 Advances in Bioscience and Biotechnology, 2012, 3, 805-813 ABB http://dx.doi.org/10.4236/abb.2012.326100 Published Online October 2012 (http://www.SciRP.org/journal/abb/) Increased resistance to apoptosis during differentiation and syncytialization of BeWo choriocarcinoma cells Bih-Rong Wei1,2,3, Chuan Xu1,2, Neal S. Rote1,2* 1Department of Reproductive Biology, Case Western Reserve University School of Medicine, Case Western Reserve University, Cleveland, USA 2Department of Obstetrics and Gynecology, University Hospitals Case Medical Center, Cleveland, USA 3SAIC-Frederick, Bethesda, USA Email: weib@mail.nih.gov, *neal.rote@case.edu Received 15 August 2012; revised 20 September 2012; accepted 30 September 2012 ABSTRACT Transition from mononuclear villous cytotrophoblast into multinuclear syncytiotrophoblast in the human placenta is accompanied by changes in apoptosis- related proteins and an apparent increased resistance to induced apoptosis. We investigated the specific nature and timing of changes in Bcl-2, Bax, p53, and caspases 3 and 8 in forskolin-treated BeWo chorio- carcinoma cells, a model for villous cytotrophoblast differentiation. BeWo cells were treated with forsko- lin or vehicle alone for up to 72 h and evaluated at 24 h intervals for syncytialization and quantitative ex- pression specific apoptosis-related proteins and mRNAs. Syncytialization was quantified using fluo- rescent staining of intercellular membranes and enu- meration of the percentage of nuclei in multinucleate cells, and differential localization of apoptosis-related proteins to multinuclear or mononuclear cells was determined by quantitative immunofluorescence. For- skolin treatment for up to 72 h resulted in 80% syn- cytialization, increased expression of Bcl-2 protein (P < 0.01) and mRNA (P < 0.05), and significantly de- creased expression of protein and mRNA for Bax, p53, and caspases 3 and 8. Syncytialized cells expressed higher levels of Bcl-2 protein concurrent with in- creased resistance to cisplatin-induced apoptosis. Thus, syncytialization of BeWo cells was accompanied by altered transcription of apoptotic-related proteins characteristic of increased apoptosis resistance sec- ondary to increased expression of the anti-apoptotic protein Bcl-2 and diminish expression of pro-apop- totic proteins. Keywords: BeWo; Trophoblast; Placenta; Caspase 8; Caspase 3; Bcl-2; Intercellular Fusion 1. INTRODUCTION The surface of the human placenta is composed of mul- tinucleate syncytiotrophoblast that expands throughout pregnancy by intercellular fusion from an underlying feeder layer of mononuclear cells (villous cytotro- phoblast). Transition from mononuclear villous cytotro- phoblast into multinuclear syncytiotrophoblast was ac- companied by a variety of changes in the level and activ- ity of apoptosis-related proteins [1,2]. Normal placental syncytiotrophoblast expressed antiapoptotic and pro- apoptotic proteins, as well as traditional indicators of apoptosis [3-8]. Some nuclei were positive for terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL), suggesting DNA fragmentation. Areas of the syncytium contained both anti-apoptotic proteins (Bcl-2 and myeloid cell leukemia sequence 1 [Mcl-1]) and pro- apoptotic Bak [1,3-9]. Activated caspase 8 and Fas-as- sociated death domain-like interleukin-1β-converting en- zyme-inhibitory protein (c-FLIP), an inhibitor of cas- pase 8, appeared to be expressed in villous cytotro- phoblast and syncytiotrophoblast [5,6,10], whereas acti- vated caspases 3 and 8 were found only in the syncyti- otrophoblast [3-5,10-12]. In spontaneously differentiat- ing isolated term villous cytotrophoblast, however, p53, procaspase 3, and activated caspases 3 and 8 were re- duced [13,14]. Efflux of the membrane phospholipid phosphatidylserine (PS) is a classical characteristic of apoptosis and also a necessary component of syncyti- otrophobl ast fo rmation [15,16]. Alterations in levels of apoptotic-related proteins may indicate decreased sensitivity to exogenous inducers of apoptosis. Although the syncytiotrophoblast in the hu- man placenta is exposed to circulating maternal immune effector cells, the antigenically foreign fetal-placental unit appears normally resistant to immune rejection. In- creased resistance to apoptosis may be one of several *Corresponding author. OPEN ACCESS  B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 806 complementary survival mechanisms that protect the syncytiotrophoblast. Experiments to test whether the syncytiotrophoblast is relatively apoptosis-resistant have been equivocal. In vitro treatment of mononuclear vil- lous cytotrophoblast and syncytiotrophoblast with stauro- sporine preferentially induced TUNEL-positive apoptotic nuclei in mononuclear cytotrophoblast, suggesting in- creased syncytiotrophoblast resistance to apoptosis [17]. In the same study, however, treatment with a combina- tion of tumor necrosis factor-alpha (TNF-α) and inter- feron-gamma (IFN-γ) induced near equal percentages of apoptotic nuclei in both cell types. Exposure of villous explant cultures to TNF-α or IFN-γ resulted in a greater apoptosis in the syncytium than in villous cytotro- phoblast [18,19]. The concurrent change