Paper Menu >>
Journal Menu >>
 Vol.2, No.7, 707-712 (2010) Natural Science http://dx.doi.org/10.4236/ns.2010.27087 Copyright © 2010 SciRes. OPEN ACCESS Synthesis and characterization of an amphiphilic chitosan bearing octyl and methoxy polyethylene glycol groups Guanghua Liu, Jianqun Gan, Aimin Chen, Qian Liu, Xusheng Zhao* Guangzhou Institute of Chemistry, Chinese Academy of Sciences, Guangzhou, China; *Corresponding Author: zhao7503@yahoo.com Received 14 April 2010; revised 18 May 2010; accepted 25 May 2010. ABSTRACT An amphiphilic N-octyl-O-methoxy poly (ethyl- ene glycol) chitosan was successfully prepared by grafting successively octyl groups onto amino groups at chitosan’s C-2 position as hy- drophobic moieties and methoxy polyethylene glycol (MPEG) groups onto hydroxyl groups at C-6, C-3 as hydrophilic ones. A certain amount of -NH2 was retained in the structure of chitosan derivatives through protection by phthalic an- hydride. The chemical structures and degree of N-and O-substitution of chitosan derivatives were characterized by FTIR, 1H NMR, GPC and elemental analysis, respectively. The amphiphi- lic property for convenient self-assembly and the preserved -NH2 groups for progressive chemical cross-linking make the resultant N- ocyl-O-MPEG chitosan soluble in water and po- tentially applicable in preparing stable chitosan hollow microspheres, a demanding drug-carrier in medical and pharmaceutical sciences. Keywords: Amphiphilic Groups; Chitosan Derivatives; Protection; Hollow Microsperes 1. INTRODUCTION In recent years, micelles and hollow microspheres pre- pared from self-assemblies of amphiphilic polymers have attracted great attention due to a variety of applica- tions in DNA, antigens, delivery carriers for drugs and protection proteins or/and enzymes, especially for con- trolled or sustained drug-delivery systems [1]. The unique core-shell architecture is composed of hydropho- bic segment, which acted as internal core, and hydro- philic segment which acted as surrounding corona in aqueous medium [2]. The hydrophobic core provides a loading space for water-insoluble drugs and stabilizes them, whereas the hydrophilic shell protects encapsu- lated drugs [3]. Additionally, the modification of hydro- philic shell affects pharmacokinetic behavior, such as prolonged circulation time, target release [4] and con- trolled drug release by using stimuli-sensitive copoly- mers [5]. In comparison with common polymers, chitosan, the second most abundant natural biopolymer only to cellu- lose [6] and known as its excellent property in non-tox- icity, biodegradability, good biocompatibility, etc. [7], would exhibit special advantages in drug delivery sys- tem. The active hydroxyl and amino groups within chi- tosan could be easily modified to endow it with new or improved properties, and at the same time, keeping its original physiochemical and biochemical properties [6]. As a result, various amphiphilic chitosan derivatives and hollow microspheres prepared from them have been ex- tensively published [6]. For examples, highly N-methy- lated modified chitosan possessing hydrophobic -N (CH3)2, -NH(CH3) and hydrophilic -N+(CH3)3 groups was reported by Sieval et al. which is soluble in water at a broad pH value [8]. Through emulsion-crosslinking, Peng et al. prepared hollow microspehres from N-me- thylated chitosan with diameters range from 2-5 um [9]. Liu et al. synthesized an amphiphilic carboxymethyl- hexanoyl chitosan [10] which formed hollow nanocap- sules with 20-200 nm in diameter [11]. As a demanding drug-carrier in medical and pharmaceutical sciences, stable chitosan hollow microsphere is probably one of the most valuable candidates. Presently, the hydrophobic groups of amphiphilic chi- tosan derivatives ever reported generally contain long alkyl [12,13], long acyl [10] and aryl [14], while the hydrophilic ones include carboxymethyl [10], sulfate [12], phosphate [15,16], N-trimethyl [17] and polyeth- ylene glycol [14,18]. To prepare amphiphilic chitosan  G. H. Liu et al. / Natural Science 2 (2010) 707-712 Copyright © 2010 SciRes. OPEN ACCESS 708 hollow microspheres as drug carriers, the substituents, especially the hydrophilic groups, should be non-toxic and biocompatible. It’s impossible to form stable core- hell structure through nothing but self-assembly of am- phiphilic chitosan. Amino groups within the backbone of chitosan should be crosslinked chemically to form stable and hard shell thereafter self-assembly process. That’s to say, adequate primary amino groups (-NH2) existed in amphiphilic chitosan are necessary for preparing hollow microspheres. To the best of our knowledge, most re- ports ever reported about amphiphilic chitosan deriva- tives did not pay much attention to this problem. In this work, we report a novel amphiphilic N-octyl O-methoxy polyethylene glycol (MPEG) chitosan pos- sessing adequate primary amino groups. PEG chain has been proved to be non-toxic, very flexible and highly hydrated, soluble in water and majority organic solvents, and is the most widely used biocompatible polymer for chemical and biological applications [19]. Through the effective protection of -NH2 groups within the backbone of chitosan during the modification process, the chitosan derivatives synthesized in this study preserve a certain amount of -NH2 groups for latter chemical crosslinking to prepare eventually sizecontrollable hollow micro- spheres. 2. EXPERIMENTAL 2.1. Materials Chitosan (Mr = 1 × 105, degree of deacetylation (DD) = 95%) was provided by Zhejiang Aoxing Biochemical Co. Ltd (China). Octylaldehyde was obtained from Guang- zhou Damo Chemical Co. Ltd (China). Methoxy poly- ethylene glycol (MPEG, Mr = 1900) was purchased from Alfa Aesar. Methyl iodide was ordered from Shanghai Jingchun Chemical Reagent Factory (China). Dimethyl- formamide (DMF) was distilled under reduced pressure and stored over molecular sieves. Dialysis tube was achieved from SPECTRUM (Canada) with a molecular weight cut-off (MWCO) of 6000~8000. Ag2O was fresh- ly prepared from AgNO3 and KOH in our laboratory. All other materials used in this study were of analytical grade and without further purification. 