 American Journal of Plant Sciences, 2012, 3, 1252-1259 http://dx.doi.org/10.4236/ajps.2012.39151 Published Online September 2012 (http://www.SciRP.org/journal/ajps) Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses* Julie Knudson1, Paul Meiman1#, Cynthia Brown2, George Beck2, Mark Paschke1, Edward Redente1 1Department of Forest and Rangeland Stewardship, Colorado State University, Colorado, USA; 2Department of Bioagricultural Sciences and Pest Management, Colorado State University, Colorado, USA. Email: #Paul.Meiman@ColoState.edu Received July 24th, 2012; revised August 22nd, 2012; accepted August 31st, 2012 ABSTRACT Chemical restrictions, ecological concerns, liability issues, and public sentiment present challenges to land managers attempting to control highly invasive plants like Canada thistle (Cirsium arvense [L.] Scop.). Although herbicide appli- cation can be an effective control strategy, increasing limitations force managers of sensitive environments (e.g., na- tional parks, wildlife refuges, protected water-bodies or waterways) to search for effective control alternatives. A greenhouse study was conducted to test the effectiveness of clipping (to simulate field mowing) and grass seeding as alternatives for Canada thistle control. Two native North American grasses (western wheatgrass [Pascopyrum smithii {Rydb.} A. Löve] and streambank wheatgrass [Elymus lanceolatus {Scribn. & J. G. Sm.}Gould ssp. lanceolatus]) and one sterile hybrid cross between common wheat (Triticum aestivum L.) and tall wheatgrass (Thinopyrum ponticum [Podp.] Z. W. Liu & R. C. Wang) called Regreen™ were used. The effects of clipping and grass seeding on Canada this- tle growth, and the effect of Canada thistle on grass growth, were evaluated using 14 unique treatments applied to pot- ted Canada thistle and grass plants. Clipping inhibited Canada thistle growth (by 60%), while grass seeding had no ef- fect. Presence of Canada thistle inhibited grass growth for all seeding treatments except when Regreen™ and western wheatgrass were seeded together with Canada thistle. Planting multiple species for restoration of Canada thistle-infested sites may be important (Regreen™ + western wheatgrass treatment), and cutting Canada thistle may be useful for reduc- ing its growth in restored areas. Keywords: Invasive Species; Mowing; Competition; Revegetation 1. Introduction Canada thistle (Cirsium arvense L.), a highly invasive non-native perennial plant, continues to challenge land managers throughout the United States. While noxious weed legislation was first enacted against Canada thistle in Vermont in 1795 [1], truly successful Canada thistle control strategies for sensitive settings are still lacking. Herbicides such as picloram have proven quite successful in less regulated upland areas [2-5] but many areas in- fested with Canada thistle require an alternative control because of issues related to aquatic habitat, herbicide restrictions, threats to endangered species, liability, or human/animal-sensitivity [6-9]. These issues often occur on wildlife refuges, natural areas, state and national parks, near protected water-bodies or waterways, or on other private or public conservation lands, and create special challenges for successful weed management. The re- cently released herbicide aminopyralid may be a viable alternative for managers facing herbicide restrictions be- cause of its lower toxicity to non-target organisms [10], but additional research on this new product is necessary, and it is of little use to managers working in areas where herbicide application is prohibited. Control methods for Canada thistle commonly used or tested in sensitive settings include hand pulling, digging, biological control, grazing, cultivation, fertilizer addition, aquatic labeled herbicides, “natural product” herbicides (e.g., acetic acid), mowing, or revegetation. Unfortu- nately, when used alone, most of these methods have de- monstrated limited utility for significant, long-term Can- ada thistle reduction [11-15]. Two control measures that may be useful in combination, however, are mowing and revegetation. Mowing is currently recommended for Canada thistle control on many weed management information websites [16-18] and is popular with landowners enrolled in the Conservation Reserve Program in the United States [19]. While the utility of strategically-timed mowing to pre- *Mention of trade names and companies is solely to inform the reader and does not constitute endorsement by Colorado State University nor does it imply criticism of similar products or companies not mentioned. #Corresponding author. Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1253 vent seed production is well documented [20-22], re- search on mowing as a standalone strategy for Canada thistle management has shown mixed results. One study found mowing conducted twice per year for 2 years tem- porarily decreased Canada thistle shoot density for the following 2 years, but by the third year shoot density in the mowed treatments was not different from the un- mowed control [23]. Another study found that mowing three times annually for 2 years reduced Canada thistle density by 85% at a subirrigated site, but did not provide significant control at a dryland site [2]. Others report that on sites that were otherwise similar, a twice per year mowing treatment resulted in a reduction of Canada this- tle density to 35% of its initial density one year later on a high density site (30 shoots·m−2), but had no effect on a lower density site (16 shoots·m−2) [24]. Mowing an in- fested alfalfa (Medicago sativa L.) field twice annually produced an 86% reduction in Canada thistle density af- ter the first year [25]. This treatment provided complete control after 4 years, demonstrating that desirable com- petitive plant species (e.g. alfalfa) may enhance the suc- cess of mowing. Overall however, these variable results demonstrate the need for further evaluation of mowing as part of a successful control strategy. Revegetation has shown promise as a secondary tool for long-term control of Canada thistle following other control measures, and is becoming increasingly popular as a component of integrated weed management pro- grams. Regardless of the target weed, a lack of compete- tion from desirable plants post-treatment often leads to reestablishment of unwanted weeds [26,27]. Several forbs and grasses have shown promise as effective com- petitors with Canada thistle, including tall fescue (Sche- donorus phoenix [Scop.] Holub), crownvetch (Securigera varia [L.] Lassen), sudangrass (Sorghum bicolor [L.] Moench ssp. drummondii [Nees ex Steud.] de Wet & Harlan), annual ragweed (Ambrosia artemisiifolia L.), common sunflower (Helianthus annuus L.), alfalfa, bien- nial sweetclover (Melilotus sp.), and a mixture of peren- nial ryegrass (Lolium perenne L.) and white clover (Tri- folium repens L.) [1,28-32]. One study reported that the use of competitive grasses for Canada thistle control was as effective as herbicide application over a 3-year period [33]. Unfortunately, most plants tested thus far as com- petitors against Canada thistle are non-native species. While use of non-natives is understandable because they are frequently more aggressive, seeding non-native plants is often strongly discouraged or prohibited in sensitive settings. Clearly, land managers of sensitive settings face a variety of challenges when attempting Canada thistle control. The lack of effective long-term control measures combined with the lack of research specific to their needs translates into limited options for people tasked with re- storing infested lands. The following experiment was conducted to test the effectiveness of clipping (to simulate field mowing) and seeding plant species acceptable for use in sensitive set- tings as control measures for Canada thistle. Specifically, the objectives of this study were to: 1) determine the ef- fects of clipping and grass competition on Canada thistle growth, 2) compare the effectiveness of the different grass seeding treatments for reducing Canada thistle growth, and 3) determine the effect of Canada thistle on the growth of each of the seeded grasses. Two North American native grasses (western wheatgrass [Pascopy- rum smithii {Rydb.} A. Löve] and streambank wheat- grass [Elymus lanceolatus {Scribn. & J.G. Sm.} Gould ssp. lanc eola tus ]) and one sterile commercial hybrid cross between common wheat (Triticum aestivum L.) and tall wheatgrass (Thinopyrum ponticum [Podp.] Z. W. Liu & R. C. Wang) called Regreen™ were chosen for this re- search. We hypothesized that both clipping and grass compe- tition would reduce Canada thistle shoot biomass, and that the effect of the two factors together would be greater than either alone. We also predicted that the grass seeding treatments containing Regreen™ would reduce Canada thistle growth more than the native grasses seeded alone with Canada thistle because of the more aggressive nature of Regreen™. Additionally, we predi- cted that grass growth would be inhibited by the presence of Canada thistle. 2. Materials and Methods A greenhouse study was conducted on potted Canada thistle plants treated with combinations of clipping (used to simulate mowing) and grass seeding. Two response variables were measured: Canada thistle shoot biomass and grass shoot biomass. Canada thistle biomass was analyzed using a two by five factorial design consisting of two levels of Canada thistle clipping (clipped, un- clipped) and five levels of grass seeding (no grass, streambank wheatgrass, western wheatgrass, Regreen™, or western wheatgrass + Regreen™). Grass biomass was analyzed using a three by four factorial design consisting of three levels of Canada thistle (clipped, unclipped, ab- sent) and four levels of grass seeding (streambank wheat- grass, western wheatgrass, Regreen™, or western wheat- grass + Regreen™). Combined, these two analyses invest- tigated a total of 14 treatment combinations applied to Canada thistle or grass plants (6 replicates per treatment) grown in potting soil for 51 weeks in a greenhouse. 2.1. Selection of Grass Species Western wheatgrass and streambank wheatgrass were chosen for their aggressive underground growth habit, early spring germination prior to Canada thistle growth, Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1254 wide geographic and habitat range throughout the west- ern United States, drought and extreme temperature tol- erance, and broad availability [34-36]. These two species have demonstrated prior success for control of other weeds including Russian knapweed (Acroptilon repens [L.] DC.) [37,38], leafy spurge (Euphorbia esula L.) [39], cheatgrass (Bromus tectorum L.), and musk thistle (Car- duus nutans L.) [40,41]. Western wheatgrass has also provided effective control on sites infested with multiple weed species [42]. Most importantly, western wheatgrass has previously demonstrated some success against Can- ada thistle [33]. The hybrid grass Regreen™ was chosen for its ability to establish aggressive root growth more quickly than native grasses, and its sterility increases acceptability for use in sensitive settings. 