in apoptotic-related proteins and the process of differentiation and fusion of the vil- lous cytotrophoblast into the syncytiotrophoblast sug- gests a role for apoptotic mechanisms in trophoblast dif- ferentiation. The potential contribution of caspase-8 to trophoblast differentiation has been discussed thoroughly in two recent reviews [20,21]. Activation of caspase-8 was proposed as an indicator of villous cytotrophoblast differentiation and intercellular fusion [22]. However, studies of placental villi and in vitro differentiation of villous cytotrophoblast have not confirmed activation of caspase-8 during differentiation [11,13,14]. To understand the role of apoptotic-related proteins in villous cytotrop hoblast d ifferen tiation , the specific n ature and timing of the changes must be defined. The current data are unclear because of controversial and frequently contradictory results. We hypothesized that increased resistance to apoptotic injury was an outcome of this process, and that syncytialization would be accompanied by a concurrent decrease in levels of pro-apoptotic path- way components and an increase in anti-apoptotic factors. We used BeWo cells, a controllable model of villous cytotrophoblast differentiation and syncytialization. In this study, progressive changes in expression of Bcl-2, Bax, p53, caspases 3 and 8 mRNA and protein were cor- related with formation of syncytia and increased resis- tance to cisplatin-induced apoptosis. Reduction in the level of caspase-3 and -8 protein levels was not accom- panied by a concomitant increase in activated caspase-8, but was the apparent effect of diminished transcription. 2. MATERIALS AND METHODS 2.1. Cell Culture and Fusion Induction BeWo, a continuous human choriocarcinoma cell line (CCL 98; ATCC, Rockville, MD), was maintained in F12K medium (Cellgro, Herndon, CA) supplemented with 10% fetal bovine serum (FBS, Invitrogen, Carlsbad, CA, cat # 10438-034) and a 1× mixture of penicillin G sodium, streptomycin sulfate, and L-glutamine (Invitro- gen) [15]. For assays of intercellular fusion BeWo cells were transferred to MEM (Cellgro) containing the same supplements. The human choriocarcinoma lines JAR and JEG-3 were maintained in RPMI-1640 medium (Cellgro) and MEM medium, respectively, with 10% FBS, penicil- lin G sodium, streptomycin sulfate, and L-glutamine. BeWo cells undergo in vitro differentiation and inter- cellular fusion during treatment with 10 µM of forskolin, an activator of adenylate cyclase [16]. A stock solution of 10 mM of forskolin (Sigma-Aldrich Corp, St. Louis, MO) was prepared in dimethyl sulfoxide (DMSO, Sigma-Aldrich, cat # P4393). Forskolin was added to each choriocarcinoma cell line to a final concentration of 10 µM for up to 72 h with daily replacement with fresh medium and forskolin. An equal volume of DMSO was used as the vehicle control. C2C12 (ATCC, cat # CRL- 1772), a murine myoblast cell line, was cultured in Dul- becco’s modified Eagle’s minimal essential medium with the same supplements. To induce fusion, culture medium was switched to differentiation medium in which 2% normal horse serum (Invitrogen, cat # 16050-114) was substituted for FBS [23]. Cells were cultured in different- tiation medium for three days followed by two days in culture medium. Hematoxylin staining was performed to evaluate the degree of intercellular fusion. 2.2. Quantifying Intercellular Fusion Intercellular fusion of choriocarcinoma cell lines was quantified using anti-E-cadherin staining to visualize in- tercellular membranes, as previously described in detail [16]. Cultures were ev aluated at 24, 48, or 72 h after ad- dition of forskolin. Cells grown on cover slips (104 cells seeded per cover slip) in 24-well plates were fixed with 4% formaldehyde for 20 min followed by permeabiliza- tion with 0.5% Triton X-100 (Sigma-Aldrich, cat # T8787) for 5 min at 4˚C. Cells were blocked in 2% goat serum (Sigma-Aldrich, cat # G9023) and 2% bovine se- rum albumin (BSA; Sigma-Aldrich, cat # A7906) in phos- phate-buffered saline (PBS) for 30 min at room tempera- ture. Anti-E-cadherin (Table 1) was added for 1 h at room temperature, followed by washing and the additio n of a FITC-conjugated secondary antibody for 1 h at room temperature. The cover slips were mounted onto slides using DAPI-containing mounting medium (Vector Labo- ratories, Burlingame, CA). The staining patterns were observed and recorded using a Nikon Eclipse80i micro- scope equipped with blue and green filters. For each cover slip, 10 fields were randomly chosen and photographed at the magnification of 200×. In each field, the total num- bers of DAPI stained nuclei were counted and the per- entage of nuclei in multinucleate cells was determined. c Copyright © 2012 SciRes. OPEN ACCESS 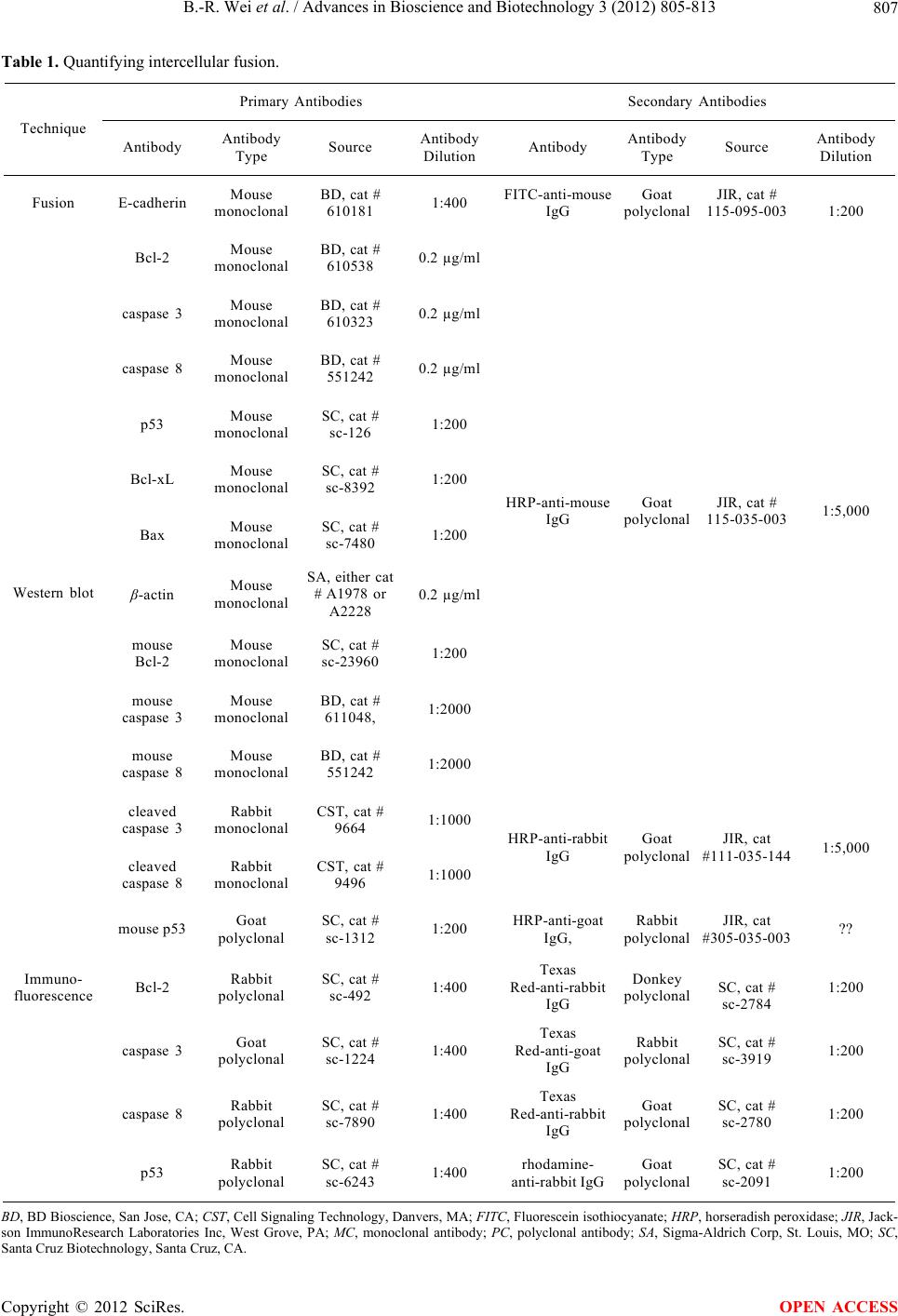 B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 Copyright © 2012 SciRes. 