2.2. Synthetic Procedures 2.2.1. Synthesis of N-Octyl Chitosan (NOC) N-octyl chitosan was prepared as reported previously (Scheme 1). Briefly, chitosan (5.0 g, 31 mmol) was sus- pended in 60 ml methanol with active stirring at room temperature before the addition of octaldehyde (5.0 ml, 31 mmol). Then, KBH4 (2.5 g) dissolved in 10 ml water was added dropwise to the mixture after 24 h. The reac- tion solution was neutralized with hydrochloric acid and O HO OH O n NH 22)KBH 4 /H 2 O , rt 1)CH 3 (CH 2 ) 6 CHO O HO OH O NH(CH 2 ) 7 CH 3 O HO OH O n-x NH 2 x phthalic anhydride DMF, 130℃, 6h O HO OH O NH(CH 2 ) 7 CH 3 O HO OH O n-x N x O O 1) MPEGI, Ag 2 O 60℃,16h 2) NH 2 NH 2 .H 2 O H 2 O 90℃, 15h O RO OR O NH(CH 2 ) 7 CH 3 O RO OR O n-x NH 2 x R= (CH 2 CH 2 O) m CH 2 CH 2 OCH 3 or H Scheme 1. Synthetic procedure of N-octyl-O-MPEG chitosan. the product was precipitated with methanol after a fur- ther 24 h continuous stirring. The precipitate was filtered, washed repeatedly with methanol and water and then dried under vacuum at 60℃ overnight to give 7.0 g N-octyl chitosan. Degree of N-substitution of octyl was 54.3% from element analysis. 2.2.2. Synthesis of N-Octyl-N-Phthaloyl Chitosan (NONPC) The synthesis of N-octyl-N-phthaloyl chitosan (NONPC) is described briefly as follows. A mixture of N-octyl chi- tosan (3.0 g, 13.8 mmol) and phthalic anhydride (PHA) (2.8 g, 3 mol equiv to amino) in 30 ml DMF was heated to 130℃ under nitrogen with magnetic stirring. After 6 h, the brown precipitate obtained by pouring the solution into ice-water was collected by filtration, extracted with ethanol in Soxhlet’s apparatus for 48 h, and dried under vacuum at 50℃ to give 3.6 g NONPC products. 2.2.3. Synthesis of Methoxy Polyethylene Glycol Lodide (MPEGI) Methoxy polyethylene glycol iodide (MPEGI) was syn- thesized according to the reports by Gorochovceva and Makuska [19]. Briefly, MPEG-1900 (19 g, 10 mmol), triphenylphosphite (8.0 ml, 30 mmol) and methyl iodide (4.3 ml, 30 mmol) were mixed and stirred in a three- neck flask at 120℃ for 6 h. Then, the reaction mixture was cooled, dissolved in toluene, precipitated in 500 ml diethyl ether, and then filtered, washed several times with diethyl ether and dried in air to give pale yellow product. The yield of the product was 92%. 2.2.4. Synthesis of N-Octyl-O-MPEG Chitosan (NOOMC) The synthetic procedure of N-octyl-O-MPEG chitosan (NOOMC) is carried out by using NONPC, MPEGI and Ag2O with different grams or molar ratios as the starting materials (Table 1). Firstly, NONPC, MPEGI and Ag2O were mixed and heated at 60℃ for 16 h with magnetic stirring. Then, hydrazine monohydrate (80%, 40 ml or 20 ml) and distilled water (80 ml or 40 ml) were poured  G. H. Liu et al. / Natural Science 2 (2010) 707-712 Copyright © 2010 SciRes. OPEN ACCESS 709 709 into the mixture and the temperature was raised to 90℃ and maintained for 15 h before the mixture was filtered to remove solid impurity. The filtrate solution was dia- lyzed against water for 72 h and then concentrated before the solid residue was dried in vacuum at 50℃ to give final products of N-octyl-O-MPEG chitosan (NOO- MC). Depending on different amount of hydrazine monohydrate and distilled water used during the synthe- sis, the products of NOOMC with two different degree of substitutions (DS) of MPEG were labeled as NOOMC (I) and NOOMC (II), respectively. 