Paper Menu >>
Journal Menu >>
 Advances in Materials Physics and Chemistry, 2012, 2, 63-67 doi:10.4236/ampc.2012.24B018 Published Online December 2012 (http://www.SciRP.org/journal/ampc) Preparation and Property Analysis of Melamine Formaldehyde Foam Dongwei Wang, Xiaoxian Zhang, Song Luo, Sai Li School of Chemical Engineering, Sichuan University, Chengdu, China Email: wangdongwei820920@126.com, lisai@scu.edu.cn Received 2012 ABSTRACT Melamine formaldehyde (MF) foam is kind of fire-retardant material and has great potential in acoustic and thermal insulation area. In this article, MF resin foam was prepared by microwave radiation. We discussed the thermal stability of MF foam and the effect of different emulsifiers on its morphology, apparent density, fire-retardancy and mechanical property. The decomposition temperature of MF foam we prepared is nearly 400℃ and the constitution of residue after combustion is made up of carbon and graphite. Emul- sifier influenced the apparent density of MF foam and using coemulsifiers can get flexible foam with uniform cell size, good mor- phology and low apparent density. When the fire-retardant MF foam’s apparent density is low of 5.53 kg/cm-3, its value of LOI can reach 32.4. The mechanical property of foam is consistent with apparent density. Keywords: Melamine Formaldehyde Resin; Foam; Emulsifier; Morphology 1. Introduction MF foam is a kind of thermosetting plastic and has attracted much attention all over the world. Because of its properties as the low density, corrosion resisting, good autologous fire-re- tardancy and high thermal stability it can be used for a long time in the environment of temperature as high as 150℃ [1]. Up to now, only several companies including BASF, Illtec and Zhong Yuan Da Hua CO. can produce flexible MF foam [2]. This kind of MF foam has a skeleton of length/diameter (L/D) to 15 or even higher and can be practically used in acoustic area [3-4] due to its three-dimensional network structure and high cell-opening ratio. Many researchers have done lots of re- searches to solve its rigidity problem. For example, glycerol [5], isocyanate [6] as well as substituted melamine [7] had been added into MF resin to improve its flexibility. As we all know, emulsifier can reduce the interfacial tension and increase the contact area between foaming agent and MF resin which is benefit to foaming. But different kinds of emulsifiers had dif- ferent fields of application and apparent density caused by emulsifiers will affect the fire-retardancy and mechanical