807 OPEN ACCESS Table 1. Quantifying intercellular fusion. Primary Antibodies Secondary Antibodies Technique Antibo d y Antibo d y Type Source Antibo d y Dilution Antibo d y Antibo d y Type Source Antibo d y Dilution Fusion E-cadherin Mouse monoclonal BD, cat # 610181 1:400 FITC -a n ti- mouse IgG Goat polyclonal JIR, cat # 115-095-003 1:200 Bcl-2 Mouse monoclonal BD, cat # 610538 0.2 µg/ml caspase 3 Mouse monoclonal BD, cat # 610323 0.2 µg/ml caspase 8 Mouse monoclonal BD, cat # 551242 0.2 µg/ml p53 Mouse monoclonal SC, cat # sc-126 1:200 Bcl-xL Mouse monoclonal SC, cat # sc-8392 1:200 Bax Mouse monoclonal SC, cat # sc-7480 1:200 β -actin Mouse monoclonal SA, either cat # A19 78 or A2228 0.2 µg/ml mouse Bcl-2 Mouse monoclonal SC, cat # sc-23960 1:200 mouse caspase 3 Mouse monoclonal BD, cat # 611048, 1:2000 mouse caspase 8 Mouse monoclonal BD, cat # 551242 1:2000 HRP- a nti- mouse IgG Goat polyclonal JIR, cat # 115-035-003 1:5,000 cleaved caspase 3 Rabbit monoclonal CST, cat # 9664 1:1000 cleaved caspase 8 Rabbit monoclonal CST, cat # 9496 1:1000 HRP- a nti- rabbit IgG Goat polyclonal JIR, cat #111-035-144 1:5,000 Western blot mouse p53 Goat polyclonal SC, cat # sc-1312 1:200 HRP- a nti- goat IgG, Rabbit polyclonal JIR, cat #305-035-003 ?? Immuno- fluorescence Bcl-2 Rabbit polyclonal SC, cat # sc-492 1:400 Texas Red-anti-rabbit IgG Donkey polyclonal SC, cat # sc-2784 1:200 caspase 3 Goat polyclonal SC, cat # sc-1224 1:400 Texas Red-anti-goat IgG Rabbit polyclonal SC, cat # sc-3919 1:200 caspase 8 Rabbit polyclonal SC, cat # sc-7890 1:400 Texas Red-anti-rabbit IgG Goat polyclonal SC, cat # sc-2780 1:200 p53 Rabbit polyclonal SC, cat # sc-6243 1:400 rhodamine- anti-rabbit IgGGoat polyclonal SC, cat # sc-2091 1:200 BD, BD Bios cience, San Jo se, CA; CST, Cell Signaling Technology, Danvers, MA; FITC, Fluorescein isothiocyanate; HRP, horseradish peroxidase; JIR, Jack- son ImmunoResearch Laboratories Inc, West Grove, PA; MC, monoclonal antibody; PC, polyclonal antibody; SA, Sigma-Aldrich Corp, St. Louis, MO; SC, anta Cruz Biotechnology, Santa Cruz, CA. S  B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 808 2.3. Western Blot Analysis Cells were lysed in buffer (20 mM Tris, pH 7.4, 125 mM NaCl, 20 mM NaF, 0.1% SDS, 10% glycerol, 0.5% so- dium deoxylate, 1% Triton X-100, 1 mM PMSF, 2 µg/ml aprotinin, leupeptin, and pepstatin) and centrifuged [16]. The lysates were separated in a 4% - 20% gradient gel (Invitrogen) and transferred onto Immobolin PVDF membranes (Millipore, Billerica, MA). The membranes were blocked in 3% BSA and incubated with the appro- priate primary antibodies and horseradish peroxidase- conjugated antibodies (see Table 1). Addition al antibod- ies that reacted more strongly against murine Bcl-2, cas- pase 3, caspase 8, and p53 were used in Western blots of lysates from C2C12 cells. Membranes were stripped with Restore Western blot stripping buffer (Pierce, Rockford, IL) and reblotted with additional antibodies. Protein bands were quantified by densitometry of autoradio- grams using a Scion Image Program (Scion Corporation, Frederick, MD). The density of each band was normal- ized against the paired β-actin band, and the ratio of the expression level of each target protein in forskolin- treated cells to vehicle-treated cells was calculated. 2.4. Real-Time PCR Total RNA was extracted from the cells u sing an RNeasy mini kit (Qiagen, Valencia, CA) according to the manu- facturer’s manual. Total cDNA was reverse transcribed using the SuperScript II First-Strand Synthesis system for RT-PCR (Invitrogen). Briefly, 5 µg of total RNA was mixed with 50 ng of random hexamers and dNTP and denatured at 65˚C for 5 min. A cDNA mixture contain- ing RT buffer, 5 mM MgCl2, 10 mM DTT, 2 U RNas- eOUT, and 10 U SuperScript reverse transcriptase was added to the denatured RNA. Reverse transcription was performed at 25˚C for 10 min followed by 50˚C for 50 min. RNA was digested from cDNA by adding RNaseH at 37˚C for 20 min. Real time PCR was carried out in a 25 µl mixture of 1 µl cDNA, 2 µl 20mM primer pair, 12.5 µl CYBR green PCR master mix (Applied Biosys- tems, Foster City, CA), and 9.5 µl of water. Each reac- tion was performed in triplicate. Primers used in this study include: p53 (forward 5’-CCC AGC CAA AGA AGA AAC CA-3”; reverse 5’-GTT CCA AGG CCT CAT TCA GCT-3’), Bax-1 (forward 5’-CAA ACT GG T GCT CAA GGC CC-3’; reverse 5’-GCA CTC CCG CAC AAA GAT G-3’), Bcl-2 (forward 5’-CAG ATG CAC CTG ACG CCC TT-3’; reverse 5’-CCC AGC CTC CGT TAT CCT GGA-3’), Bcl-xL (forward 5’-GGG GTA AAC TGG GGT CGC ATT-3’; reverse 5’-CTT GCG AAG TTG GCG TCC A-3’), caspase 3 (forward 5’-AGA ACT GGA CTG TGG CAT TGA-3’; reverse 5’-GCT TGT CGG CAT ACT GTT TCA G-3’), caspase 8 (forward 5’AGG AGG AGA TGG AAA GGG AAC TT-3’; reverse 5’-ACC TCA ATT CTG ATC TGC TCA CTT CT-3’), and 18S control (forward 5’-CGG CTA CCA CAT CCA AGG AA-3’; reverse 5’-GCT GGA ATT ACC GCG GCT-3’). RT-PCR was performed using a 7500 Real Time PCR System (Applied Biosystems) with a program of 95˚C for 5 min followed by 40 cycles of 95˚C for 15 s and 60˚C for 1 min. The 7500 System SDS software (Applied Biosystems) was used to analyze the data related to the 18S RNA control. The ratio of the mRNA level in forskolin to vehicle treated cells was calculated as 2(CtDMSO-Ctforskolin). 