2.3. Characterization 1H NMR spectra were recorded at 400MHz using a Bruker DRX-400 spectrometer. CF3COOD and D2O were used as the solvents for chitosan and its derivatives, respectively. Elemental analysis was determined using an Elementar Vario EL III analyzer. FTIR spectra were obtained from Fourier transformation infrared spec- trometer (Analect RFX-65A) in KBr discs. Molecular weight of the amphiphilic chitosan was determined using Waters 515-410 gel permeation chromatography (GPC). 3. RESULTS AND DISCUSSION 3.1. Synthesis of the Chitosan Derivatives Generally, amphiphilic chitosan could be prepared through introducing the hydrophilic moiety first and the hydrophobic one later [12,13], or in the reverse order [11,20]. The latter procedure was adopted in this inves- tigation. As shown in Scheme 1, the amphiphilic chito- san derivatives were synthesized through the following three steps. At the first step, hydrophobic octyl groups were introduced by Schiff reaction prior to MPEG. Oc- tylaldhyde residue was found very easy to be removed with heterogeneous reaction, and rather difficult to be excluded when Schiff reaction is carried out at the last step. Meanwhile, if MPEG was introduced onto hy- droxyl firstly, the molecule weight of the resultant O-MPEG chitosan would be much higher than chitosan, and the mole ratio of octylaldehyde and O-MPEG chito- san is difficult to calculate and control in Schiff reaction. Table 1. Amounts of reactants in synthesizing NOOMC with different degree of substitutions. No. NONPC/g MPEGI/g Ag2O/g m(NONPC): m(MPEGI): m(Ag 2O) * product (I) 1.5 10.2 1.2 1:1:1 NOOMC(Ⅰ) (II) 0.8 10.8 1.3 1:2:2 NOOMC()Ⅱ NONPC, N-octyl-N-phthaloyl chitosan; MPEGI, methoxy polyethyl- ene glycol iodide; NOOMC, N-octyl-O-MPEG chitosan. * m repre- sents mole ratio. It has been estimated that amino groups at C-2 show higher nucleophilic activity than hydroxyl groups (either C-3 or C-6) in the main backbone of chitosan [21]. Thereby, to avoid the grafting of MPEG onto amino groups which retained in N-octyl chitosan, it is necessary to protect amino groups in advance by the use of phthalic anhydride. This protection method was reported earlier in 1991 by Nishimura et al. [22], which has been applied to various region-selective modifications of chitosan [21,23]. A key point for this pathway is the fact that chitosan is insoluble in several organic solvents, such as DMF, while N-phthaloyl derivative of chitosan is soluble [18,22]. N- octyl chitosan, just like chitosan, has also a bad solubility in organic solvent. It was found in our experiment that N-octyl-N-phthaloyl chitosan is soluble in DMF, which is of great significance for further reaction with MPEG io- dide. Accordingly, N-phthaloyl group is the prerequisite for both protection of active amino in chitosan and solubi- lization of the product in DMF. As mentioned above, a certain amount of primary amino is indispensable