prop- erty. The corresponding basic research on foaming happens of MF foam with different emulsifiers has not been seen in recent papers. In this paper, we discussed the cell morphology with different emulsifiers under the radiation of microwave and ap- parent density, fire-retardancy and mechanical property caused by emulsifiers. These experimental results will provide signifi- cant theoretical foundation for producing superior MF foam. 2. Experiment 2.1. Materials Melamine (99%), formaldehyde(37%), NaOH, petroleum ether (30~60℃), dimethyl silicone oil (H-201), tween-80, Octyl phenol ethoxylates (OP-10) and sodium dodecylbenzenesul- fonate (SDBS), acetic acid and so on are analytically pure, they are all purchased from KeLong Reagent Corporation Chengdu. 2.2. Prepar ation of MF Foam Melamine and 37% formaldehyde with the molar ratio of 1:3 were added to flask with three necks equipped with motor stir- rer, thermometer and condenser and heated to about 60℃ and kept for a few minutes until the solution became clear. Then 10%NaOH was dripped to adjust the pH of the solution to 8.5, and emulsifier can be added with the mass ratio 2% of the resin. And then the solution was kept to the temperature of about 85 ℃ for properly 2~3h till the viscosity of the system up to 2000 cm•Pa and cooled to room temperature and MF resin was ob- tained. Took a certain amount of MF resin and about 3% acetic acid and 10% petroleum ether the mass ratio of the MF resin together were added and agitated vigorously to uniform and put into the microwave oven to foam without restraint of volume for about 5 min. Finally the MF foam was put into air oven at 120℃ to wipe off water, residual formaldehyde and for further cross-linking to transfer ether bond to methylene [8]. 2.3. Te st and Analysis Fourier transform Infrared (FTIR) spectra were measured in the spectral range from 400 to 4000 cm-1, on a Nicollet 380 FT-IR, KBr tablet, Thermo Electron Corporation spectrometer, USA. Thermo gravimetric analysis (TG) was carried out with a Seta- ram Setsys TG-DTA, samples were heated from room tem- perature to 700℃ in a 50 ml/min flow of N2 and heating rates of 10℃/min. Differential Scanning Calorimeter, DSC, GRN1- CDR-4P system, samples were heated from room temperature to 360℃ in a 50 ml/min flow of N2 and heating rates of 10℃ Copyright © 2012 SciRes. AMPC 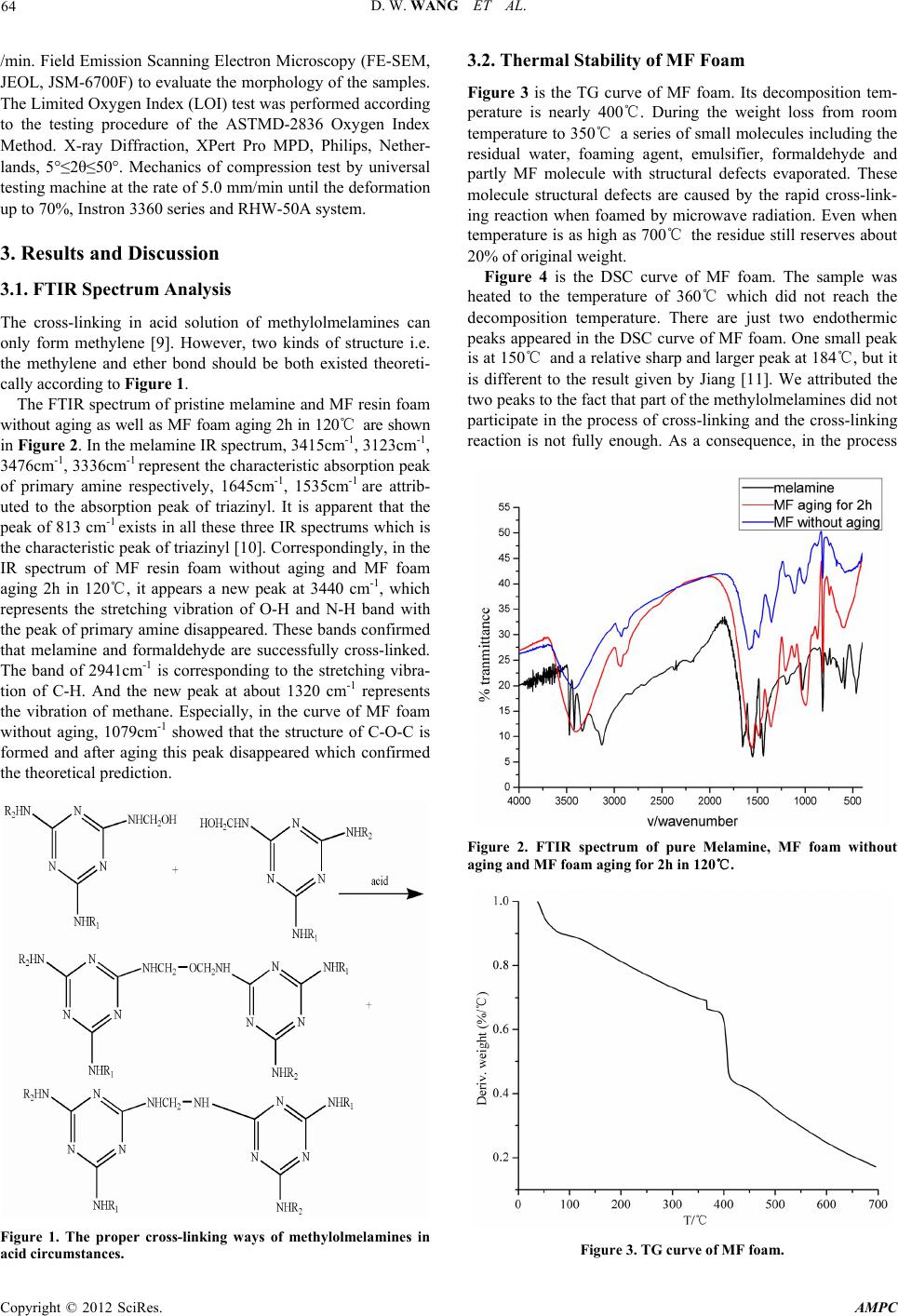 D. W. WANG ET AL. 64 /min. Field Emission Scanning Electron Microscopy (FE-SEM, JEOL, JSM-6700F) to evaluate the morphology of the samples. The Limited Oxygen Index (LOI) test was performed according to the testing procedure of the ASTMD-2836 Oxygen Index Method. X-ray Diffraction, XPert Pro MPD, Philips, Nether- lands, 5°≤2θ≤50°. Mechanics of compression test by universal testing machine at the rate of 5.0 mm/min until the deformation up to 70%, Instron 3360 series and RHW-50A system. 