Paper Menu >>
Journal Menu >>
 Creative Education 2012. Vol.3, Supplement, 76-79 Published Online December 2012 in SciRes (http://www.SciRP.org/journal/ce) DOI:10.4236/ce.2012.37B019 Copyright © 2012 SciRes. 76 The Development of PAT (Process Analytical Technology) for Drug Production and the Requirements for Domestic Pharmaceutical Engineering Education Shun Yao, Hang Song, Qiang Cheng, Bin g Liang School of Chemical Engineering, Sichuan University, Chengdu, China Email: Cusack@scu.edu.cn, Hangsong@vip.sina.com, Chengqiang@scu.edu.cn, Liangbing@sina.com Received 2012 After decades of practice and development, the GMP of drug production has entered the cGMP (Current Good Manufacturing Practice) era. In 21st century, PAT has become one of the new trends in the reform of the United States cGMP. In order to learn from foreign pharmaceutical quality control and production management experience, in this paper, the concept of PAT was introduced, the background of drug pro- duction PAT and its significance, the development trends of domestic pharmaceutical production PAT were reviewed. The domestic pharmaceutical engineering education under the new situation of PAT was discussed with emphasis. It was pointed out that to meet the requirements of the development of PAT, China's pharmaceutical engineering education needs constant adjustment and reform. Keywords: cGMP; Process Analytical Technol ogy; Drug Quali t y Con t r ol ; Pharmaceutical Engineering Education Introduction The process industries are those industries where the primary production processes are either continuous, or occur on a batch of materials that is indistinguishable. At present, process man- ufacturing is common in the pharmaceutical, chemical, food, beverage, and biotechnology industries. PAC (Process Analyti- cal Chemistry) and PAT (Process Analytical Technology) are two important conceptions related to the quality analysis and control in process industries. In 1987, Callis firstly divided PAC into 5 stages on the basis of various analytical operation areas and ways [1], which are named as off-line, at-line, on-line, in-line and noninvasive analysis. On-line analysis also can be divided into intermittent and continuous analysis. PAC is the art and science of making measurements for the purpose of control of large scale chemical processes. The first real-time measurements in a production environment were made with modified laboratory instrumentation. In the last decade, mul- ti-functional, automatic and portable instrumentations have been developed for immediate analysis. Under the background, PAT emerges at the right moment. Quite different from the common techniques, it becomes a framework for innovative development, manufacture and quality assurance in September 2005 defined by FDA. Current level of product quality is gen- erally adequate for the intended use, but the process by which the producers achieve relative level of quality is inefficient. So FDA mainly wants to propel pharmaceutical manufacturers through this framework to adopt innovative technologies for process control without fearing production delays or validation risks. Four PAT tools are needed for generation of process un- derstanding as follows [2]: (1) Multivariate data acquisition and anal ysis tools; (2) Process analyzers; (3) Process monitoring and control tools; (4) Continuous improvement and knowledge management tools. On the basis of guiding principle of “Quality by Design (QbD)”, the FDA is encouraging pharmaceutical industry to build up a system for “designing, analyzing and controlling manufacturing through timely measurements (during processing) of critical quality and performance attributes of raw and in-process materials and processes, with the goal of ensur- ing final product quality.” To be clearer, it is urgent to adopt industrial process analytical technology and improve quality and production efficiency in the first half of 21st c entury. PAT Development Situation both at Home and Abroad During the last 20 years, the number of papers about PAT & PAC indexed in Web of knowledge is rapidly increasing (shown in Figure 1). This obvious trend has proved that real- time monitoring and analysis techniques are attracting great interest from academic and industrial circles. Actively advo- cated by FDA, more and more pharmaceutical factories begin to pay attention to the importance of PAT in the production process of drugs, and make effort to realize