Paper Menu >>
Journal Menu >>
 Materials Sciences and Applications, 2010, 1, 109-117 doi:10.4236/msa.2010.13019 Published Online August 2010 (http://www.SciRP.org/journal/msa) Copyright © 2010 SciRes. MSA 109 Preparation of Micron-Sized Di-Functional Magnetic Composite Polymer Particles Hasan Ahmad1, Tania Tofaz1, Mohammad Wali Ullah Oli1, Mohammad Abdur Rahman1, Mohammad Abdul Jalil Miah1, Klaus Tauer2 1Department of Chemistry, Rajshahi University, Rajshahi, Bangladesh; 2MPI of Colloid and Interfaces, Am Mühlenberg, Golm, Germany. Email: samarhass@yahoo.com, Klaus.tauer@mpikg.mpg.de Received May 22nd, 2010; revised June 17th, 2010; accepted July 7th, 2010. ABSTRACT In this investigation micron-sized monodisperse magnetic composite polymer particles with amino and amide functional groups were prepared considering their applications in biotechnology. First, polystyrene/poly (acrylic acid-acrylam- ide-N-N-methylene-bis-acrylamide) [PS/P(AA-AAm-MBAAm)] composite polymer particles were prepared by seeded copolymerization. The carboxyl groups present on or near the particles surface were modified by amine-nucleophile, ethylene diamine (EDA), through pre-activation with dicyclohexyl carbodiimide as coupling agent. The aminated parti- cles were then magnetically modified and named as aminated-Fe3O4 composite particles. Formation of such magnetic composite particles was confirmed by scanning electron micrographs, FTIR-spectra and magnetic susceptibility meas- urement. The produced composite particles were paramagnetic. To see the relative hydrophilic character of the parti- cles surface the adsorption behavior of trypsin (TR) as biomolecule was studied on PS particles and aminated-Fe3O4 composite particles. The magnitude of adsorbed TR on PS particles was higher than that on aminated-Fe3O4 composite particles. Keywords: Aminated Fe3O4 Composite, Amino and Amide Groups, Trypsin, Hydrophilic 1. Introduction In recent years polymer particles with well defined parti- cle size and various reactive functional groups on the sur- face are attracting much interest because of their applica- tions in a broad range of fields, e.g. as binders in paints, adhesives, paper coating, textile and as a solid support in the biochemical and biological field as well as in cataly- sis and calibration standards [1-5]. The functional groups include hydroxyl [6-8], aldehyde [9-11], carboxyl [12- 16], amino [17-19], epoxy [20-22], cyano [23], sulfhydr- yl [24] and chloromethyl groups [25-27]. A variety of methods are available to produce monodispersed parti- cles in the micrometer-size range. Of these, dispersion polymerization in non-aqueous media allows successful preparation of micron-sized polymer particles with ease [28-33]. However, this methodology has suffered draw- backs when functional monomers were employed [34, 35]. These limitations include polydispersity, odd-shaped particles and in extreme case coagulation during polymer- ization. Moreover, the presence of functional comono- mers in dispersion copolymerization produces polymer particles with limited number of functional groups on the surface. To overcome these limitations dispersion pol- ymerization followed by seeded copolymerization is exp- ected to be suitable to prepare functional polymer parti- cles in the micrometer-size range. The carboxyl group is one of the most widely studied functional groups for polymer particles intended for bio- medical or diagnostic applications. This functional group can undergo easy derivatization using water/oil- soluble coupling agents. In a previous work H. Ahmad et al. rep- orted the amination of carboxyl functionalized submic- ron-sized copolymer latex particles using amine-nucleo- philes [36]. The present work is an extension of this early work, emphasizing the precipitation of magnetic nano- particles on the surface of micron-sized aminated com- posite polymer particles to diversify their applications in magnetic support based separation in various fields of biotechnology and biomedicine, like cell separation [37], enzyme immobilization [38,39], drug delivery [40], and protein separation [41,42]. Most of the works available in the literature are concerned with preparation of nano to submicron-sized magnetic latex particles [43-50] and a few with micron-sized latex particles [51,52] bearing id- entical functional group. To prepare magnetic latex par- ticles numerous routes have been investigated. They in- clude the precipitation of iron oxide submicron particles  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles 110 in presence of water soluble polymers [53-55], polymer- ization of suitable monomer mixtures in presence of iron oxide particles stabilized by surfactants [56-58] and layer-by-layer self-assembly of alternating layers of pol- yelectrolytes and magnetic nanoparticles onto colloidal templates [59]. In a recent work, preparation of submicr- on-sized sulfate- and carboxyl-functionalized magnetite/ polystyrene spheres is reported for further deposition of gold nanoparticles [60]. Despite the success of these appr- oaches little attention has been made for the preparation of di-functional micron-sized magnetic latex particles. The present work aims to describe the preparation of monodispersed micron-sized magnetic composite partic- les bearing amino and amide groups on or near the parti- cles surface. The produced di-functional magnetic latexes, named as aminated-Fe3O4 latexes, would offer advantage- es in biomedical applications as amino functional group can be utilized for coupling of affinity ligands or binding of biomolecules and the remaining amide functional group can simultaneously be used to modify the particles surface or even can act as spacer among the immobilized biomolecules preventing self reaction. 2. Experimental Details 2.1 Materials Styrene (S) and acrylic acid (AA) purchased from Fluka, Chemika, Switzerland, were distilled under reduced pre- ssure to remove inhibitors and preserved in the refrigera- tor until use. Acrylamide (AAm) from LOBA Chem. In- dia and N,N'-methylene-bis-acrylamide (MBAAm) from Sigma, Chemical Company, USA, a crosslinking agent were used without any purification. 2,2′-Azobisisobuty- ronitrile (AIBN) and potassium persulfate (KPS) both from LOBA Chem. India, were recrystallized from etha- nol and distilled water respectively and preserved in the refrigerator before use. Poly(N-vinylpyrrolidone) (PVP) from Fluka, Chemika, Switzerland of molecular weight 3.6 × 105 g/mol was used as a polymeric stabilizer. Tri- caprylylmethylammonium chloride (aliquate336) from Fl- uka, Chemika, Switzerland was used as a cationic co- stabilizer. The biomolecule used was trypsin (TR) from E. Merck, Germany, used as adsorbent on the surface of polymer particles without any purification. Dicyclohexyl carbodiimide (DCC), ethylene diamine (EDA) all from LOBA Chem. India, were used as supplied. Ferric chlo- ride hexahydrate (FeCl3·6H2O), ferrous sulphate (FeSO4), sodium hydroxide (NaOH), oleic acid and other chemi- cals were of analytical grade. Deionized water was dis- tilled using a glass (Pyrex) distillation apparatus. Scanning Electron Microscope, SEM (LEO Electron Microscopy Ltd., UK); Burker Proton NMR, 250 MHz; FTIR (8044 Simadzu, Japan); Centrifuge machine from Kokuson Corporation, Tokyo, Japan; Helios gamma sin- gle-beam UV-Visible spectrophotometer from Unicam, UK; HI 9321 Microprocessor pH-meter and conductivity meter both from HANNA instruments, Portugal were us- ed in this study. NICOMP 380 (USA) particles sizer was used to measure hydrodynamic diameter of the polymer particles. Sherwood Scientific Magnetic Susceptibility Ba- lance was used for susceptibility measurement. Thermal analysis was carried out using thermogravimetry (TG) analyser from Seiko Instruments Inc EXSTAR 6000 TG/ DTA 6300. 2.2 Preparation of Polystyrene (PS) Seed Particles Polystyrene (PS) seed particles were prepared by disper- sion polymerization of 10 g styrene in presence of PVP (0.432 g) and aliquate336 (0.123 g) using 0.162 g AIBN as oil soluble initiator. The dispersion media used was ethanol (65 mL) and the polymerization was carried out in a 250 mL three necked round bottom flask under a nitrogen atmosphere at 70℃ for 24 h. The conversion was nearly 100% as measured gravimetrically. 2.3 Preparation of Micron-Sized Composite Polymer Particles Micron-sized PS/P(AA-AAm-MBAAm) composite pol- ymer particles were prepared by seeded copolymeriza- tion of AA, AAm and MBAAm in presence PS seed par- ticles. Prior to the seeded copolymerization PS seed par- ticles were transferred into water and magnetically stirred with AA, AAm and MBAAm comonomers at room temperature for 12 h in order to minimize the formation of soluble copolymer. The reaction flask was then trans- ferred to a preheated thermostat water bath maintained at 70℃. The polymerization reaction was carried out under a nitrogen atmosphere for 12 h using KPS as an initiator. Polymerization conditions are shown in Table 1. 2.4 Measurement of Hydrodynamic Particle Size PS/P(AA-AAm-MBAAm) composite polymer particles were washed by serum replacement to remove any unrea- cted monomer and initiator fragments. A dilute dispers- ion of approximately 0.01% polymer solid was prepared, Table 1. Preparation of PS/P(AA-AAm-MBAAm)a) compo- site polymer particlesb) Ingredients PS dispersionc) (g) 29.5130 AA (g) 0.8881 AAm (g) 0.8759 MBAAm (g) 0.036 KPS (g) 0.29 Water (g) 60.0 a)Core/shell ratio: 1/0.6; b)70˚C, N2, 120 rpm, 24 h; c)Polymer solid, 4.89 g/L Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles111 pH was adjusted using acid/alkali solution and intensity weighted average hydrodynamic size was measured by NICOMP particle size. Each sample was measured tw- ice and the average value is reported. The deviation be- tween successive measurements was less than ± 5%. 