3. Results and Discussion 3.1. FTIR Spectrum Analysis The cross-linking in acid solution of methylolmelamines can only form methylene [9]. However, two kinds of structure i.e. the methylene and ether bond should be both existed theoreti- cally according to Figure 1. The FTIR spectrum of pristine melamine and MF resin foam without aging as well as MF foam aging 2h in 120℃ are shown in Figure 2. In the melamine IR spectrum, 3415cm-1, 3123cm-1, 3476cm-1, 3336cm-1 represent the characteristic absorption peak of primary amine respectively, 1645cm-1, 1535cm-1 are attrib- uted to the absorption peak of triazinyl. It is apparent that the peak of 813 cm-1 exists in all these three IR spectrums which is the characteristic peak of triazinyl [10]. Correspondingly, in the IR spectrum of MF resin foam without aging and MF foam aging 2h in 120℃, it appears a new peak at 3440 cm-1, which represents the stretching vibration of O-H and N-H band with the peak of primary amine disappeared. These bands confirmed that melamine and formaldehyde are successfully cross-linked. The band of 2941cm-1 is corresponding to the stretching vibra- tion of C-H. And the new peak at about 1320 cm-1 represents the vibration of methane. Especially, in the curve of MF foam without aging, 1079cm-1 showed that the structure of C-O-C is formed and after aging this peak disappeared which confirmed the theoretical prediction. Figure 1. The proper cross-linking ways of methylolmelamines in acid circumstances. 3.2. Thermal Stability of MF Foam Figure 3 is the TG curve of MF foam. Its decomposition tem- perature is nearly 400℃. During the weight loss from room temperature to 350℃ a series of small molecules including the residual water, foaming agent, emulsifier, formaldehyde and partly MF molecule with structural defects evaporated. These molecule structural defects are caused by the rapid cross-link- ing reaction when foamed by microwave radiation. Even when temperature is as high as 700℃ the residue still reserves about 20% of original weight. Figure 4 is the DSC curve of MF foam. The sample was heated to the temperature of 360℃ which did not reach the decomposition temperature. There are just two endothermic peaks appeared in the DSC