2.2. Source of Plant Material Western wheatgrass seed (variety “Arriba”) was obtained from Pawnee Buttes Seed Inc. (Greeley, CO, USA). Streambank wheatgrass seed (variety “Sodar”) was ob- tained from Granite Seed (Lehi, UT, USA). Regreen™ seed was obtained from Rainier Seed Company (Daven- port, WA, USA). Canada thistle horizontal roots were collected from a site near Fort Collins, Colorado, USA (lat 40˚33'46"N, long 105˚00'24"W; 1491 m above sea level). Horizontal roots were collected 26 August 2004, placed in sealed plastic bags with soil collected from the same location, and transported to the laboratory in a cooler. The bagged soil was moistened and stored at 6˚C in the dark for 8 weeks to prevent sprouting before use in the experiment. Plant nomenclature follows the USDA PLANTS Database [36]. 2.3. Plant Preparation and Treatment On 23 October 2004, refrigerated Canada thistle hori- zontal root sections were cut into 2.5-cm long pieces (diameter ranging from 0.2 to 0.65 cm) with a minimum of one bud per piece. Pieces were soaked in 2.54 cm of tap water in a refrigerated (6˚C) covered tray in the dark for 28 h, then planted in rows at a depth of approximately 1.3 cm in 25- × 52-cm flat plastic trays filled with 2.5 cm of wetted Scotts MetroMix 350 potting soil (Sun Gro Horticulture, Bellevue, WA, USA). Each tray was tho- roughly watered after planting and kept moist. Germinated horizontal root pieces were transferred to 164 ml conetainers in December 2004. In late January 2005, the plants were transferred to 1.3-L pots and grown for 7 weeks. Surviving plants were then transferred to 3.8-L plastic pots filled with Scotts MetroMix 350 pot- ting soil (1 plant per pot), and grown there for the re- mainder of the experiment. After 10 weeks of postemer- gence growth, 12 grass seeds were added to 48 of the 60 Canada thistle pots (Canada thistle control pots had no seeded grasses). Each pot assigned to a grass seeding treatment received 12 grass seeds (six of each species for the two-species treatment). Western wheatgrass and stream- bank wheatgrass seeds were planted approximately 1.3 cm deep, while Regreen™ seeds were planted approxi- mately 0.6 cm deep. More seeds were planted than needed to assure establishment of a sufficient number of plants. Twenty four grass control pots of the same size and growth medium as the Canada thistle pots were seeded with only grasses as described above. The result was six replicate pots for each treatment combination. Six weeks after initial seeding, adequate grass seed germination was obtained and pots were thinned to four grass plants per pot. Regreen™ + western wheatgrass treatments were thinned to two grass plants per species in each pot. Because of limited project resources, only one grass species combination treatment was evaluated. Western wheatgrass was chosen for this combination with Regreen™ because of its more aggressive rhizome- tous growth habit than streambank wheatgrass [34]. At 17 weeks of Canada thistle growth when plants were blooming, the single clipping treatment was performed, cutting plants with hand shears 9 cm above the soil sur- face to simulate mowing. This clipping height was cho- sen because it is the approximate mowing height used in field studies for mowing Canada thistle [2,43]. Clipped shoot biomass from each Canada thistle plant was placed in individual paper bags, dried at 55˚C to constant mass and weighed to determine Canada thistle shoot biomass. Grasses were not clipped. Experimental plants grew in the greenhouse for an ad- ditional 34 weeks post-clipping at approximately 22˚C ± 5˚C, and were watered as needed and weeded for nonex- perimental species. Plants received natural light supple- mented with 400-W high pressure sodium vapor bulbs (1.5 m above greenhouse benches) to obtain a 16-h pho- toperiod. Pot locations on the greenhouse bench were re-randomized and pots were moved every 6 weeks to minimize effects of potential differences in light or tem- perature. For the final harvest, experimental plants were clipped at the soil surface and separated into grass shoot biomass or Canada thistle shoot biomass for each pot and placed in separate paper bags. It was not possible to ac- curately separate root biomass when multiple species grew in the same pot, thus root biomass was not consi- dered further. Plant material was dried at 55˚C to con- stant mass and weighed to determine shoot biomass for each plant species. 