2.5 Amination of PS/P (AA-AA-MBAAm) Composite Particles PS/P(AA-AA-MBAAm) latex particles were transferred from water to ethanol medium by ultra-centrifugal was- hing at 12,000 rpm and the solid content of the latex was adjusted to about 2%. EDA was covalently bonded to the carboxyl groups of composite particles by the following method [36]. Pre-activation method: 50 g of latex dispersion was mi- xed with 2.1166 g coupling agent (DCC) in a reagent bottle, magnetically stirred at 25˚C for 1 h. The activated carboxyl group was then reacted with 0.6154 g EDA at 25˚C for 3 h and then again at 0˚C for 24 h. 2.6 Preparation of Aminated-Fe3O4 Composite Particles 2.5 g of modified aminated composite particles were taken in a round bottom flask containing 70 mL water. The dispersion was cooled (0-5˚C) in an ice-water bath under a nitrogen gas bubbling. 0.5 g (3 mmol) of FeCl3·6H2O and 0.2558 g (2.7 mmol) of FeSO4 were dissolved in 25 mL water. Then the iron solution was added to the flask con- taining the aminated composite dispersion. A light brown color mixture was formed. The ice bath was removed and the flask was immersed in a preheated water bath at 85˚C. Immediately, 2.9123 mL of 5 M aqueous NaOH solution was added. The reaction mixture gradua- lly turned to black. The mixture was kept stirring at 85˚C for 2 h. Oleic acid (0.01456 g, 2.26 mmol) was slowly added towards the end of the process to stabilize the ami- nated-Fe3O4 dispersion. Then the dispersion was cooled to room temperature. The resultant magnetic particles were centrifuged and thoroughly washed with deionized distilled water, followed by 0.1 M HCl and again by de- ionized distilled water. 2.7 Measurement of Magnetic Susceptibility Aminated-Fe3O4 composite particles were washed, sepa- rated from their dispersion by centrifugation and dried in oven at 80˚C for 2 h. The dried powders were placed in a pre-weighed sample tube and measured the magnetic susceptibility (g) using the following equation: 9 0 10 m RRLC g where, C is calibration constant of the balance, L is length of the sample, R0 and R are reading of the empty and sample tubes and M is weight of the sample (dry basis) in C.G.S unit. 2.8 Thermogravimetric Analysis (TGA) of Aminated-Fe3O4 Composite Polymer Particles Thermal properties of the dried aminated-Fe3O4 powder was measured by heating samples under flowing nitrogen atmosphere from 40˚C to 600˚C at a heating rate of 20˚C /min and the weight loss was recorded. 2.9 Adsorption of Trypsin (TR) on PS Seed and Aminated-Fe3O4 Composite Polymer Particles A mixture of 20 mL was prepared from purified polymer dispersion (polymer solid 0.15 g) and TR (200 mg/g). The pH value of the mixture was immediately adjusted at the isoelectric point (pH 10.0) using a buffer solution. In each measurement the dispersion-TR mixture was al- lowed to stand at 25˚C for 45 min. Polymer particles were then separated by centrifugation at 10,000 rpm for 10 min. In order to remove wafting particles completely, the supernatant was centrifuged once more. The concen- tration of TR in the supernatant was determined by a UV-visible spectrophotometer at the wavelength of 280 nm. The magnitude of TR adsorbed was calculated by subtracting the concentration of TR in the medium from that of the initial concentration. Calibration curve was used for this purpose. 3. Results and Discussion Figures 1(a) and (b) shows the SEM photographs of PS and PS/P(AA-AAm-MBAAm) composite polymer parti- cles. The average diameters and coefficient of variations are 1.47 µm and 5.87% for PS seed and 1.51 µm and 2.43% for PS/P(AA-AAm-MBAAm) composite particles. The average size of PS/P(AA-AAm-MBAAm) composite particles increased slightly after seeded copolymerization. Both the PS seed particles and the composite polymer par- ticles are fairly monodispersed and spherical, having a sm- ooth homogeneous surface. Submicron-sized P(AA-AAm- MBAAm) copolymer particles are not observed. This suggests that seeded copolymerization of AA, AAm and MBAAm proceeds smoothly on PS seed particles, with a) b) 2.5 µm 2.5 µm a) b) 2.5 µm2.5 µm 2.5 µm2.5 µm (a) (b) 2.5 μm2.5 μm Figure 1. SEM photographs of (a) PS seed; (b) PS/P(AA- AAm-MBAAm) composite polymer particles Copyright © 2010 SciRes. MSA 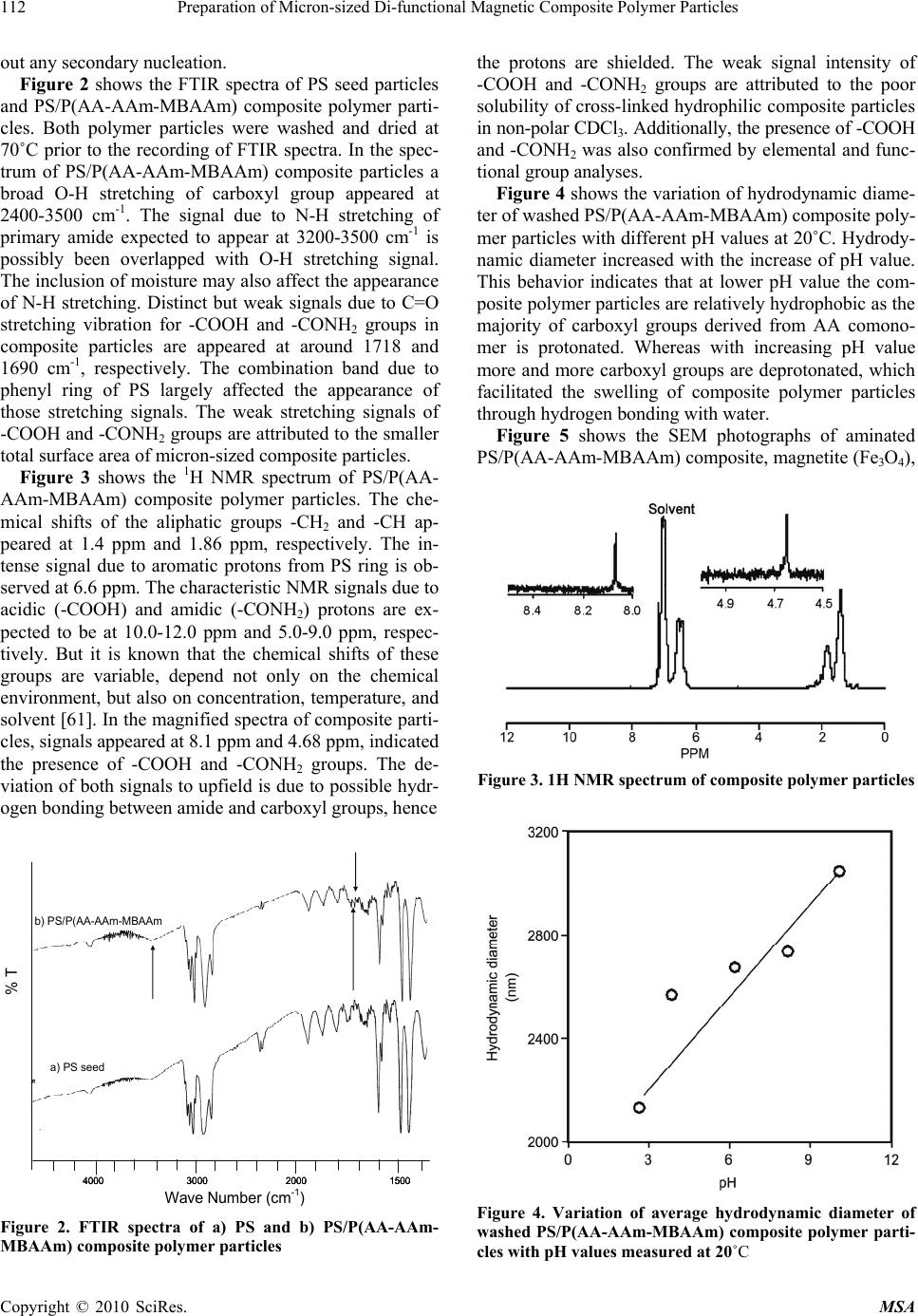 Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles 112 out any secondary nucleation. Figure 2 shows the FTIR spectra of PS seed particles and PS/P(AA-AAm-MBAAm) composite polymer parti- cles. Both polymer particles were washed and dried at 70˚C prior to the recording of FTIR spectra. In the spec- trum of PS/P(AA-AAm-MBAAm) composite particles a broad O-H stretching of carboxyl group appeared at 2400-3500 cm-1. The