curve of MF foam. One small peak is at 150℃ and a relative sharp and larger peak at 184℃, but it is different to the result given by Jiang [11]. We attributed the two peaks to the fact that part of the methylolmelamines did not participate in the process of cross-linking and the cross-linking reaction is not fully enough. As a consequence, in the process Figure 2. FTIR spectrum of pure Melamine, MF foam without aging and MF foam aging for 2h in 120℃. Figure 3. TG curve of MF foam. Copyright © 2012 SciRes. AMPC 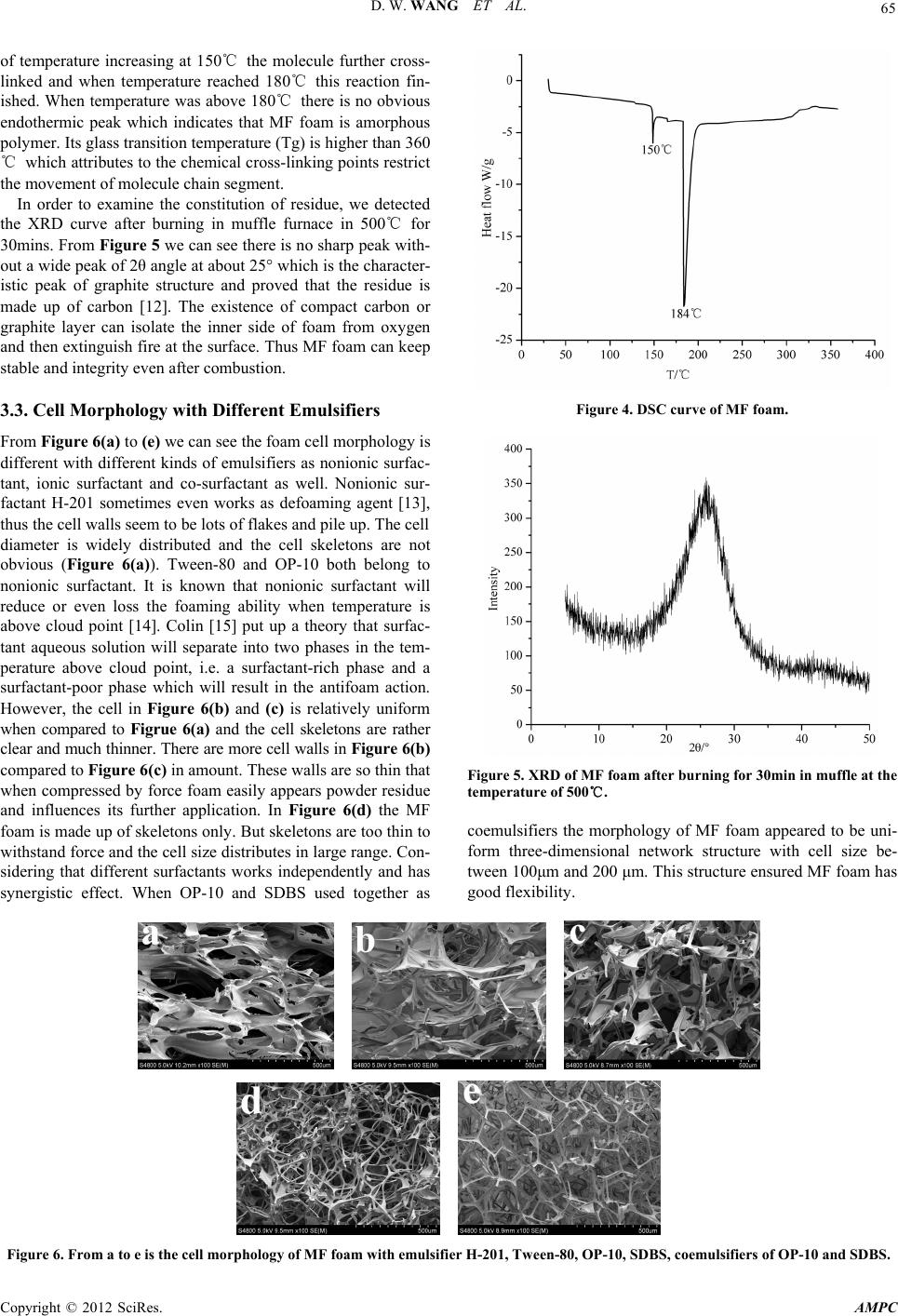 D. W. WANG ET AL. Copyright © 2012 SciRes. AMPC 65 of temperature increasing at 150℃ the molecule further cross- linked and when temperature reached 180℃ this reaction fin- ished. When temperature was above 180℃ there is no obvious endothermic peak which indicates that MF foam is amorphous polymer. Its glass transition temperature (Tg) is higher than 360 ℃ which attributes to the chemical cross-linking points restrict the movement of molecule chain segment. In order to examine the constitution of residue, we detected the XRD curve after burning in muffle furnace in 500℃ for 30mins. From Figure 5 we can see there is no sharp peak with- out a wide peak of 2θ angle at about 25° which is the character- istic peak of graphite structure and proved that the residue is made up of carbon [12]. The existence of compact carbon or graphite layer can isolate the inner side of foam from oxygen and then extinguish fire at the surface. Thus MF foam can keep stable and integrity even after combustion. 3.3. Cell Morphology with Different Emulsifiers Figure 4. DSC curve of MF foam. From Figure 6(a) to (e) we can see the foam cell morphology is different with different kinds of emulsifiers as nonionic surfac- tant, ionic surfactant and co-surfactant as well. Nonionic sur- factant H-201 sometimes even works as defoaming agent [13], thus the cell walls seem to be lots of flakes and pile up. The cell diameter is widely distributed and the cell skeletons are not obvious (Figure 6(a)). Tween-80 and OP-10 both belong to nonionic surfactant. It is known that nonionic surfactant will reduce or even loss the foaming ability when temperature is above cloud point [14]. Colin [15] put up a theory that surfac- tant aqueous solution will separate into two phases in the tem- perature above cloud point, i.e. a surfactant-rich phase and a surfactant-poor phase which will result in the antifoam action. However, the cell in Figure 6(b) and (c) is relatively uniform when compared to Figrue 6(a) and the cell