2.4. Statistical Analyses Two dependent variables were analyzed: Canada thistle shoot biomass and grass shoot biomass. Canada thistle biomass was analyzed using a two by five factorial de- Copyright © 2012 SciRes. AJPS 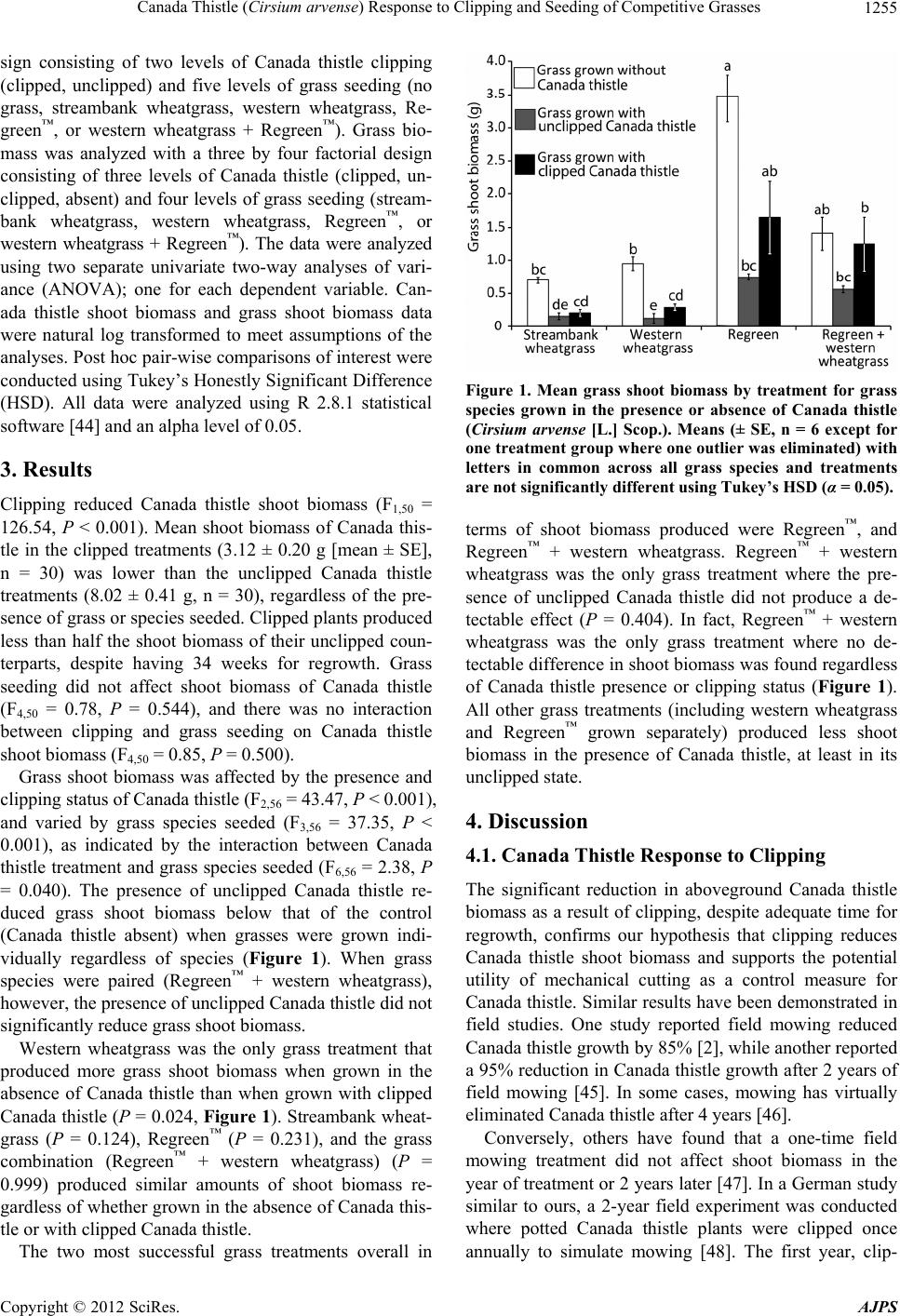 Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1255 sign consisting of two levels of Canada thistle clipping (clipped, unclipped) and five levels of grass seeding (no grass, streambank wheatgrass, western wheatgrass, Re- green™, or western wheatgrass + Regreen™). Grass bio- mass was analyzed with a three by four factorial design consisting of three levels of Canada thistle (clipped, un- clipped, absent) and four levels of grass seeding (stream- bank wheatgrass, western wheatgrass, Regreen™, or western wheatgrass + Regreen™). The data were analyzed using two separate univariate two-way analyses of vari- ance (ANOVA); one for each dependent variable. Can- ada thistle shoot biomass and grass shoot biomass data were natural log transformed to meet assumptions of the analyses. Post hoc pair-wise comparisons of interest were conducted using Tukey’s Honestly Significant Difference (HSD). All data were analyzed using R 2.8.1 statistical software [44] and an alpha level of 0.05. 3. Results Clipping reduced Canada thistle shoot biomass (F1,50 = 126.54, P < 0.001). Mean shoot biomass of Canada this- tle in the clipped treatments (3.12 ± 0.20 g [mean ± SE], n = 30) was lower than the unclipped Canada thistle treatments (8.02 ± 0.41 g, n = 30), regardless of the pre- sence of grass or species seeded. Clipped plants produced less than half the shoot biomass of their unclipped coun- terparts, despite having 34 weeks for regrowth. Grass seeding did not affect shoot biomass of Canada thistle (F4,50 = 0.78, P = 0.544), and there was no interaction between clipping and grass seeding on Canada thistle shoot biomass (F4,50 = 0.85, P = 0.500). Grass shoot biomass was affected by the presence and clipping status of Canada thistle (F2,56 = 43.47, P < 0.001), and varied by grass species seeded (F3,56 = 37.35, P < 0.001), as indicated by the interaction between Canada thistle treatment and grass species seeded (F6,56 = 2.38, P = 0.040). The presence of unclipped Canada thistle re- duced grass shoot biomass below that of the control (Canada thistle absent) when grasses were grown indi- vidually regardless of species (Figure 1). When grass species were paired (Regreen™ + western wheatgrass), however, the presence of unclipped Canada thistle did not significantly reduce grass shoot biomass. Western wheatgrass was the only grass treatment that produced more grass shoot biomass when grown in the absence of Canada thistle than when grown with clipped Canada thistle (P = 0.024, Figure 1). Streambank wheat- grass (P = 0.124), Regreen™ (P = 0.231), and the grass combination (Regreen™ + western wheatgrass) (P = 0.999) produced similar amounts of shoot biomass re- gardless of whether grown in the absence of Canada this- tle or with clipped Canada thistle. The two most successful grass treatments overall in Figure 1. Mean grass shoot biomass by treatment for grass species grown in the presence or absence of Canada thistle (Cirsium arvense [L.] Scop.). Means (± SE, n = 6 except for one treatment group where one outlier was eliminated) with letters in common across all grass species and treatments are not significantly different using Tukey’s HSD (α = 0.05). terms of shoot biomass produced were Regreen™, and Regreen™ + western wheatgrass. Regreen™ + western wheatgrass was the only grass treatment where the pre- sence of unclipped Canada thistle did not produce a de- tectable effect (P = 0.404). In fact, Regreen™ + western wheatgrass was the only grass treatment where no de- tectable difference in shoot biomass was found regardless of Canada thistle presence or clipping status (Figure 1). All other grass treatments (including western wheatgrass and Regreen™ grown separately) produced less shoot biomass in the presence of Canada thistle, at least in its unclipped state. 4. Discussion 4.1. Canada Thistle Response to Clipping The significant reduction in aboveground Canada thistle biomass as a result of clipping, despite adequate time for regrowth, confirms our hypothesis that clipping reduces Canada thistle shoot biomass and supports the potential utility of mechanical cutting as a control measure for Canada thistle. Similar results have been demonstrated in field studies. One study reported field mowing reduced Canada thistle growth by 85% [2], while another reported a 95% reduction in Canada thistle growth after 2 years of field mowing [45]. In some cases, mowing has virtually eliminated Canada thistle after 4 years [46]. Conversely, others have found that a one-time field mowing treatment did not affect shoot biomass in the year of treatment or 2 years later [47]. In a German study similar to ours, a 2-year field experiment was conducted where potted Canada thistle plants were clipped once annually to simulate mowing [48]. The first year, clip- Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1256 ping resulted in an increase in Canada thistle shoot bio- mass compared to the unclipped control. It was only in the second year that the clipped Canada thistle plants responded in a manner similar to our study, where clip- ped plants produced less shoot biomass than unclipped controls. One difference between the German study and ours was the height of clipping. Clipping height was 30 cm in their study [48], leaving a significant amount of photo- synthesizing foliage behind, which likely facilitated first year regrowth. Those authors theorized that their mild clipping treatment mimicked moderate herbivory, where plant growth would be stimulated by removal of top growth. In our study, Canada thistle was clipped at a standard mowing height of 9 cm, leaving little foliage behind. This more substantial removal of plant material likely inhibited regrowth by reducing photosynthetic