Paper Menu >>
Journal Menu >>
 Advances in Ma terials Physics and Che mist ry, 2012, 2, 78-83 doi:10.4236/ampc.2012.24B022 Published Online December 2012 (htt p://www.SciRP.org/journal/ampc) Copyright © 2012 SciRes. AMPC Impacts of Melt Spinning and Element Substitution on Electrochemical Characteristics of the La–Mg–Ni-based A2B7-Type Alloys Yang-huan Zhang 1,2, Ho ng-wei Sha ng1,2, Ying Cai1, Zhong -hui H ou1, Guo-fang Zhang1,2, Dong -liang Zhao2 1Key Laboratory of Integrated Exploi tati on of Baiyun Obo Multi-Met al Resources, Inner Mongolia University of Science and Technology Baoto u, China 2Department of Function al Ma terial Resea r ch , Cent ral Iron and Steel Resea rc h Institute, Beijing, China Email: zyh59@yahoo.com.cn, shanghongwei86@126.com, caiying@imust.cn, hzh329@yahoo.cn, afang1001@126.com, dlzhao@sina.com Received 2012 ABSTRACT The partial substitution of Zr for La has been per for med in order to ameliorate th e elect roch e mical hydrogen storage performances of La–Mg–Ni based A2B7-type electrode alloys. The melt spinning technology was used to prepare the La0.75−xZrxMg0.25Ni3.2Co0.2Al0.1 (x=0, 0.05, 0.1, 0.15, 0.2) electrode alloys. The impacts of the melt spinning and the substituting La with Zr on the structures and the electrochemical h ydrogen sto rage char acteri stics of th e alloys were systemically investigated. The anal ysis of XRD and TEM reveals that t he as-cast and spun alloys have a multiphase structure, composing of two main phases (La, Mg)2Ni7 and LaNi5 as wel l as a re- sidu al phase LaNi2. The electro chemical measurement in dicates that b oth the substitution of Zr for La and the melt spinning ameli- orate the electr ochemical c ycle stabi lity of the all oys dramatically. Furthermore, the high rate discharge ability (HRD) of the as -spun (10 m/s) alloys notably declines with growing the amount of Zr substitution, while it first augments and then falls for the (x=0.1) alloy with rising the spinning rate. Keywords: A2B7-Type Alloy; Substituting La with Zr; Melt-Spinning; Elect rochemical Characteri s tics 1. Introduction With rapid development of electric equipments, the requirement for new electrode materials with superior performances, espe- cially high discharge capacity and electrochemical hydrogen storage kin etics, has become more and more pressing. The rare earth-based AB5-type alloys, although have been industrialized in large scale in China and Jap an, are suffe ri ng a severe frustra- tion on account of their limited d ischarge capa cit y of about 330 mAh/g. La–Mg–Ni-system AB3 and A2B7-type alloys have been considered to be the most promising candidates as the negative electrode materials of Ni–MH rechargeable battery in virtue of their higher discharge capacities (380–410 mAh/g) and low production costs since Kadir et al. [1] and Kohno et al. [2] reported their research results. The National High Technol- ogy Research and Development Program of China (for short “863” Program) provides powerful financial support in order to promote the industrialization of these new-type alloys. Such a lot of efforts have been dedicated to realizing this target and dramatic progress has been achieved, about which Liu et al. have published a perfect summari zation recentl y [3,4]. Howev- er, the Chinese researchers in this area were deeply frustrated by a fact that the produ ction of the new t ype alloys