 Advances in Microbiology, 2012, 2, 317-326 http://dx.doi.org/10.4236/aim.2012.23038 Published Online September 2012 (http://www.SciRP.org/journal/aim) Molecular Profiling of Drug Resistant Isolates of Mycobacterium tuberculosis in North India Dinesh K. Tripathi1*, Kanchan Srivastava2*, Surya Kant2, Kishore K. Srivastava1# 1Department of Microbiology, Central Drug Research Institute, Lucknow, India 2Department of Pulmonary Medicine, Chhatrapati Shahuji Maharaj Medical University, Lucknow, India Email: #kishore@cdri.res.in Received May 9, 2012; revised June 7, 2012; accepted June 18, 2012 ABSTRACT Multidrug-resistant tuberculosis (MDR-TB) is a major public health problem because treatment is complicated, cure rates are well below those for drug susceptible tuberculosis (TB), and patients may remain infectious for months or years despite receiving the best available therapy. To gain a better understanding of MDR-TB, we characterized isolates recovered from 69 patients with MDR-TB, by use of IS6110 restriction fragment-length polymorphism (RFLP) analysis; spacer oligonucleotide genotyping (i.e. spoligotyping). Clinical isolates from patients with tuberculosis have been con- sidered to contain clonally expanded Mycobacterium tuberculosis (MTB) strain. Over the years, the identification method based on IS6110 insertion sequences has been established as the standard for typing strains of MTB. IS6110 RFLP fingerprinting is very convincing when it is applied to classify MTB isolates harboring a large number of IS6110 in their chromosomes. Therefore, in the present study we have characterized the isolates from the patients suffering from MDR TB, on the basis of conserved Variable Number Tandem Repeats (VNTR), Direct Repeats (DR) and Inser- tion Sequences (IS) IS6110 elements. The polymorphic data showed significant level of dissimilarities among all the MDR isolates of MTB. Comparative studies with the DR and VNTR data substantiate th at polymorphism occur among MDR-TB cases as shown by the number of repeats present in different clinical isolates. Keywords: Mycobacterium; Drug Resistance; IS6110; Polymorphism 1. Introduction Although Tuberculosis (TB) is a preventable and treat- able disease, it remains one of the leading infectious dis- eases worldwide. As a result of inadequate treatment, the proportion of patients with MDR-TB is constantly in- creasing, and the extensively drug resistant TB (XDR-TB) has become a new global threat. One important advance in the field of tuberculosis research has been the devel- opment of molecular techniques that allow the id entifica- tion and tracking of individual strains of MTB. This new discipline, the molecular epidemiology of tuberculosis, began with the identification of IS6110, a novel myco- bacterial insertion sequence which formed the basis of a reproducible genotyping technique for MTB [1]. The spread of MDR-TB, due to emergence of MTB isolates has increased worldwide and reached epidemic proportion in many countries [2-4]. MDR-TB, which is caused by MTB, isolates that are resistant to, at least, Rifampicin (RIF) and Isoniazid (INH), is a serious public health hazard [5,6]. Treating MDR-TB can be difficult because loss of use of the 2 most potent anti-TB drugs (i.e., INH and RIF) means that only less MDR-TB can be cured by short-course chemotherapy [7-11], for other patients, bacillary growth is merely suppressed as long as treatment is continued [9,11]. Furthermore, 8% - 35% of patients with MDR-TB have persistently active disease that is refractory to multidrug therapy [12-16]. Conse- quently, in most studies, the cure rates for MDR-TB re- main well below those for drug-susceptible TB, and mortality rates may be substantial, even among HIV- neg ative patients [12]. In addition, patients with MDR-TB those do not respond to treatmen t are a co nstan t so urce of transmission of multidrug-resistant MTB [17-20]. In con- trast to most bacteria, for MTB acquisition of drug resis- tance does not occur as a result of horizontal transfer of resistance-bearing genetic elements. Rather, acquisition of drug resistance by MTB results from mutations (caused by nucleotide substitutions, insertions, or deletions) in specific resistance-determining regions of the genetic targets (or their promoters) or activating enzymes of anti-TB chemotherapeutic agents [21]. Inadequate ther- apy or sub therapeutic drug level may provide a selective growth advantage and, thus, may favor the growth of a resistant phenotype that can ultimately predominate in *Authors have equally contributed. #Corresponding author. C opyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. 