car- bohydrate production. Removal of plant top-growth through activities such as mowing is believed to weaken Canada thistle plants by inhibiting photosynthetic carbo- hydrate production and transport, while also forcing de- pletion of root carbohydrate reserves to support regrowth after cutting [49]. This may explain the inability of the clipped plants to fully recover after the second clipping in the German study [48]. Another difference between the German study and ours was the timing of clipping. Root carbohydrate reserves of Canada thistle are considered to be lowest at the initiation of flowering [46,50]. In our study, Canada thistle was clipped when 99% of the plants were flowering. Clipping our plants when carbohydrate reserves were low may have enhanced the effectiveness of our clipping treatment. In the German study, plants were clipped in June each year, before flowering [48]. In their study, flowering oc- curred later in the growing season, with only 6% of con- trol heads beginning to flower by August the first year, and 43% of control heads beginning to flower by August the second year [48]. With earlier flowering the second year, root carbohydrate reserves may have been lower at the time of clipping than the first year, perhaps also con- tributing to the decrease in biomass of clipped plants the second year. It may be that height of clipping and timing of clipping relative to flowering play a critical role in the relative success of clipping treatments for control of Canada thistle. 4.2. Canada Thistle Response to Grass Seeding Although the presence of grass plants did not inhibit Canada thistle shoot growth in this study (contrary to the hypothesis), the effect of the grasses on Canada thistle root growth was not assessed. A key requirement for grass species selection in this study was an aggressive underground growth habit. Native grasses such as alkali sacaton (Sporobolus airoides [Torr.] Torr.) have been shown in greenhouse studies to inhibit Canada thistle root growth while demonstrating no effect on Canada thistle shoot biomass [51]. A 1-yr greenhouse experiment may be too short for root competition to result in changes in aboveground Canada thistle biomass. Canada thistle shoot biomass fluctuated significantly for the first 2 years of a field study when grown in the presence of plant competitors, and those authors concluded that plant com- petitors require more than two seasons of growth before they can effectively suppress Canada thistle [28]. Con- versely, others report that seeding the competitive exotic grasses perennial ryegrass (Lolium perenne L.), Italian ryegrass (Lolium perenne L. ssp. multiflorum [Lam.] Husnot), and orchardgrass reduced shoot biomass of pot- ted Canada thistle plants each year of their 2-year study [52]. The grasses for this experiment were also selected for their drought tolerance, a benefit untested under green- house conditions, but potentially important under hot, dry field conditions. For example, western wheatgrass estab- lishes and maintains cover across a range of soil moisture availabilities [53], while Canada thistle growth may be suppressed when soil moisture availability is reduced [54]. Hot, dry years may allow grasses such as western wheatgrass to gain a foothold over Canada thistle under field conditions. 4.3. Canada Thistle Response to Grass Seeding × Clipping Contrary to our hypothesis that the combined effect of clipping and grass competition on Canada thistle biomass would be greater than either factor alone, there was no synergistic effect of clipping and grass seeding in our study, although different results may be expected in field trials. One field study demonstrated that seeding peren- nial grasses and mowing twice annually for 3 years can reduce Canada thistle density by more than 90% [33]. Other testing of grass seeding and field mowing for con- trol of Canada thistle has found that while the compete- tive ability of the grasses was important for controlling Canada thistle in the early stages of the experiment, as the age of the grass stand increased and mowing contin- ued, the effects of mowing became more important than competition between the grass and Canada thistle [22]. 4.4. Grass Response to Canada Thistle Presence and Species of Grass Seeded The negative effect of unclipped Canada thistle on shoot biomass of each of the single-species grass treatments in our study was hypothesized and expected because pre- vious researchers have demonstrated the negative im- pacts Canada thistle can have on the growth of other plants [55-57]. What was surprising in our study was the Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1257 failure of Canada thistle to reduce shoot biomass of the paired grass species (Regreen™ + western wheatgrass), regardless of Canada thistle clipping status. The mecha- nism driving the resilience of the paired grass treatment is unclear, but one likely explanation is the increased func- tional group diversity [58,59] resulting from the pres- ence of an annual and a perennial grass. Another possi- bility, although more difficult to substantiate, is the cu- mulative effect of allelochemicals produced by both Re- green™ and western wheatgrass. Common wheat (one of the hybrid components of Regreen™) is believed to have allelopathic potential against weeds in cropping systems [60], and there is some evidence of phytotoxic