signal due to N-H stretching of primary amide expected to appear at 3200-3500 cm-1 is possibly been overlapped with O-H stretching signal. The inclusion of moisture may also affect the appearance of N-H stretching. Distinct but weak signals due to C=O stretching vibration for -COOH and -CONH2 groups in composite particles are appeared at around 1718 and 1690 cm-1, respectively. The combination band due to phenyl ring of PS largely affected the appearance of those stretching signals. The weak stretching signals of -COOH and -CONH2 groups are attributed to the smaller total surface area of micron-sized composite particles. Figure 3 shows the 1H NMR spectrum of PS/P(AA- AAm-MBAAm) composite polymer particles. The che- mical shifts of the aliphatic groups -CH2 and -CH ap- peared at 1.4 ppm and 1.86 ppm, respectively. The in- tense signal due to aromatic protons from PS ring is ob- served at 6.6 ppm. The characteristic NMR signals due to acidic (-COOH) and amidic (-CONH2) protons are ex- pected to be at 10.0-12.0 ppm and 5.0-9.0 ppm, respec- tively. But it is known that the chemical shifts of these groups are variable, depend not only on the chemical environment, but also on concentration, temperature, and solvent [61]. In the magnified spectra of composite parti- cles, signals appeared at 8.1 ppm and 4.68 ppm, indicated the presence of -COOH and -CONH2 groups. The de- viation of both signals to upfield is due to possible hydr- ogen bonding between amide and carboxyl groups, hence b) PS/P(AA-AAm-MBAAm 40003000 20001500 a) PS seed b) PS/P(AA-AAm-MBAAm 40003000 2000150040003000 20001500 a) PS seed Wave Number ( cm - 1 ) % T Figure 2. FTIR spectra of a) PS and b) PS/P(AA-AAm- MBAAm) composite polymer particles the protons are shielded. The weak signal intensity of -COOH and -CONH2 groups are attributed to the poor solubility of cross-linked hydrophilic composite particles in non-polar CDCl3. Additionally, the presence of -COOH and -CONH2 was also confirmed by elemental and func- tional group analyses. Figure 4 shows the variation of hydrodynamic diame- ter of washed PS/P(AA-AAm-MBAAm) composite poly- mer particles with different pH values at 20˚C. Hydrody- namic diameter increased with the increase of pH value. This behavior indicates that at lower pH value the com- posite polymer particles are relatively hydrophobic as the majority of carboxyl groups derived from AA comono- mer is protonated. Whereas with increasing pH value more and more carboxyl groups are deprotonated, which facilitated the swelling of composite polymer particles through hydrogen bonding with water. Figure 5 shows the SEM photographs of aminated PS/P(AA-AAm-MBAAm) composite, magnetite (Fe3O4), Figure 3. 1H NMR spectrum of composite polymer particles Figure 4. Variation of average hydrodynamic diameter of washed PS/P(AA-AAm-MBAAm) composite polymer parti- cles with pH values measured at 20˚C Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles113 and aminated-Fe3O4 composite particles. All the particles were washed by serum replacement prior to the observa- tion by SEM. It is to be mentioned that after three wash- ings the supernatant obtained from the aminated-Fe3O4 composite particles was clear indicating no leaching of Fe3O4 particle. The observed magnetite (Fe3O4) particles were prepared by co-precipitation of Fe2+ (aq) and Fe3+ (aq) in presence of NaOH and aminated-Fe3O4 composite particles were prepared by the impregnation of iron ions and precipitation of Fe3O4 on aminated composite poly- mer particles. The mean diameter of aminated-Fe3O4 composite particles is 1.60 µm which is greater than the mean diameter of aminated composite polymer particles (1.54 µm). It is also observed that after amination the average size of composite particles slightly increased as compared to that before amination (1.51 µm). Similar inc- rease in average size after amination was also observed in our previous work [36]. The surface of the aminated composite polymer particles is smooth but the surface of the aminated-Fe3O4 composite particles is not smooth with many small Fe3O4 particles attached to the surface of aminated composite particles. Additionally, the preci- pitation of Fe3O4 particles on aminated composite surface was confirmed by the measurement of magnetic suscep- tibility and the value was recorded as positive (2.4768 × 10-4). This result suggests that the prepared aminated- Fe3O4 composite particles are paramagnetic. Prior to this measurement the dispersion was centrifuged and dried at low temperature (80˚C) to prevent any crystal deforma- tion of the impregnated Fe3O4 particles. Figure 6 shows the FTIR spectra of magnetite (Fe3O4) and aminated-Fe3O4 composite particles. In the FTIR sp- ectra of Fe3O4, the characteristic stretching vibrations due to Fe-O bonds of Fe3O4 particles appeared at 582.5 and 439.7 cm-1 