relative proposed PAT applications. For instance, AstraZeneca has completed the construction of PAT system in one of its factory, and near infrared (NIR) spectroscopy is employ ed to control blending of active pharmaceutical ingredients (APIs) and employ an open-ended coaxial probe in situ monitoring and control of moisture content in pharmaceutical powder processes. Pfizer is handling its quality control through "Right First Time" (RFT) program, a global quality and efficiency improvement compo- nent of PAT. Sanofi-Aventis has successfully applied PAT tools in production process of solid oral dosage forms, includ- ing monitoring impurities and thickness uniformity of tablets. 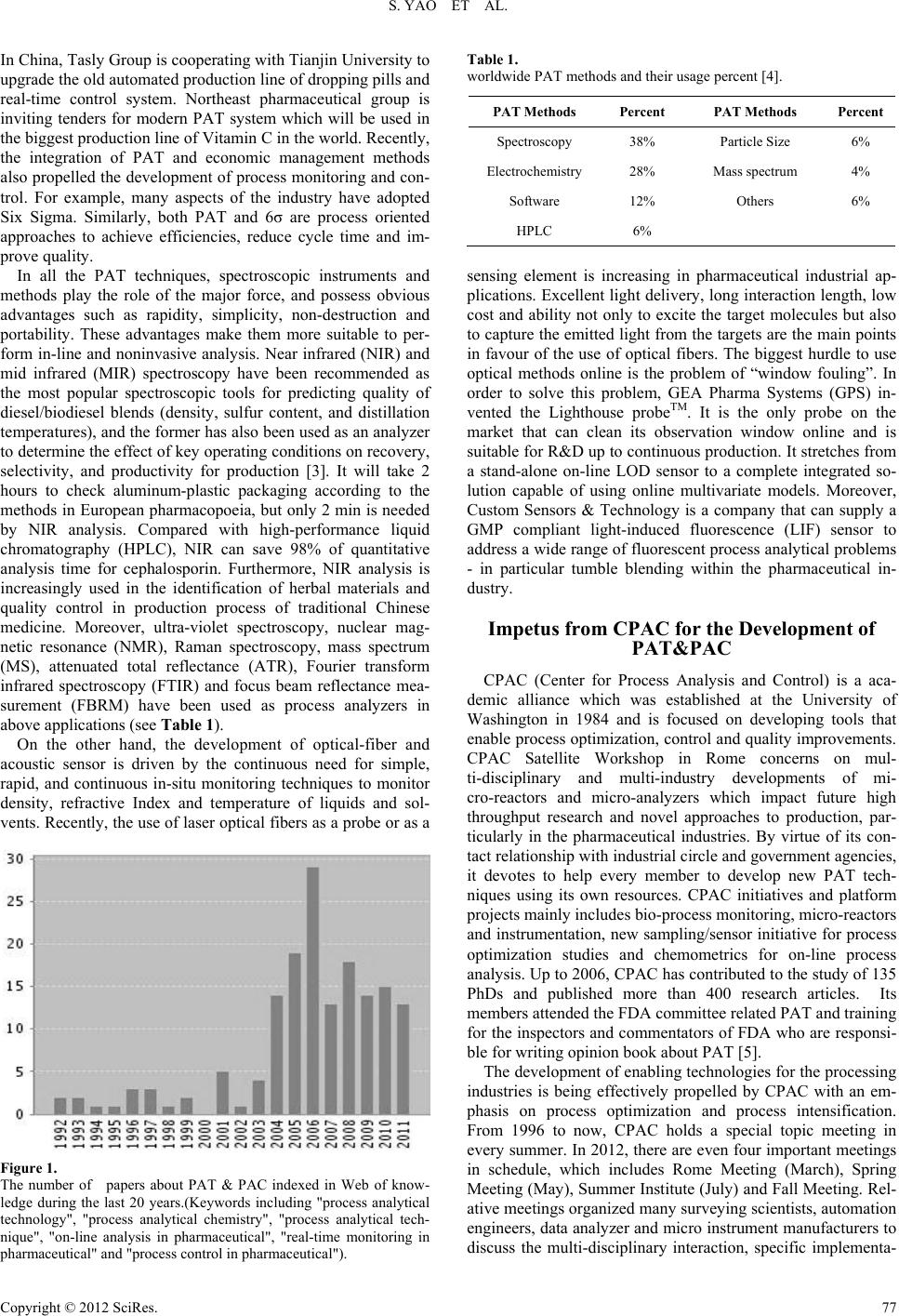 S. YAO ET AL. Copyright © 2012 SciRes. 77 In China, Tasly Group is cooperating with Tianjin University to upgrade the old automated production line of dropping pills and real-time control system. Northeast pharmaceutical group is inviting tenders for modern PAT system which will be used in the biggest production line of Vitamin C in the world. Recently, the integration of PAT and economic management methods also propelled the development of process monitoring and con- trol. For example, many aspects of the industry have adopted Six Sigma. Similarly, both PAT and 6σ are process oriented approaches to achieve efficiencies, reduce cycle time and im- prove quality. In all the PAT techniques, spectroscopic instruments and methods play the role of the major force, and possess obvious advantages such as rapidity, simplicity, non-destruction and portability. These advantages make them more suitable to per- form in-line and noninva sive analysis. Near infrared (NIR) and mid infrared (MIR) spectroscopy have been recommended as the most popular spectroscopic tools for predicting quality of diesel/biodiesel blends (density, sulfur content, and distillation temperatures), and the former has also been used as an analyzer to determine the effect of key operating conditions on recovery, selectivity, and productivity for production [3]. It will take 2 hours to check aluminum-plastic packaging according to the methods in European pharmacopoeia, but only 2 min is needed by NIR analysis. Compared with high-performance liquid chromatography (HPLC), NIR can save 98% of quantitative analysis time for cephalosporin. Furthermore, NIR analysis is increasingly used in the identification of herbal materials and quality control in production process of traditional Chinese medicine. Moreover, ultra-violet spectroscopy, nuclear mag- netic resonance (NMR), Raman spectroscopy, mass spectrum (MS), attenuated total reflectance (ATR), Fourier transform infrared spectroscopy (FTIR) and focus beam reflectance mea- surement (FBRM) have been used as process analyzers in above applications (see Table 1). On the other hand, the development of optical-fiber and acoustic sensor is driven by the continuous need for simple, rapid, and continuous in-situ monitoring techniques to monitor density, refractive Index and temperature of liquids and sol- vents. Recently, the use of laser optical fibers as a probe or as a Figure 1. The number of papers about PAT & PAC indexed in Web of know- ledge during the last 20 years.(Keywords including "process analytical technology", "process analytical chemistry", "process analytical tech- nique", "on-line analysis in pharmaceutical", "real-time monitoring in pharmaceutical" and "process control in pharmaceutical"). Table 1. worldwide PAT methods and their usage percent [4]. PAT Methods Percent PAT Methods Percent Spectroscopy 38% Particle Size 6% Electrochemistry 28% Mass spec trum 4% Software 12% Others 6% HPLC 6% sensing element is increasing in pharmaceutical industrial ap- plications. Excellent light delivery, long interaction length, low cost and ability not only to excite the target molecules but also to capture the emitted light from the targets are the main points in favour of the use of optical fibers. The biggest hurdle to use optical methods online is the problem of “window fouling”. In order to solve this problem, GEA Pharma Systems (GPS) in- vented the Lighthouse probeTM. It is the only probe on the market that can clean its observation window online and is suitable for R&D up to continuous production. It stretches from a stand-alone on-line LOD sensor to a complete integrated so- lution capable of using online multivariate models. Moreover, Custom Sensors & Technology is a company