2.5. Quantitative Immunofluorescence BeWo cells were grown and stained with anti-E-cadherin, as described above. After washing, the cells were treated for 1 h at room temperature with a primary antibody against an apoptosis-related protein, followed by wash- ing and the addition of an appropriate fluorescent secon- dary antibody (see Table 1). All digital images were taken using a Nikon Eclipse 80i microscope equipped with blue, green, and red filters. Fluorescent staining was quantified using MetaVue, Meta Imaging Series® 6.1(Universal Imaging Corpora- tion, Downingtown, PA) software. Fused and non-fused cells were identified and delineated in mask images based on nuclear staining with DAPI and intercellular membrane staining with anti-E-cadherin. These deline- ated areas were transferred to a companion red fluoro- chrome image for quantitative image analysis. Each companion area was required to be in exact register with the mask image. Thresholds were set for analysis and the fluorescent density of each region quantified. Quantita- tive data were expressed as the ratios of fluorescent in- tensity between mononuclear and syncytial cells in the same culture and represent the analysis of three inde- pendent experiments. The total number (n) of measure- ments in those experiments ranged from 20 to 39 (mean = 28.9, median = 29). 2.6. Apoptosis Induction BeWo cells were grown on cover slips in 24 well plates and treated with forskolin for 48 h as described above. The culture medium was removed and replaced with medium containing cisplatin (Sigma-Aldrich, cat# P4394) at a concentration of 10 µM for 16 h. Medium was re- moved and cells were washed once with PBS and stained with blue-fluorescent Hoechst 33342 and green-fluores- cent YO-PRO (Invitrogen). Hoechst 33342 brightly stains condensed chromatin in apoptotic cells and dimly stains the chromatin in live cells. YO-PRO can enter apoptotic cells but not the live cells. Diluted Hoechst 33342 (5 µg/ml) and YO-PRO (0.1 µM) were added to the cells and incubated for 30 min on ice. Cells were Copyright © 2012 SciRes. OPEN ACCESS  B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 809 washed and cover slips were mounted onto slides using VECTASHIELD mounting medium (Vector Labs). The staining patterns were recorded immediately using a Nikon Eclipse80i microscope equipped with blue and green filters and a Cool SNAP Photometrics camera. 2.7. Statistical Analysis All quantitative data were expressed as mean ± standard deviation and analyzed using one way analysis of vari- ance/least significant difference (Tukey). 3. RESULTS The BeWo model is a highly reproducible model of vil- lous cytotrophoblast differentiation and syncytialization. Forskolin induced progressive intercellular fusion; the number of nuclei in multinucleate cells were 20.8% ± 15.9% at 24 h (10.7 % ± 2.6% in the vehicle-treated con- trol; P = NS), 65.2% ± 5.2% at 48 h (10.9% ± 2.6% in control, P < 0.01; P < 0.01 compared with 24 h forsko- lin-treated cells), and 80.2% ± 4.1% at 72 h (8.9% ± 1.3% in control, P < 0.01; P < 0.01 compared with 48 h forskolin treated cells). These data are in very close agreement with our previous observations [16] and those of other investigators [24-27]. As we have cautioned in the past, maximum rates of intercellular fusion are not observed using media F-12, but require the use of media F-12K or MEM [28]. Bcl-2 expression was increased during forskolin- driven differentiation of BeWo compared with the vehi- cle-treated controls (Figure 1(a)). Increased transcription preceded elevations in protein levels (Figure 1(b)); mRNA levels were increased over controls by 48 h (7.7 fold increase, P < 0.01) and remained at a steady level through 72 h. A resultant significant increase in protein levels occurred by 72 h (6.9 fold, P < 0.01). Expression of Bcl-xL (B-cell lymphoma-extra large), an antiapop- totic protein in the Bcl-2 protein family, was not affected by forskolin treatment of BeWo cells (data not shown). Expression of Bax, a pro-apoptotic protein, was sup- pressed during differentiation of BeWo cells (Figure 1(a)). Transcription of Bax was significantly decreased by 24 h (P < 0.01) and remained suppressed throughout all time points (Figure 1(c)). The level of protein was significantly decreased (P < 0.01) by 72 h of forskolin- induced differentiation. Thu s the estimated ratio of Bcl-2 to Bax was progressively increasing throughout the dif- ferentiation process; 2.2 at 24 h, 5.0 at 48 h, and 10.0 at 72 h. Treatment with forskolin also suppressed transcription of p53 mRNA (Figure 1(d)) at all time points: 39% at 24 h (P < 0.01), 56% at 48 h (P < 0.01), and 75% at 72 h (P < 0.01) (Figure 1(c)). Levels of p53 protein were also reduced significantly (Figure 1(d)) by 39% at 24 h (P < 0.01), 52% at 48 h (P < 0.01), and 62% at 72 h (P < 0.01). Expression of the pro-forms of the effector caspase 3 (procaspase 3) and initiator caspase 8 (procaspase 8) were diminished during differentiation of BeWo cells (Figure 1(a)). The level of procaspase 3 mRNA was significantly decreased by 24 h of treatment (52% de- crease, P < 0.01), without any significant further de- crease thereafter (Figure 1(e)). Protein was also signifi- cantly decreased by 24 h (29% decrease, P < 0.05) and diminished further by 72 h (64% decrease, P < 0.05 compared to 24 h). Altered expression of procaspase 8 was delayed in comparison to procaspase 3; mRNA was reduced by 42% at 