 Journal of Cancer Therapy, 2012, 3, 822-830 http://dx.doi.org/10.4236/jct.2012.325104 Published Online October 2012 (http://www.SciRP.org/journal/jct) Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study*# Kazuhiko Sato1, Yoshio Mizuno1, Masahiro Kato2, Takahiro Shimo2, Jun Kubota2, Naoko Takeda1,3, Yuko Inoue3, Hiroshi Seto4, Tomohiko Okawa5 1Department of Breast Oncology, Tokyo-West Tokushukai Hospital, Tokyo, Japan; 2Department of Radiation Oncology, To- kyo-West Tokushukai Hospital, Tokyo, Japan; 3Inoue Ladies Clinic, Tokyo, Japan; 4Seto Hospital, Saitama, Japan; 5Health Evalua- tion Center, Utsunomiya Memorial Hospital, Tochigi, Japan. Email: kazsato.boc@gmail.com Received August 27th, 2012; revised September 29th, 2012; accepted October 8th, 2012 ABSTRACT Background: Previous research has compared the efficacies of accelerated partial breast irradiation (APBI) and whole- breast irradiation (WBI). APBI immediately after surgery may provide more benefit after intraoperative insertion of catheters. Although balloon catheter-based APBI is available in the US, it is difficult in Japanese women, who have relatively small breasts. With the applicators being implanted during tumor removal, APBI can be started immediately after surgery. The aim of this study was to assess the safety and efficacy of APBI using the intraoperative open-cavity implant technique. Method: Patients (age ≥ 40 years) with invasive breast cancer (diameter ≤ 3 cm) were enrolled. Be- fore lumpectomy, the insertion of applicators and delivery doses were simulated by computed tomography (CT). After confirmation of free margins and negative sentinel nodes (SNs) using frozen section analysis, applicators were inserted. Postoperative CT-based dose distribution analysis was performed using dose-volume histograms. APBI was started on the day of surgery, delivering 32 Gy in 8 fractions over the following 5 - 6 days, and it covered a distance of 2 cm from tumor margins. This observational study was approved by the institutional review board of our hospital. Results: From October 2008 to July 2012, 157 women (160 lesions) were enrolled (age 55.0 years, <40:9, SN+: 25, for patients’ re- quest). The mean number of applicators used was 6.4 (2 - 15) and mean planning target volume was 35.8 cm3 (6.5 - 137.1 cm3). All radiotherapy-related toxicities were mild. However, 12 patients (7.5%) experienced wound breakdown because of surgical site infection. Two patients developed ipsilateral breast tumor recurrence (1 marginal, 1 at a distant site). Conclusions: Despite the small number of participants and a short follow-up period, our results suggest that this technique could be helpful in establishing clinical safety and efficacy. Keywords: Breast Cancer; Partial Breast Irradiation; Multicatheter Brachytherapy 1. Introduction Breast-conserving therapy (BCT), which comprises lumpectomy followed by whole-breast irradiation (WBI), is as effective as mastectomy in patients with early breast cancer [1,2]. Recent meta-analyses of trials comparing lumpectomy alone with lumpectomy + WBI demon- strated not only a 3-fold higher local relapse rate with the omission of radiation following lumpectomy but also a statistically significant compromise in overall survival [3-5]. On the basis of these data, breast-conserving sur- gery followed by WBI has become the accepted standard of treatment. However, irradiation of the entire breast on a daily basis for 4 - 6 weeks is time consuming for the radiation center, and 15% - 30% of patients who undergo lumpectomy do not receive WBI because of physician bias, patient age, fear of radiation treatment, distance from a radiation treatment facility, or socioeconomic factors [6-11]. Moreover, radiation-induced lung injury and late cardiovascular toxicity are further concerns regarding WBI. An alternative method to WBI is there- fore warranted. Local recurrence after BCT is most likely to occur in the vicinity of the lumpectomy site [12-15]. If the radia- *Funding: this study was not funded by any public or private grant. #Disclosure: all authors declare no conflict of interest. Copyright © 2012 SciRes. JCT  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study 823 tion target can be confined to an