respectively, as also reported elsewhere [62, 0.2 µm a)a) 2.5 µ m2.5 µm a)b) 0.2 µm0.2 µm0.2 µm a)a) 2.5 µ m2.5 µm2.5 µm2.5 µ m a)b) (a) (b) 2.5 μm 0.2 μm b) 2.5 µm c) 2.5 µm b) 2.5 µm c) 2.5 µm b) 2.5 µm c) 2.5 µm (c) 2.5 μm Figure 5. SEM photographs of (a) aminated PS/P(AA-AAm- MBAAm) composite polymer; (b) magnetite (Fe3O4); (c) ami- nated-Fe 3O4 composite particles 63]. The broad band at 3330.8 cm-1 can be assigned to the stretching vibration of surface water molecules or the envelope of hydrogen bonded surface OH groups. In am- inated-Fe3O4 composite particles a combined strong sig- nal for N-H and -OH stretching derived from amide, car- boxyl and surface water molecules is appeared at 3404.1 cm-1. The stretching signal due to Fe-O is also observed in aminated-Fe3O4 composite particles at around 439 cm-1 and the signal appeared at 540 cm-1 broadened due to the out of plane C-H bending vibration for substituted phenyl ring of PS. The above results also confirmed that the Fe3O4 particles have successfully been precipitated on the aminated composite polymer particles. TGA was us- ed to determine the total inorganic material i.e. iron oxide content in aminated-Fe3O4 composite particles. TGA curve is illustrated in Figure 7. It is expected that as theaminated-Fe3O4 composite particles are heated from ambient temperature to 600˚C, the organic part of the 50010001500200030004000 a) Fe 3 O 4 particles b) Aminated- Fe 3 O 4 composite 50010001500200030004000 a) Fe 3 O 4 particles b) Aminated- Fe 3 O 4 composite 50010001500200030004000 50010001500200030004000 a) Fe 3 O 4 particles b) Aminated- Fe 3 O 4 composite Wave Number ( cm - 1 ) % T Figure 6. FTIR spectra of a) magnetite (Fe3O4); b) amina- ted-Fe3O4 composite polymer particles 0 20 40 60 80 100 120 0100200 300 400500 600 Temperature (°C) Weight loss (%) 443.9 °C 8.8 % 0 20 40 60 80 100 120 0100200 300 400500 600 Temperature (°C) Weight loss (%) 443.9 °C 8.8 % Figure 7. TGA thermogram of aminated-Fe3O4 composite polymer particles Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles 114 composite would be burned and the percentage of the remaining part after calcination would give the iron ox- ide content. As such TGA thermogram indicates that about 8.8% (w/w) magnetic Fe3O4 nano-particles are pre- cipitated on the aminated composite polymer particles. Considering the larger average size of the composite par- ticles, the percentage of magnetic iron oxide is pretty high to modify the particles surface [64]. Figure 8 shows the adsorption behaviors of TR on the PS seed and aminated-Fe3O4 composite particles. It is well known that magnitude of adsorption is largely dep- endent on the degree of hydrophobic interaction between the particle surface and biomolecules and also on the to- tal particle surface area. Since the sizes of the seed and aminated-Fe3O4 composite particles are not the same as measured from the SEM photographs, so the adsorption behavior of TR has been presented per unit area of the particles. The magnitude of adsorption on the aminated- Fe3O4 composite particles is lower than that on PS seed particles. As already mentioned, in order to use polymer particles, as a carrier for biomolecules, the particle sur- face should be sufficiently hydrophilic with limited am- ount of hydrophobicity. It is well known that the strong hydrophobic interaction of biomolecules with solid sur- face results in the loss of native conformation as well as the activity of adsorbed biomolecule. 4. Conclusions A synthetic rout for the preparation of monodisperse mi- cron-sized magnetic composite polymer particles with amine and amide functional groups on or near the surface has been developed. The process involved the prepara- tion of micron-sized PS latex particle by dispersion poly- merization followed by seeded copolymerization with AA, AAm and MBAAm, surface modification with EDA and subsequent precipitation of iron ions to form Fe3O4. 0.00 0.07 0.14 0.21 0.28 0.35 PS Magnitude of adsorbed TR (mg m -2 ) Aminated-Fe 3 O 4 composite 0.00 0.07 0.14 0.21 0.28 0.35 PS Magnitude of adsorbed TR (mg m -2 ) Aminated-Fe 3 O 4 composite Figure 8. Magnitude of trypsin (TR) adsorbed on PS seed and aminated-Fe3O4 composite particles measured under the constant concentration against the total solid content at 25˚C. pH: 10.0; particles: 0.15 g; TR: 200 mg/g of particles The presence of amino and amide groups improved hy- drophilicity and hence could prevent nonspecific adsorp- tion of proteins. 