that can supply a GMP compliant light-induced fluorescence (LIF) sensor to address a wide range of fluorescent process analytical problems - in particular tumble blending within the pharmaceutical in- dustry. Impetus from CPAC for the Development of PAT&PAC CPAC (Center for Process Analysis and Control) is a aca- demic alliance which was established at the University of Washington in 1984 and is focused on developing tools that enable process optimization, control and quality improvements. CPAC Satellite Workshop in Rome concerns on mul- ti-disciplinary and multi-industry developments of mi- cro-reactors and micro-analyzers which impact future high throughput research and novel approaches to production, par- ticularly in the pharmaceutical industries. By virtue of its con- tact relationship with industrial circle and government agencies, it devotes to help every member to develop new PAT tech- niques using its own resources. CPAC initiatives and platform projects mainly includes bio-process monitoring, micro-reactors and instrumentation, new sampling/sensor initiative for process optimization studies and chemometrics for on-line process analysis. Up to 2006, CPAC has contributed to the study of 135 PhDs and published more than 400 research articles. Its members attended the FDA committee related PAT and training for the inspectors and commentators of FDA who are responsi- ble for writing opinion book about PAT [5]. The development of enabling technologies for the processing industries is being effectively propelled by CPAC with an em- phasis on process optimization and process intensification. From 1996 to now, CPAC holds a special topic meeting in every summer. In 2012, there are even four important meetings in schedule, which includes Rome Meeting (March), Spring Meeting (May), Summer Institute (July) and Fall Meeting. Rel- ative meetings organized many surveying scientists, aut omation engineers, data analyzer and micro instrum ent manufacturers to discuss the multi-disciplinary interaction, specific implementa-  S. YAO ET AL. Copyright © 2012 SciRes. 78 tion program, and new conceptions together with advanced techniques, etc. Through advocating such activities, wisdom is pooled for mutual benefit and the consciousness of PAT has been popularly accepted and grown stronger. Need for Domesti c E ducation to ADAPT International PAT Requirment Since pharmaceutical engineering specialty was established among Chinese higher learning institutions in 1998, there have been 150 universities enrolling students and 12-15 thousand graduates in every year. Educational levels are ranging from college students to doctoral students. University type includes comprehensive universities, universities of science and engi- neering, medical and pharmaceutical universities, normal uni- versities, and even universities of post and telecommunication. Because of the short history of Chinese pharmaceutical engi- neering education, it is inevitable there are some problems. Teaching level, teacher resource and experience are very vari- ous, and some course contents follow and copy those of medi- cine and pharmacy-related specialties. Both engineering cha- racteristics and the relationship with pharmaceutical industry are not enough. In this regard, the writers believe that it is ne- cessary to take the following countermeasures to solve above problems. 1) Closely follow the developments of the pharmaceutical industry, strengthen the construction of pharmaceutical engineering education PAT is entering the stage of industrialization and globaliza- tion. In the next few decades, the implementation of PAT will be an important change occurring in pharmaceutical field, which will have significant impact on relative industry. Current pharmaceutical companies need a long process to fully under- stand and implement PAT program. So it is urgent to increase inputs in the education and training of pharmaceutical engi- neering talents. In t he present situation, a few domestic colleges and universities open the course of PAT and PAC for the edu- cation of pharmaceutical engineering. Process analytical che- mistry is generally introduced as a branch of analytical chemi- stry briefly, but few Chinese teaching materials have been pub- lished or used. It is worth mentioning that Capital Normal University has began its positive attempt, and PAC becomes an elective spe- cialized course for the students (2007 senior students) major in applied