48 h (P < 0.01 compared to control) with no significant further reduction thereafter, and pro- tein was progressively reduced over the 72 h time span; 21% reduction at 48 h (P < 0.01 versus control, P < 0.05 versus 24 h) and 46 % reduction at 72 h (P < 0.01 versus control, P < 0.01 versus 24 h) (Figure 1(f)). We also measured levels of active caspases 3 and 8 to determine whether loss of procaspase proteins resulted from active- tion (Figure 1(g)). No active caspase 3 bands were ob- served in forskolin-treated BeWo cells. Although a very small amount of activated caspase 8 was observed in BeWo treated with DMSO, the lev el did not significantly fluctuate during treatment with forskolin. The active fragments of both were readily observed after treating BeWo cells for 3 h with staurosporine, a known inducer of apoptosis (Figure 1(g)) [16] . To analyze the relationship between changes in apop- tosis-related protein expression and intercellular fusion, we determined the distribution of protein between mononuclear and multinuclear cells by quantitative im- munofluorescence (Figures 2(a) and (b)). Although we performed multiple time points with and without forsko- lin treatment (0, 24, 48, and 72 h), only the 72 h samples are presented. Bcl-2 was the only protein in our study that increased during differentiation. Microscopically, the fluorescent signal for Bcl-2 appeared more intense in syncytial cells than in mononuclear cells, which was confirmed by quantification (Figure 2(c)). Increased expression of Bcl-2 was related to syncytialization, whether spontane- ous (DMSO control) or forskolin-induced; fluorescence in DMSO treated cells was more intense (P < 0.01) in fused cells (90.4 ± 12.3) than mononuclear cells (77.6 ± 12.0). The same relationship was observed in cells treated with forskolin: mononuclear cells, 75.2 ± 9.6; fused cells, 97.5 ± 16.0 (P < 0.01). Treatment with for- skolin appeared to augment the fluorescent intensity in populations of fused cells (P = 0.05), but not mononu- clear cells (P = NS). Increased expression of Bcl-2 pro- tein in syncytial cells was confirmed by immunoperoxi- dase labeling of human placental villi, in which intense Copyright © 2012 SciRes. OPEN ACCESS 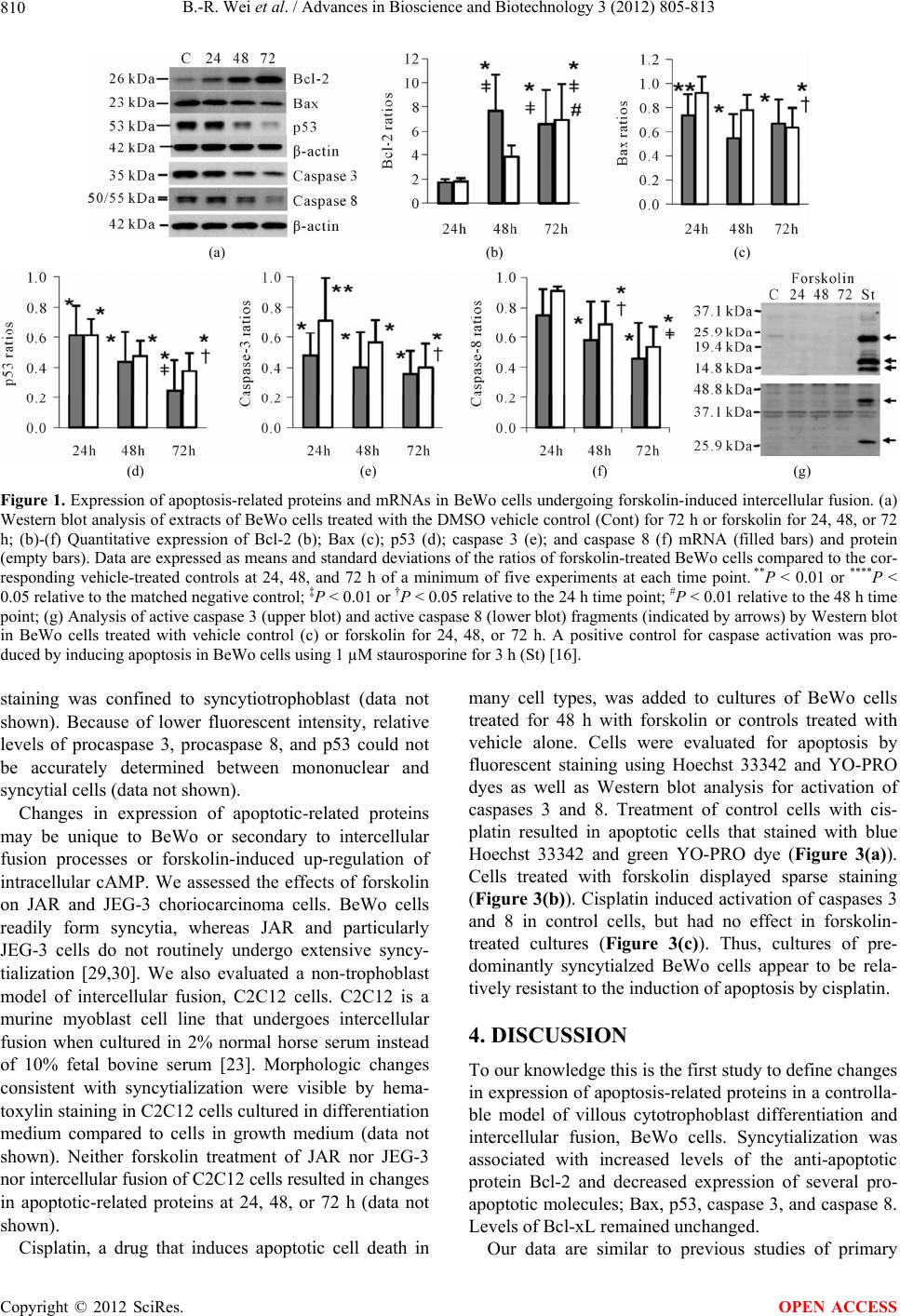 B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 Copyright © 2012 SciRes. 810 (a) (b) (c) (d) (e) (f) (g) Figure 1. Expression of apoptosis-related proteins and mRNAs in BeWo cells undergoing forskolin-induced intercellular fusion. (a) Western blot analysis of extracts of BeWo cells treated with the DMSO vehicle control (Cont) for 72 h or forskolin for 24, 48, or 72 h; (b)-(f) Quantitative expression of Bcl-2 (b); Bax (c); p53 (d); caspase 3 (e); and caspase 8 (f) mRNA (filled bars) and protein (empty bars). Data are expressed as means and standard deviations of the ratios of forskolin-treated BeWo cells compared to the cor- responding vehicle-treated controls at 24, 48, and 72 h of a minimum of five experiments at each time point. **P < 0.01 or ****P < 0.05 relative to the matched negative control; ‡P < 0.01 or †P < 0.05 relative to the 24 h time point; #P < 0.01 relative to the 48 h time point; (g) Analysis of active caspase 3 (upper blot) and active caspase 8 (lower blot) fragments (indicated by arrows) by Western blot in BeWo cells treated with vehicle control (c) or forskolin for 24, 48, or 72 h. A positive control for caspase activation was pro- duced by inducing apoptosis in BeWo cells using 1 µM staurosporine for 3 h (St) [16]. staining was confined to syncytiotrophoblast (data not shown). Because of lower fluorescent intensity, relative levels of procaspase 3, procaspase 8, and p53 could not be accurately determined between mononuclear and syncytial cells (data not shown). Changes in expression of apoptotic-related proteins may be unique to BeWo or secondary to intercellular fusion processes or forskolin-induced up-regulation of intracellular cAMP. We assessed the effects of forskolin on JAR and JEG-3 choriocarcinoma cells. BeWo cells readily form syncytia, whereas JAR and particularly JEG-3 cells do not routinely undergo extensive syncy- tialization [29,30]. We also evaluated a non-trophoblast model of intercellular fusion, C2C12 cells. C2C12 is a murine myoblast cell line that undergoes intercellular fusion when cultured in 2% normal horse serum instead of 10% fetal bovine serum [23]. Morphologic changes consistent with syncytialization were visible by hema- toxylin staining in C2C12 cells cultured in differentiation medium compared to cells in growth medium (data not shown). Neither forskolin treatment of JAR nor JEG-3 nor intercellula r fusion of C2C1 2 cells result ed in ch an g es in apoptotic-related proteins at 24, 48, or 72 h (data not shown). Cisplatin, a drug that induces apoptotic cell death in many cell types, was added to cultures of BeWo cells treated for 48 h with forskolin or controls treated with vehicle alone. Cells were evaluated for apoptosis by fluorescent staining using Hoechst 33342 and YO-PRO dyes as well as Western blot analysis for activation of caspases 3 and 8. Treatment of control cells with cis- platin resulted in apoptotic cells that stained with blue Hoechst 33342 and green YO-PRO dye (Figure 3(a)). Cells treated with forskolin displayed sparse staining (Figure 3(b)). Cisplatin induced activation of caspases 3 and 8 in control cells, but had no effect in forskolin- treated cultures (Figure 3(c)). Thus, cultures of pre- dominantly syncytialzed BeWo cells appear to be rela- tively resistant to the induction of apoptosis by cisplatin. 