allelo- chemical production by western wheatgrass [61,62]. This study also elucidated the potential utility of me- chanical cutting of Canada thistle to enhance grass growth when only a single desirable species is seeded. While shoot biomass growth of all of the single grass species was inhibited by the presence of unclipped Can- ada thistle, clipping Canada thistle resulted in greater grass biomass for each single species treatment equiva- lent to grass biomass in the complete absence of Canada thistle (with the exception of western wheatgrass). Grass growth may benefit from Canada thistle cutting not only because of the weakened state of the Canada thistle plants, but also from the decrease in competitive plant canopy. The lack of benefit to western wheatgrass from Can- ada thistle clipping may be a product of short study dura- tion. Regreen™ establishes and produces growth more quickly than many species considered for revegetation [63]. Streambank wheatgrass also establishes and ma- tures more quickly than western wheatgrass [34]. It may take longer for the benefits of Canada thistle clipping to translate into increased growth of western wheatgrass. While the seeded grasses did not significantly inhibit Canada thistle shoot biomass in this study, it is probable that a field study, performed over a longer duration, would more clearly determine the utility of these grasses for restoration of Canada thistle infested sites. The ag- gressive underground growth of these grasses may not translate to observable aboveground effects on Canada thistle for several seasons (regardless of additional con- trol measures used), and the characteristics of these grasses for which they were initially chosen (such as drought tolerance and early season germination) may translate to further advantages over Canada thistle in a field setting. The results of this experiment demonstrate the poten- tial for both mechanical cutting and grass seeding as ef- fective tools for restoration of Canada thistle infested sites. The one-time clipping event resulted in a decrease in Canada thistle biomass, and has implications for the use of mowing as a field control measure. As revegeta- tion tools, the grasses used in this experiment proved to be tolerant of Canada thistle presence, and the combina- tion of Regreen™ and western wheatgrass demonstrated the ability to grow equally well regardless of Canada thistle presence or cutting status. This is an important finding, as it is generally considered that almost any con- trol measure for Canada thistle requires multiple applica- tions, with complete eradication impossible or at least requiring multiple seasons. Thus any useful revegetation species must be capable of growing in concert with Can- ada thistle until it can be controlled. The success of this combination grass seeding also emphasizes the potential importance of the synergistic effects of using more than one species for restoration of Canada thistle infested sites. 5. Acknowledgements We thank the Colorado Agricultural Experiment Station, (Dr. Lee Sommers, Director) and Colorado State Uni- versity, Fort Collins, CO, USA for funding this experi- ment. We also thank Dr. Phil Westra, Dr. Paul Duffy, and Lonnie Pilkington for their assistance and contributions to this project, as well as Rainier Seed Company and Granite Seed Company for donating grass seed. REFERENCES [1] F. Detmers, “Canada Thistle (Cirsium arvense Tourn.), Field Thistle, Creeping Thistle,” Technical Bulletin 414, University of Ohio Agricultural Experiment Station, Co- lumbus, 1927. [2] K. G. Beck and J. Sebastian, “Combining Mowing and Fall-Applied Herbicides to Control Canada Thistle (Cir- sium arvense),” Weed Technology, Vol. 14, No. 2, 2000, pp. 351-356. doi:10.1614/0890-037X(2000)014[0351:CMAFAH]2.0.C O;2 [3] W. W. Donald, “Retreatment with Fall-Applied Herbicides for Canada Thistle (Cirsium arvense) Control,” Weed Sci- ence, Vol. 41, No. 3, 1993, pp. 434-440. [4] L. E. Foote, D. L. Kill and C. S. Williams, “Canada This- tle Control on Roadsides,” Weed Science, Vol. 18, No. 2, 1970, pp. 307-310. [5] S. C. Peterson and J. V. Parochetti, “Canada Thistle (Cir- sium arvense) Control in Timothy (Phleum pratense) and Red Clover (Trifolium pretense) Sward,” Weed Science, Vol. 26, 1978, pp. 215-220. [6] J. Krueger-Mangold, R. L. Sheley and B. D. Roos, “Maintaining Plant Community Diversity in a Waterfowl Production Area by Controlling Canada Thistle (Cirsium arvense) Using Glyphosate,” Weed Technology, Vol. 16, No. 2, 2002, pp. 457-463. doi:10.1614/0890-037X(2002)016[0457:MPCDIA]2.0.C O;2 [7] R. G. Lym and C. G. Messersmith, “Survey for Picloram in North Dakota Groundwater,” Weed Technology, Vol. 2, No. 2, 1988, pp. 217-222. Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses 1258 [8] K. M. Norgaard, “The Politics of Invasive Weed Man- agement: Gender, Race and Risk Perception in Rural California,” Rural Sociology, Vol. 72, No. 3, 2007, pp. 450-477. doi:10.1526/003601107781799263 [9] US Environmental Protection Agency, “Endangered Spe- cies Protection Bulletin,” 2011. http://www.epa.gov/oppfead1/endanger/bulletins.htm [10] US Environmental Protection Agency, “Aminopyralid Pesticide Fact Sheet,” Office of Pesticide Programs, Ar- lington, 2005. [11] J. Jacobs, J. Sciegienka and F. Menalled, “Ecology and Management of Canada Thistle [Cirsium arvense (L.) Scop.],” Invasive Species Technical Note MT-5, Natural Resource Conservation Service, US Department of Agri- culture, Bozeman, 2006. [12] J. Radhakrishnan, J. R. Teasdale and C. B. Coffman, “Agricultural Applications of Vinegar,” Proceedings of the 57th Annual Northeastern Weed Science Society Meeting, Baltimore, 6-9 January 2003, p. 63. [13] P. E. Reece and R. G. Wilson, “Effect of Canada Thistle (Cirsium arvense) and Musk Thistle (Carduus nutans) Control on Grass Herbage,” Weed Science, Vol. 31, No. 4, 1983, pp. 488-492. [14] C. C. Reed, D. L. Larson and J. L. Larson, “Canada This- tle Biological Control Agents on Two South Dakota Wildlife Refuges,” Natural Areas Journal, Vol. 26, No. 1, 2006, pp. 47-52. doi:10.3375/0885-8608(2006)26[47:CTBCAO]2.0.CO;2 [15] R. L. Zimdahl and G. Foster, “Canada Thistle (Cirsium arvense) Control with Disking and Herbicides,” Weed Technology, Vol. 7, No. 1, 1993, pp. 146-149. [16] Plant Conservation Alliance, “Canada Thistle,” 2011. http://www.nps.gov/plants/ALIEN/fact/ciar1.htm [17] US Army Corps of Engineers, “Canada Thistle (Cirsium arvense) Mechanical Control,” 2009. http://el.erdc.usace.army.mil/emrrp/emris/emrishelp4b/mo wing_canada_thistle.htm [18] Wisconsin Department of Natural Resources, “Canada Thistle,” 2004. http://dnr.wi.gov/invasives/fact/canada_thistle.htm [19] C. Holen, H. Person, B. Holder, R. Severson and M. Halstvedt, “Canada Thistle Control on Conservation Re- serve Program Land,” In: H. Kandel, R. Severson and P. Glogoza, Eds., Report: 2006 On-Farm Cropping Trials Northwest and West Central Minnesota, Crookston, 2007, pp. 35-36. [20] L. A. Derscheid and R. E. Schultz, “Achene Development of Canada Thistle and Perennial Sowthistle,” Weeds, Vol. 8, No. 1, 1960, pp. 55-62. doi:10.2307/4040507 [21] R. J. Moore, “The Biology of Canadian Weeds. 13. Cir- sium arvense (L.) Scop.,” Canadian Journal of Plant Sci- ence, Vol. 55, No. 4, 1975, pp. 1033-1048. doi:10.4141/cjps75-163 [22] F. P. Thrasher, C. S. Cooper and J. M. Hodgson, “Compe- tition of Forage Species with Canada Thistle, as Affected by Irrigation and Nitrogen Levels,” Weeds, Vol. 11, No. 2, 1963, pp. 136-138. doi:10.2307/4040707 [23] R. F. Pywell, M. J. Hayes, J. B. Tallowin, K. J. Walker, W. R. Meek, C. Carvell, L. A. Warman and J. M. Bullock, “Minimizing Environmental Impacts of Grassland Weed Management: Can Cirsium arvense Be Controlled with- out Herbicides?” Grass and Forage Science, Vol. 65, No. 2, 2010, pp. 159-174. doi:10.1111/j.1365-2494.2010.00735.x [24] A. J. Bicksler and J. B. Masiunas, “Canada Thistle (Cir- sium arvense) Suppression with Buckwheat or Sudangrass Cover Crops and Mowing,” Weed Technology, Vol. 23, No. 4, 2009, pp. 556-563. doi:10.1614/WT-09-050.1 [25] J. M. Hodgson, “The Nature, Ecology and Control of Canada Thistle,” Technical Bulletin 1386, US Department of Agriculture, Washington DC, 1968. [26] R. L. Sheley and M. F. Carpinelli, “Creating Weed-Re- sistant Plant Communities Using Niche-Differentiated Non- native Species,” Rangeland Ecology & Management, Vol. 58, No. 5, 2005, pp. 480-488. doi:10.2111/03-142.1 [27] A. J. Travnicek, R. G. Lym and C. Prosser, “Fall-Pre- scribed Burn and Spring-Applied Herbicide Effects on Canada Thistle Control and Soil Seedbank in a Northern Mixed-Grass Prairie,” Rangeland Ecology & Management, Vol. 58, No. 4, 2005, pp. 413-422. doi:10.2111/1551-5028(2005)058[0413:FBASHE]2.0.CO ;2 [28] B. N. Ang, L. T. Kok, G. I. Holtzman and D. D. Wolf, “Competitive Growth of Canada Thistle, Tall Fescue, and Crownvetch in the Presence of a Thistle Defoliator, Cas- sida rubiginosa Muller (Coleoptera: Chrysomelidae),” Bio- logical Control, Vol. 4, No. 3, 1994, pp. 277-284. doi:10.1006/bcon.1994.1035 [29] A. J. Bicksler, “Canada Thistle Management Systems for Sustainable and Organic Farms,” Ph.D. Dissertation, Uni- versity of Illinois at Urbana-Champaign, Urbana, 2009. [30] M. G. Cripps, G. R. Edwards, G. W. Bourdot, D. J. Saville, H. L. Hinz and S. V. Fowler, “Effects of Pasture Compe- tition and Specialist Herbivory on the Performance of Cirsium arvense,” Biocontrol Science and Technology, Vol. 20, No. 6, 2010, pp. 641-656. doi:10.1080/09583151003695407 [31] P. D. Ominski, M. H. Entz and N. Kenkel, “Weed Sup- pression by Medicago sativa in Subsequent Cereal Crops: A Comparative Survey,” Weed Science, Vol. 47, No. 3, 1999, pp. 282-290. [32] L. G. Perry, S. A. Cronin and M. W. Paschke, “Native Cover Crops Suppress Exotic Annuals and Favor Native Perennials in a Greenhouse Competition Experiment,” Plant Ecology, Vol. 204, No. 2, 2009, pp. 247-259. doi:10.1007/s11258-009-9588-1 [33] R. G. Wilson and S. D. Kachman, “Effect of Perennial Grasses on Canada Thistle (Cirsium arvense) Control,” Weed Technology, Vol. 13, No. 1, 1999, pp. 83-87. [34] D. Ogle, “Western Wheatgrass Plant Guide,” Idaho State Office, Natural Resource Conservation Service, US De- partment of Agriculture, Washington DC, 2000. [35] D. Ogle, “Streambank Wheatgrass Plant Guide,” Idaho State Office, Natural Resource Conservation Service, US Department of Agriculture, Washington DC, 2006. [36] US Department of Agriculture, “The PLANTS Database,” 2009. http://plants.usda.gov Copyright © 2012 SciRes. AJPS  Canada Thistle (Cirsium arvense) Response to Clipping and Seeding of Competitive Grasses Copyright © 2012 SciRes. AJPS 1259 [37] L. J. Benz, K. G. Beck, T. D. Whitson and D. W. Koch, “Reclaiming Russian Knapweed Infested Rangeland,” Journal of Range