area of the breast, the treatment course can be accelerated. Accordingly, the efficacy of accelerated partial breast irradiation (APBI) has been investigated as an alternative method to WBI in patients with early-stage breast cancer. Several radio- therapy techniques including multicatheter interstitial brachytherapy, balloon catheter brachytherapy, 3-di- mensional conformal radiation therapy, and intraopera- tive radiotherapy (IORT) have been reported. These techniques, which are radiobiologically equivalent to conventional WBI, are capable of accurate delivery of radiation to the tissue surrounding the lumpectomy cavity. Despite the absence of long-term data from any large randomized clinical trial, there have been many prospec- tive and retrospective studies providing encouraging re- sults to support the view that APBI is a viable alternative to WBI. Among these techniques, multicatheter interstitial brachy- therapy has been used the longest and most frequently, but it requires considerable skill to deliver radiation to the target. Breast brachytherapy using a balloon-based brachytherapy device has been rapidly incorporated into clinical practice, especially after it was granted Food and Drug Administration (FDA) approval [16]. Although intracavitary techniques can achieve accu- rate anatomic delivery with less of a learning curve for medical personnel, they are considered by some to incur the risk of increased toxicity in the skin and the chest wall because of excessive doses received by those re- gions, especially in Asian women, who generally have small breasts [17]. More recently, concerns have been reported on the use of balloon-based brachytherapy in APBI. Compared with WBI, balloon-based brachyther- apy in APBI shows a 2-fold increased risk for subsequent mastectomy as well as postoperative and radiation-re- lated complications after 5 years [18]. With regard to the detection of cancer cells and the lack of necessity for additional surgery for catheter inser- tion, it might be of benefit for patients to start APBI im- mediately following lumpectomy with simultaneous multicatheter insertion during primary surgery after free margins are confirmed by frozen section analysis. Never- theless, there is considerable risk of inaccurate radiation administration because of the change in the lumpectomy cavity during treatment, and of the side effect of delayed wound healing. Therefore, the aim of this registry trial was to report our preliminary findings of an assessment of the safety and efficacy of APBI. 2. Patients and Methods 2.1. Patients Eligibility criteria for patient selection included age ≥ 40 years, histologically documented breast cancer, unifocal disease, maximum tumor diameter ≤3.0 cm on pre- operative imaging [mammography, ultrasonography, and breast magnetic resonance imaging (MRI)], negative mar- gins, and sentinel nodes (SNs) negative for metastases confirmed by frozen section analysis during surgery. Eligibility criteria also excluded any prior treatment including chemotherapy and hormonal therapy. All pa- tients provided written informed consent, and this registry study was approved by the institutional review board of our hospital. 2.2. Treatment Schema Before surgery, the insertion of applicators and delivery doses were simulated by computed tomography (CT) (Figures 1(a) and (b)). Sentinel node biopsy using a blue dye and a radioisotope as well as lumpectomy with tumor-free margins of approximately 1 cm were per- formed. After confirmation of absence of metastases in SNs by frozen section analysis, patients were enrolled in the study. The surgical margins of the lumpectomy cavity were also confirmed as being free by specimen mam- mography and frozen section analysis, with surgical hemoclips being placed at the 3, 6, 9, and 12 O’clock positions. Applicators for the introduction of iridium wires were inserted according to the preoperative CT- based simulation (Figure 2). The lumpectomy cavity was identified on postoperative CT scans with the help of hemoclips. The clinical target volume was the estimated tumor volume plus a margin of 10 mm. The planning target volume (PTV) was defined as the estimated tumor volume plus a 20 mm margin, i.e., clipped margin plus 10 