318 persons in whom the disease was originally caused by drug susceptible isolates [5]. Moreover, in patients with MDR-TB, selection for additional mutations may be ac- complished by adding a single drug to a failing regimen [20]. In the human lung, selection of drug-resistan ce mu- tations in MTB occurs predominantly within lung cavi- ties for which high bacterial loads, active mycobacterial replication, and reduced exposure to host defense mecha- nisms have been reported [20-22]. Because MTB in spu- tum samples obtained from patients originates from lung cavities, molecular analysis of serially recovered sputum isolates allowed us to study aspects of the genetic evolu- tion of drug resistance in the human host. RIF and INH are two crucial bactericidal drugs helps in clearing nearly 80% MTB cells primarily in the cavi- ties. Other drugs, Ethambutol (EMB) and Pyrazinamide (PYZ) are supporting drugs during the initial phase [23, 24]. Therefore, immediate identification of these resistant isolates is very important for adjustments in treatment [25-27]. RIF were introduced in1972 as an anti TB drug and has excellent sterilizing activity. It acts by bind ing to the β-subunit of RNA polymerase (rpoB) [28], the en- zyme responsible for transcription and expression of mycobacterial genes, resulting in inhibition of the bacte- rial transcription activity and thereby killing the organ- ism. Mutations in the 81-bp core region of rpoB were reported to be responsible for resistance in at least 95% of the isolates [27,29,30]. This region is located between codons 507 to 533 with the most common changes in codons Ser531.eu, H is526Tyr and Asp16 Val [30,31]. The INH enters the bacterial cell as prodrug it is acti- vated to a toxic substance in the cell by a catalase p erox- ides encoded by a katG gene [32] and subsequently af- fects intracellular targets such as mycolic acid biosynthe- sis, an important component of the cell wall, which eventually results in loss of cellular integrity and the bacterial death. Genetic and biochemical studies have shown that re- sistance to EMB is mediated by mutations in the embB gene, which encodes arabinisyl transferase, an integral membrane protein that is inhibited by the drug. Various studies have identified five mutations in codo n 306 ATG of the embB gene that alter its first or third base ATG to GTG CTG/ATA ATC or to ATT, resulting in three dif- ferent amino acid substitutions (Met to Val, Leu or Ile) in the EMB resistant isolates. These five mutations are as- sociated with 70% - 90% of all embB resistant isolates [11,33,34]. The early and rapid detection of multidrug resistance is essential for efficient treatment and control of MTB. The culture based methods for detection of MTB infection and drug susceptibility testing usually take more than a month, due to the slow growth of this bacterium. The use of molecular methods for the identi- fication of mutations in the genes may offers means for rapid screening of the drug resistance among the MTB isolates and initiation of early treatment [27,28,30]. In the above context, we in the present study have typed the drug resistant isolates on the basis of DR and VNTR and compared those with the standard IS6110. 2. Materials and Methods 2.1. Collection of MTB Isolates Three consecutive morning sputum samples from each patient were collected in properly labeled screw cap dis- posable plastic bottles after oral gurgling with normal water. Sputum samples were processed and stained for Acid Fast Bacilli (AFB). One sputum sample from each smear positive patient was processed, inoculated on Lowenstein Jensen (LJ) slants and incubated in auto- mated culture system at 37˚C for six weeks (Table 1). The preliminary identification of mycobacterium iso- lates depends on their growth on LJ slants. Specific iden- tification is accomplished by the performance of Ziehl- Neelsen (Z-N) stain and battery of bioch emical tests. The positive