 J. Biomedical Science and Engineering, 2010, 3, 892-899 doi:10.4236/jbise.2010.39119 Published Online September 2010 (http://www.SciRP.org/journal/jbise/ JBiSE ). Published Online September 2010 in SciRes. http://www.scirp.org/journal/jbise Cytotoxic and inhibitory activity of ceramide on cancer cell lines Muthana Ibrahim Maleek Department of Biology, College of Science, University of Baghdad, Baghdad, Iraq. Email: fadhilsalih@gmail.com Received 21 January 2010; revised 15 February 2010; accepted 15 February 2010. ABSTRACT The inhibitory activity of ceramide on cancer cells was evaluated and its cytotoxicity on different cancer cell lines was measured. Ceramide was separated fro- m bovine brain and spinal cord, and extracted by or- ganic solvents. The crude extract was purified by us- ing silicic acid column. Detection and identification of purified extract were carried out by using three ass- ays: visualization, spectrophotometry and infrared. Cytotoxic effect of different concentrations (7, 15, 30 and 60 μM) of ceramide on HEp-2, RD , AMGM5, REFAM3 and AMN3 cancer cell lines was studied. Results showed that ceramide at 30 μM imposed cy- totoxic activity on all cancer cell lines especially on AMGM5. Effect of ceramide at 30 μM on cell divi- sion of human lymphocyte was also examined. Sig- nificant reductions in mitotic and blast indices were observed. In addition, No genotoxic effects or chrom- osomal aberration were detected in lymphocyte chr- omosomes when ceramide was tested in vitro. Keywords: Ceramide; Cancer Cell Line; Cytotoxicity; Genotoxicity; Chromosomal Aberration 1. INTRODUCTION Cancer is a clonal disorder characterized by genetic in- stability and shift in the control mechanism that govern cell proliferation and differentiation [1,2]. It is a world- wide health problem, with a geographical variation all over the world [3]. Conventional cancer therapy is based on surgery, radi- otherapy, chemotherapy or combinations of them. Gen- erally, surgery and radiotherapy are preferred in local- ized tumors and chemotherapy is used when cancer cells are spread through the body [4]. The introduction of can- cer chemotherapy in the 5th and 6th decades of the last century has resulted in the development of curative the- rapeutic interventions for patients with several types of solid tumors and hematopoietic neoplasms. However, im- portant obstacles were encountered in the use of chemo- therapy that included toxicity to the normal tissues of the body and the presence of mutations that confer resis- tance to these chemotherapeutic agents [5]. Therefore, cancer patients as well as many physicians began to re- quest natural products for treatment due to their multiple effects in treating the disease, relieving patient’s symp- toms in addition to improved safety and lower cost [6]. Natural products have long been a fertile source of cure for cancer, which is projected to become