mm. Because PTV excluded the skin, the skin over the tumor was partially removed to a depth of approximately 5 mm. Dose distribution analysis was performed on the basis of postoperative CT using dose-volume histograms (Figure 3). Irradiation plans were created using the Nucletron PLATO treatment planning system (Version UPS: 11.3; Nucletron Trading BV, Veenendaal, The Netherlands). The plans were made by 2 dosimetrists (A.K. and T.S.) and approved by a radiation oncologist (M.K.). Patients received high- dose-rate (HDR) brachytherapy using a 192Ir source. Radiation coverage of 4 Gy does not reach surround- ing tissue < 5 mm from the skin surface and the posterior chest wall, and a radiation dose of ≥3 Gy does not directly expose these organs. APBI was started on the day of primary surgery, delivering 32 Gy in 8 fractions over 5 - 6 days. Fractions delivered twice daily were separated by an interval of at least 6 h. 2.3. Assessment of Outcomes The prospective follow-up policy was designed so that Copyright © 2012 SciRes. JCT 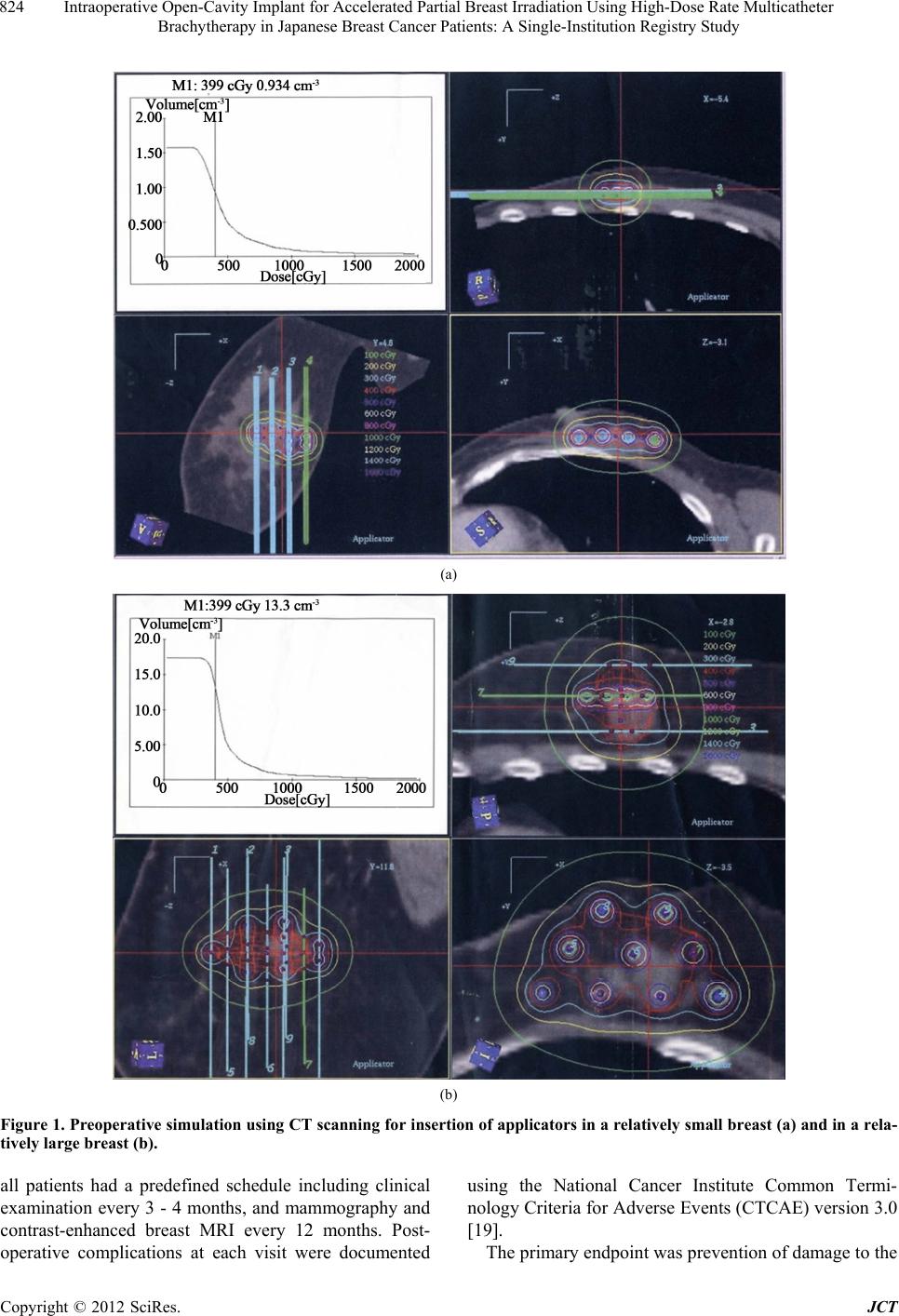 Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study Copyright © 2012 SciRes. JCT 824 (a) (b) Figure 1. Preoperative simulation using CT scanning for insertion of applicators i n a relative ly small breast (a) and in a rela - tively large breast (b). all patients had a predefined schedule including clinical examination every 3 - 4 months, and mammography and contrast-enhanced breast MRI every 12 months. Post- operative complications at each visit were documented using the National Cancer Institute Common Termi- nology Criteria for Adverse Events (CTCAE) version 3.0 [19]. The primary endpoint was prevention of damage to the  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study 825 (a) (b)(c) (f) (e) (d) Figure 2. Implantation of the multicatheter using the open-cavity method. Figure 3. Optimization of treatment plans with dose-volume histograms. conserved breast as assessed by acute and late toxicities: radiation dermatitis, wound infection, skin breakdown, and fat necrosis requiring multiple aspirations. Secondary outcomes were ipsilateral breast recurrence (ILBR) and cosmesis evaluated by the Harvard/National Surgical Adjuvant Breast and Bowel Project (NSABP) criteria [20]. Annual mammography and breast MRI were per- formed for the detection of locoregional recurrence. 3. Results 3.1. Study Enrolment A total of 157 consecutive patients with 160 lesions were treated with BCT using intraoperative open-cavity im- plantation (IOCI) from October 2008 to July 2012. The median follow-up time was 918 days (52 - 1424 days). Patient characteristics and demographics (mean age, 55.0 Copyright © 2012 SciRes. JCT  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study 826 years) are listed in Table 1. The most common path- ological finding was invasive ductal carcinoma. Most tumors (92.5%) were 2 cm in diameter, whereas 88.1% were hormone receptor positive. Thirteen patients (8.1%) required excisional biopsy for a definitive diagnosis. Most patients lived too far from our institution to permit frequent follow-up. Table 1. Patient demographics (n = 157) and tumor charac- teristics (n = 160). Characteristics n % Mean age (range, years) 55.0 (30 - 92) <40 years 9 5.7 40 - 69 years 133 84.7 >70 years 15 9.6 Pathological tumor stage pTis 17 10.6 pT1 131 81.9 pT2 12 7.5 Grading (nuclear grade) Grade I 107 66.9 Grade II 31 19.4 Grade III 1 0.6 NA 21 13.1 Number of lymph node involved 0 135 84.4 1 - 3 17 10.6 >3 8 5.0 Margins at first excision Free 142 88.7 Suspicious 7 4.4 DCIS only 11 6.9 Hormone receptor status (>10%) ER+ or PgR+ 141 88.1 ER− and PgR− 19 11.9 Her2/neu status + (IHC 3+ or FISH+) 15 9.4 ‒ (IHC 3‒ and FISH‒) 128 80.0 NA 17 10.6 Abbreviations: DCIS, ductal carcinoma in situ; ER, estrogen receptor; PgR, progesterone receptor. Her2/neu, human epidermal growth factor receptor 2; IHC, immunohistochemistry; FISH, fluorescence in situ hybridization. Overall, 23 patients (14.6%) did not meet the criteria because of various factors, including 25 with SNs positive for metastases, 9 under 40 years of age, and 1 who had previously received WBI before APBI. 149 patients (94.3%) underwent adjuvant systemic therapy. 