cultures include growth in LJ medium after de- contamination of sputum samples and incubation at 37˚C for 4 - 6 weeks . MTB isolates recovered from 69 HIV-negative, and smear positive cases of both sexes, age varied from 18 to 62 years with MDR-TB that was refractory to chemo- therapy given for >12 months. All subjects were selected from Department of Pulmonary Medicine, CSM Medical University, Lucknow and residents from the peripheral region of Uttar Pradesh attending OPD of CSMMU, UP. Drug susceptibility was tested every 2 - 3 months. For all patients, treatment regimens were adjusted on the basis of the results of these evalu ation s, at month intervals. W e performed a detailed microbiological analysis of MTB isolates recovered from these patients. History relevant to tuberculosis such as time and duration, AFB load, out- come of Patients was recorded in predesigned data sheet (Table 2). 2.2. Drug susceptibility Testing (DST) The phenotypic resistance of all isolates was determined at baseline. Resistance to RIF and INH was in LJ me- dium that contained 2 µg/ml RIF or 0.1 µg /ml. INH. Table 1. Results of culture of smear (AFB) positive sputum specimen (n = 69). S. No.Results of Culture Number Percentage (%) 1 Growth of Mycobacteria 69 87.34 2 Contamination 03 03.94 3 No growth of Myc obacteria 04 05.06 Total 76 100 Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. Copyright © 2012 SciRes. AiM 319 Table 2. Profile of selected tuberculosis (TB) patients. S. No. Age Range (in Years) Total M/Fa Ratio Duration of Treatment (in Months) Mean Sputum AFB Loadb Patients Outcome Status Alive/Deceasedc 1 19 - 30 12 8 /4 18 - 55 (36.5) 1.0 - 2.5 10/2 2 31 - 40 13 11/2 21 - 82 (51.5) 2.0 - 2.5 08/5 3 41 - 50 26 24/2 20 - 54 (37.0) 2.0 - 2.5 19/7 4 51 - 62 18 15/3 06 - 14 (10.0) 2.5 - 3.0 11/07 Note: aMale/Female: (M/F); bSputum smears were recorded as having 1 - 4, 4 - 40, or 140 bacilli/high power fields, and they were given a score of 1, 2, or 3; cDuring the study. Other anti tuberculosis agents were also determined on LJ medium that contained critical concentration of 7.5 µg/ml EMB, 10 µg/ ml Streptomycin(SM), 5 µg/ml, Kanamycin (KM), 2 µg/ml Ofloxacin (Ofx), 10 µg/ ml Ethionamide (ETO). Phenotypic susceptib ility testing for PZA was not performed. All inoculated LJ drug and con- trol media were incubated at 37˚C for 3 weeks. The me- dia were examined at 48 h then weekly. The reading for drug susceptibility were taken at 3 weeks after that drug deterioration and the growth on control and drug con- taining media were recorded according to Kent and Kubica [35]. Drug resistance was expressed in proportion method, where a strain is considered to be drug resistant if the number of colonies that grow on a drug containing media is 1 % or more of colonies that grow on a drug free media. The control media must show good grow th at least 50 to 150 colonies MTB H37Rv strain has been used as a control st rain (Tables 3 and 4). 2.3. DNA Isolation The mycobacteria were cultured in LJ medium for 3 weeks. The cells were harvested, and chromosomal DNA was extracted by an enzymatic lysis method [36,37]. The bacteria were pelleted by centrifugation and resuspended in a 10 mM Tris-HCl-1 mM EDTA buffer (pH 8.0) [36]. Cell walls were digested with Lysozyme (10 mg/ml), Proteinase K (10 mg/ml), and 10% SDS. DNA was ex- tracted using 0.3 M cetyltrimethylammonium bromide (CTAB) and 5M NaCl, purified by Phenol chloroform extraction. DNA was precipitated by adding 1 volume of isopropanol to the aqueous supernatant. After 30 min incubation at –20˚C the mixture was centrifuged for 15 min. at 10,000 × g, the pellet was washed once with 70% ethanol, air-dried and finally suspended in Mili Q water [38]. 