as the n ega- tive electro de in Ni –MH battery has not been found in China as a result of little poor electrochemical cycle stability of the elec- trode alloys. A serious challenge faced by researchers keeps intact, enhancing the cycle stability of the alloy without reduc- ing its dischar ge capacity. The element substitution has been regarded as one of the ef- fective methods for improving the overall properties of the hydrogen storage alloys. In addition, the preparation technology is also extremely important for improving the performances of the alloys. Therefore, it is expected that the combinatio n of an optimized amount of Zr substitution with a proper melt spin- ning technique may yield an alloy with high di scharge capacit y and good cycling stability. The A2B7-type La0.75−xZrxMg0.25Ni3.2 Co0.2Al0.1 (x=0 -0.2) alloys were prepared by melt spinning, and a systematic investigation on the effects of the substitution of Zr for La and the melt spinning on the electrochemical cycle stability and kinetics of the electrode alloys has been per- formed. 2. Experimental The chemical compositions of the alloys were La0.75-xZrxMg0.25 Ni3.2Co0.2Al0.1 (x=0, 0.05, 0.1, 0.15, 0.2). For convenience, the alloys were denoted with Zr content as Zr0, Zr0.05, Zr0.1, Zr0.15 and Zr0.2, respectively. The alloy ingots were prepared using a vacuum induction furnace in a helium atmosphere under a pressure of 0.04 MPa. A part o f the as-cast alloys was r e-melted and spun by melt-spinning with a rotating copper roller. The spinning rate was approximately expressed b y the l inear veloc- ity of the copper roller. The spinning rates used in the experi- ment were 5, 10, 15 and 20 m/s, respecti vely. The phase structures and compositions of the as-cast and *Supported by National Natural Science Foundation s of China (51161015 and 50961009), National 863 plans projects of China (2011AA03A408) , Natural Science Foundatio n of Inner Mongo lia, China (2011Z D10 and 2010ZD05).  Y.-H. ZHANG ET AL. Copyright © 2012 SciRes. AMPC 79 spun alloys were characterized by XRD (D/max/2400). The thin film samples of the as-spun alloys were prepared by ion etching for observing the morphology with HRTEM (JEM- 2100F). The round electrode pellets in a 15 mm diameter were pre- pared by cold pressing a mixture of alloy powder and carbonyl nickel powder in the weight ratio of 1:4 under a 35 MPa pres- sure. Aft er d ried for 4 h, th e electrod e pellets were immers ed in a 6 M KOH solution for 24 h in order to wet the electrodes fully befor e the electr ochemical measu r ement. Electrochemical measurements were performed at 30°C by using a tri-electrode open cell, consisting of a working elec- trode (the metal h ydride elect rode), a sintered Ni(OH)2/NiOOH counter electrode and a Hg/HgO reference electrode, which were immersed in a 6M KOH el ectrol yte. The vol tage between the negative electrode and the reference electrode was defined as the disch arge voltage. In every cycle, t he alloy electro de was firstly charged with a constant current density of 300 mA/g. After restin g 15 min, it was discharged at th e same current den- sity to cut-off vo ltage of –0.500 V. The electrochemical impedance spectra (EIS), the hydrogen diffusion in alloy bulk and the Tafel polarization curves of the alloys were measured by an electrochemical workstation (PARSTAT 2273). The EIS of the alloy electrodes were meas- ured in the frequency range from 10 kHz to 5 mHz at 50% depth of discharge (DOD). The Tafel polarization curves were measured i n t he p o tenti al ran ge of –1.2 to +1.0 V (vs. Hg/HgO) with a scan rate of 5 mV/s also at 50% depth of discharge. For the potentiostatic discharge, the test electrodes in the fully charged state were discharged at 500 mV potential steps for 5000s, using the electrochemistry corrosion software (Corr- Ware). 