4. DISCUSSION To our knowledge this is the first study to define changes in expression of apoptosis-related proteins in a controlla- ble model of villous cytotrophoblast differentiation and intercellular fusion, BeWo cells. Syncytialization was associated with increased levels of the anti-apoptotic protein Bcl-2 and decreased expression of several pro- apoptotic molecules; Bax, p53, casp ase 3, and caspase 8. Levels of Bcl-xL remained unchanged. Our data are similar to previous studies of primary OPEN ACCESS 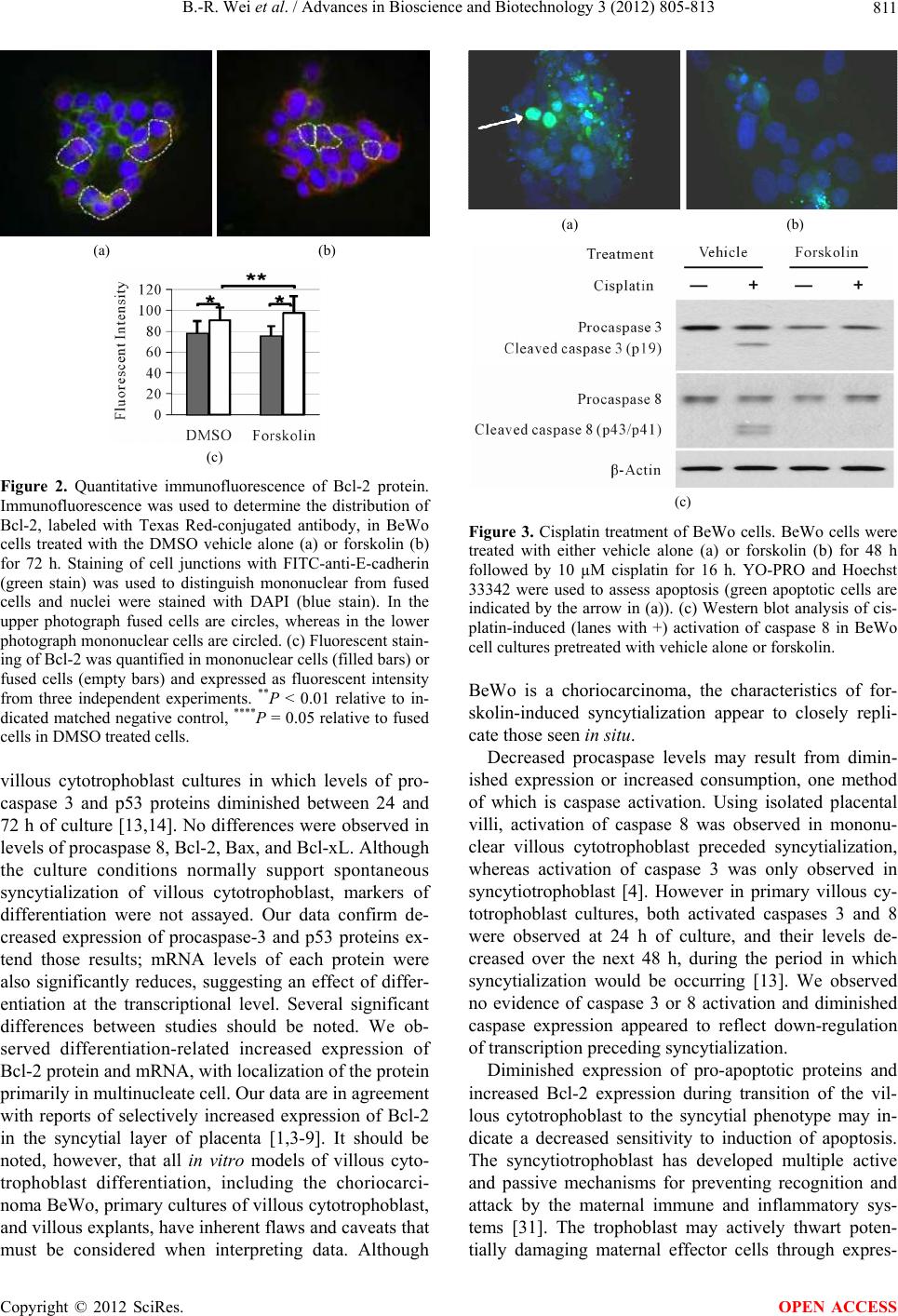 B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 811 (a) (b) (c) Figure 2. Quantitative immunofluorescence of Bcl-2 protein. Immunofluorescence was used to determine the distribution of Bcl-2, labeled with Texas Red-conjugated antibody, in BeWo cells treated with the DMSO vehicle alone (a) or forskolin (b) for 72 h. Staining of cell junctions with FITC-anti-E-cadherin (green stain) was used to distinguish mononuclear from fused cells and nuclei were stained with DAPI (blue stain). In the upper photograph fused cells are circles, whereas in the lower photograph mononuclear cells are circled. (c) Fluorescent stain- ing of Bcl-2 was quantified in mononuclear cells (filled bars) or fused cells (empty bars) and expressed as fluorescent intensity from three independent experiments. **P < 0.01 relative to in- dicat ed matched n egative con trol, ****P = 0.05 relative to fused cells in DMSO treated cells. villous cytotrophoblast cultures in which levels of pro- caspase 3 and p53 proteins diminished between 24 and 72 h of culture [13,14]. No differences were observed in levels of procaspase 8, Bcl-2, Bax, and Bcl-xL. Although the culture conditions normally support spontaneous syncytialization of villous cytotrophoblast, markers of differentiation were not assayed. Our data confirm de- creased expression of procaspase-3 and p53 proteins ex- tend those results; mRNA levels of each protein were also significantly reduces, suggesting an effect of differ- entiation at the transcriptional level. Several significant differences between studies should be noted. We ob- served differentiation-related increased expression of Bcl-2 protein and mRNA, with localization of the protein primarily in multinu cleate cell. Our data are in agreement with reports of selectively increased expression of Bcl-2 in the syncytial layer of placenta [1,3-9]. It should be noted, however, that all in vitro models of villous cyto- trophoblast differentiation, including the choriocarci- noma BeWo, primary cultures of villous cytotrop hoblast, and villous explants, have inherent flaws and caveats that must be considered when interpreting data. Although (a) (b) (c) Figure 3. Cisplatin treatment of BeWo cells. BeWo cells were treated with either vehicle alone (a) or forskolin (b) for 48 h followed by 10 µM cisplatin for 16 h. YO-PRO and Hoechst 33342 were used to assess apoptosis (green apoptotic cells are indicated by the arrow in (a)). (c) Western blot analysis of cis- platin-induced (lanes with +) activation of caspase 8 in BeWo cell cultures pretreated with vehicle alone or forskolin. BeWo is a choriocarcinoma, the characteristics of for- skolin-induced syncytialization appear to closely repli- cate those seen in situ. Decreased procaspase levels may result from dimin- ished expression or increased consumption, one method of which is caspase activation. Using isolated placental villi, activation of caspase 8 was observed in mononu- clear villous cytotrophoblast preceded syncytialization, whereas activation of caspase 3 was only observed in syncytiotrophoblast [4]. However in primary villous cy- totrophoblast cultures, both activated caspases 3 and 8 were observed at 24 h of culture, and their levels de- creased over the next 48 h, during the period in which syncytialization would be occurring [13]. We observed no evidence of caspase 3 or 8 activation and diminished caspase expression appeared to reflect down-regulation of transcription preceding syncytialization. Diminished expression of pro-apoptotic proteins and increased Bcl-2 expression during transition of the vil- lous cytotrophoblast to the syncytial phenotype may in- dicate a decreased sensitivity to induction of apoptosis. The syncytiotrophoblast has developed multiple active and passive mechanisms for preventing recognition and attack by the maternal immune and inflammatory sys- tems [31]. The trophoblast may actively thwart poten- tially damaging maternal effector cells through expres- Copyright © 2012 SciRes. OPEN ACCESS  B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 812 sion of Fas ligand (FasL) on the cell surface and secre- tion of solub le apop tosis-inducing FasL into th e maternal circulation [32]. The syncytiotrophoblast also expresses high levels of four different receptors for