2.4. PCR Amplification The primers (Table 5) were used to amplify the flanking regions of the VNTR, DR and IS6110 insertion sequence [38]. PCR was performed using an automated gradient thermal cycler (Bio-Rad) and all reaction buffers con- tained 10 m M Tris/HC 1 (p H 8.3), 50 mM KC1, 1 - 5 m M Table 3. Drug resistance and susceptibility profile for 177 patients. Name of drugs No. of sensitive strains (%) No. of resistant strains (%) Isoniazid (INH) 110 (62.14) 67 (37.85) Rifampicin (RI F) 101 (57.06) 76 (42.93) Streptomycin (SM) 155 (87.57) 22 (12.42) Ethambutol (EMB) 163 (92.09) 14 (07.90) Pyrazinamide (PZA) Not done -- Ethionamide (ETO) All (100) Nil Kanamycin (KM) 174 (98.0) 03 (1.69) Capriomycin (CM) 173 (97.74) 04 (2.26) Amikacin (AM) All Nil Ofloxacin (Ofx) All Nil Cycloserine (CS) All Nil p-amino salicylic acid (PAS) None -- Sensitive to all drugs None* -- Resistance to any drugs -- All Total-177 *MDR: Multi-drug resistant: Resistance to both Isoniazid and Rifampicin with or without Resistance to other drug. Table 4. Multidrug resistance pattern of clinical isolates to anti tuberculosis drugs. S. No.No. of drugs Name of drug No. of resistant strains Total (%) 69:177 1 2 Drugs*RIF + INH 30 (43.47) RIF + SM 21 (30.43) RIF + EMB 05 (07.24) 81.15:31.63 2 3 Drugs*RIF + INH + SM 07 (10.14) *RIF + INH + SM 04 (05.79) 15.93:06.21 3 4 Drugs*RIF + INH + EMB + SM 02 (02.89) 02.89:01.12 Rifampicin (RIF); Is oniazid (INH); Strepto mycin (SM); Ethambutol (EMB).  D. K. TRIPATHI ET AL. 320 Table 5. PCR primers used for gene amplification. S. No. Primer Sequence 1 DR0272 F-5’AGCGATCCTGCTGGTGG3’ R-3’TGCTGTTAGGGTCAAACG5’ 2 DR0642 F-5’CCACTAGCAGATGGCCGTT3’ R-3’GCTCCAAGCGTAGTGATCCT5’ 3 DR2068 F-5’CACGACGTAGACGAATGC3’ R-3’ATGACACGCTTTCTGCCC5’ 4 DR3074 F-5’GTCACGATTGACACGCGGT3’ R-3’CATGGCCTCCGTTGTACTC5’ 5 DR3319 F-5’TGGTAGGTCTGGTTCCGC3’ R-3’ATGTGCATCCTCAACGGG5’ 6 DR3991 F-5’CCAACCTAGGCGTGTTCG3’ R-3’GATGTTCACCCCGAATGG5’ 7 DR4110 F-5’TTTAGACGATCGCACCGC3’ R-3’AACGGAATCGTGGTCAGC5’ 8 VNTR4052 F-5’GAGCCAAATCAGGTCCGG3’ R-3’GAGGTATCAACGGGCTTGT5’ 9 VNTR4120 F-5’GTTCACCGGAGCCAACC3’ R-3’GAGGTGGTTTCGTGGTCG5’ 10 VNTR4156 F-5’ACCGCAAGGCTGATGATCC3’ R-3’GTGCATCTCGTCGACTTCC5’ 11 VNTR4348 F-5’ACAAGGAGAGCGGTGTCG3’ R-3’CATCCTGTAGATGGCGGC5’ 12 IS6110 F-5’CCTGCGAGCGTAGGCGTCGG3’ R-3’CTCGTCCAGCGCCGCTTCGG5’ MgCl2, 0.2 mM of each dNTP (Fermentas, USA), 2 - 5 units Taq polymerase (Fermentas, USA), l µM of each primer, and 100 ng template DNA in a final volume of 100 µl. The amplification profile consisted of a denatura- tion step at 95˚C for five minutes, followed by 30 cycles with denaturation at 95˚C for one minute, primer anneal- ing at 65˚C, 67˚C and 55˚C for one minute, and exten- sion at 72˚C for one minute. The PCR products were electrophoresed through 1.5% - 2% agarose gels and stained with ethidium bromide. Visualization was done on a UV light illuminator (Chemidoc) the copy number of the amplified products was inferred from the differ- ence between the molecular weights of the amplified products of the samples and those of the H37Rv strain. To estimate the length of the amplified products were used to compare with standard molecular weight markers (Fermentas, USA). In the present study we have characterized 69 isolates from the patients suffering from MDR TB, on the basis of conserved VNTR, DR and IS6110 elements. The sets of DNA primers (VNTR = #4, DR = #7 and IS6110 = #1) were designed from the MTB genome and were used to amplify the genomic DNAs of isolates. Sequences of primers listed below were used for VNTR, DR, and IS elements. The position of each locus is reported earlier [39-41]. 3. Results 3.1. Patients A total of 177 sputum smear positive pulmonary tuber- culosis patients were studied. Out of 177, 76 RIF resis- tant cases were selected. Among 76 cases 58 were male and remaining 18 were female (76.31% and 23.68%). All of them were in the age group of 19 - 62 years (Table 2). Of the 76 cases, 60 (78.94%) were in low income group and only 16 (21.05%) from middle-income group. Ma- jority, of the patients came from urban area. Of these 76 smear positive cases, culture for Mycobacteria were positive in 69 (87.34%) cases, contaminatio n in 3 (3.97%) and no growth of Mycobacteria in 4 (5.06%) cases (Ta- ble 1). Some of the patients were mono drug resistant initially but they converted into MDR cases. Study was carried out on 69 RIF resistant and other drugs resistant cases. During the study period, sixty nine patients previously had TB; none of the patients had extra pulmonary TB and Diabetes mellitus. Most patients excreted large num- bers of bacilli in sputum (median score, 2.0) (Table 2), some patients died during the study, most likely as a re- sult of cachexia and/or chronic respiratory failure. Pa- tients who died had more extensive disease compared with patients who survived. At the time that TB was originally diagnosed, all patien ts were treated with World Health Organization category I therapy (i.e., treatment with INH, RIF, PZA and EMB for 2 months, followed by treatment with either INH and RIF or INH, RIF, and PZA for an additional 4 months) for varying lengths of time [36]. Once MDR-TB was diagnosed, the patients were switched to treatment regimens tailored to the phe- notypic drug-susceptibility profile of their isolates. At entry to the study, therapy was again adjusted according to phenotypic drug susceptibility, treatment history, and the side effect profile. 