3. Results and Discussion 3.1. Structural characteristics The XRD pattern s o f the as-cast and spun La0.75-xZrx Mg0.25Ni3.2 Co0.2Al0.1 (x=0-0.2) alloys are presented in Fi gure 1. It is evi- dent that the melt spinning and Zr substitution bring on an ob- vious broadening of the major diffraction peaks of the alloys, to be ascribed to the refined grain and the stored stress in the grains by melt spinning and Zr substitution. It is found that the as-cast and spun alloys hold a multiphase structure, composing of t wo major phases ( La, Mg)2Ni7 and LaN i5 as well a s a resid- ual phase LaN i2. The structures of the alloys maintain almost unaltered after partially substituted by Zr. The lattice parame- ters together with the abundances of the (La, Mg)2Ni7 and LaNi 5 majo r phases in the alloys, which were calculated by Jade 6.0 software based on the XRD d ata, are listed in Ta ble 1 . It is found that the Zr substitution arouses an increase of the LaNi 5 phase and a decrease of the (La, Mg)2Ni7 p hase. Mean- while, it also leads to a d ecrease of th e lat tice co n stan ts an d cell volume of the two major phases, which is due to the fact that the atom radius of Zr is smaller than that of La. Furthermore, the shrink of the cell volume, incurred by the substitution, just i- fies the successful alloying of Zr with (La, Mg)2Ni7 and LaNi5 major phases. The TEM micrographs of the as-spun (10 m/s) La0.75-xZrx Mg0.25Ni3.2Co0.2Al0.1 (x=0-0.2) alloys are demons trated in Fig- ure 2. It can be seen from Figure 2 th at the ch ange of th e mor- phologies of the alloys seems to be equivocal with the various of Zr content. However, the amplified morphologies of Figure 2 reveal that the as-spun Zr0 and Zr0.1 alloys display an entire crystalline structure. And some crystal defects such as subgrains and grain boundaries can be seen clear ly. It is evid ent that Z r0.2 alloy displays an obvious amorphous-like structure, and the nanocrystalline region, the amorphous region as well as the transition region can be clearly viewed from the amplified morphology of Figure 2(c), which seems to be conflicting with the result in Figure 1 due to no amorphous phase is d etected by XRD observation. One probable reason is that the amorphous- like phase for ms at so me s elective locations in the as-spun alloy and its amount is very small, thus, the XRD patterns could not detect i ts presence. Figure 1.XRD profiles of the as-cast spun alloys: (a) Zr0.1, (b) As-spun (10 m/s) Ta ble 1. Lattice parameters, abundances of LaNi5 and (La, Mg)2Ni7 major phases. Spinni ng rate Alloys Major phases Lattice c onsta nts Cell volume Phase abund-ance A(nm) C(nm) V (nm3) (wt.%) 10 m/s Zr0 (La, Mg)2Ni7 0.5199 2.4406 0.5712 72.31 LaNi5 0.5192 0.4180 0.0976 25.24 Zr0.05 (La, Mg)2Ni7 0.5142 2.4362 0.5578 66.03 LaNi5 0.5130 0.4107 0.0936 31.02 Zr0.1 (La, Mg)2Ni7 0.5071 2.4308 0.5413 64.25 LaNi5 0.5059 0.4082 0.0905 34.08 Zr0.15 (La, Mg)2Ni7 0.5031 2.4251 0.5315 60.97 LaNi5 0.5011 0.4053 0.0881 36.50 Zr0.2 (La, Mg)2Ni7 0.4963 2.4174 0.5156 58.02 LaNi5 0.4933 0.4021 0.0847 39.32 Figure 2. TEM micrographs of the as-spun (10 m/s) alloys: (a) Zr 0 alloy, (b) Zr0.1 alloy, (c ) Zr0.2 alloy. 