the major cause of death in this century [7]. Therefore, a natural product, ceramide, was tested for its inhibitory and cytotoxic ef- fects on tumor cells. The combination of sphingosine plus fatty acid is kno- wn as ceramide (n-acylsphingosine), sphingosine or rela- ted bases are important membrane components in animal cells. They are present in large amounts especially in br- ain and nerve tissue. Sphingolipids can be found in all eukaryotic cells and the major phosphosphingolipid, sp- hingomyelin, is very abundant in the outer leaflet of the plasma membrane constituting of about 30% of all lipids [8]. However, altogether over 300 sphingolipids with dis- tinct head groups have been identified in eukaryotic cells. In most mammalian cells, the structure is based on the long chain of sphingosine base, which is subsequenttly acylated to form ceramide (Figure 1) and more complex sphingolipids are formed by the addition of polar head groups to the C1 position of ceramide [9]. Sphingom- yelins as phospholipids are formed when ceramide reacts with phosphatidylcholine to form sphingomyelin plus diacylglycerol. This occurs mainly in the Golgi appara- tus and to a lesser extent in the plasma membrane. In organelles involved in secretory and endocytic processes sphingomyeline is restricted to the luminal aspect [10]. Ceramide was confirmed to function as a second me- ssenger in several cellular processes, including apoptosis, growth suppression, differentiation, transformation, proli- feration, regulate cell-cell, cell-substrate interactions and  M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 893 Sphingosine O + ║ CH3(CH2)14C---S----CoA Palmitoyl-CoA ↓ Ceramide (N-palmitoylsphingosine) Figure 1. Formation of ceramide [10]. cell senescence [11]. In turn, aberrant metabolism of cer- amide has been associated with inflammation, tumori ge- nesis, diabetes, and neurodegenerative disorders [12,13]. Ceramide has emerged as a novel class of signaling mol- ecules that also regulate phagocytosis [14]. The aim of this study is to purify and characterize the ceramide that was isolated from bovine brains and to study its ability to inhibit tumor cells in vitro. 2. MATERIALS AND METHODS 2.1. Chemicals and Culture Media Ciramide was separated and purified according to Dasgu- pa et al. [15] and Dasgupta and Hogan, [16]. A modified method of Bischel and Austin [17] was used to visualize ceramide. The modification offered more sensitive and stable color reaction in practice (down to 5-10 μg). Silica gel G thin-layer chromatography and standard solvent systems (chloroform: methanol: acetic acid; 95: 4.5:0.5) were employed to visualize ceramide. Chromat- ograms were dried in a ventilated hood and sprayed with the clorox reagent (5ml of clorox was added to 50 ml benzene and then 5ml glacial acetic acid). The clorox re- agent must be used immediately. After spraying, chro- matograms were air dried in