TNF-related apoptosis-inducing ligand (TRAIL) [33]. Macrophages are susceptible to apoptosis induced by TRAIL, thus over-expression of TRAIL may be an active mechanism that is important for maintaining the immune privilege status of the placenta. Members of the Bcl-2 family are divided into two sub groups according to their roles in apoptosis; anti-apop- totic proteins (e.g. Bcl-2 and Bcl-xL) and pro-apoptotic proteins (e.g., Bax, Bad [Bclantagonist of cell death], Bid). The anti-apoptotic activity of Bcl-2 is often de- pendent on its relative concentration to Bax, so that an increasing Bcl-2/Bax ratio is indicative of resistance to apoptosis [34,35]. In other cell models over- expression of Bcl-2 inhibited Bax-induced activation of caspase 3 and rescued cells from apoptosis [36]. Syncytialized BeWo cells expressed increased levels of Bcl-2 and re- duced protein level of Bax resulting in an increased Bcl-2/Bax ratio. Indeed, syncytialized BeWo cells were more resistant to cisplatin-induced apoptosis than were mononuclear BeWo cells. Changing levels of Bcl-2 and Bax protein appeared to reflect altered transcription. Transcription of Bcl-2 and Bax are regulated by p53, which generally suppresses the expression of anti-apoptotic proteins and stimulates ex- pression of pro-apoptotic molecules, such as Bax [37]. Decreased expression of p53 during BeWo cell sy- ncytialization precedes significant changes in Bcl-2 ex- pression, which may indicate a causal interrelationship. Increased resistance to apoptosis appears to specifi- cally relate to trophob last syncytialization, rather than be a general phenomenon of intercellular fusion. Under normal physiologic conditions very few cells undergo intercellular fusion; one o f which is the sk eletal myoblast that fuses to form multinuclear myotubes. Fusion of a myoblast model, C2C12 cells, was not accompanied by alterations in expression of apoptosis-related proteins. Induced resistance to apoptosis is more critical for the syncytiotrophoblast, which is derived from immunologi- cally foreign fetal tissue and a more likely target for ma- ternal immune cells. 5. ACKNOWLEDGEMENTS The authors thank Liping Luo for her skillful technical assistance in portions of this study. This work was supported in part by grants RO1- HD043566 and R21-HD052803 from the National Institutes of Health to NSR. REFERENCES [1] Straszewski-Chavez, S.L., Abrahams, V.M. and Mor, G. (2005) The role of apoptosis in the regulation of tro- phoblast survival and differentiation during pregnancy. Endocrine Reviews, 26, 877-897. doi:10.1210/er.2005-0003 [2] Heazell, A.E.P. and Crocker, I.P. (2008) Live and let die—Regulation of villous trophoblast apoptosis in nor- mal and abnormal pregnancies. Placenta, 29, 772-783. doi:10.1016/j.placenta.2008.07.003 [3] Huppertz, B., Frank, H.G., Kingdom, J.C., Reister, F. and Kaufmann, P. (1998) Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochemistry and Cell Biology, 110, 495-508. doi:10.1007/s004180050311 [4] Huppertz, B., Frank, H.G., Reister, F., Kingdom, J., Korr, H. and Kaufmann, P. (1999) Apoptosis cascade pro- gresses during turnover of human trophoblast: Analysis of villous cytotrophoblast and syncytial fragments in vitro. Laboratory Investigation, 79, 1687-1702. [5] Ratts, V.S., Tao, X.J., Webster, C.B., et al. (2000) Ex- pression of BCL-2, BAX and BAK in the trophoblast layer of the term human placenta: A unique model of apoptosis within a syncytium. Placenta, 21, 361-366. doi:10.1053/plac.1999.0486 [6] Huppertz, B., Tews, D.S. and Kaufmann, P. (2001) Apoptosis and syncytial fusion in human placental tro- phoblast and skeletal muscle. International Review of Cytology, 205, 215-253. doi:10.1016/S0074-7696(01)05005-7 [7] Danihel, L., Gomolcak, P., Korbel, M., et al. (2002) Ex- pression of proliferation and apoptotic markers in human placenta during pregnancy. Acta Histochemica, 104, 335- 338. doi:10.1078/0065-1281-00683 [8] Ishihara, N., Matsuo, H., Murakoshi, H., Laoag-Fernan- dez, J.B., Samoto, T. and Maruo, T. (2002) Increased apoptosis in the syncytiotrophoblast in human term pla- centas complicated by either preeclampsia or intrauterine growth retardation. American Journal of Obstetrics and Gynecology, 186, 158-166. doi:10.1067/mob.2002.119176 [9] Ishihara, N., Matsuo, H., Murakoshi, H., Laoag-Fernan- dez, J., Samoto, T. and Maruo, T. (2000) Changes in pro- liferative potential, apoptosis and Bcl-2 protein expres- sion in cytotrophoblasts and syncytiotrophoblast in hu- man placenta over the course of pregnancy. Endocrine Journal, 47, 317-327. doi:10.1507/endocrj.47.317 [10] Ka, H. and Hunt, J.S. (2003) Temporal and spatial pat- terns of expression of inhibitors of apoptosis in human placentas. American Journal of Pathology, 163, 413-422. doi:10.1016/S0002-9440(10)63671-1 [11] Black, S., Kadyrov, M., Kaufmann, P., Ugele, B., Emans, N. and Huppertz, B. (2004) Syncytial fusion of human trophoblast depends on caspase 8. Cell Death and Dif- ferentiation, 11, 90-98. doi:10.1038/sj.cdd.4401307 [12] Huppertz, B., Frank, H.G. and Kaufmann, P. (1999) The apoptosis cascade-morphological and immunohisto-che- mical methods for its visualization. Anatomy and Embry- ology, 200, 1-18. doi:10.1007/s004290050254 [13] Yusuf, K., Smith, S.D., Sadovsky, Y. and Nelson, D.M. Copyright © 2012 SciRes. OPEN ACCESS  B.