chemistry and teacher in 2010. In this process, they found that some problems that exist in the curriculum and teaching contents: ① PAC is an interdisciplinary course involved in process engineering, analytical chemistry, control engineering, systems engineering, instrumental analysis and testing, information science, applied mathematics and other disciplines, which requires students with quite a discipline basis. But the relevant professional students are short of groundwork supported by corresponding courses; ② the course contents mainly come from foreign textbooks and journal articles, such as the monograph “Process analytical technology” edited by Katherine A. Bakeev and published by John Wiley & Sons, or those reviews published in the American Journal of Analytical Chemistry. The domestic classes have not the appropriate teaching materials. ③ PAC & PAT are closely related to the actual production process, so a common university laboratory can not meet the requirements of the relative corollary experi- ments wel l. Ideal teachi ng experime nts need to be designed and conducted with the help from industrial quality control depart- ment; On the other hand, laboratory conditions are needed to be improved and the actual environment of industrial scene needs to be simulated. Above explorations have provided a valuable reference for solving the problems in the PAT teaching contents and course settings. PAT pharmaceutical engineering education has lagged be- hind relative technical development, so it is necessary to intro- duce PAT concept into pharmaceutical engineering education and find the gap between the requirements of PAT and existing curriculum contents as soon as possible. It is needed to deeply analyze the similarities and differences between domestic and foreign pharmaceutical engineering curriculums and contents. Under the joint promotion from educational guidanc e com mit- tee, universities and pharmaceutical industry circle, it is hopeful and achievable to improve and reform the pharmaceutical en- gineering education and training to meet the CGMP require- ments at all levels. (2) Pharmaceutical industry should play a more active role in t he P AT pharmaceutical engineering education Traditionally, the task of personnel training and basic science education is mainly taken by universities. Meanwhile enter- prises are responsible for the induction training and training for new technologies. PAT in the pharmaceutical industry obtains its continuous development in the GMP process. As the initial proponents and the ultimate implementers of PAT, the enter- prises need to play its due role in pharmaceutical engineering education. Referring to the practice experience of CPAC in recent 20 years, it is best to establish the relevant departments to organize and promote the cooperation among industrial, academia and research people for PAT R&D and education. Only on the basis of the actual requirements in the industrial production, the production practice and field experience, good teaching books can be written to satisfy the requirements from teachers and students to combine the advantages of the re- sources and teachers, unless they will be completely divorced from reality and useless. As another way to establish relation- ship with universities, the enterprises should provide more in- ternship trainee opportunities and establish related bases for students to help them improve understanding of the PAT apart from classrooms, just like the efforts from academic unions, industry and enterprises of CPAC in these 20 years. There was also a foreign example that University of Dallas (Texas) coo- perated with the Dow Chemical Company to publish the text- book of “Process Analytical Chemistry (PAPAC)” and distri- buted it to teachers to help them teach PAC to the students in 2002. The course materials are written based on PAC R & D team's knowledge and experience in Dow Chemical Company [6]. (3) Raise the level of teachers and cultivate teach- er-engineer complex talents The team of qualified teachers is essential for training high-level talent. Due to historical reason of pharmaceutical engineering, in Chinese colleges and universities,the ma jority of teaching staff is short of engineering concept,which is mostly with pharmacy background. Teaching of drug analysis is very different from which of process analysis. On the other hand, some teaching staff majoring in engineering control are inapprehensive for the specificity of drug production. PAT involves many disciplines, and needs a strong foundation and knowledge in different practical fields. Facing the shortage of professional PAT teaching personnel in the domestic pharma-  S. YAO ET AL. Copyright © 2012 SciRes. 