for the preparing of amphiphilic chitosan hollow microspheres. It is identified from Scheme 1 that the content of primary amino of the final product is determined by the first step reaction. Hence, primary amino cannot be modified and replaced com- pletely with octyl groups. In our study, to assure an in- complete substitution reaction on amino groups, we adopted an appropriate molar ratio of chitosan to octy- laldehyde in Schiff reaction. It is expected that we could get amphiphilic chitosan possessing different amount of active amino groups. That is, a certain extent of primary amino content could be preserved through controlling the molar ratio of the starting materials. Further investi- gations are currently under way in our laboratory. Compared with Schiff reaction at the first step, the amino groups retained in N-octyl chitosan should be modi- fied completely with phthalimide groups to avoid their participation in further reaction. Threefold excess of phthalic anhydride was used in our experiment to protect amino as complete as possible. The course of the etherifi- cation of N-octyl-N-phthaloyl with MPEGI was similar to that described by Natalijia Gorochovceva et al. [18] where Ag2O is a suitable and effective catalyzer for etherification. The reaction between chitosan hydroxyl groups and MPEGI is irreversible and proceeds till the consumption of active functional groups. In our study, the degree of sub- stitution (DS) of MPEG is represented from the molar ratio of MPEGI to N-octyl-N-phthaloyl chitosan. N-octyl- O-MPEG chitosan with different DS of MPEG is illus- trated from the molar ratio of the above two reagents. 3.2. Characterization of Chitosan Derivatives The degree of substitution (DS) of chitosan derivatives was calculated by comparing C and N molar ratio ob- 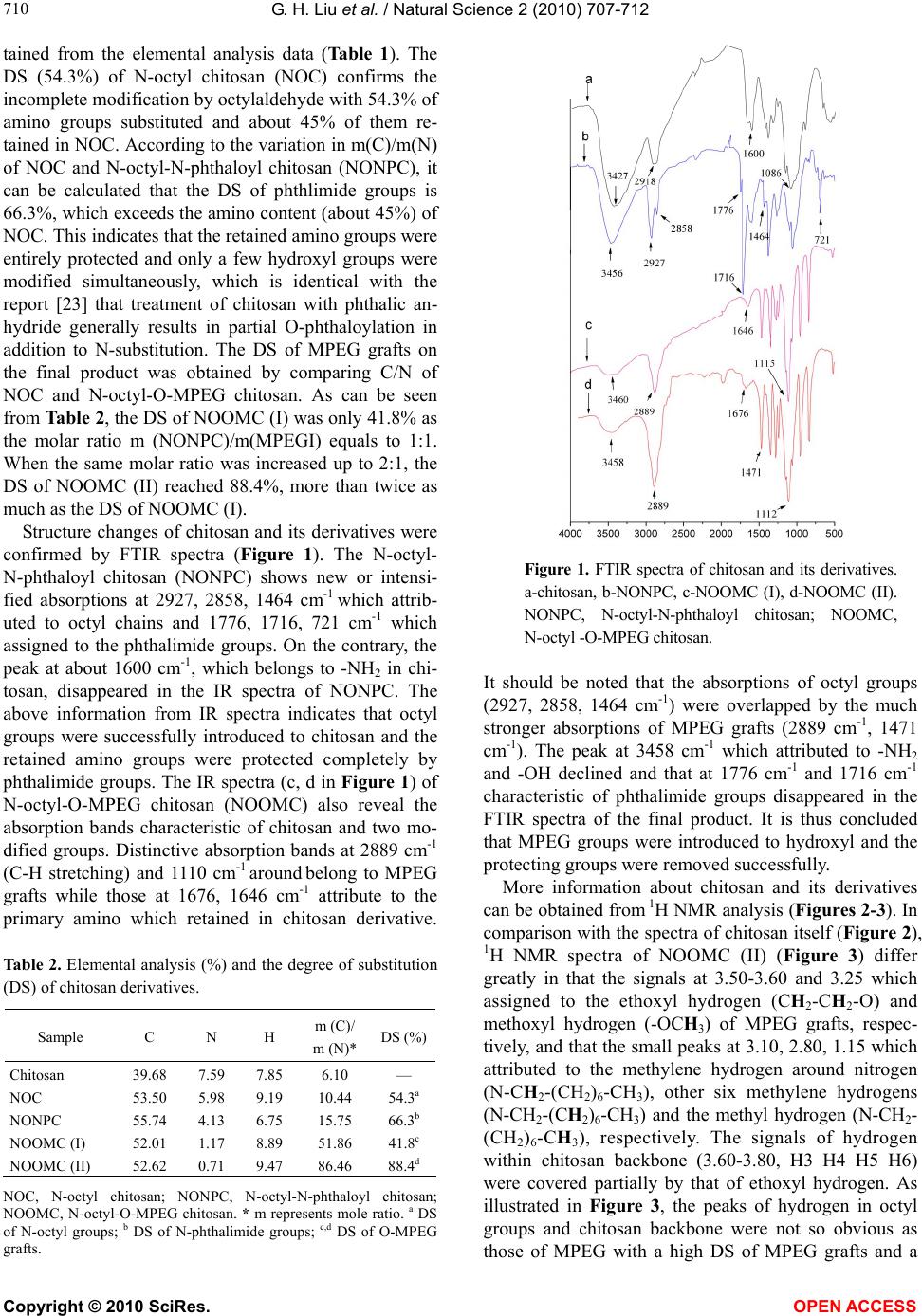 G. H. Liu et al. / Natural Science 2 (2010) 707-712 Copyright © 2010 SciRes. OPEN ACCESS 710 tained from the elemental analysis data (Table 1). The DS (54.3%) of N-octyl chitosan (NOC) confirms the incomplete modification by octylaldehyde with 54.3% of amino groups substituted and about 45% of them re- tained in NOC. According to the variation in m(C)/m(N) of NOC and N-octyl-N-phthaloyl chitosan (NONPC), it can be calculated that the DS of phthlimide groups is 66.3%, which exceeds the amino content (about 45%) of NOC. This indicates that the retained amino groups were entirely protected and only a few hydroxyl groups were modified simultaneously, which is identical with the report [23] that treatment of chitosan with phthalic an- hydride generally results in partial O-phthaloylation in addition to N-substitution. The DS of MPEG grafts on the final product was obtained by comparing C/N of NOC and N-octyl-O-MPEG chitosan. As can be seen from Table 2, the DS of NOOMC (I) was only 41.8% as the molar ratio m (NONPC)/m(MPEGI) equals to 1:1. When the same molar ratio was increased up to 2:1, the DS of NOOMC (II) reached 88.4%, more than twice as much as the DS of NOOMC (I). Structure changes of chitosan and its derivatives were confirmed by FTIR spectra (Figure 1). The