137 patients (86.7%) received hormonal therapy, 48 (30.4%) received che- motherapy, and 9 (5.7%) were administered adjuvant tras- tuzumab. Two patients underwent additional WBI after APBI because of the high risk of local recurrence. 3.2. Implantation Valuables and Dosimetric Analyses The mean number of applicators used was 6.5 (2 - 15) and the distribution of row for implantation was 1 per 62 lesions (38.8%), 2 for 88 lesions (50.0%), and 3 for 10 lesions (6.2%). Tabular dose-volume histograms were obtained for all patients. Dose hotspots according to the volume encompassed by the 150% of isodose surface, dose homogeneity index, and organs at risk are shown in Table 2. 3.3. Adverse Events: Acute and Late Toxicities All acute and late toxicities were generally mild, with no grade 3 or 4 toxicities, and no patient required secondary surgery because of toxicity. Table 3 presents an analysis of variables associated with Grade 0 vs. Grade 1 - 2 skin toxicity. The distribution of patients according to late skin tox- icity score at final follow-up was also analyzed. There were no cases of Grade 3 or 4 skin toxicity, but 2 patients developed seroma requiring multiple aspirations because of fat necrosis, and 10 patients developed wound infec- Table 2. Dosimetric parameters and doses according to organs at risk. PTV V100 V150 DNR Mean volume (cm3) 35.8 29.2 9.6 0.30 Min volume (cm3) 6.5 2.6 1.2 0.10 Max volume (cm3) 137.1 106.5 59.3 0.56 Breast (volume, cm3) Cavity* (volume, cm3) Skin (max dose, Gy) Lung (max dose, Gy) Mean 322.1 18.2 2.0 2.1 Min 47.8 0.3 1.0 0.4 Max 949.4 75.4 12.4 3.5 Abbreviations: PTV, planning target volume; V100/150, volume encompassed by the 100%/150% isodose surface; DNR, dose nonuniformity ratio (V150/V100). *clipped margin. Copyright © 2012 SciRes. JCT  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study Copyright © 2012 SciRes. JCT 827 registry system and had a relatively short follow-up period, the number of patients enrolled was larger than that in previously published reports on Asian populations. tion followed by wound breakdown. However, most pa- tients achieved either good or excellent cosmetic out- come (Figure 4). HDR multicatheter brachytherapy is the most fre- quently used method for APBI, and a balloon-type model is commercially available in the US. After approval of this device by FDA, there has been a dramatic increase in the use of the implantable balloon brachytherapy catheter [21]. Balloon-type brachytherapy may require less of a learning curve for medical personnel and have high reproducibility, but is not suitable for most Japanese 3.4. ILBR and Patterns of Failure At final follow-up, 157 patients remained alive. One patient died from a nonoriginal breast cancer-related cause without any local recurrence, and local failure occurred in 3 patients, including 2 ILBRs (1 tumor bed failure and 1 distant-site in-breast failure) and 1 lymph node recurrence without distant metastasis. One of these ILBRs was regarded as a tumor bed failure observed adjacent to the initial lumpectomy cavity 12 months after APBI in a patient who had positive lymph nodes and excised margin involvement. After a second breast-conserving surgery with WBI, there has been no sign of recurrence. The other recurrence regarded as a distant-site in-breast failure was revealed as a tumor growing approximately 1 cm distant from the lumpectomy cavity and which had already been observed on contrast- enhanced MRI before initial surgery. Salvage glandectomy was performed 24 months after initial surgery. Table 3. Acute and chronic toxicity after treatment. Acute Toxicity (CTCAE v3.0[19]) N % Hemorrhage Grade 1 2 1.3 Wound infection Grade 2 1 0.6 Radiation dermatitis Grade 1 18 11.3 Grade 2 4 2.5 Chronic Toxicity Seroma requiring multiple aspirations or fat necrosis 2 1.3 Wound infection with wound breakdown 10 6.3 4. Discussion This observational study has confirmed that APBI using multicatheter brachytherapy is safe and represents an effective alternative to WBI followed by lumpectomy in Japanese breast cancer patients. Although this was a single-institution study using a consecutive patient Figure 4. Patients achieving good or excellent cosmetic outcome after APBI.  