5. Acknowledgements This work was supported by the financial grant from MOSICT, Dhaka (Economic code: 3-2605-3993-5921). REFERENCES [1] C. Picot, “Position Paper: Functional Polymer Latexes,” Polymer for Advanced Technologies, Vol. 6, No. 7, 1995, pp. 427-434. [2] Q. Wang, S. Fu and T. Yu, “Emulsion Polymerization,” Progress in Polymer Science, Vol. 19, No. 4, 1994, pp. 703-753. [3] J-W. Kim, J-G. Park, J-H. Ryu, I-S. Chang and K-D. Suh, “Monodisperse Chloromethyl-Functionalized Macropor- ous Polymer Particles by Seeded Polymerization in Aqueous Media,” Colloid and Polymer Science, Vol. 283, No. 11, 2005, pp. 1233-1240. [4] M-O. Jung, J-H. Ryu, J-G Park, J-B. Jun and K-D. Suh, “Surface Modification of Monodisperse-Crosslinked Polymeric Microspheres Using a Redox Initiation Sys- tem,” Journal of Applied Polymer Science, Vol. 100, No. 2, 2006, pp. 1349-1356. [5] H. Ahmad, M. M. Rahman and M. A. J. Miah, “Prepara- tion of Micron-sized Epoxy Functionalized Polystyrene Particles and Adsorption of Biomolecules,” Journal of Polymer Materials, Vol. 21, No. 1, 2005, pp. 27-36. [6] B. Charleux, C. Pichot and M. F. Llauro, “Styrene- terminated Poly(vinyl alcohol) Macromonomers: 2. Free- radical (Co)polymerization Studies and Application to the Functionalization of Latex Particles,” Polymer, Vol. 34, No. 2, 1993, pp. 4352-4359. [7] H. Ahmad, M. A. J. Miah, M. S. Pervin and M. M. Rah- man, “Composite Polymer Particles Prepared with Dif- ferent Amounts of Acrylic Acid and Their Usefulness as Carriers for Biomolecules,” Colloid and Polymer Science, Vol. 281, No. 10, 2003, pp. 988-992. [8] A. M. I. Ali, P. Pareek, L. Sewell, A. Schmid, S. Fujii, S. P. Armes and I. M. Shirley, “Synthesis of Poly (2-hy- droxypropyl methacrylate) Latex Particles Via Aqueous Dispersion Polymerization,” Soft Matter, Vol. 3, No. 5, 2007, pp. 1003-1013. [9] S. Margel and E. Wiesel, “Acrolein Polymerization: Mon- odisperse, Homo and Hybrido Microspheres, Synthesis, Mechanism, and Reactions,” Journal of Polymer Science, Polymer Chemistry Edition, Vol. 22, No. 1, 1984, pp. 145- 158. [10] M. Okubo, Y. Kondo and M. Takahashi, “Production of Submicron-Size Monodisperse Polymer Particles Having Aldehyde Groups by Seeded Aldol Condensation,” Col- loid and Polymer Science, Vol. 271, No. 2, 1993, pp. 109- 113. [11] P. Lei, J. Xu, Q. Wang and C. Wu, “Surface Functionali- zation of Polymer Latex Particles: 4. Tailor-Making of Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles115 Aldehyde-functional Poly(methylstyrene) Latexes in an Emulsifier-Free System, Langmuir, Vol. 16, No. 9, 2000, pp. 4141-4147. [12] K. Kang, C. Kan, Y. Du and D. Liu, “Synthesis and Properties of Soap-Free Poly(Methyl Methacrylate-Ethyl Acrylate-Methacrylic Acid) Latex Particles Prepared by Seeded Emulsion Polymerization,” European Polymer Journal, Vol. 41, No. 3, 2005, pp. 439-445. [13] X. Z. Kong and E. Ruckenstein, “Monodisperse Core/ shell Latex Particles Containing Carboxylic Acid Groups and their Optimum Acid Content for Pore Generation,” Journal of Applied Polymer Science, Vol. 71, No. 9, 1999, pp. 1455-1460. [14] J.-S. Song, L. Chagai and M. A. Winnik, “Monodisperse Micrometer-Size Carboxyl-functionalized Polystyrene Par- ticles Obtained by Two-Stage Dispersion Polymeriza- tion,” Macromolecules, Vol. 19, No. 17, 2006, pp. 5729- 5737. [15] H. Ahmad, M. M. Hossain, M. M. Rahman and M. A. J. Miah, “Monodispersed Carboxylated Composite Polymer Microspheres and Physical Immobilization of Bio- molecules,” Polymer Journal, Vol. 39, No. 5, 2007, pp. 428-434. [16] T. Ding, E. S. Daniels, M. S. El-Aasser and A. Klein, “Synthesis and Characterization of Functionalized Poly- mer Latex Particles through a Designed Semicontinuous Emulsion Polymerization Process,” Journal of Applied Polymer Science, Vol. 97, No. 1, 2005, pp. 248-256. [17] T. Delair, V. Marguet, C. Pichot and B. Mandrand, “Syn- thesis and Characterization of Cationic Amino Function- alized Polystyrene Latexes,” Colloid and Polymer Sci- ence, Vol. 272, No. 8, 1994, pp. 962-970. [18] I. Miraballes-Martínez, A. Martín-Molina, F. Galisteo- González and J. Forcada, “Synthesis of Amino-Function- alized Latex Particles by a Multistep Method,” Journal of Polymer Science Polymer Chemistry Edition, Vol. 39, No. 17, 2001, pp. 2929-2936. [19] Z. Ma, Y. Guan and H. Liu, “Synthesis and Characteriza- tion of Micron-Sized Monodisperse Superparamagnetic Polymer Particles with Amino Groups,” Journal of Poly- mer Science Polymer Chemistry, Vol. 43, No. 15, 2005, pp. 3433-3439. [20] D. Horace and P. Caporal, “Reactive Poly(Lucidly Meth- acrylate) Microspheres Prepared by Dispersion Polymeri- zation,” Journal of Polymer Science Polymer Chemistry, Vol. 38, No. 21, 2000, pp. 3855-3863. [21] J. M. Jin, J. M. Lee, M. H. Ha, K. Lee and S. Choe, “Highly Crosslinked Poly(Glycidyl Methacrylate-co-Di- vinyl Benzene) Particles by Precipitation Polymeriza- tion,” Polymer, Vol. 48, No. 11, 2007, pp. 3107-3115. [22] B. Gong, J. Zhu, L. Li, K. Qiang and L. Ren, “Synthesis of Non-Porous Poly(Glycidyl Methacrylate-co-Ethylene- Dimethacrylate) Beads and their Application in Separa- tion of Biopolymers,” Talanta, Vol. 68, No. 3, 2006, pp. 666-672. [23] X. Lu, D. Huang, X. Yang and W. Huang, “Preparation