N-octyl- N-phthaloyl chitosan (NONPC) shows new or intensi- fied absorptions at 2927, 2858, 1464 cm-1 which attrib- uted to octyl chains and 1776, 1716, 721 cm-1 which assigned to the phthalimide groups. On the contrary, the peak at about 1600 cm-1, which belongs to -NH2 in chi- tosan, disappeared in the IR spectra of NONPC. The above information from IR spectra indicates that octyl groups were successfully introduced to chitosan and the retained amino groups were protected completely by phthalimide groups. The IR spectra (c, d in Figure 1) of N-octyl-O-MPEG chitosan (NOOMC) also reveal the absorption bands characteristic of chitosan and two mo- dified groups. Distinctive absorption bands at 2889 cm-1 (C-H stretching) and 1110 cm-1 around belong to MPEG grafts while those at 1676, 1646 cm-1 attribute to the primary amino which retained in chitosan derivative. Table 2. Elemental analysis (%) and the degree of substitution (DS) of chitosan derivatives. Sample C N H m (C)/ m (N)* DS (%) Chitosan 39.68 7.59 7.85 6.10 — NOC 53.50 5.98 9.19 10.44 54.3a NONPC 55.74 4.13 6.75 15.75 66.3b NOOMC (I) 52.01 1.17 8.89 51.86 41.8c NOOMC (II) 52.62 0.71 9.47 86.46 88.4d NOC, N-octyl chitosan; NONPC, N-octyl-N-phthaloyl chitosan; NOOMC, N-octyl-O-MPEG chitosan. * m represents mole ratio. a DS of N-octyl groups; b DS of N-phthalimide groups; c,d DS of O-MPEG grafts. Figure 1. FTIR spectra of chitosan and its derivatives. a-chitosan, b-NONPC, c-NOOMC (I), d-NOOMC (II). NONPC, N-octyl-N-phthaloyl chitosan; NOOMC, N-octyl -O-MPEG chitosan. It should be noted that the absorptions of octyl groups (2927, 2858, 1464 cm-1) were overlapped by the much stronger absorptions of MPEG grafts (2889 cm-1, 1471 cm-1). The peak at 3458 cm-1 which attributed to -NH2 and -OH declined and that at 1776 cm-1 and 1716 cm-1 characteristic of phthalimide groups disappeared in the FTIR spectra of the final product. It is thus concluded that MPEG groups were introduced to hydroxyl and the protecting groups were removed successfully. More information about chitosan and its derivatives can be obtained from 1H NMR analysis (Figures 2-3). In comparison with the spectra of chitosan itself (Figure 2), 1H NMR spectra of NOOMC (II) (Figure 3) differ greatly in that the signals at 3.50-3.60 and 3.25 which assigned to the ethoxyl hydrogen (CH2-CH2-O) and methoxyl hydrogen (-OCH3) of MPEG grafts, respec- tively, and that the small peaks at 3.10, 2.80, 1.15 which attributed to the methylene hydrogen around nitrogen (N-CH2-(CH2)6-CH3), other six methylene hydrogens (N-CH2-(CH2)6-CH3) and the methyl hydrogen (N-CH2- (CH2)6-CH3), respectively. The signals of hydrogen within chitosan backbone (3.60-3.80, H3 H4 H5 H6) were covered partially by that of ethoxyl hydrogen. As illustrated in Figure 3, the peaks of hydrogen in octyl groups and chitosan backbone were not so obvious as those of MPEG with a high DS of MPEG grafts and a  G. H. Liu et al. / Natural Science 2 (2010) 707-712 Copyright © 2010 SciRes. OPEN ACCESS 711 711 O CH 2 OH NH 2 O OH 1 2 3 4 5 6 H1 H3 H4 H5 H6 H2 COCH 3 n 7 6 5 4 3 2 1 Figure 2. 1H NMR spectra of chitosan. Figure 3. 