a hood to remove all unbou- nd Cl2. In addition, paper chromatograms were also used for ceramide visulization. They were rinsed twice with tap water for 1-2 min duration, and allowed to air dry until damp. They were then sprayed with benzidine re- agent (0.5 g benzidine and 0.2 g of KI dissolved in 50 ml of 50% ethanol) and then filtered. This solution was kept out of direct light and used within 2h after preparation. Alternatively, paper chromatograms may be dipped in the benzidine reagent. Two more ways were also used for visualization purposes; spectrophotometry assay and In- frared assay, as described by Mc Murry [18]. Rosswell Park Memorial Institute (RPMI)-1640 med- ium [19] was used as cell line growth medium. It was ki- ndly provided by ICCMGR. All chemicals, solvents and reagents were supplied by BDH (UK) unless otherwise stated. 2.2. Cell Lines 1) Human larynx epidermoid carcinoma (HEp-2) cell line was kindly provided by the Iraqi Center for Cancer and Medical Genetics Research (ICCMGR). This human cell line was originally obtained from a 57-year-old man with a primary tumor of the larynx. Its biological and chemical sensitivity has been very well documented [20-23]. 2) Rhabdomyosarcoma (RD) cell line was kindly pro- vided by ICCMGR. This human cell line was derived from a biopsy specimen obtained from a pelvic rhabdo- myosarcoma of a 7-year-old 3) Ahmed-Majeed-glioblastoma-multiforme-2005 (AM- GM5) cell line was kindly provided by Dr. A. Al-Sham- ery from ICCMGR. This human cell line was obtained from a human cerebral glioblastoma multiforme (GBM) of a 72-year-old Iraqi male who underwent surgery for intracranial tumor. Morphologic examination, immuno- cytochemical staining, growth kinetics, and karyotypic characteristics of this cell line were studied at ICCMGR [A. Al-Shamery, personal communication]. The cultured cells were spindle-like or polyhedral in shape. The po- pulation doubling time was 28 hours. The chromosomal number varied between 38 and 46, with mode chromo- somal number of 42. In addition, chemotherapeutic drug sensitivity was studied for AMGM5 cells at ICCMGR and the cells appeared to be resistant to Cisplatin, Vin- cristine and Etoposide. 4) Ahmed-Mohammed-Nahi-2003 (AMN3) cell line was kindly provided by ICCMGR. This murine mamm- ary adenocarcinoma cell line was derived from a sponta- neous mammary adenocarcinoma of female BALB/c mice [25]. 5) Rat Embryo fibroblasts (REFAM3) cell line was established and kindly provided by Dr. A. Al-Shamery from ICCMGR. Cells of this normal murine cell line were a mixture of fibroblastic and epithelial cells with normal chromosomal picture [25]. Cell lines were maintained as described by Freshney [19]. 