3.2. Phenotypic and Genotypic Resistance Profile of M. tuberculosis All 69 isolates displayed phenotypic resistance to RIF and taken together the isolates from all 69 patients were highly resistant to many of the most potent first and sec- ond line agents. Identification tests for Mycobacterium isolates were done in accordance with the standard pro- cedures. Tables 3 and 4 show the sensitivity and resis- tance pattern of 69 strains of MTB to 4 anti tuberculosis drugs. All strains were resistant to one or more drugs. Highest mono drug resistance (42.93%) was found in RIF either alone or in combination with other drugs [43.47%]. Our study identified 30 isolates were resistant to both INH and RIF; the other 39 isolates were resistant to all the three and four drugs tested (Tables 3 and 4). Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. Copyright © 2012 SciRes. AiM 321 Genotypic results are adding power to this approach that based on the detection of DNA po lymorphism within the DR cluster and VNTR- PCR are gold standard tech- niques for strain typing (Table 5 ) and for the stud y of the global molecular epidemiology of MTB (Tables 6 and 7). These tools provide information like latent infection, Strain-specific patterns, and drug resistance in various isolates. The Polymorphic data showed significant level of dissimilarities among all the MDR isolates of MTB. Out of 69 patients, a number of VNTR’s were detected, without showin g any standard prof ile. The polymorphism of each tandem repeat locus was found to be different; they had moderate or high allelic diversity which are useful for the differentiation of MTB strains. Molecular genotyping based on VNTR-PCR analyses has several advantages over standard IS6110 RFLP and other typing methods. Five types of DR’s were amplified with each other of the primer sets used. When compared the DR and VNTR data, we could only observe that polymor- phism occur among clinical isolates of MDR-TB and there are number of fingerprints presen t (Figures 1 and 2, Table 7). 4. Discussion In 1993, the National Tuberculosis Program (NTP) in India was strengthened in the form of Revised National Tuberculosis Control Program (RNTCP). Like HIV- AIDS, threat perception due to occurrence of multidrug resistance has assumed considerable gravity in con- structing the epidemic situation analysis and appropriate intervention. In this study drug resistance of MTB to at least one drug were found in all selected cases. This situation is highly alarming. Resistances (37.85%) were found in INH which is the most popular drug, followed by RIF (42.9%) cases. Resistances to SM were found in 12.42% cases and to EMB 7.90% cases [42-44]. The efficiency of current tuberculosis control program in any Table 6. Grouping of clinical isolates on the basis of IS6110. Groups No. of Patient Samples in Which IS6110 Positive No. of Patients Samples in Which IS6110 Negative A 1, 2, 9, 10, 17, 19, 23, 34 - B 3, 4, 20, 21, 22 - C 5, 11, 13, 14, 15, 16, 18, 24, 25, 26, 27, 28 to33, 35, 38 to 46, 48 to 63, 65 to 71 - D 6 - E 7 - F - 8, 36, 37, 47, 64 Table 7. Polymorphism in pulmonary isolates with various DRs and VNTRs. S. No. Primers Name (DR/VNTR) Polymorphism Shows in Patients Samples Direct Repeats (DR) Band Size of Primers 1 DR0272 305 kb 3 - 8, 18 - 22 2 DR0642 231 kb 1 - 6, All bands are of same size. 3 DR2068 336 kb 6 - 12, All bands are of same size. 4 DR3074 172 kb 6 - 12, All bands are of same size. 5 DR3319 574 kb 2 - 8, 2 - 5, 7 - 9. 