skeletons are rather clear and much thinner. There are more cell walls in Figure 6(b) compared to Figure 6(c) in amount. These walls are so thin that when compressed by force foam easily appears powder residue and influences its further application. In Figure 6(d) the MF foam is made up of skeletons only. But skeletons are too thin to withstand force and the cell size distributes in large range. Con- sidering that different surfactants works independently and has synergistic effect. When OP-10 and SDBS used together as Figure 5. XRD of MF foam after burning for 30 min in muffle at the temperature of 500℃. coemulsifiers the morphology of MF foam appeared to be uni- form three-dimensional network structure with cell size be- tween 100μm and 200 μm. This structure ensured MF foam has good flexibility. Figure 6. From a to e is the cell morphology of MF foam with emulsifier H-201, Tween-80, OP-10, SDBS, coemulsifiers of OP-10 and SDBS. 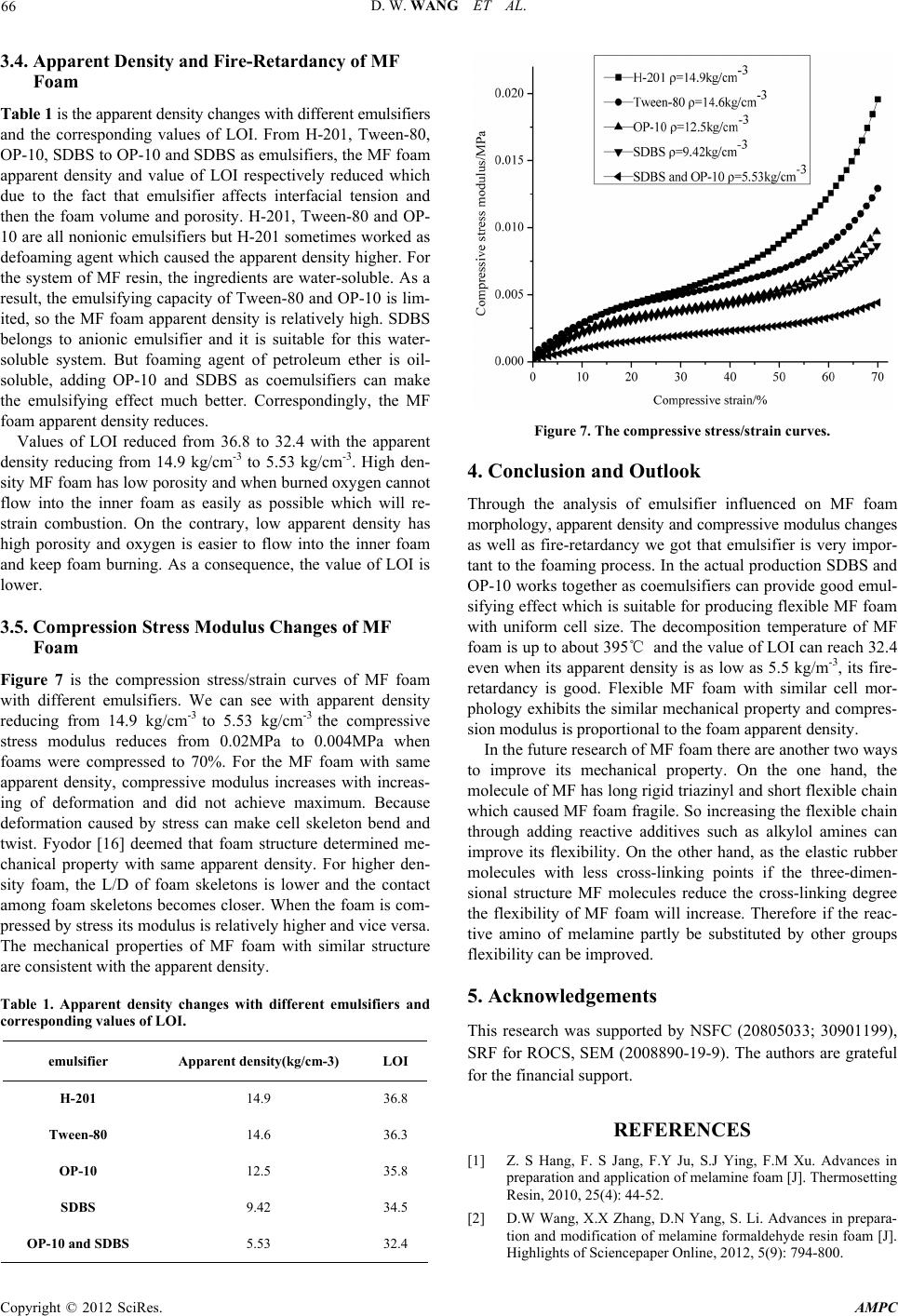 D. W. WANG ET AL. 66 3.4. A pparent Density and Fire-Retardancy of MF Foam Table 1 is the apparent density changes with different emulsifiers and the corresponding values of LOI. From H-201, Tween-80, OP-10, SDBS to OP-10 and SDBS as emulsifiers, the MF foam apparent density and value of LOI respectively reduced which due to the fact that emulsifier affects interfacial tension and then the foam volume and porosity. H-201, Tween-80 and OP- 10 are all nonionic emulsifiers but H-201 sometimes worked as defoaming agent which caused the apparent density higher. For the system of MF resin, the ingredients are water-soluble. As a result, the emulsifying capacity of Tween-80 and OP-10 is lim- ited, so the MF foam apparent density is relatively high. SDBS belongs to anionic emulsifier and it is suitable for this water- soluble system. But foaming agent of petroleum ether is oil- soluble, adding OP-10 and SDBS as coemulsifiers can make the emulsifying effect much better. Correspondingly, the MF foam apparent density reduces. Values of LOI reduced from 36.8 to 32.4 with the apparent density reducing from 14.9 kg/cm-3 to 5.53 kg/cm-3. High den- sity MF foam has low porosity and when burned oxygen cannot flow into the inner foam as easily as possible which will re- strain combustion. On the contrary, low apparent density has high porosity and oxygen is easier to flow into the inner foam and keep foam burning. As a consequence, the value of LOI is lower. 