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study 828 women, who have relatively small breasts. Although the balloon-type method can deliver a more accurate radi- ation dose, especially in the area up to 1 cm beyond the lumpectomy cavity, the actual radiation field would be restricted by the distance between the skin and chest wall, especially in patients with small breasts. Catheter insertion is generally performed on confir- mation of a definitive pathology report. If margin status and nodal involvement are examined using frozen section analysis, catheter insertion can be done during primary surgery and radiation therapy can start immediately postoperatively. The aims of IORT are the same as ours in terms of the management of lumpectomy margins. This is more convenient for patients, but APBI should be started without waiting for confirmation of detailed pathology. The benefit of this new method is that catheter inser- tion is based on preoperative CT dosimetric analysis, but accurate and safe insertion could also be achieved using an open cavity method. APBI offers more efficacious tumor control than postoperative WBI because several reports have found that fluid in the lumpectomy cavity is conducive to the growth of residual cancer cells [22], and also that delayed radiation therapy might offer less tumor control, especially in patients receiving chemotherapy [23,24]. However, several safety issues must be ad- dressed: 1) This technique delivers radiation immediately after surgery, leading to delay in the wound-healing process. According to IORT data about safety, that delay might not have to be concerned [25-27]; 2) This study was conducted in an Asian population and its findings cannot yet be generalized; 3) Infection is an issue be- cause of the relatively long length of the catheters used. In this small cohort, all adverse events were mild and second surgery was not required in any case. Moreover, our cohort included 2 patients receiving WBI after APBI and 1 patient who previously received WBI. Smith et al. recently reported a double opportunity to perform mastectomy on patients receiving APBI rather than on those receiving WBI [18]. However, this report was based on a retrospective review of a Medicare billing claims database and did not address the reasons for mastectomy. In the US, balloon-type APBI is the most common procedure in brachytherapy-based APBI. The present report was not based on prospective competitive trials, and cannot therefore be applied to other APBI methods. APBI might offer better local control than WBI because of the direct delivery of radiation exposure to the lumpectomy cavity. Moreover, APBI can avoid injury to normal tissues such as the skin, ribs, lung, heart, and chest wall, i.e., it is a safer procedure than WBI. There are several reports including randomized clinical trials on the efficacy of APBI [28-30]. However, we await the results of a large trial comparing the efficacy of APBI with WBI, i.e., NSABP B39/Radiation Therapy On- cology Group 0413 Phase III trial. We did not publish our data until the median follow-up period had passed 2.5 years, because the median follow-up period of most reports on APBI-induced late toxicities was longer than 20 months [31-33] and the peak hazard of recurrence is between 12 and 24 months [34]. Hazard likelihood de- creases steadily between 2 and 5 years [34]. Our study included 2 cases of ipsilateral breast tumor recurrence (IBTR) but only 1 of these was a true recur- rence—the so-called tumor bed recurrence. The other was located distant to the lumpectomy cavity, which was evident in contrast breast MRI before the first surgery (Figure 3). Therefore, it is important to confirm unifocal disease at the time of the first surgery. According to the Early Breast Cancer Tria lists Col- laborative Group database, 1 death from breast cancer could be avoided for every 4 recurrences, the so-called “1-in-4 rule” [4,5]. IBTR after APBI should be avoidable. There are several factors influencing local recurrence, including patient age, tumor subtype, margin status, tumor features (size, unicentricity, existence of extensive intraductal components), nodal status, types of systemic therapy [35-37]. Before obtaining the results from a large randomized trial, the correct selection of candidates would be crucial after consideration of the above factors [38]. The biological features of the tumor would be also related to IBTR risk [39-42] and we might have to consider the use of APBI on the basis of individual factors. Further research on APBI using the IOCI tech- nique is needed to establish its clinical efficacy. 