6 DR3991 534 kb 2 - 9, All bands are of same size. 7 DR4110 531 kb 18 - 24 Variable Number Tandem Repeats (VNTR) 8 VNTR4052 879 kb 4 - 10 9 VNTR4120 447 kb 5 - 11 10 VNTR4156 704 kb 6 - 12 11 VNTR4348 516 kb 7 - 14 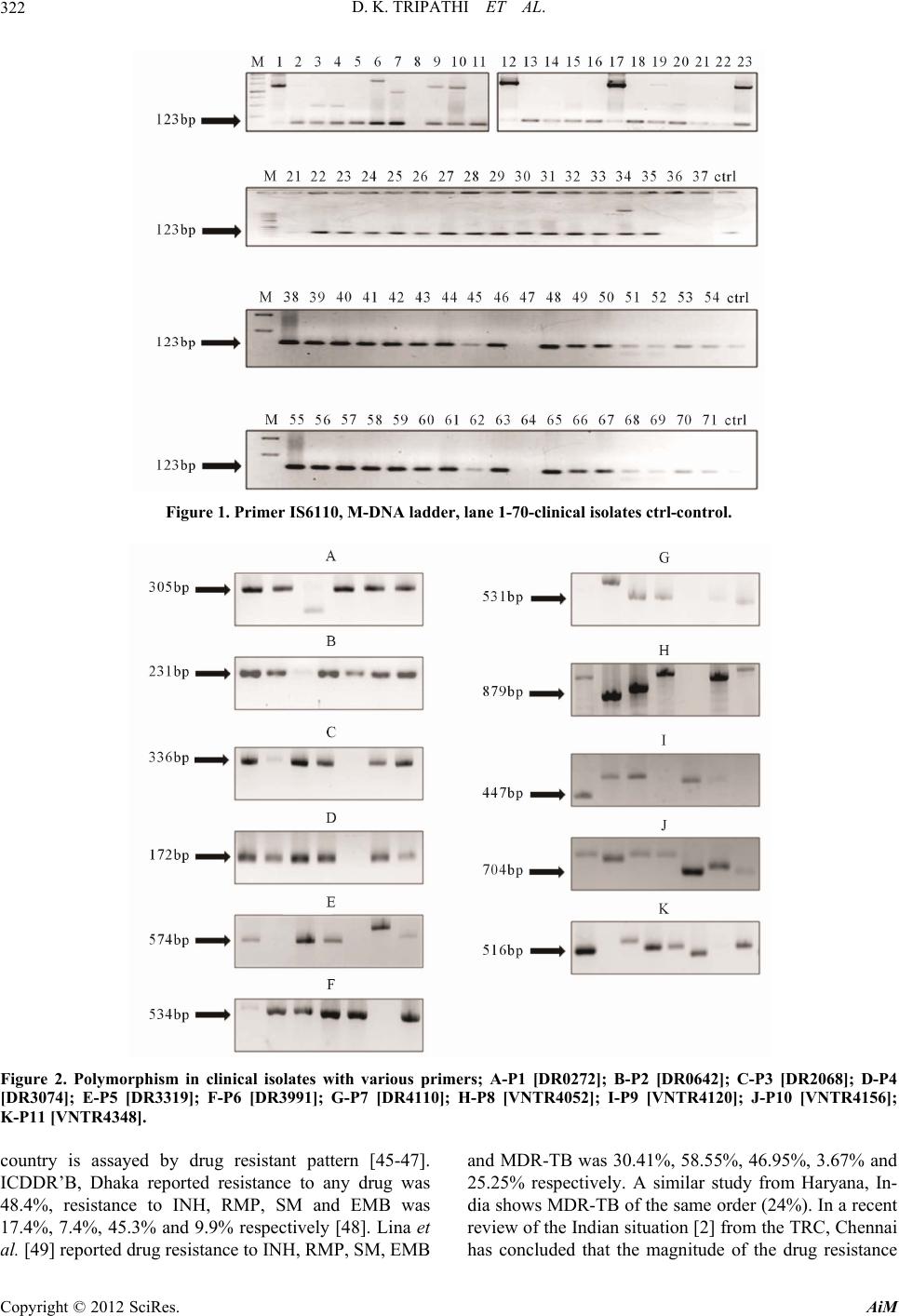 D. K. TRIPATHI ET AL. 322 Figure 1. Primer IS6110, M-DNA ladder, lane 1-70-clinical isolates ctrl-control. Figure 2. Polymorphism in clinical isolates with various primers; A-P1 [DR0272]; B-P2 [DR0642]; C-P3 [DR2068]; D-P4 [DR3074]; E-P5 [DR3319]; F-P6 [DR3991]; G-P7 [DR4110]; H-P8 [VNTR4052]; I-P9 [VNTR4120]; J-P10 [VNTR4156]; K-P11 [VNTR4348]. country is assayed by drug resistant pattern [45-47]. ICDDR’B, Dhaka reported resistance to any drug was 48.4%, resistance to INH, RMP, SM and EMB was 17.4%, 7.4%, 45.3% and 9.9% respectively [48]. Lina et al. [49] reported drug resistance to INH, RMP, SM, EMB and MDR-TB was 30.41%, 58.55%, 46.95%, 3.67% and 25.25% respectively. A similar study from Haryana, In- dia shows MDR-TB of the same order (24%). In a recent review of the Indian situation [2] from the TRC, Chennai has concluded that the magnitude of the drug resistance Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. 323 problem is principally due to acquired resistance (re- placed in recent times by the term drug resistance among previously treated cases). In New Delhi, a similar extent of acquired drug resistance was reported. Institute of Thoracic medicine in Chennai had shown acquired resis- tance of about 63% among patients from District Tuber- culosis Centers of Tamil Nadu. Resistance to INH and RIF (MDR TB) was of the order of 20.3%. It was con- sidered 53% that initial drug resistance in India (freshly defined as, drug resistance among new cases) could be at a lower order than similarly placed countries globally, as distinct from the acquired drug resistance situation given above. There could be 5% - 10% resistance to INH, [20, 29,50,51]. This could be reflecting the primary drug re- sistance problem in the Indian context [2,52-54]. In this study 69 isolates resistant to two or more of the tested drug was identified. This is comparable to what has been reported in the neighboring countries, with re- sistance to INH and RIF being more common than resis- tance to EMB. The simultaneous resistance to INH and EMB that was detected in (3%) of the isolates is in agreement with previous reports [9,15,51], and the si- multaneous resistance to RIF and EMB detected in (7.24%) of the isolates is consistent with a previous study [7-9]. Resistance to RIF is increasing because of wide- spread application that results in selection of resistant mutants, and is seen in cases noncompliant with TB treatment [51,52]. In this context, resistance to RIF can be assumed to be a surrogate marker for MDR-TB [11, 48]. Phenotypic susceptibility testing for PZA was not performed, because the results of this test can be difficult to reproduce and may not correlate well with drug sus- ceptibility in vivo [12,13]. In conclusion, our results of MDR-TB underline the importance of strengthening classical case finding and treatment of smear-positive patients according to the on- going DOTS program. The introduction of the rapid, specific and technically affordable molecular techniques can be used and interpreted in conjunction with conven- tional methods to detect more active cases of MDR-TB cases. The Polymerase Chain Reaction (PCR) appears to be a simple and accurate method that allows genotyping to be undertaken more quickly and in a less costly man- ner. It is applicable for direct detection in stained sputum smear preparations, which help in reducing the time needed for bacterial growth, and should facilitate the adequate choice of anti tuberculosis therapy [1,14,40,] that limits the extent and severity of MDR-TB transmis- sion and infection. INH and RIF’s resistance in MTB complex (MTC) isolates are mainly based on mutations in a limited num- ber of genes. However, mutation frequencies vary in dif- ferent mycobacterial populations. In this work, we ana- lyzed the distribution of resistance-associated mutations in MTB. The application of DNA fingerprinting can pro- vide valuable insights into the p athogenesis of tuberculo- sis and may help in identifying strains of MTB with spe- cific properties such as virulence and failure of drug re- sponse. Most of the epidemiological applications of RFLP analysis have used an insertion sequence known as IS6110 [39,52-55]. It was initially described by Thierry et al. [56] and has been shown to be distributed through- out