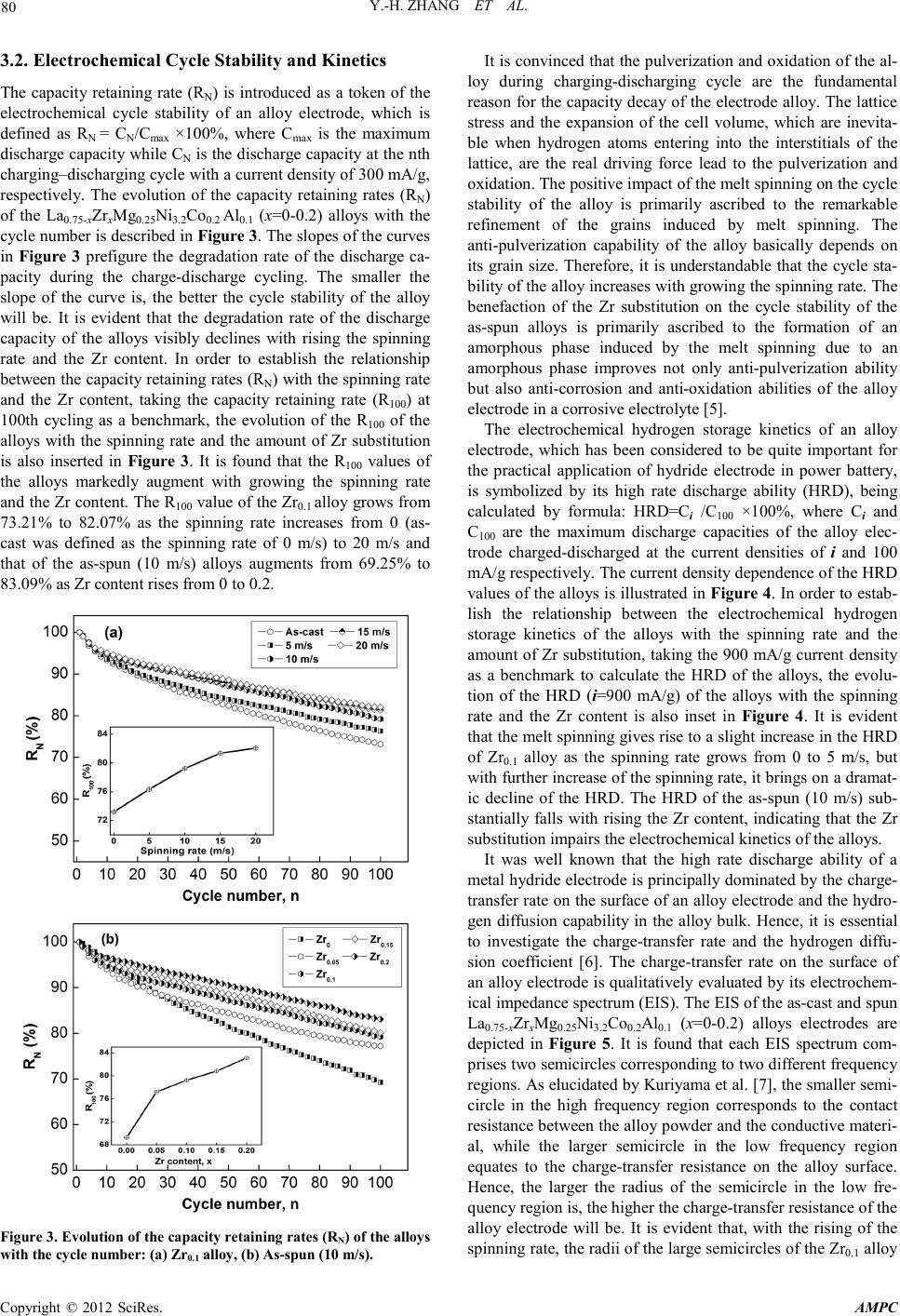 Y.-H. ZHANG ET AL. Copyright © 2012 SciRes. AMPC 80 3.2. Electroche mical Cycle Stability and Kine t ics The capacity retaining rate (RN) is introduced as a token of the electrochemical cycle stability of an alloy electrode, which is defined as RN = CN/Cmax ×100%, where Cma x is the maximum disch arge capacity while CN is the disch arge capacity at the nth charging–discharging cycle with a current density of 300 mA/g, respecti vely. The evolution of the capacity retaining rates (RN) of the La0.75-xZrxMg0.25Ni3.2Co0.2 Al0.1 (x=0-0.2) alloys with the cycle number is described in Figure 3. The slopes of the cur ves in Figure 3 prefigure the degradation rate of the discharge ca- pacity during the charge-discharge cycling. The smaller the slope of the curve is, the better the cycle stability of the alloy will be. It is evident that the degradatio n rate of the discharge capacity of the alloys visibly declines with rising the spinning rate and the Zr content. In order to establish the relationship between th e capacity retaini ng rates (RN) with the spinning rate and the Zr content, taking the capacity retaining rate (R100) at 100th cycling as a benchmark, the evolution of the R100 of the alloys with the spinning rate and the amount of Zr substitution is also inserted in Figure 3. It is found that the R100 values of the alloys markedly augment with growing the spinning rate and the Zr content. The R100 value of the Zr0.1 alloy grows from 73.21% to 82.07% as the spinning rate increases from 0 (as- cast was defined as the spinning rate of 0 m/s) to 20 m/s and that of the as-spun (10 m/s) alloys augments from 69.25% to 83.09% as Zr content rises from 0 to 0.2. Figure 3. Evolutio n of the capaci ty re taini ng ra tes (RN) of the alloy s with the cycle number: (a) Zr0.1 alloy, (b) As-spun (10 m/s). It is convinced that the pulverization and oxidation of the al- loy during charging-discharging cycle are the fundamental reason for the capacity decay of the el ectrode alloy. The lattice stress and the expansion of the cell volume, which are inevita- ble when hydrogen atoms entering into the interstitials of the lattice, are the real driving force lead to the pulverization and oxidation. The positive impact of the melt spinning on the cycle stability of the alloy is primarily ascribed to the remarkable refinement of the grains induced by melt spinning. The anti-pulverization capability of the alloy basically depends on its grain size. Therefore, it is understandable that the cycle sta- bility of the alloy increases with growing the spinning rate. The benefaction of the Zr substitution on the cycle stability of the as-spun alloys is primarily ascribed to the formation of an amorphous phase induced by the melt spinning due to an amorphous phase improves not only anti-pulverization ability but also anti-corrosion and anti-oxidation abilities of the alloy electrode in a corrosive electrolyte [5]. The electrochemical hydrogen storage kinetics of an alloy electrode, which has been considered to be quite important for the practical application of hydride electrode in