2.3. Assay of Ceramide Toxicity Confluent monolayers were treated as in subculture .The growth medium was decanted off and the cell sheet wash- ed twice with PBS and trypsinized using trypsin-versene (Gibco, Canada) [19]. When the cells are in exponential growth, medium was removed and ceramide at varying concentrations (7, 15, 30 and 60 μM) in serum free me- dium (SFM) plus DMSO (1μl /ml) [26] were added to the wells, five replicates were used for each concentra- tion of ceramide. Twenty columns were used as control (cells treated with SFM plus DMSO only). Plates were 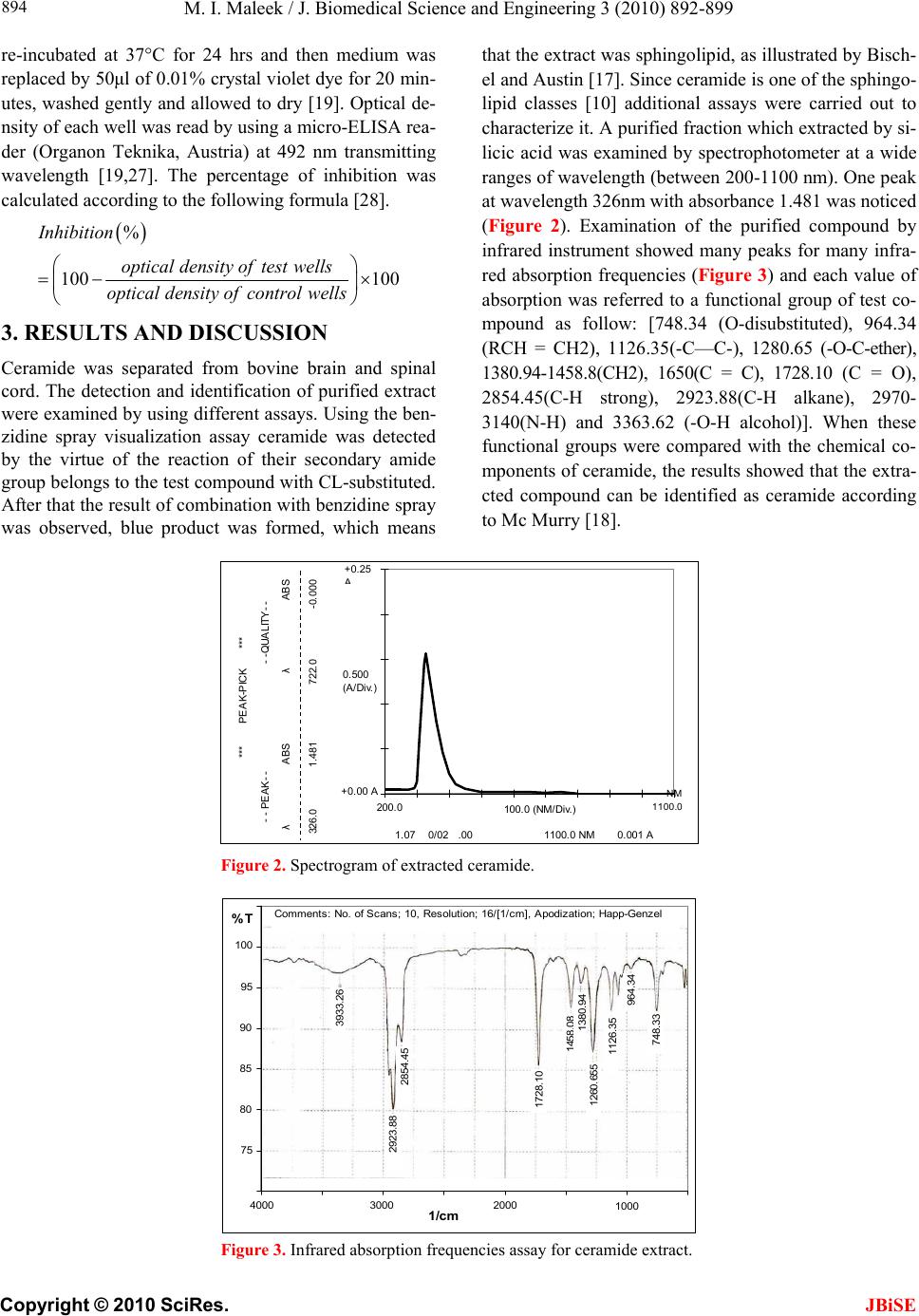 M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. 894 re-incubated at 37°C for 24 hrs and then medium was replaced by 50μl of 0.01% crystal violet dye for 20 min- utes, washed gently and allowed to dry [19]. Optical de- nsity of each well was read by using a micro-ELISA rea- der (Organon Teknika, Austria) at 492 nm transmitting wavelength [19,27]. The percentage of inhibition was calculated according to the following formula [28]. JBiSE % 100 100 Inhibition opticaldensity oftestwells opticaldensity ofcontrolwells that the extract was sphingolipid, as illustrated by Bisch- el and Austin [17]. Since ceramide is one of the sphingo- lipid classes [10] additional assays were carried out to characterize it. A purified fraction which extracted by si- licic acid was examined by spectrophotometer at a wide ranges of wavelength (between 200-1100 nm). One peak at wavelength 326nm with absorbance 1.481 was noticed (Figure 2). Examination of the purified compound by infrared instrument showed many peaks for many infra- red absorption frequencies (Figure 3) and each value of absorption was referred to a functional group of test co- mpound as follow: [748.34 (O-disubstituted), 964.34 (RCH = CH2), 1126.35(-C—C-), 1280.65 (-O-C-ether), 1380.94-1458.8(CH2), 1650(C = C), 1728.10 (C = O), 2854.45(C-H strong), 2923.88(C-H alkane), 2970- 3140(N-H) and 3363.62 (-O-H alcohol)]. When these functional groups were compared with the chemical co- mponents of ceramide, the results showed that the extra- cted compound can be identified as ceramide according to Mc Murry [18]. 3. RESULTS AND DISCUSSION Ceramide was separated from bovine brain and spinal cord. The detection and identification of purified extract were examined by using different assays. Using the ben- zidine spray visualization assay ceramide was detected by the virtue of the reaction of their secondary amide group belongs to the test compound with CL-substituted. After that the result of combination with benzidine spray was observed, blue product was formed, which means 100.0 NM/Div. NM 1100.0 326.0 1.481 722.0 -0.000 λ ABS λ ABS - - PEAK- - - -QUALITY- - *** PEAK-PICK *** 200.0 1.07 0/02 .00 1100.0 NM 0.001 A +0.00 A 0.500 (A/Div.) +0.25 Figure 2. Spectrogram of extracted ceramide. 100 95 90 85 80 75 %T 3000 1000 4000 2000 1/cm 3933.2 2854.4 1728.1 1458.08 1380.9 1260.65 1126.3 2923.8 964.3 748.3 Comments: No. of Scans; 10, Resolution; 16/[1/cm], Apodization; Happ-Genzel Figure 3. Infrared absorption frequencies assay for ceramide extract. 