5. Acknowledgements The authors would like to thank Enago (www.enago.jp) for the English language review. This study was pre- sented in part at the 2010 the American Society of Clini- cal Oncology-Breast Annual Meeting (poster) and at the 2011 European Breast Cancer Meeting (poster). REFERENCES [1] B. Fisher, S. Anderson, J. Bryant, et al., “Twenty-Year Follow-Up of a Randomized Trial Comparing Total Ma- stectomy, Lumpectomy, and Lumpectomy plus Irradi- ation for the Treatment of Invasive Breast Cancer,” The New England Journal of Medicine, Vol. 347, 2002, pp. 1233-1241. doi:10.1056/NEJMoa022152 [2] U. Veronesi, N. Cascinelli, L. Mariani, et al., “Twenty- Year Follow-Up of a Randomized Study Comparing Breast-Conserving Surgerywith Radical Mastectomy for Early Breast Cancer,” The New England Journal of Medi- cine, Vol. 347, 2002, pp. 1227-1232. doi:10.1056/NEJMoa020989 Copyright © 2012 SciRes. JCT  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study 829 [3] V. Vinh-Hung and C. Verschraegen, “Breast-Conserving Surgery with or without Radiotherapy: Pooled-Analysis for Risks of Ipsilateral Breast Tumor Recurrence and Mortality,” Journal of the National Cancer Institute, Vol. 96, No. 2, 2004, pp. 115-1121. doi:10.1093/jnci/djh013 [4] M. Clarke, R. Collins, S. Darby, et al., “Effects of Radio- therapy and of Differences in the Extent of Surgery for Early Breast Cancer on Local Recurrence and 15-Year Survival: An Overview of the Randomised Trials,” Lan- cet, Vol. 366, No. 9503, 2005, pp. 2087-2106. [5] Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), S. Darby, P. McGale, C. Correa, et al., “Ef- fect of Radiotherapy after Breast-Conserving Surgery on 10-Year Recurrence and 15-Year Breast Cancer Death: Meta-Analysis of Individual Patient Data for 10,801 Women in 17 Randomised Trials,” Lancet, Vol. 378, No. 9804, 2011, pp. 1707-1716. doi:10.1016/S0140-6736(11)61629-2 [6] D. C. Farrow, W. C. Hunt and J. M. Samet, “Geographic Variation in the Treatment of Localized Breast Cancer,” The New England Journal of Medicine, Vol. 326, 1992, pp. 1097-1101. doi:10.1056/NEJM199204233261701 [7] D. A. Lazovich, E. White, D. B. Thomas, et al., “Under- utilization of Breast-Conserving Surgery and Radiation Therapy among Women with Stage I or II Breast Can- cer,” The Journal of the American Medical Association, Vol. 266, No. 24, 1991, pp. 3433-3438. doi:10.1001/jama.1991.03470240055032 [8] B. A. Mann, J. M. Samet, W. C. Hunt, et al., “Changing Treatment of Breast Cancer in New Mexico from 1969 through 1985,” The Journal of the American Medical As- sociation, Vol. 259, No. 23, 1988, pp. 3413-3417. doi:10.1001/jama.1988.03720230023024 [9] R. Ballard-Barbash, A. L. Potosky, L. C. Harlan, et al., “Factors Associated with Surgical and Radiation Therapy for Early Stage Breast Cancer in Older Women,” Journal of the National Cancer Institute, Vol. 88, No. 11, 1996, pp. 716-726. doi:10.1093/jnci/88.11.716 [10] W. F. Athas, M. Adams-Cameron, W. C. Hunt, et al., “Travel Distance to Radiation Therapy and Receipt of Radiotherapy Following Breast-Conserving Surgery,” Journal of the National Cancer Institute, Vol. 92, No. 3, 2000, pp. 269-271. doi:10.1093/jnci/92.3.269 [11] A. T. Schroen, D. R. Brenin, M. D. Kelly, et al., “Impact of Patient Distance to Radiation Therapy on Mastectomy Use in Early-Stage Breast Cancer Patients,” Journal of Clinical Oncology, Vol. 23, No. 28, 2005, pp. 7074-7080. doi:10.1200/JCO.2005.06.032 [12] B. Fisher and S. Anderson, “Conservative Surgery for the Management of Invasive and Noninvasive Carcinoma of the Breast: NSABP Trials,” National Surgical Adjuvant Breast and Bowel Project,” World Journal of Surgery, Vol. 18, No. 1, 1994, pp. 63-69. doi:10.1007/BF00348193 [13] K. Holli, R. Saaristo, J. Isola, et al., “Lumpectomy with or without Postoperative Radiotherapy for Breast Cancer with Favourable Prognostic Features: Results of a Rando- mized Study,” British Journal of Cancer, Vol. 84, No. 2, 2001, pp. 164-169. doi:10.1054/bjoc.2000.1571 [14] The Uppsala-Orebro Breast Cancer Study Group, “Sector Resection with or without Postoperative Radiotherapy for Stage I Breast Cancer: A Randomized Trial,” Journal of the National Cancer Institute, Vol. 82, No. 23, 1990, p. 1851. doi:10.1093/jnci/82.23.1851 [15] R. M. Clark, P. B. McCulloch, M. N. Levine, et al., “Randomized clinical Trial to Assess the Effectiveness of Breast Irradiation following Lumpectomy and Axillary Dissection for Node-Negative Breast Cancer,” Journal of the National Cancer Institute, Vol. 84, No. 9, 1992, pp. 683-689. doi:10.1093/jnci/84.9.683 [16] G. L. Smith, Y. Xu, T. A. Buchholz, et al., “Brachyther- apy for Accelerated Partial-Breast Irradiation: A Rapidly Emerging Technology in Breast Cancer Care,” Journal of Clinical Oncology, Vol. 29, No. 2, 2010, pp. 157-165. doi:10.1200/JCO.2009.27.0942 [17] V. Zannis, P. Beitsch, F. Vicini, et al., “Descriptions and Outcomes of Insertion Techniques of a Breast Brachy- therapy Balloon Catheter in 1403 Patients Enrolled in the American Society of Breast Surgeons MammoSite Breast Brachytherapy Registry Trial,” The American Journal of Surgery, Vol. 190, No. 4, 2005, pp. 530-538. doi:10.1016/j.amjsurg.2005.06.007 [18] G. L. Smith, Y. Xu, T. A. Buchholz, et al., “Association between Treatment with Brachytherapy vs Whole-Breast Irradiation and Subsequent Mastectomy, Complications, and Survival among Older Women with Invasive Breast Cancer,” The Journal of the American Medical Associa- tion, Vol. 307, No. 17, 2012, pp. 1827-1837. doi:10.1001/jama.2012.3481 [19] Common Terminology Criteria for Adverse Events (CTCAE) Version 3.0. http://ctep.cancer.gov/protocolDevelopment/electronic_ap plications /docs/ctcaev3.pdf [20] M. Trombetta, T. B. Julian, Y. Kim, et al., “The Alle- gheny General Modification of the Harvard Breast Cos- mesis Scale for the Retreated Breast,” Oncology, Vol. 23, No. 11, 2009, pp. 954-956. [21] B. K. Mary, “Dramatic Increase in the Use of Implantable Accelerated Partial Breast Irradiation after Breast-Con- serving Surgery,” CA: A Cancer Journal for Clinicians, Vol. 61, No. 4, 2011, pp. 205-206. doi:10.3322/caac.20122 [22] B. Belletti, J. S. Vaidya, S. D’Andrea, et al., “Targeted Intraoperative Radiotherapy Impairs the Stimulation of Breast Cancer Cell Proliferation and Invasion Caused by Surgical Wounding,” Clinical Cancer Research, Vol. 14, No. 5, 2008, pp. 1325-1332. doi:10.1158/1078-0432.CCR-07-4453 [23] J. Huang, L. Barbera, M. Brouwers, et al., “Does Delay in Starting Treatment Affect the Outcomes of Radiotherapy? A Systematic Review,” Journal of Clinical Oncology, Vol. 21, No. 3, 2003, pp. 555-563. doi:10.1200/JCO.2003.04.171 [24] R. S. Punglia, A. M. Saito, B. A. Neville, et al., “Impact of Interval from Breast Conserving Surgery to Radio- Copyright © 2012 SciRes. JCT  Intraoperative Open-Cavity Implant for Accelerated Partial Breast Irradiation Using High-Dose Rate Multicatheter Brachytherapy in Japanese Breast Cancer Patients: A Single-Institution Registry Study Copyright © 2012 SciRes. JCT 830 therapy on Local Recurrence in Older Women with Breast Cancer: Retrospective Cohort Analysis,” British Medical Journal, Vol. 340, 2010, p. 845. doi:10.1136/bmj.c845 [25] U. Kraus-Tiefenbacher, L. Bauer, A. Scheda, et al., “Long-Term Toxicity of an Intraoperative Radiotherapy Boost Using Low Energy X-Rays during Breast-Con- serving Surgery,” International Journal of Radiation Oncology, Biology and Physics, Vol. 66, No. 2, 2006, pp. 377-381. doi:10.1016/j.ijrobp.2006.05.042 [26] M. C. Leonardi, G. B. Ivaldi, L. Santoro, et al., “Long- Term Side Effects and Cosmetic Outcome in a Pool of Breast Cancer Patients Treated with Intraoperative Ra- diotherapy with Electrons as Sole Treatment,” Tumori, Vol. 98, No. 3, 2012, pp. 324-330. [27] E Sperk, G Welzel, A Keller, et al., “Late Radiation Tox- icity after Intraoperative Radiotherapy (IORT) for Breast Cancer: Results from the Randomized Phase III Trial TARGIT A,” Breast Cancer Research and Treatment, Vol. 135, No. 1, 2012, pp. 253-256. doi:10.1007/s10549-012-2168-4 [28] H. M. Kuerer, T. B. Julian, E. A. Strom, et al., “Acceler- ated