the MTB complex. Spoligotyping, in addition to IS6110 RFLP, can be useful in determining more distant relationships among isolates. In our current study, the relative instability of IS6110 RFLP was found in one of two MDR outbreak strains; however, not fewer than four of nine of the IS6110 RFLP patterns showed a minor and different al- teration. Therefore, the transposition rate may be strongly related to the M. tuberculosis genotype represented. DNA fingerprinting of MTB has been shown to be a powerful epidemiologic tool because it exploits variabil- ity in both the no. and genomic position of insertion se- quences and tandem repeats to generate strain specific patterns [2,54,56]. The integration of VNTR-typing with conventional approaches has the potential to be a powerful new tech- nology, which provides a robust and high resolution tool for the molecular epidemiology of the MTB complex. The direct repeat (DR) locus is the characteristic of the MTB complex. The DR locus consists of multiple tan- dem 36-bp repeats interspersed with variable spacers of about equal size. Polymorphism of the DR locus (ab- sence or presence of single Direct variant repeat DVR), has been exploited widely for distinguishing among clinical isolates of the MTB by using spacer oligonucleo- tide typing., In the present study we have used all the three control group of genes and tried to d emonstrate the differences among clinical isolates of MDR TB Isolates. In the present study, polymorphic data showed signifi- cant level of dissimilarities among all the MDR isolates of MTB. Out of 69 patients, a number of VNTRs were detected, without sh owing any standard profile. Similar ly two types of DRs were amplified with each of the primer sets used (Table 5). When we compared the DR and VNTR data we could only claim that polymorphism oc- cur among clinical isolates of MDR-TB and since there are numb er of finger p rints pre se nt [53-55 ]. Over the past decade, much has been learned of the drug targets and mechanisms of resistance to first-line and several second-lines anti tuberculosis agents (Table 4) [42,43,53,54]. As mentioned above, MTB generally acquires drug resistance via de novo nsSNP, small dele- tions, or insertions in specific chromosomal loci, unlike most other pathogenic bacteria, which often acquire drug resistance via horizontal transfer. This attribute of MTB drug resistance, coupled with fast and efficient DNA Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. 324 sequencing methods, makes studying drug resistance highly amenable for molecular epidemiologic investiga- tions [41,46,47,54,57]. Molecular epidemiologic studies on drug resistance have generally sought to examine the nature (e.g., genotype-specific mutations, association of specific mutations with p henotypic resistance) and extent (e.g., prevalence of specific mutations in a population) of drug resistance and patient risk factors (e.g., HIV) for acquiring resistance. The report by Bifani et al. [57,58] provides an example of a study of the nature and evolu- tion of drug resistance during a clonal MDR-TB out- break. 5. Conclusions MTB is an obligate pathogen that does not naturally rep- licate outside of its host environment. As such, MTC members are believed to have coevolved with hominids for millions of years. Consequently, it is very possible that, unlike other opportunistic patho gens, viable tube rcle bacilli encode the minimum ensemble of virulen ce genes required for successful infection, replication, and dis- semination. Thus, the relative success of one clonal MTB family over another might rely on the relationship be- tween levels of gene expression and environmental fac- tors and the host. Strain analysis, together with virulence studies, will help pinpointing isolates associated with higher morbid- ity and mortality, with the aim of directing efforts to limit the spread of those strains within th e region. 