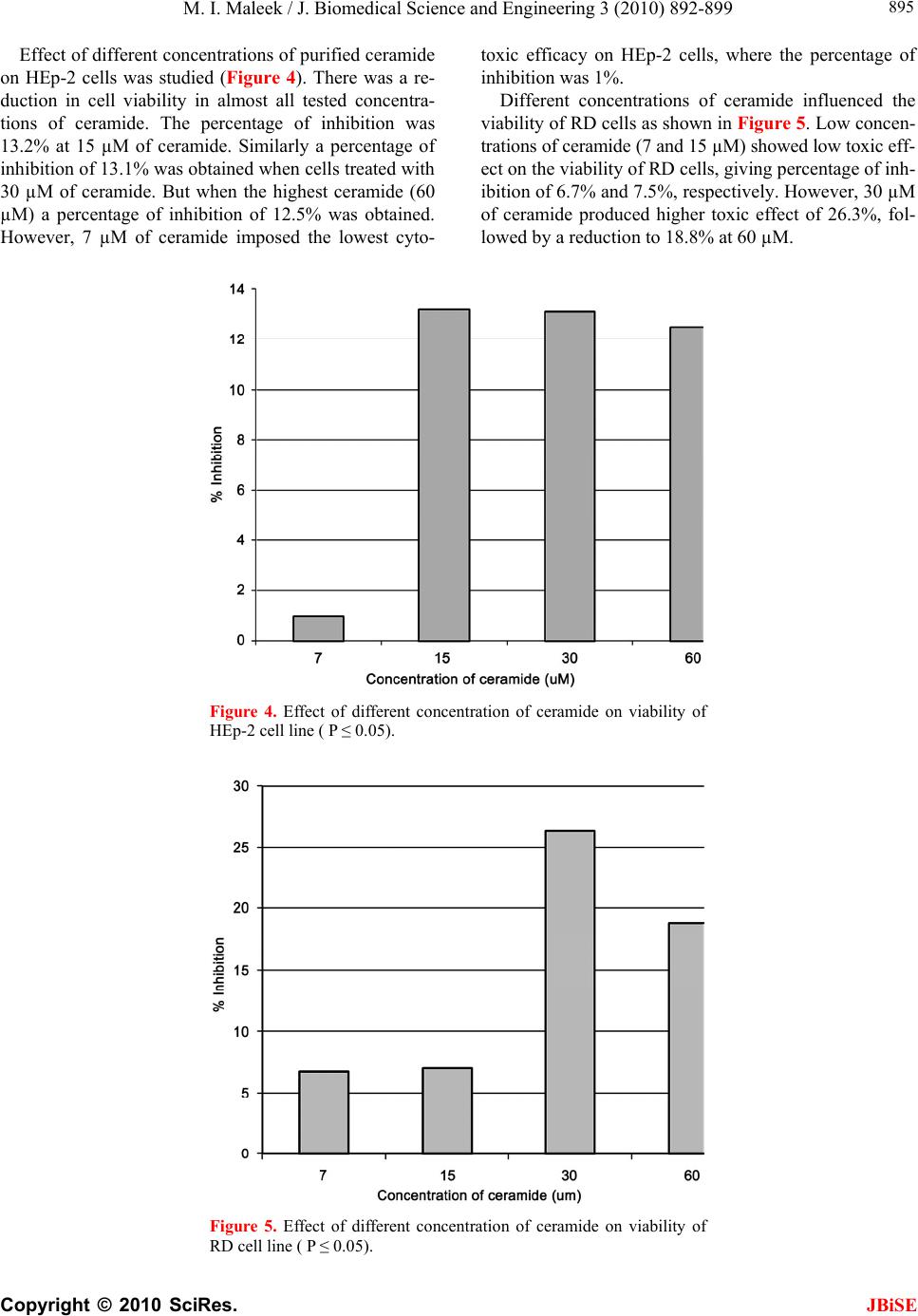 M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 895 Effect of different concentrations of purified ceramide on HEp-2 cells was studied (Figure 4). There was a re- duction in cell viability in almost all tested concentra- tions of ceramide. The percentage of inhibition was 13.2% at 15 µM of ceramide. Similarly a percentage of inhibition of 13.1% was obtained when cells treated with 30 µM of ceramide. But when the highest ceramide (60 µM) a percentage of inhibition of 12.5% was obtained. However, 7 µM of ceramide imposed the lowest cyto- toxic efficacy on HEp-2 cells, where the percentage of inhibition was 1%. Different concentrations of ceramide influenced the viability of RD cells as shown in Figure 5. Low concen- trations of ceramide (7 and 15 µM) showed low toxic eff- ect on the viability of RD cells, giving percentage of inh- ibition of 6.7% and 7.5%, respectively. However, 30 µM of ceramide produced higher toxic effect of 26.3%, fol- lowed by a reduction to 18.8% at 60 µM. Figure 4. Effect of different concentration of ceramide on viability of HEp-2 cell line ( P ≤ 0.05). Figure 5. Effect of different concentration of ceramide on viability of RD cell line ( P ≤ 0.05). 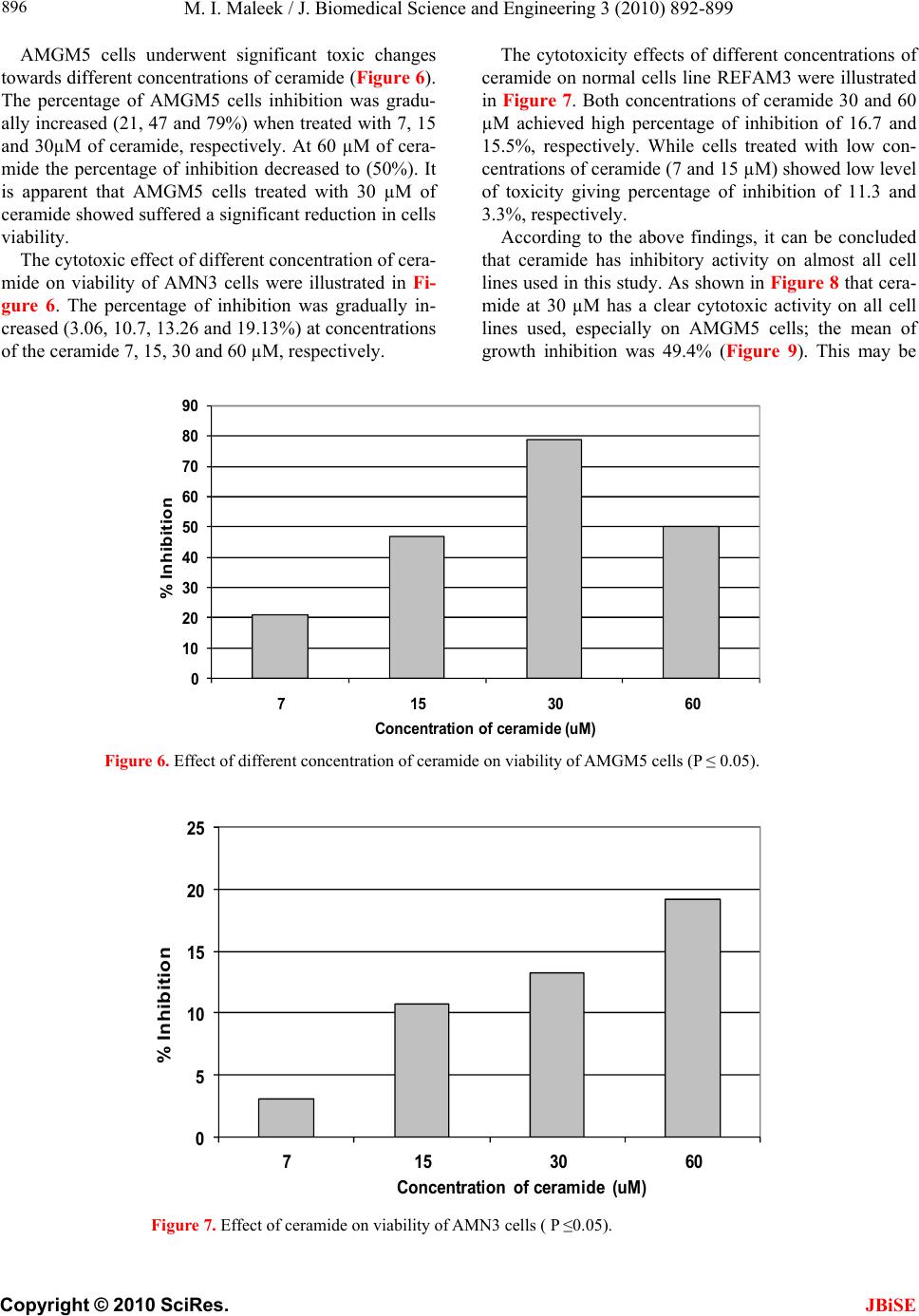 M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 896 AMGM5 cells underwent significant toxic changes towards different concentrations of ceramide (Figure 6). The percentage of AMGM5 cells inhibition was gradu- ally increased (21, 47 and 79%) when treated with 7, 15 and 30µM of ceramide, respectively. At 60 µM of cera- mide the percentage of inhibition decreased to (50%). It is apparent that AMGM5 cells treated with 30 µM of ceramide showed suffered a significant reduction in cells viability. The cytotoxic effect of different concentration of cera- mide on viability of AMN3 cells were illustrated in Fi- gure 6. The percentage of inhibition was gradually in- creased (3.06, 10.7, 13.26 and 19.13%) at concentrations of the ceramide 7, 15, 30 and 60 µM, respectively. The cytotoxicity effects of different concentrations of ceramide on normal cells line REFAM3 were illustrated in Figure 7. Both concentrations of ceramide 30 and 60 µM achieved high percentage of inhibition of 16.7 and 15.5%, respectively. While cells treated with low con- centrations of ceramide (7 and 15 µM) showed low level of toxicity giving percentage of inhibition of 11.3 and 3.3%, respectively. According to the above findings, it can be concluded that ceramide has inhibitory activity on almost all cell lines used in this study. As shown in Figure 8 that cera- mide at 30 µM has a clear cytotoxic activity on all cell lines used, especially on AMGM5 cells; the mean of growth inhibition was 49.4% (Figure 9). This may be 0 10 20 30 40 50 60 70 80 90 7153060 % Inhibition Con centratio n of cer am id e (uM) Figure 6. Effect of different concentration of ceramide on viability of AMGM5 cells (P ≤ 0.05). 