Management, Vol. 52, No. 4, 1999, pp. 351-356. doi:10.2307/4003545 [38] R. M. Bottoms and T. D. Whitson, “A Systems Approach for the Management of Russian Knapweed (Centaurea repens),” Weed Technology, Vol. 12, No. 2, 1998, pp. 363-366. [39] R. G. Lym, “The Biology and Integrated Management of Leafy Spurge (Euphorbi a esula) on North Dakota Range- land,” Weed Technology, Vol. 12, No. 2, 1998, pp. 367- 373. [40] K. K. Rose, A. L. Hild, T. D. Whitson, D. W. Koch and L. Van Tassell, “Competitive Effects of Cool-Season Grasses on Reestablishment of Three Weed Species,” Weed Tech- nology, Vol. 15, No. 4, 2001, pp. 885-891. doi:10.1614/0890-037X(2001)015[0885:CEOCSG]2.0.C O;2 [41] T. D. Whitson and D. W. Koch, “Control of Downy Brome (Bromus tectorum) with Herbicides and Perennial Grass Competition,” Weed Technology, Vol. 12, No. 2, 1998, pp. 391-396. [42] R. G. Wilson, S. B. Orloff, D. L. Lancaster, D. W. Kirby and H. L. Carlson, “Integrating Herbicide Use and Peren- nial Grass Revegetation to Suppress Weeds in Noncrop Areas,” Invasive Plant Science and Management, Vol. 3, No. 1, 2010, pp. 81-92. doi:10.1614/IPSM-09-008.1 [43] K. G. Beck and J. Sebastian, “An Integrated Canada This- tle Management System Combining Mowing with Fall- Applied Herbicides,” Proceedings of the 46th Annual Western Society of Weed Science Meeting, Tucson, 9-11 March 1993, pp. 102-104. [44] R Development Core Team, “R: A language and Environ- ment for Statistical Computing,” R Foundation for Statis- tical Computing, Vienna, 2009. [45] R. L. Amor and R. V. Harris, “Control of Cirsium arvense (L.) Scop. by Herbicides and Mowing,” Weed Research, Vol. 17, No. 5, 1977, pp. 303-309. doi:10.1111/j.1365-3180.1977.tb00483.x [46] F. A. Welton, V. H. Morris and A. J. Hartzler, “Organic Food Reserves in Relation to the Eradication of Canada Thistles,” Technical Bulletin 441, University of Ohio Ag- ricultural Experiment Station, Columbus, 1929. [47] C. W. Grekul and E. W. Bork, “Fertilization Augments Canada Thistle (Cirsium arvense L. Scop) Control in Temperate Pastures with Herbicides,” Crop Protection, Vol. 26, No. 4, 2007, pp. 668-676. doi:10.1016/j.cropro.2006.06.005 [48] S. Kluth, A. Kruess and T. Tscharntke, “Influence of Me- chanical Cutting and Pathogen Application on the Per- formance and Nutrient Storage of Cirsium arvense,” Journal of Applied Ecology, Vol. 40, No. 2, 2003, pp. 334-343. doi:10.1046/j.1365-2664.2003.00807.x [49] C. M. Boerboom and D. L. Wyse, “Response of Canada Thistle (Cirsium arvense) and Birdsfoot Trefoil (Lotus corniculatus) to Bentazon,” Weed Science, Vol. 36, No. 2, 1988, pp. 250-253. [50] A. C. Arny, “Variations in the Organic Reserves in Un- derground Parts of Five Perennial Weeds from Late April to November,” Technical Bulletin 84, University of Min- nesota Agricultural Experiment Station, St Paul, 1932. [51] A. Ferrero-Serrano, T. R. Collier, A. L. Hild, B. A. Mealor and T. Smith, “Combined Impacts of Native Grass Competition and Introduced Weevil Herbivory on Canada Thistle (Cirsium arvense),” Rangeland Ecology & Man- agement, Vol. 61, No. 5, 2008, pp. 529-534. doi:10.2111/07-142R.1 [52] J. Friedli and S. Bacher, “Direct and Indirect Effects of a Shoot-Base Boring Weevil and Plant Competition on the Performance of Creeping Thistle, Cirsium arvense,” Bio- logical Control, Vol. 22, No. 3, 2001, pp. 219-226. doi:10.1006/bcon.2001.0971 [53] L. M. Laurialt, R. E. Kirksey and D. M. VanLeeuwen, “Performance of Perennial Cool-Season Forage Grasses in Diverse Soil Moisture Environments, Southern High Plains, USA,” Crop Science, Vol. 45, No. 3, 2005, pp. 909-915. doi:10.2135/cropsci2004.0280 [54] W. W. Donald and T. Prato, “Efficacy and Economics of Herbicides for Canada Thistle (Cirsium arvense) Control in No-Till Spring Wheat (Triticum aestivum),” Weed Sci- ence, Vol. 40, No. 2, 1992, pp. 233-240. [55] G. M. Bendall, “The Allelopathic Activity of Californian Thistle (Cirsium arvense (L.) Scop.) in Tasmania,” Weed Research, Vol. 15, No. 2, 1975, pp. 77-81. doi:10.1111/j.1365-3180.1975.tb01102.x [56] W. J. Stachon and R. L. Zimdahl, “Allelopathic Activity of Canada Thistle (Cirsium arvense) in Colorado,” Weed Science, Vol. 28, No. 1, 1980, pp. 83-86. [57] R. G. Wilson, “Effect of Canada Thistle (Cirsium arvense) Residue on Growth of Some Crops,” Weed Science, Vol. 29, No. 2, 1981, pp. 159-164. [58] M. L. Pokorny, R. L. Sheley, C. A. Zabinski, R. E. Engel, T. J. Svejcar and J. J. Borkowski, “Plant Functional Group Diversity as a Mechanism for Invasion Resistance,” Resto- ration Ecology, Vol. 13, No. 3, 2005, pp. 448-459. doi:10.1111/j.1526-100X.2005.00056.x [59] J. L. Funk, E. E. Cleland, K. N. Suding and E. S. Zavaleta, “Restoration Through Reassembly: Plant Traits and Inva- sion Resistance,” Trends in Ecology and Evolution, Vol. 23, No. 12, 2008, pp. 695-703. doi:10.1016/j.tree.2008.07.013 [60] Y. Ma, “Allelopathic Studies of Common Wheat (Triticum aestivum L.),” Weed Biology and Management, Vol. 5, No. 3, 2005, pp. 93-104. doi:10.1111/j.1445-6664.2005.00164.x [61] U. G. Bokhari, “Allelopathy among Prairie Grasses and Its Possible Ecological Significance,” Annals of Botany, Vol. 42, No. 1, 1978, pp. 127-136. [62] R. K. Kohli, H. P. Singh and D. R. Batish, “Allelopathy in Agroecosystems,” Haworth Press, New York, 2001. [63] D. Glen, “Regreen: A Cool Season, Soil Stabilizing Cover Crop,” HybriTech Seed International, Inc., Wichita, 1992.
|