp. 862-867, 2006. [11] J. Y. Lee, Y. G. Liao, R. Nagahata, and S. Horiuchi, "Effect of metal nanoparticles on thermal stabilization of polymer/metal nanocomposites prepared by a one-step dry process," Polymer, vol. 47, pp. 7970-7979, 2006. [12] J. Varga, "Polypropylene: structure andmorphology," ed. by J. Kar ge r-Kocsis, Ch apman & Hall, Lond on, 1995. [13] J. Junkasem, J. Menges, and P. Supaphol, "Mechanical properties  Y J.-Y. LEE ET AL. Copyright © 2012 SciRes. AMPC 114 of injection-molded is otactic polypropylene/roselle fiber compo- sites," J. Appl. Polym. Sci., vol. 101, pp. 3291-3300, 2006. [14] X. Sun, H. Li, J. Wang, and S. Yan, "Shear-induced interfacial structure of isotactic polypropylene (iPP) in iPP/fiber compo- sites," Macrom olec ules, vol. 39, pp . 8720-8726, 2006. [15] J. H. Flynn, "Degradation kinetics applied to lifetime predictions of polymers," Polym. Eng. Sci., vol. 20, pp. 675-677, 1980. [16] J. Y. Lee, H. K. Lee, M. J. Shim, and S. W. Kim, "Thermal de- composition Characteristics of epoxy network chemically toughened with liquid rubber using dynamic TG analysis," J. Ind. Eng. Chem. , vol. 6, pp. 250-255, 2000. |