1H NMR spectra of N-octyl-O-MPEG chitosan (NOOMCⅡ). much large number of hydrogen it contained. Molecular weight of the amphiphilic chitosan (NOOMC (I)) was measured by gel permeation chro- matography (GPC). The results showed that Mw of NOOMC (I) is near 1 × 105 and Mz is about 1.6 × 105, which is a little bit smaller than that calculated by Element Analysis (2 × 105). It might be caused by the existence of trace amount of unreacted MPEGI after dialysis or cleavage of chitosan chains during modifi- cations. 4. CONCLUSIONS N-octyl-O-MPEG chitosan (NOOMC), was synthesized for the first time with fair production yield and high de- gree of substitution of amphiphilic groups. The degree of substitution of octyl groups reached 54.3% while that of MPEG grafts amounted to 41.8%-88.4% depending on the variation of N-octyl-N-phthaloyl chitosan to meth- oxy polyethylene glycol iodide molar ratio. With the introduction of the amphiphilic groups, the chitosan de- rivatives were endowed with good solubility in water and potential self-assembly properties. Significantly, the effective protection of amino groups was successfully adopted to retain a certain amount of -NH2 in the deriva- tives, which is indispensable for preparing hollow mi- crospheres. 5. ACKNOWLEDGEMENTS The authors express their gratitude to National Natural Science Foun- dations of China and Natural Science Foundation of Guangdong Prov- ince, China, for financial support. REFERENCES [1] Peng, X.H., Zhang, L.N. and Kennedy, J.F. (2006) Release behavior of microspheres from cross-linked N- methylated chitosan encapsulated ofloxacin. Carbohy- drate Polymers, 65(3), 288-295. [2] Ye, Y.Q., Yang, F.L., Hu, F.Q., Du, Y.Z., Yuan, H. and Yu, H.Y. (2008) Core-modified chitosan-based polyme- ric micelles for controlled release of doxorubicin. Inter- national Journal of Pharmaceutics, 352(1-2), 294-301. [3] van Nostrum, C.F. (2004) Polymeric micelles to deliver photosensitizers for photodynamic therapy. Advanced Drug Delivery Reviews, 56(1), 9-16. [4] Maeda, H., Wu, J., Sawa, T., Matsumura, Y. and Hori, K. (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. Journal of Controlled Release, 65(1-2), 271-284. [5] Liu, X.M., Wang, L.S., Wang, L., Huang, J.C. and He, C.B. (2004) The effect of salt and pH on the phase- transition behaviors of temperature-sensitive copolymers based on N-isopropylacrylamide. Biomaterials, 25(25), 5659-5666. [6] Mourya, V.K. and Inamdar, N.N. (2008) Chitosan-mo- difications and applications: Opportunities galore. Rea- ctive & Functional Polymers, 68(6), 1013-1051. [7] Chandy, T. and Sharma, C.P. (1990) Chitosan - as a Biomaterial. Biomaterials Artificial Cells and Artificial Organs, 18(1), 1-24. [8] Sieval, A.B., Thanou, M., Kotze, A.F., Verhoef, J.E., Brussee, J. and Junginger, H.E. (1998) Preparation and NMR characterization of highly substituted N-trimethyl chitosan chloride. Carbohydrate Polymers, 36(2-3), 157- 165. [9] Peng, X.H. and Zhang, L. (2005) Surface fabrication of hollow microspheres from N-methylated chitosan cross- linked with gultaraldehyde. Langmuir, 21(3), 1091- 