power battery, is symbolized by its high rate discharge ability (HRD), being calculated by formula: HRD=Ci /C100 ×100%, where Ci and C100 are the maximum discharge capacities of the alloy elec- trode charged-discharged at the current densities of i and 100 mA/g respectivel y. The current density dependence of the HRD values of the allo ys is illustrated in Figure 4. In order to estab- lish the relationship between the electrochemical hydrogen storage kinetics of the alloys with the spinning rate and the amount of Zr substitution, taking the 900 mA/g current density as a benchmark to calculate the HRD of the alloys, the evolu- tion of the HRD (i=900 mA/g) of the alloys with the spinning rate and the Zr content is also inset in Figure 4. It is evident that the melt spinning gives rise to a slight increase in the HRD of Zr0.1 alloy as the spinning rate grows from 0 to 5 m/s, but with further increase of the spinning rate, it brings on a dramat- ic decline of the HRD. The HRD of the as-spun (10 m/s) sub- stantially falls with rising the Zr content, indicating that the Zr substitution impairs the electrochemical kinetics of the alloys. It was well known that the high rate discharge ability of a metal hydrid e electrod e is principally dominated by the ch arge- transfer rate on the surface of an alloy electrode and the hydro- gen diffusion capability in the alloy bulk. Hence, it is essential to investigate the charge-transfer rate and the hydrogen diffu- sion coefficient [6]. The charge-transfer rate on the surface of an alloy electrod e is qualitatively evaluated by its electrochem- ical impedan ce sp ectrum (EIS). The EIS of the as-cast and spun La0.75-xZrxMg0.25Ni3.2Co0.2Al0.1 (x=0-0.2) alloys electrodes are depicted in Figure 5. It is found that each EIS spectrum com- prises two semicircl es corresponding to two different frequency regions. As elucidated by K uriyama et al . [7], the smaller semi- circle in the high frequency region corresponds to the contact resistan ce between the all oy powder and the co nduct ive materi- al, while the larger semicircle in the low frequency region equat es to the charge-trans fer resistance on the alloy surface. Hence, the larger the radius of the semicircle in the low fre- quency region is, the h igher the charge-transfer resistance of th e alloy electrode will be. It is evident that, with the rising of the spinning rate, the radii of th e large semicircles of the Zr0.1 alloy  Y.-H. ZHANG ET AL. Copyright © 2012 SciRes. AMPC 81 in the low frequency first shrink and then expand, and those of the as-spun (10 m/s) alloys always swell with growing the Zr content. The hydrogen diffusion coefficients in the alloys can be derived by measuring the semilogarithmic curves of anodic curren t versus working duration o f an alloy as depicted in Fig- ure 6. Based on the model founded by White et al. [8], the dif- fusion coefficient (D) of the hydrogen atoms in the bulk of the alloy can be calculated by following formulae: 2 0 22 6 log log()2.303 s FD D iCC t da a π =±−− (1) 2 2 2.303 logad i Ddt π = − (2) In (2), logdi dt is the slope of the linear region of Figure 6, which can be gained by origin 7.5 software in a walk. The alloy particle radius (a) is supposed to be a=15 μm. Thus, hydrogen diffusion coefficient D can be easily o btained, and the results are also presented in Figure 6. It is evident that with