of Narrow or Monodisperse Polymer Microspheres with Cyano Group by Distillation-Precipitation Polymeriza- tion,” Polymer Bulletin, Vol. 56, No. 2-3, 2006, pp. 171- 178. [24] T. Delair, C. Pichot and B. Mandrand, “Synthesis and Characterization of Cationic Latex Particles Bearing Sul- fhydryl Groups and their Use in the Immobilization of Fab Antibody Fragments (1),” Colloid and Polymer Sci- ence, Vol. 272, No. 1, 1994, pp. 72-81. [25] T. Bahar and A. Tuncel, “Monodisperse Poly (p-chloro- methylstyrene) Microbeads by Dispersion Polymeriza- tion,” Polymer Engineering and Science, Vol. 39, No. 10, 1999, pp. 1849-1855. [26] H. Ahmad, N. Saito, Y. Kagawa and M. Okubo, “Prepa- ration of Micrometer-Sized Monodisperse ‘Janus’ Com- posite Polymer Particles Having Temperature-Sensitive Polymer Brushes at Half of the Surface by Seeded Atom Transfer Radical Polymerization,” Langmuir, Vol. 24, No. 3, 2008, pp. 688-691. [27] S. Baruch-Sharon and S. Margel, “Preparation and Char- acterization of Core-Shell Polystyrene/Polychloromethyl- styrene and Hollow Polychloromethylstyrene Microme- ter-sized Particles of Narrow-size Distribution,” Colloid and Polymer Science, Vol. 287, No. 7, 2009, pp. 859-869. [28] K. E. J. Barrett, “Dispersion Polymerization in Organic Meidia,” British Polymer Journal, Vol. 5, No. 4, 1973, pp. 259-271. [29] C. M. Tseng, Y. Y. Lu, M. S. El-Aasser and J. W. Van- derhoff, “Uniform Polymer Particles by Dispersion Po- lymerization in Alcohol,” Journal of Polymer Science Polymer Chemistry Edition, Vol. 24, No. 11, 1986, pp. 2995-3007. [30] T. Okaya, K. Kikuchi, A. Sukuki and N. Ikeda, “Disper- sion Polymerization of Vinyl Acetate in a Mixture of Ethanol and Water,” Polymer International, Vol. 54, No. 1, 2005, pp. 143-148. [31] D. Hérault, C. Saluzzo and M Lemaire, “Preparation of Monodisperse Enantiomerically Pure Glycidyl Methacry- late–Ethylene Glycol Dimethacrylate Copolymers in Dispersion Copolymerization: Functionalization,” Reac- tive and Functional Polymers, Vol. 66, No. 5, 2006, pp. 567-577. [32] S. Jiang, E. D. Sudol, V. L. Dimonie and M. S. El-Aasser, “Kinetics of Dispersion Polymerization of Methyl Methacrylate and n-Butyl Acrylate: Effect of Initiator Concentration, Macromolecules, Vol. 40, No. 14, 2007, pp. 4910-4916. [33] H. Ahmad, B. M. Chaki, M. M. Rahman, M. A. J. Miah and K. Tauer, “Solvency Effect of the Dispersion Me- dium on the Radical Polymerization of Styrene in Non- Aqueous Dispersion Media,” E-Polymers, No. 080, 2007. http://www.e-polymers.org [34] J. S. Song, F. Tronc and M. A. Winnik, “Two-Stage Dis- persion Polymerization toward Monodisperse, Controlled Micrometer-Sized Copolymer Particles,” Journal of Ameri- can Chemical Society, Vol. 126, No. 21, 2004, pp. 6562- 6563. [35] M. Yasuda, H. Seki, H. Yokoyama, H. Ogino, K. Ishimi Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles 116 and H. Ishikawa, “Simulation of a Particle Formation Stage in the Dispersion Polymerization of Styrene” Mac- romolecules, Vol. 34, No. 10, 2001, pp. 3261-3270. [36] H. Ahmad, M. E. Hossain, M. A. Rahman, M. M. Rah- man, M. A. J. Miah and K. Tauer, “Carboxyl Functional- ized Poly(Methyl Methacrylate-Acrylic Acid-Ethylene Glycol Dimethacrylate) Copolymer Particles and Their Amination with Amine-Nucleophiles,” E-Polymers, No. 096. 2008. http//www.e-polymers.org [37] P. Kronick and R. W. Gilpin, “Use of Superparamagnetic Particles for Isolation of Cells,” Journal of Biochemical and Biophysical Methods, Vol. 12, No. 1-2, 1986, pp. 73-80. [38] X. Li and Z. H. Sun, “Synthesis of Magnetic Polymer Microspheres and Application for Immobilization of Pro- teinase of Balillus Sublitis,” Journal of Applied Polymer Science,” Vol. 58, No. 11, 1995, pp. 1991-1997. [39] X. Liu, Y. Guan, R. Shen and H. Liu, “Immobilization of Lipase onto Micron-Size Magnetic Beads,” Journal of Chromatography B, Vol. 822, No. 1-2, 2005, pp. 91-97. [40] T. K. Jain, M. A. Morales, S. K. Sahoo, D. L. Seslie- Pelecky and V. Labhasetwar, “Iron Oxide Nanoparticles for Sustained Delivery of Anticancer Agents,” Molecular Pharmaceutics, Vol. 2, No. 3, 2005, pp. 194-205. [41] A. M. Schmidt, “Thermoresponsive Magnetic Colloids,” Colloid and Polymer Science, Vol. 285, No. 9, 2007, pp. 953-966. [42] A. Elaissari and V. Bourrel, “Thermosensitive Magnetic Latex Particles for Controlling Protein Adsorption and Desorption,” Journal of Magnetism and Magnetic Mate- rials, Vol. 225, No. 1-2, 2001, pp. 151-155. [43] F. Sayer, G. Güven and E. Pişkin, “Magnetically Loaded Poly(methyl methacrylate-co-acrylic acid) Nano-particles,” Colloid and Polymer Science, Vol. 284, No. 9, 2006, pp. 965-978. [44] M. Lattuada and T. A. Hatton, “Functionalization of Monodisperse Magnetic Nanoparticles,” Langmuir, Vol. 23, No. 4, 2007, pp. 2158-2168. [45] J-J. Yuan, S. P. Armes, Y. Takabayashi, K. Prassides, C. A. P. Leite, F. Galembeck and