1095. [10] Liu, T.Y., Chen, S.Y., Lin, Y.L. and Liu, D.M. (2006) Synthesis and characterization of amphiphatic carboxy- methyl-hexanoyl chitosan hydrogel: Water-retention abi- lity and drug encapsulation. Langmuir, 22(23), 9740- 9745. [11] Liu, K.H., Chen, S.Y., Liu, D.M. and Liu, T.Y. (2008) Self-assembled hollow nanocapsule from amphiphatic carboxymethyl-hexanoyl chitosan as drug carrier. Ma- cromolecules, 41(17), 6511-6516. [12] Zhang, C., Ping, Q.N., Zhang, H.J. and Jian, S. (2003) Preparation of N-alkyl-O-sulfate chitosan derivatives and micellar solubilization of taxol. Carbohydrate Polymers, 54(2), 137-141. [13] Zhang, C., Ding, Y., Yu, L.L. and Ping, Q.N. (2007) Po- lymeric micelle systems of hydroxycamptothecin based on amphiphilic N-alkyl-N-trimethyl chitosan derivatives. Co- lloids and Surfaces B-Biointerfaces, 55(2), 192-199. [14] Yoksan, R. and Chirachanchai, S. (2008) Amphiphilic  G. H. Liu et al. / Natural Science 2 (2010) 707-712 Copyright © 2010 SciRes. OPEN ACCESS 712 chitosan nanosphere: Studies on formation, toxicity, and guest molecule incorporation. Bioorganic & Medicinal Chemistry, 16(5), 2687-2696. [15] Jayakumar, R., Nagahama, H., Furuike, T. and Tamura, H. (2008) Synthesis of phosphorylated chitosan by novel method and its characterization. International Journal of Biological Macromolecules, 42(4), 335-339. [16] Jayakumar, R., Egawa, T., Furuike, T., Nair, S.V. and Tamura, H. (2009) Synthesis, characterization, and ther- mal properties of phosphorylated chitin for biomedical applications. Polymer Engineering and Science, 49(5), 844-849. [17] Zhang, C., Ding, Y., Ping, Q.E. and Yu, L.L. (2006) Novel chitosan-derived nanomaterials and their micelle- forming properties. Journal of Agricultural and Food Chemistry, 54(22), 8409-8416. [18] Gorochovceva, N. and Makuska, R. (2004) Synthesis and study of water-soluble chitosan-O-poly (ethylene glycol) graft copolymers. European Polymer Journal, 40(4), 685-691. [19] Kulbokaite, R., Ciuta, G., Netopilik, M. and Makuska, R. (2009) N-PEG’ylation of chitosan via “click chemistry” reactions. Reactive & Functional Polymers, 69(10), 771-778. [20] Ngawhirunpat, T., Wonglertnirant, N., Opanasopit, P., Ruktanonchai, U., Yoksan, R., Wasanasuk, K. and Chi- rachanchai, S. (2009) Incorporation methods for cholic acid chitosan-g-mPEG self-assembly micellar sys- tem containing camptothecin. Colloids and Surfaces B-Bio- interfaces, 74(1), 253-259. [21] Makuska, R. and Gorochovceva, N. (2006) Regiose- lective grafting of poly(ethylene glycol) onto chitosan through C-6 position of glucosamine units. Carbohydrate Polymers, 64(2), 319-327. [22] Nishimura, S.I., Kohgo, O., Kurita, K. and Kuzuhara, H. (1991) Chemospecific manipulations of a rigid polysa- ccharide-syntheses of novel chitosan derivatives with excellent solubility in common organic-solvents by regi- oselective chemical modifications. Macromolecules, 24(17), 4745-4748. [23] Hu, Y.Q., Jiang, H.L., Xu, C.N., Wang, Y.J. and Zhu, K.J. (2005) Preparation and charac terization of poly (ethylene glycol)-g-chitosan with water- and organo- solubility. Carbohydrate Polymers, 61(4), 472-479. |