the rising of the spinning rate, the D values of the Zr0.1 alloy first mount up and then fall, and those of the as-spun (10 m/s) alloys mo- notonously drop with rising the Zr content. Figure 4. Evolution of the high rate discharge ability (HRD) of the alloys with the discharge current density: (a) Zr0.1 alloy, (b) As- spun (10 m/s). Figure 5. Electrochemical impedance spectra (EIS) of the alloy electrodes: (a) Zr0.1 alloy, (b) As spun (10 m/s). Figure 6. Semilogarithmic curves of anodic current vs. time res- ponses of the alloys: (a) As-spun (10 m/s), (b) Zr0.15 alloy.  Y.-H. ZHANG ET AL. Copyright © 2012 SciRes. AMPC 82 The limiting current density (IL), another important electro- chemical kinetics parameter which is mainly dominated by the hydrogen diffusion in the bulk of the alloy during anodic pola- rization [9], can be obtained by measuring the Tafel polariza- tion curves of an alloy. The Tafel po larizatio n curves of La0.75-x ZrxMg0.25Ni3.2Co0.2Al0.1 (x=0-0.2) alloys are depicted in Figure 7, from which a clear inflection point in each anodic polariza- tion curve can be seen, there being a limiting value of the cur- rent density which is defined as limiting current density (IL). It indicates that an oxidation reacti on took place on the sur face of the alloy electrode, and the generated oxidation product resists further penetration of hydrogen atoms [10]. The IL va lues of the alloys as a function of the spinning rate and the Zr content are also inset in Figure 7. It is visible that th e IL values of the Zr0.1 alloy first rise and then decline, and those of the as-spun (10 m/s) alloys always decrease wit h growing t he Zr co ntent. Figure 7. Tafel polarization curves of the as-cast and spun alloys: (a) Zr0.1 alloy, (b) As-spun (10 m/s). Based on the investigation of the electrochemical kinetics, some elucidation s can be provided as the reasons why the HRD of the Zr0.1 alloy has a ma xi mum val u e with the variation of the spinning rate. Upon the refined microstructure by melt spinning, a lot of new crystallites and grain boundaries are generated, which may act as the fast diffusion paths for hydrogen absorp- tion [11], enhancing the HRD of the alloy. However, it must be mentioned that the refinement of the grains resulted from the melt spinning severely impairs the charge-transfer rate on the alloy surface due to the fact that the refined grains effectively prohibit the pulverization of the alloy particles, a lower new surface o f the al loy electrode being formed, decreasing th e rate of charge transfer at the alloy-electrolyte interface. It is the above-mentioned contrary impacts engendered by melt spin- ning that lead to a max imum HRD of the alloys. Furthermore, the d ecreased HRD of th e as-spun (10 m/s) alloys by Zr substi- tution is ascribed to the formation of a amorphous-like structur e which not only increases the charge-transfer resistance of the alloy electrodes but also hinders the hydrogen diffusion from inner of the bulk to the surface, and subsequently results in the drop of the electroch emical ki netic property. 