A. L. Lewis, “Synthesis of Biocompatible Poly[2-(methacryloyloxy)ethyl phosphoryl- choline]-Coated Magnetite Nanoparticles, Langmuir, Vol. 22, No. 26, 2006, pp. 10989-10993. [46] R. A. Wassel, B. Grady, R. D. Kopke and K. J. Dormer, “Dispersion of Super Paramagnetic Iron Oxide Nanopar- ticles in Poly(d,l-lactide-co-glycolide) Microparticles,” Colloids and Surfaces A: Physicochemical and Engineer- ing Aspects, Vol. 292, No. 2-3, 2007, pp. 125-130. [47] H. Mouaziz, S. Braconnot, F. Ginot and A. Elaïssari, “Elaboration of Hydrophilic Aminodextran Containing Submicron Magnetic Latex Particles,” Colloid and Poly- mer Science, Vol. 287, No. 3, 2009, pp. 287-297. [48] H. Mouaziz, R. Veyret, A. Theretz, F. Ginot and A. Elaïssari, “Aminodextran Containing Magnetite Nanopar- ticles for Molecular Biology Applications: Preparation and Evaluation,” Journal of Biomedical Nanotechnology, Vol. 5, No. 2, 2009, pp. 172-181. [49] F. Sauzedde, A. Elaïssari and C. Pichot, “Hydrophilic Magnetic Polymer Latexes. 1. Adsorption of Magnetic Iron Oxide Nanoparticles onto Various Cationic Latexes,” Colloid and Polymer Science, Vol. 277, No. 9, 1999, pp. 846-855. [50] E. Pollert, K. Knížek, M. Maryško, A. Lančok, J. Bo- háček, D. Horák and M. Babič, “Magnetic Poly(glycidyl methacrylate) Microspheres Containing Maghemite Pre- pared by Emulsion Polymerization,” Journal of Magnet- ism and Magnetic Materials Vol. 306, No. 2, 2006, pp. 241-247. [51] C. Yang, H. Liu, Y. Guan, J. Xing, J. Liu and G. Shan, “Preparation of Magnetic Poly(Methylmethacrylate-Divinyl- benzene-Glycidylmethacrylate) Microspheres by Spraying Suspension Polymerization and their Use for Protein Ad- sorption,” Journal of Magnetism and Magnetic Materials, Vol. 293, No. 1, 2005, pp. 187-192. [52] D. Horak, M. Babic, H. Mackova and M. J. Benes, “Preparation and Properties of Magnetic Nano- and Mi- crosized Particles for Biological and Environmental Separations,” Journal of Separation Science, Vol. 30, No. 11, 2007, pp. 1751-1772. [53] D. Horak, M. Babic, H. Mackova and M. J. Benes, “Preparation and Properties of Magnetic Nano- and Mi- crosized Particles for Biological and Environmental Separations,” Journal of Separation Science, Vol. 30, No. 11, 2007, pp. 1751-1772. [54] C. C. Berry, S. Wells, S. Charles and A. S. G. Curtis, “Dextran and Albumin Derivatised Iron Oxide Nanopar- ticles: Influence on Fibroblasts in Vitro,” Biomaterials, Vol. 24, No. 25, 2003, pp. 4551-4557. [55] A. L. Autenshlyus, N. A. Brusenstov and A. Lockshim, “Magnetic-sensitive Dextran-Ferrite Immunosorbents (for Diagnostic and Therapy),” Journal of Magnetism and Magnetic Materials, Vol. 122, No. 1-3, 1993, pp. 360- 363. [56] D. Charmot, “Preparation of Monodisperse, Magnetizable, Composite Metal/Polymer Microspheres,” Progress in Colloid and Polymer Science, Vol. 79, 1989, pp. 94-100. [57] L. P. Ramirez and K. Landfester, “Magnetic Polystyrene Nanoparticles with a High Magnetite Content Obtained by Miniemulsion Processes,” Macromolecular Chemistry and Physics, Vol. 204, No. 1, 2003, pp. 22-31. [58] S. Lu and J. Forcada, “Preparation and Characterization of Magnetic Polymeric Composite Particles by Minie- mulsion Polymerization,” Journal of Polymer Science Polymer Chemistry Edition, Vol. 44, No. 13, 2006, pp. 4187-4203. [59] F. Caruso, A. S. Susha, M. Giersig and H. Mohwald, “Magnetic Core-shell Particles: Preparation of Magnetite Multilayers on Polymer Latex Microspheres,” Advanced Materials, Vol. 11, No. 11, 1999, pp. 950-953. [60] S.-Z. Hsiao, J.-L. Ou, Y. Sung, C.-P. Chang and M.-D. Ger, “Preparation of Sulfate- and Carboxyl-functionalized Magnetite/Polystyrene Spheres for Further Deposition of Gold Nanoparticles,” Colloid and Polymer Science, Vol. 288, No. 7, 2010, pp. 787-794. Copyright © 2010 SciRes. MSA  Preparation of Micron-sized Di-functional Magnetic Composite Polymer Particles Copyright © 2010 SciRes. MSA 117 [61] D. L. Pavia, G. M. Lampman, G. S. Kriz and J. A. Vy- vyan, “Introduction to Spectroscopy,” Harcourt Brace College Publishers, New York, 2008. [62] J. L. Zhang, R. S. Srivastava and R. D. K. Misra, “Core-Shell Magnetite Nanoparticles Surface Encapsu- lated with Smart Stimuli-Responsive Polymer: Synthesis, Characterization, and LCST of Viable Drug-targeting De- livery System,” Langmuir, Vol. 23, No. 11, 2007, pp. 6342-6351. [63] M. Ma, Y. Zhang, W. Yu, H. Y. Shen, H. Q. Zhang and N. Gu, “Preparation and Characterization of Magnetite Nanoparticles Coated by Amino Silane,” Colloids and Surfaces A: Physicochemical and Engineering Aspects, Vol. 212, No. 2-3, 2003, pp. 219-226. [64] P. Govindaiah, Y. J. Jung, J. M. Lee, T-J. Park, D. Y. Ryu, J. H. Kim and I. W. Cheong, “Monodisperse and Fluo- rescent Poly(Styrene-co-Methacrylic Acid-co-2-Naphthyl Methacrylate)/Fe3O4 Composite Particles,” Journal of Colloid and Interface Science, Vol. 343, No. 2, 2010, pp. 484-490. |