0 5 10 15 20 25 7 153060 % Inhibition Concentration of ceramide (uM) Figure 7. Effect of ceramide on viability of AMN3 cells ( P ≤0.05).  M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 897 Figure 8. Effect of different concentration of ceramide on viability of REFAM3 cells ( P ≤ 0.05). Figure 9. Relationship between growth inhibition of cell lines at different concentrations of ce- ramide ( P ≤ 0.05). due to the cellular nature of AMGM5 cell line. As men- tioned earlier that AMGM5 is a human cell line origi- nated from a human cerebral glastoblastoma multiforme (GBM). In addition, ceramide was proved to bind vig- orously to CD95 receptors on cerebral cells, thus this specificity of engagement may initiate multiple signaling pathways that lead to activate caspases which is respon- sible for apoptosis [29-31]. This conclusion agreed with that of Seumois et al. [32] who recently demonstrated on the de novo generation that ceramide contributes to spo- ntaneous neutrophil apoptosis via caspase activation. On the other hand, the addition of ceramide to REFAM3, a normal cell line which is a mixture of fibroblastic and epithelial cells with normal chromosomal picture, showed low cytotoxic effect of ceramide (Figure 9 and 10). This may be attributed to the ability of these cells to generate ceramide-1-phosphate (cer-1-p) by the action of ceramide kinase [33]. Subsequently, cer-1-p in plasma membrane can be hydrolyzed by phosphatases [34]. Re- cently, Rile et al. [35] reported that cer-1-p can also be 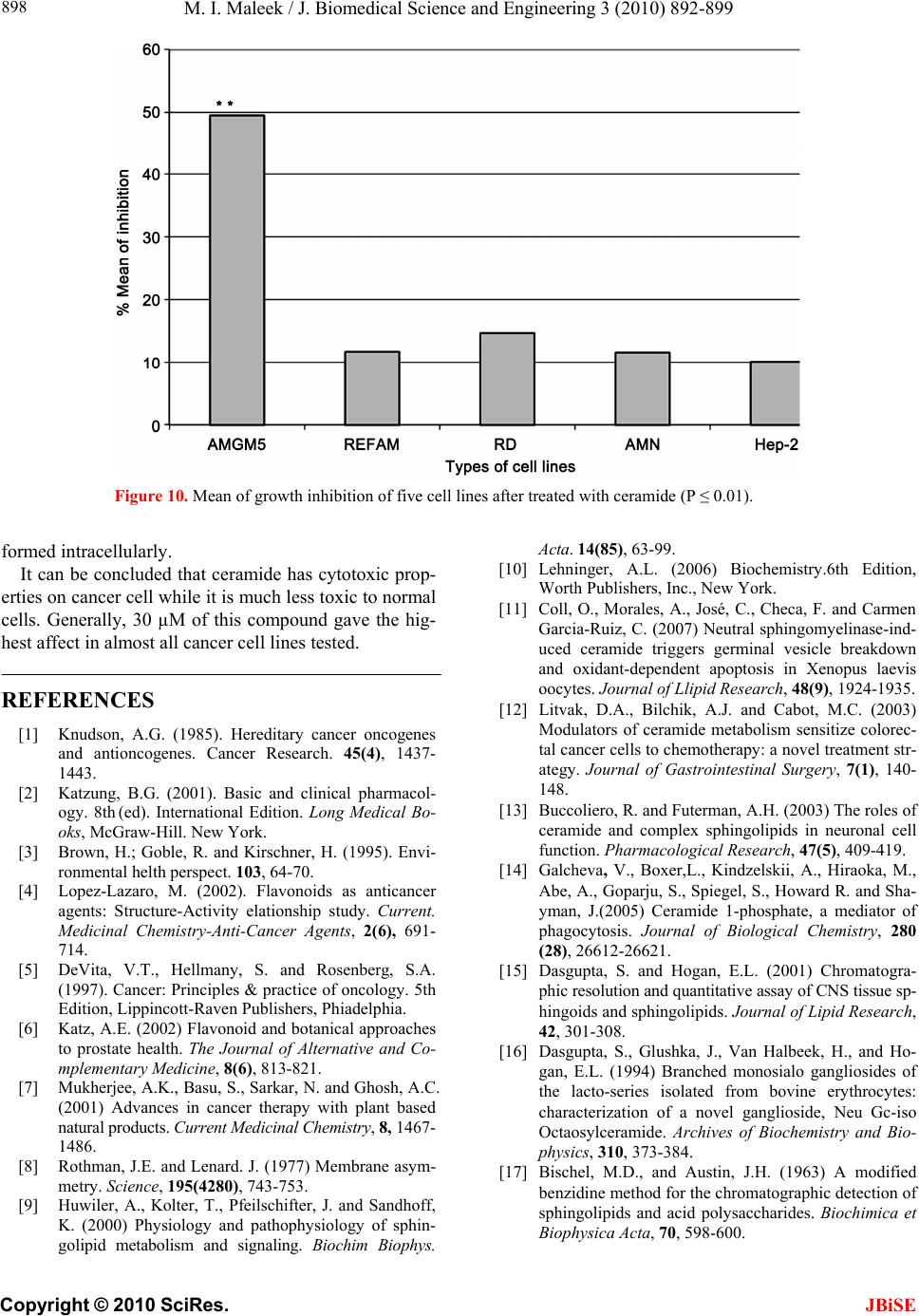 M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 898 Figure 10. Mean of growth inhibition of five cell lines after treated with ceramide (P ≤ 0.01). formed intracellularly. It can be concluded that ceramide has cytotoxic prop- erties on cancer cell while it is much less toxic to normal cells. Generally, 30 µM of this compound gave the hig- hest affect in almost all cancer cell lines tested. REFERENCES [1] Knudson, A.G. (1985). Hereditary cancer oncogenes and antioncogenes. Cancer Research. 45(4), 1437- 1443. [2] Katzung, B.G. (2001). Basic and clinical pharmacol- ogy. 8th (ed). International Edition. Long Medical Bo- oks, McGraw-Hill. New York. [3] Brown, H.; Goble, R. and Kirschner, H. (1995). Envi- ronmental helth perspect. 103, 64-70. [4] Lopez-Lazaro, M. (2002). Flavonoids as anticancer agents: Structure-Activity elationship study. Current. Medicinal Chemistry-Anti-Cancer Agents, 2(6), 691- 714. [5] DeVita, V.T., Hellmany, S. and Rosenberg, S.A. (1997). Cancer: Principles & practice of oncology. 