3.5. Compression Stress Modulus Changes of MF Foam Figure 7 is the compression stress/strain curves of MF foam with different emulsifiers. We can see with apparent density reducing from 14.9 kg/cm-3 to 5.53 kg/cm-3 the compressive stress modulus reduces from 0.02MPa to 0.004MPa when foams were compressed to 70%. For the MF foam with same apparent density, compressive modulus increases with increas- ing of deformation and did not achieve maximum. Because deformation caused by stress can make cell skeleton bend and twist. Fyodor [16] deemed that foam structure determined me- chanical property with same apparent density. For higher den- sity foam, the L/D of foam skeletons is lower and the contact among foam skeletons becomes closer. When the foam is com- pressed by stress its modulus is relatively higher and vice versa. The mechanical properties of MF foam with similar structure are consistent with the apparent density. Table 1. Apparent density changes with different emulsifiers and corresponding values of LOI. emulsifier Apparent density(kg/cm-3) LOI H-201 14.9 36.8 Tween-80 14.6 36.3 OP-10 12.5 35.8 SDBS 9.42 34.5 OP-10 and SDBS 5.53 32.4 Figure 7. The compressive stress/strain curves. 4. Conclusion and Outlook Through the analysis of emulsifier influenced on MF foam morphology, apparent density and compressive modulus changes as well as fire-retardancy we got that emulsifier is very impor- tant to the foaming process. In the actual production SDBS and OP-10 works together as coemulsifiers can provide good emul- sifying effect which is suitable for producing flexible MF foam with uniform cell size. The decomposition temperature of MF foam is up to about 395℃ and the value of LOI can reach 32.4 even when its apparent density is as low as 5.5 kg/m-3, its fire- retardancy is good. Flexible MF foam with similar cell mor- phology exhibits the similar mechanical property and compres- sion modulus is proportional to the foam apparent density. In the future research of MF foam there are another two ways to improve its mechanical property. On the one hand, the molecule of MF has long rigid triazinyl and short flexible chain which caused MF foam fragile. So increasing the flexible chain through adding reactive additives such as alkylol amines can improve its flexibility. On the other hand, as the elastic rubber molecules with less cross-linking points if the three-dimen- sional structure MF molecules reduce the cross-linking degree the flexibility of MF foam will increase. Therefore if the reac- tive amino of melamine partly be substituted by other groups flexibility can be improved. 