-R. Wei et al. / Advances in Bioscience and Biotechnology 3 (2012) 805-813 Copyright © 2012 SciRes. 813 OPEN ACCESS (2002) Trophoblast differentiation modulates the activity of caspases in primary cultures of term human tro- phoblasts. Pediatric Research, 52, 411-415. doi:10.1203/00006450-200209000-00018 [14] Hu, C., Smith, S.D., Pang, L., Sadovsky, Y. and Nelson, D.M. (2006) Enhanced basal apoptosis in cultured term human cytotrophoblasts is associated with a higher ex- pression and physical interaction of p53 and Bak. Pla- centa, 27, 978-983. doi:10.1016/j.placenta.2005.11.004 [15] Rote, N.S., Chang, J., Katsuragawa, H., Ng, A.K., Lyden, T.W. and Mori, T. (1995) Expression of phosphatidyl- serine-dependent antigens on the surface of differentiat- ing BeWo human choriocarcinoma cells. American Jour- nal of Reproductive Immunology, 33, 114-121. [16] Das, M., Xu, B. , L in, L. , Chak rabarti, S., Shiva swamy , V. and Rote, N.S. (2004) Phosphatidylserine efflux and in- tercellular fusion in a BeWo model of human villous cy- totrophoblast. Placenta, 25, 396-407. doi:10.1016/j.placenta.2003.11.004 [17] Crocker, I.P., Barratt, S., Kaur, M. and Baker, P.N. (2001) The in vitro characterization of induced apoptosis in pla- cental cytotrophoblasts and syncytiotrophoblasts. Pla- centa, 22, 822-830. doi:10.1053/plac.2001.0733 [18] Crocker, I.P., Tansinda, D.M., Jones, C.J.P. and Baker, P.N. (2004) The influence of oxygen and tumor necrosis factor-α on the cellular kinetics of term placental villous explants in culture. Journal of Histochemistry & Cyto- chemistry, 52, 749-757. doi:10.1369/jhc.3A6176.2004 [19] Balkundi, D.R., Ziegler, J.A., Watchko, J.F., Craven, C. and Trucco, M. (2003) Regulation of FasL/Fas in human trophoblasts: Possible implications for chorioamniotis. Biology of Reproduction, 69, 718-724. doi:10.1095/biolreprod.102.013102 [20] Gauster, M. and Huppertz, B. (2010) The paradox of caspase 8 in human trophoblast fusion. Placenta 31, 82-88. doi:10.1016/j.placenta.2009.12.007 [21] Rote, N.S., Wei, B-R., Xu, C. and Luo, L. (2010) Caspase 8 and human villous cytotrophoblast differentiation. Pla- centa, 31, 89-96. doi:10.1016/j.placenta.2009.12.014 [22] Gauster, M., Siwetz, M. and Huppertz, B. (2009) Fusion of villous trophoblast can be visualized by localizing ac- tive caspase 8. Placenta, 30, 547-550. doi:10.1016/j.placenta.2009.03.007 [23] Fernando, P., Kelly, J.F., Balazsi, K., Slack, R.S. and Megeney, L.A. (2002) Caspase 3 activity is required for skeletal muscle differentiation. Proceedings of the Na- tional Academy of Sciences, 99, 11025-11030. doi:10.1073/pnas.162172899 [24] Wice, B., Menton, D., Geuze, H. and Schwartz, A.L. (1990) Modulators of cyclic AMP metabolism induce syncytiotrophoblast formation in vitro. Experimental Cell Research, 186, 306-316. doi:10.1016/0014-4827(90)90310-7 [25] Mi, S., Lee, X., Li, X.-P., et al. (2000) Syncytin is a cap- tive retroviral envelope protein involved in human pla- cental morphogenesis. Nature, 403, 785-789. doi:10.1038/35001608 [26] Hu, R., Jin, H., Zhou, S., Yang, P. and Li, X. (2007) Pro- teomic analysis of hypoxia-induced responses in the syncytialization of human placental cell line BeWo. Pla- centa, 28, 399-407. doi:10.1016/j.placenta.2006.07.005 [27] Rashid-Doubell, F., Tannetta, D., Redman, C.W.G., Sar- gent, I.L., Boyd, C.A.R. and Linton, E.A. (2007) Caveo- lin-1 and lipid rafts in confluent BeWo trophoblasts: Evi- dence for Rock-1 association with caveolin-1. Placenta, 28, 139-151. doi:10.1016/j.placenta.2005.12.005 [28] Rote, N.S. (2005) Intercellular fusion of BeWo. Placenta, 26, 686. doi:10.1016/j.placenta.2004.09.003 [29] Borges, M., Bose, P., Frank, H.G., Kaufmann, P. and Pot- gens, A.J. (2003) A two-colour fluorescence assay for the measurement of syncytial fusion between trophoblast- derived cell lines. Placenta, 24, 959-964. doi:10.1016/S0143-4004(03)00173-5 [30] Al-Nasiry, S., Spitz, B., Hanssens, M., Luyten, C. and Pijnenborg, R. (2006) Differential effects of inducers of syncytialization and apoptosis on BeWo and JEG-3 choriocarcinoma cells. Human Reproduction, 21, 193- 201. doi:10.1093/humrep/dei272 [31] Jerzak, M. and Bischof, P. (2002) Apoptosis in the first trimester human placenta: the role in maintaining immune privilege at the maternal-foetal interface and in the tro- phoblast remodelling. European Journal of Obstetrics & Gynecology and Reproductive Biology, 100, 138-142. doi:10.1016/S0301-2115(01)00431-6 [32] Abrahams, V.M., Straszewski-Chavez, S.L., Guller, S. and Mor, G. (2004) First trimester trophoblast cells se- crete Fas ligand which induces immune cell apoptosis. Molecular Human Reproduction, 10, 55-63. doi:10.1093/molehr/gah006 [33] Phillips, T.A., Ni, J., Pan, G., et al. (1999) TRAIL (Apo-2L) and TRAIL receptors in human placentas: Im- plications for immune privilege. Journal of Immunology, 162, 6053-6059. [34] Stoetzer, O.J., Nüssler, V., Darsow, M., et al. (1996) As- sociation of bcl-2, bax, bcl-xL and interleukin-1 beta- converting enzyme expression with initial response to chemotherapy in acute myeloid leukemia. Leukemia, 10, S18-S22.
|