Partial Breast Irradiation after Conservative Surgery for Breast Cancer,” Annals of Surgery, Vol. 239, No. 3, 2004, pp. 338-351. doi:10.1097/01.sla.0000114219.71899.13 [29] J. S. Vaidya, D. J. Joseph, J. S. Tobias, et al., “Targeted Intraoperative Radiotherapy versus Whole Breast Radio- therapy for Breast Cancer (TARGIT-A trial): An Interna- tional, Prospective, Randomised, Non-Inferiority Phase 3 Trial,” Lancet, Vol. 376, No. 9735, 2010, pp. 91-102. doi:10.1016/S0140-6736(10)60837-9 [30] A. Ruano-Ravina, P. Cantero-Muñoz and A. Eraso Urién, “Efficacy and Safety of Intraoperative Radiotherapy in Breast Cancer: A Systematic Review,” Cancer Letters, Vol. 313, No. 1, 2011, pp. 15-25. doi:10.1016/j.canlet.2011.08.020 [31] Z. Póti, C. Nemeskéri, A. Fekésházy, et al., “Partial Breast Irradiation with Interstitial 60CO Brachytherapy Results in Frequent Grade 3 or 4 Toxicity. Evidence Based on a 12-Year Follow-Up of 70 Patients,” Interna- tional Journal of Radiation Oncology, Biology and Phys- ics, Vol. 58, No. 4, 2004, pp. 1022-1033. doi:10.1016/j.ijrobp.2003.08.013 [32] F. Perera, F. Chisela, L. Stitt, et al., “TLD Skin Dose Measurements and Acute and Late Effects after Lumpec- tomy and High-Dose-Rate Brachytherapy Only for Early Breast Cancer,” International Journal of Radiation On- cology, Biology and Physics, Vol. 62, No. 5, 2005, pp. 1283-1290. doi:10.1016/j.ijrobp.2005.01.007 [33] S. M. Bentzen and J. R. Yarnold, “Reports of Unexpected Late Side Effects of Accelerated Partial Breast Irradia- tion—Radiobiological Considerations,” International Jour- nal of Radiation Oncology, Biology and Physics, Vol. 77, No. 4, 2010, pp. 969-973. doi:10.1016/j.ijrobp.2010.01.059 [34] T. Saphner, D. C. Tormey and R. Gray, “Annual Hazard Rates of Recurrence for Breast Cancer after Primary Therapy,” Journal of Clinical Oncology, Vol. 14, No. 10, 1996, pp. 2738-2746. [35] H. A. Jones, N. Antonini, A. A. Hart, et al., “Impact of Pathological Characteristics on Local Relapse after Breast-Conserving Therapy: A Subgroup Analysis of the EORTC Boost versus No Boost Trial,” Journal of Clini- cal Oncology, Vol. 27, No. 30, 2009, pp. 4939-4947. doi:10.1200/JCO.2008.21.5764 [36] M. Sanghani, P. T. Truong, R. A. Raad, et al., “Valida- tion of a Web-Based Predictive Nomogram for Ipsilateral Breast Tumor Recurrence after Breast Conserving Ther- apy,” Journal of Clinical Oncology, Vol. 28, No. 5, 2010, pp. 718-722. doi:10.1200/JCO.2009.22.6662 [37] C. Aristei, C. Leonardi, F. Stracci, et al., “Risk Factors for Relapse after Conservative Treatment in T1-T2 Breast Cancer with One to Three Positive Axillary Nodes: Re- sults of an Observational Study,” Annals of Oncology, Vol. 22, No. 4, 2011, pp. 842-847. doi:10.1093/annonc/mdq470 [38] C. Shah, J. B. Wilkinson, M. Lyden, et al., “Predictors of Local Recurrence following Accelerated Partial Breast Irradiation: A Pooled Analysis,” International Journal of Radiation Oncology, Biology and Physics, Vol. 82, No. 5, 2012, pp. 825-830. doi:10.1016/j.ijrobp.2011.11.042 [39] P. L. Nguyen, A. G. Taghian, M. S. Katz, et al., “Breast Cancer Subtype Approximated by Estrogen Receptor, Progesterone Receptor, and HER-2 Is Associated with Local and Distant Recurrence after Breast-Conserving Therapy,” Journal of Clinical Oncology, Vol. 26, No. 14, 2008, pp. 2373-2378. doi:10.1200/JCO.2007.14.4287 [40] E. K. Millar, P. H. Graham, S. A. O’Toole, et al., “Pre- diction of Local Recurrence, Distant Metastases, and Death after Breast-Conserving Therapy in Early-Stage Invasive Breast Cancer Using a Five-Biomarker Panel,” Journal of Clinical Oncology, Vol. 27, No. 28, 2009, pp. 4701-4708. doi:10.1200/JCO.2008.21.7075 [41] Z. Gabos, J. Thoms, S. Ghosh, et al., “The Association between Biological Subtype and Locoregional Recur- rence in Newly Diagnosed Breast Cancer,” Breast Cancer Research and Treatment, Vol. 124, No. 1, 2010, pp. 187-194. doi:10.1007/s10549-010-1135-1 [42] J. A. Hattangadi-Gluth, J. Y. Wo, P. L. Nguyen, et al., “Basal Subtype of Invasive Breast Cancer Is Associated with a Higher Risk of True Recurrence after Conventional Breast-Conserving Therapy,” International Journal of Radiation Oncology, Biology and Physics, Vol. 82, No. 3, 2012, pp. 1185-1191. doi:10.1016/j.ijrobp.2011.02.061
|