5. Acknowledgements This research was supported by NSFC (20805033; 30901199), SRF for ROCS, SEM (2008890-19-9). The authors are grateful for the financial support. REFERENCES [1] Z. S Hang, F. S Jang, F.Y Ju, S.J Ying, F.M Xu. Advances in preparation and application of melamine foam [J]. Thermosetting Resin, 2010, 25(4): 44-52. [2] D.W Wang, X.X Zhang, D.N Yang, S. Li. Advances in prepara- tion and modification of melamine formaldehyde resin foam [J]. Highlights of Sciencepaper Online, 2012, 5(9): 794-800. Copyright © 2012 SciRes. AMPC  D. W. WANG ET AL. 67 [3] J Luc, R Ame´lie, D Mickael. Elastic and damping characteriza- tions of acoustical porous materials: Available experimental methods and applications to a melamine foam [J]. Applied Acoustics. 2008, 69: 1129-1140. [4] K Naoki, U Takayasu, S Yasuhiro, M Hiroshi. Investigation of non-acoustical parameters of compressed melamine foam mate- rials [J]. Applied Acoustics. 2009, 70: 595-604. [5] R.S Frank, J.M Alex. Aminotriazine-aldehyde foam modified with a primary triol [P]. US3093600, 1963-06-11. [6] I Yasno, H Shun, O Tatsnya. Melamine resin foam, process for production thereof and melamine /formaldehyde resin conden- sate [P]. US5436278, 1995-07-25. [7] J Weise, W Reuther, G Turznik, et al. Melamine resin moldings having increased elasticity [P]. US5084488, 1992-01-28. [8] I.H Anderson, M Cawley, W Steedman. Melamine Formalde- hyde resins I. an examination of some model compound systems [J]. Br. Polym. J., 1969, 1: 24-28. [9] S Kenji. Condensation of Methylolmelamine [J]. Bulletin of the chemical society of Japan, 1968, 41: 7-17. [10] F.Q Liu, K.Y Mao, D.H Zhang, X.Y Tang. Structure determina- tion of melamine resins by Fourier transform-infrared spectros- copy [J]. Analytical Chemistry, 1990, 18(5): 409-413. [11] H Jiang. Study of property of Basofil fiber [J]. Synthetic fiber, 2003, 4: 18-20. [12] L Zhou. Fire Retardancy of EVA/Multi-walled Carbon Nano- tubes Nanocomposites [J]. Chinese Plastics, 2009, 23(7): 23-29. [13] T Yasusaka, K Mitsuru. Modified silicone oil-in-water emulsion defoaming agent and defoaming method using it [J]. US 005431853A, 1995-11-7. [14] M.C Per, K Roland, S Per, K.C Hugo. Direct measurement of temperature-dependent interactions between non-ionic surfactant layers [J]. J. Chem. Soc., 1986, 82, 2735-2746. [15] A.B Colin, D Langevin. Why Do Ethoxylated Nonionic Surfac- tants Not Foam at High Temperature?[J]. Langmuir, 1997, 13(4): 599-601. [16] A.S Fyodor. Foamed Polymers. Cellular Structure and Properties [J]. Polymer science. 1983, 51: 155-219. Copyright © 2012 SciRes. AMPC |