5th Edition, Lippincott-Raven Publishers, Phiadelphia. [6] Katz, A.E. (2002) Flavonoid and botanical approaches to prostate health. The Journal of Alternative and Co- mplementary Medicine, 8(6), 813-821. [7] Mukherjee, A.K., Basu, S., Sarkar, N. and Ghosh, A.C. (2001) Advances in cancer therapy with plant based natural products. Current Medicinal Chemistry, 8, 1467- 1486. [8] Rothman, J.E. and Lenard. J. (1977) Membrane asym- metry. Science, 195(4280), 743-753. [9] Huwiler, A., Kolter, T., Pfeilschifter, J. and Sandhoff, K. (2000) Physiology and pathophysiology of sphin- golipid metabolism and signaling. Biochim Biophys. Acta. 14(85), 63-99. [10] Lehninger, A.L. (2006) Biochemistry.6th Edition, Worth Publishers, Inc., New York. [11] Coll, O., Morales, A., José, C., Checa, F. and Carmen Garcia-Ruiz, C. (2007) Neutral sphingomyelinase-ind- uced ceramide triggers germinal vesicle breakdown and oxidant-dependent apoptosis in Xenopus laevis oocytes. Journal of Llipid Research, 48(9), 1924-1935. [12] Litvak, D.A., Bilchik, A.J. and Cabot, M.C. (2003) Modulators of ceramide metabolism sensitize colorec- tal cancer cells to chemotherapy: a novel treatment str- ategy. Journal of Gastrointestinal Surgery, 7(1), 140- 148. [13] Buccoliero, R. and Futerman, A.H. (2003) The roles of ceramide and complex sphingolipids in neuronal cell function. Pharmacological Research, 47(5), 409-419. [14] Galcheva, V., Boxer,L., Kindzelskii, A., Hiraoka, M., Abe, A., Goparju, S., Spiegel, S., Howard R. and Sha- yman, J.(2005) Ceramide 1-phosphate, a mediator of phagocytosis. Journal of Biological Chemistry, 280 (28), 26612-26621. [15] Dasgupta, S. and Hogan, E.L. (2001) Chromatogra- phic resolution and quantitative assay of CNS tissue sp- hingoids and sphingolipids. Journal of Lipid Research, 42, 301-308. [16] Dasgupta, S., Glushka, J., Van Halbeek, H., and Ho- gan, E.L. (1994) Branched monosialo gangliosides of the lacto-series isolated from bovine erythrocytes: characterization of a novel ganglioside, Neu Gc-iso Octaosylceramide. Archives of Biochemistry and Bio- physics, 310, 373-384. [17] Bischel, M.D., and Austin, J.H. (1963) A modified benzidine method for the chromatographic detection of sphingolipids and acid polysaccharides. Biochimica et Biophysica Acta, 70, 598-600.  M. I. Maleek / J. Biomedical Science and Engineering 3 (2010) 892-899 Copyright © 2010 SciRes. JBiSE 899 [18] McMurry, J. (2000).Organic chemistry. 5th Edition, Brooks/Cole Publishers, Philadelphia. [19] Freshney, R.I. (1994) Culture of animal cells. 3rd. Edition, Wiley-Liss. USA, 267-308. [20] Taron, M., Plasencia, C., Abad, A., Martin, C. and Gu- illot, M. (2000). Cytotoxic effects of topotecan comb- ined with Various Active G2/M-phase anticancer dr- ugs in human tumor-derived cell lines. Invest New Drugs, 18(2), 139-147. (Pubmed Abstract). [21] Rodrigues, M., Junior, F.B. and Perussi, J.R. (2004) Dipyridamole increases the cytotoxicity of cisplatin in human larynx cancer cells in vitro. brazilian. Journal of Medical Biological Research, 37(4), 591-599. [22] Toyozumi, Y., Arima, N., Izumaru, S., Kato, S., Mo- rimatsu, M. and Nakashima, T. (2004) Loss of caspa- se-8 activation pathway is a possible mechanism for CDDP