79 ceutical engineering education, it is urgent to improve the PAT level of employed teachers and cultivate teacher - engineer composite teaching personnel. More training and re-education are much-needed for most of teachers to make them fit the role, meanwhile teachers must spontaneously devote into relative learning, training and research to improve their own level. Some experienced senior engineers al so can be hired as visiting professors to teach on the podium. In a word, only the qualified and excellent teaching team can ensure to handle the discipli- nary teaching contents a nd realize educational objectives in the greatest extent. In addition to the teachers' self-learning, the related departments need to propel the plan and organization of education, visiting and training with specific purpose. (4) Improve the conditions of PAT practice teaching PAC & PAT are closely related to the actual production process, which require related teaching to have strong practical and operational. The e xisting conditions of the vast majority of university laboratories can not meet the process analysis tech- nology requirements of the experimental course. Old experi- mental contents about off-line analysis should be decreased or deleted. Many process analysis instruments and equipments are needed; otherwise the students will always indulge in empty talk and master nothing. For instance, our department has pur- chased fiber optic spectromete r and employed it in PAT teach- ing to show its application both in the extraction process of natural medicines and synthetic process of chemical bulk drugs on our self-made industrial control simulation platform. More investment needs to be added in the laboratory construction and improvement of the experimental conditions, on the other hand educational agencies need to actively contact pharmaceutical companies, and even environmental monitoring departments to design relative teaching experiments and strengthen the prac- tical application of PAT methods. Conclusions PAT has become one of the new trends of the U.S. cGMP reform in the 21st century. It is entering the stage of industria- lization, which will have a significant impact on the pharma- ceutical industry. In order to meet the requirements of the PAT development, China's pharmaceutical engineering education needs to adapt to the development of the pharmaceutical indus- try, and continue to adjust and update the curriculum, the teaching contents and teaching modes. Pharmaceutical industry should be actively involved both in the PAT pharmaceutical engineering education and in the research program; through various forms, the knowledge basis and PAT level of pharma- ceutical engineering teachers should be greatly improved, which can make them become complex interdisciplinary talents as soon as possible; the improvement of laboratory construction, practices of teaching and learning conditions should follow up in the same pace. Only in this way, it is realistic to train more and more professionals to meet cGMP requirements. Acknowledgment The paper and relative studies were supported by the project on the construction of pharmaceutical engineering feature spe- cialty in Sichuan University which was financed by ministry of education of China (2010-2012). REFERENCES J. B. Callis, D. L. Lllma n, and B. R. Kowalski, “Process analytical chemistry,” Anal. Chem., 1987, vol. 59, pp. 624-637. J. Munson, C. F. Stanfiled, B. Gujra, “A review of process analytical technology (PAT) in th e. U.S. pharmaceutical ind ustry,” Curr Pharm Anal, 2006, vol. 2, pp.405–414. M. Blanco, M. Alcala, J. M. Gonzalez, et al., “A process analytical technology approach base on near infrared spectroscopy: Tablet hardness, content uniformity, and dissolution test measurements of intact tablets,” J. Pharm. Sci., 2006, vol. 95, pp. 2137-2144. P. B. Janardhan, “Embracing process analytical technology,” Pharma Focus Asia, 2008, Issue. 6. J. Workman, J. M. Koch, B. Lavine, R. Chrisman, "Process Analytical Chemistry," Anal. Chem. , 2009, vol. 81, pp.4623-4643 J. P. Chauvel, W.W. Henslee and L. A. Melton, “Teaching Process Analytical Chemistry,” Anal. Chem., 2002, JULY, pp.381-384 |