4. Conclusions The La-Mg-Ni system A2B7-type La0.75-xZrxMg0.25Ni3.2Co0.2 Al0.1 (x=0, 0.05, 0.1, 0.15, 0.2) electrode alloys were success- fully synthesized by melt spinning. The as-spun Zr0 alloy exhibits an enti re n anocr ystalli ne an d micr o-crystall ine struct ure, where as the as-spun alloy substituted by Zr exhibits a amorphous-like structure. Both the melt spinning and the substitution of Zr for La markedly enhance the electrochemical cycle stability of the electrode alloys. The electrochemical kinetics, including the HRD, the hydrogen diffusion coefficients (D) as well as the limiting current density (IL) of the Zr0.1 alloy first increase and then decline with growing the spinning rate, and those of the as-spun (10 m/s) alloys monotonously fall with rising the Zr content, for which the refinement of the grains and the forma- tion of the amorphous-like structure are principally responsible. 5. Acknowledgements This work is supported by National Natural Science Founda- tions of China (51161015 and 50961009), National 863 plans projects of China (2011AA03A408), Natural Science Founda- tion of Inner Mongolia, China (2011ZD10 and 2010ZD05). REFERENCES [1] K. Kadir, T. Sakai, I. Uehara, “Synthesis and structure determi- nation of a new series of hydrogen storage alloys: RMg2Ni9 (R=La, Ce, Pr, Nd, Sm and Gd) built from MgNi2 La v e s -type layers alternating with AB5 layers,” J. Alloys Compd., vol. 257, pp. 115–121, 1997. [2] T. Kohno, H. Yoshida, F. Kawashima, T. Inaba, I. Sakai, M. Yamamoto, and et al., “Hydrogen storage properties of new ternary system alloys: La2MgNi9, La5Mg2Ni23, La3MgNi14,” J. Alloys Compd., vol. 311, pp. L5–L7, 2000. [3] Y.F. Liu, H. Pan, M. Gao and Q. Wang, “Advanced hydrogen storage alloys for Ni/MH rechargeable batteries,” J. Mater. Ch em, vol. 21, pp. 4743–4755, 2011. [4] Y.F. Liu, Y.H. Cao, L. Huang, M.X. Gao, H.G. Pan, “Rare earth–Mg–Ni-based hydrogen storage alloys as negative elec- trode materials for Ni/MH batteries,” J. Alloys Compd., vol. 509, pp. 675–686, 2011. [5] Y.H. Zhang, B.W. Li, H.P. Ren, Y. Cai, X.P. Dong, X.L. Wang, “Investi gati on on structu res and electroc hemica l performanc es of the as-cast and -quenched La0.7Mg0.3Co0.45Ni2. 55–xFex (x=0–0.4) electrode alloys,” Int. J. Hydrogen Energy, vol. 32, pp. 4627–4634, 2007. [6] A. Gasiorowski, W. Iwasieczko, D. Skoryna, H. Drulis, and M.  Y.-H. ZHANG ET AL. Copyright © 2012 SciRes. AMPC 83 Jurczyk, “Hydriding properties of nanocrystalline Mg2−xMxNi al- loys synthesized by mechanical alloying (M=Mn, Al),” J. Alloys Compd., vol. 364, pp. 283–288, 2004. [7] N. Kuriyama, T. Sakai, H. Miyamura, I. Uehara I, H, Ishikawa, T. Iwasaki. “Electrochemical impedance and deteriorat ion behavior of metal hydride electrodes,” J. Alloys Compd., vol. 202, pp. 183–197, 1993. [8] G. Zhong, B.N. Popov, and R.E.White, “Electrochemical Deter- mination of the Diffusion Coefficient of Hydrogen Through an La N i 4.25Al0.75 Electrode in Alkaline Aqueous Solution,” J. Elec- trochem. Soc., vol. 142, pp. 2695–2698, 1995. [9] B.V. Ratnakumar, C. Witham, JR. R.C. Bowman, A. Hightower, and B. Fultz, “Electrochemical studies on LaNi5–xSnx metal hy- dride alloys,” J. Electrochem. Soc., vol. 143, pp. 2578–2584, 1996. [10] X.Y. Zhao, Y. Ding, L.Q. Ma, L.Y. Wang, M. Yang, and X.D. Shen, “Electrochemical properties of MmNi3.8Co0.75Mn0.4Al0.2 hydrogen storage alloy modified with nanocrystalline nickel,” Int. J. Hydrogen Energy, vol. 33, pp. 6727–6733, 2008. [11] Y. Wu, W. Hana, S.X. Zhou, M.V. Lototsky, J.K. Solberg, V.A. Yartys, “Microstructure and hydrogenation behavior of ball-milled and melt-spun Mg–10Ni–2Mm alloys,” J. Alloys Compd., vol. 466, pp. 176–181, 2008. |