resistance in human laryngeal squamous cell carcinoma, HEp-2 cells. International Journal of On- cology, 25(3), 721-728. (Pubmed Abstract). [23] Singh, S., Chhipa, R.R., Vijayakumar, M.V. and Bhat, M.K. (2005) DNA damaging drugs-induced down- regulation of Bcl-2 is essential for induction of apo- ptosis in high-risk HPV-positive HEp-2 and KB cells. Cancer Letters, 236(2), 213-221. (Article in Press). [24] McAllister, R.M., Melnyk, J., Finklestein, J.Z., Ada- ms, E.C. and Gardner, M.B. (1969) Cultivation in vitro of cells derived from a human rhabdomyosarco- ma. Cancer, 24(3), 520-526. [25] Al-Shamery, A.M.H. (2003) The study of newcastle disease virus effect in the treatment of transplanted tumors in mice. Master Of Veterinary Medicine, Col- lege of Veterinary Medicine, University of Baghdad, Baghdad, Iraq. [26] Reiners, J.J. and Russell, E.C. (1999) Aryl hy- dro- carbon receptor regulation of ceramide-induced apop- tosis in murine hepatoma 1c1c7 cells. Journal of Bio- logical Chemistry, 274(4), 2502-2510. [27] Mahony, D.E., Gilliat, E., Dawson, S., Stockdale, E. and Lee, S.H. (1989) Vero cell assay for rapid dete- ction of clostridium perfringens enterotoxin. Applied and Environmental Microbiology, 55(9), 2141-2143. [28] Chiang, W., Chang, M.Y. and Lin, C.C. (2003) In vi- tro cytotoxic antiviral and immunomodulatory effects of Plantago major and Plantago asiatica. American Journal of Chinese Medicine, 31(2), 225-234. [29] Hannun, Y.A. (1996) Functions of ceramide in coor- dinating cellular responses to stress, Science, 274(5294), 1855-1859 [30] Michihiko, M., Zhexiong, J., Shohei, Y., Ryuichi, A., Shirou, F., Satoshi, B., Sato, T.K., Naochika, D., Ts- uneyo, M., Eda, T.B., Toshiro, O. and Hisanori, U. (2005) Role of membrane sphingomyelin and cera- mide in platform formation for Fas-mediated apoptosis. Journal of Experimental Medicine, 202(2), 249-259. [31] Takeda, S.; Susumu, M.; Kiyomi, T. and Yasuyuki, I. (2006). Apoptosis Occurs via the ceramide recycling pathway in human HaCaT keratinocytes. The Journal of Biochemistry, 139(2), 255-262. [32] Seumois, G., Marianne, F., Laurent, G., Céline, F., Christophe, D., Cédric, F., Benjamin, D., Cécile, O., Alain, V., Pierre, L. and Fabrice, B. (2007) De novo C16- and C24-ceramide generation contributes to spon- taneous neutrophil apoptosis. The Journal of Leuko- cyte Biology, 81(6), 1477-1486. [33] Sugiura, M., Kono, K., Liu, H., Shimizugawa, T., Mi- nekura, H., Spiegel, S. and Kohama, T. (2002) Cera- mide Kinase, a Novel Lipid Kinase. The Journal of Biological Chemistry, 277(26), 23294-23300. [34] Boudker, O. and Futerman, A.H. (1993) Detection and characterization of ceramide-1-phosphate phosphatase activity in rat liver plasma membrane. The Journal of Biological Chemistry, 268(29), 22150-22155. [35] Rile, G.Y., Yatomi, T.T. and Ozaki, Y. (2003). Cera- mide 1-phosphate formation in neutrophils. Acta Hae- matological, 109, 76-83.
|