6. Acknowledgements The work was supported by CSIR-CDRI SIP-0026 and DST WOS-A LS-24/2008 to KS [WOS-A LS-24/2008]. This is CSIR-CDRI co mmunication #127/2012/KKS. REFERENCES [1] D. van Soolingen, P. E. W. de Hass, P. W. M. Hermans, P. M. Groenen and J. D. van Embden, “Comparison of Vari- ous Repetitive DNA Elements as Genetic Markers for Strain Differentiation and Epidemiology of M. tuberculo- sis,” Journal of Clinical Microbiology, Vol. 31, No. 8, 1993, pp. 1987-1995. [2] P. Venkataraman and Paramasivan, “Drug Resistance in Tuberculosis and Issues Related to Multidrug Resistance in Planning for TB Control in India,” The Health Admin- istrator, Special Issue on Tuberculosis, Vol. 15, 2002, pp. 127-136. [3] Centers for Disease Control and Prevention (CDC), “Ex- tensively Drug-Resistant Tuberculosis-United States,” 1993-2006. [4] D. A. Mitchison, “Drug Resistance in Tuberculosis ,” Euro- pean Respiratory Journal, Vol. 25, No. 2, 2005, pp. 376- 379. doi:10.1183/09031936.05.00075704 [5] I. Mokrousov, O. Narvskaya, E. Limeschenko, T. Otten and B. Vyshnevskiy, “Detection of Ethambutol Resistant M. tuberculosis Isolates by Multiplex Allele Specific PCR Assay Targeting embB 306 Mutations,” Journal of Clini- cal Microbiology, Vol. 40, No. 5, 2002, pp. 1617-1620. doi:10.1128/JCM.40.5.1617-1620.2002 [6] I. Mokrousov, T. Otten and M. Filipenko, “Detection of Isoniazid Resistant M. tuberculosis Isolates by Multiplex Allele Specific PCR Assay Targeting katG Codons 351 Variation,” Journal of Clinical Microbiology, V ol. 40, No. 7, 2002, pp. 2509-2512. doi:10.1128/JCM.40.7.2509-2512.2002 [7] A. Van Rie, A. Warren and R. I. Mshanga, “Analysis for a Limited Number of Gene Codons Can Predict Drug Re- sistance of M. tuberculosis in a High-Incidence Commu- nity,” Journal of Clinical Microbiology, Vol. 39, No. 2, 2001, pp. 636-641. doi:10.1128/JCM.39.2.636-641.2001 [8] L. M. Parsons, M. Salfinger and A. Coleridge, “Phenotypic and Molecu lar Ch aracteriz ation of M. tuberculosis Isolates to both Isoniazid and Ethambutol,” Antimicrobial Agents and Chemotherapy, Vol. 49, No. 6, 2005, pp. 2218-2225. doi:10.1128/AAC.49.6.2218-2225.2005 [9] Z. Yang, R. Durmaz and D. Yang, “Simultaneous Detec- tion of Isoniazid, Rifampicin, and Ethambutol by a Single Multiplex Allele Specific Polymerase Chain Reaction (PCR) Assay,” Diagnostic Microbiology & Infectious Di- sease, Vol. 53, No. 3, 2005, pp. 201-208. doi:10.1016/j.diagmicrobio.2005.06.007 [10] A. Hristea, D. Otelea, S. Paraschiv, A. Macri, C. Baicus, O. Moldovan, M. Tinischi, V. Arama and A. Streinu-Cercel, “Detection of M. tuberculosis Resistance Mutations to Rifampin and Isoniazid by Real-Time PCR,” Indian Jour- nal of Medical Microbiology, Vol. 28, No. 3, 2010, pp. 211-216. doi:10.4103/0255-0857.66474 [11] I. Mokrousov, T. Otten, B. Vyshnevskiy and O. Narvskaya, “Allele-Specific rpoB PCR Assays for Detection of Ri- fampin-Resistant M. tuberculosis in Sputum Smear,” An- timicrobial Agents and Chemotherapy, Vol. 47, No. 7, 2003, pp. 2231-2235. doi:10.1128/AAC.47.7.2231-2235.2003 [12] G. Canetti, W. Fox, A. Khomenko, H. T. Mahler, N. K. Menon, D. A. Mitchison, N. Rist and N. A. Smelev, “Ad- vances in Techniques of Testing Mycobacterial Drug Sensitivity and t he Use of Sensitivity Tests in Tuberculosis Control Programmes,” Bulletin of the World Health Or- ganization, Vol. 41, No. 1, 1969, pp. 21-43. [13] T. H. Weniger, J. Krawczyk, P. H. Supply, S. Niemann and D. Harmsen, “MIRU-VNTRplus: A Web Tool for Polyphasic Genotyping of M. tuberculosis Complex Bac- teria,” Nucleic Acids Research, Vol. 38, Suppl. 2, 2010, pp. 1-6. doi:10.1093/nar/gkq351 [14] R. Johnson, M. E. Streicher, E. G. Louw, R. M. Warren, P. D. van Helden and T. C. Victor, “Drug Resistance in M. tuberculosis,” Current Issues in Molecular Biology, Vol. 8, No. 2, 2006, pp. 97-112. [15] B. Madison, B. Robinson-Dunn and I. George, “Multi- center Evaluation of Ethambutol Susceptibili ty Testing of M. tuberculosis by Agar Proportion 15 and Radiometric Methods,” Journal of Clinical Microbiology, Vol. 40, No. Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. 325 11, 2002, pp. 3976-3979. doi:10.1128/JCM.40.11.3976-3979.2002 [16] H. Rinder, K. T. Mieskes and T. Loscher, “Heteroresis- tance in Mycobacterium tuberculosis,” The International Journal of Tuberculosis and Lung Disease, Vol. 5, No. 4, 2001, pp. 339-345. [17] V. Nikolayevskyy, Y. Balabanova, T. Simak, N. Malo- manova, I. Fedorin and F. Drobniewski, “Performance of the Genotype(R) MTBDR Plus Assay in the Diagnosis of Tuberculosis and Drug Resistance in Samara, Russian Federation,” BMC Clinical Pathology, Vol. 10, 2009, pp. 2-22. doi:10.1186/1472-6890-9-2 [18] G. C. Baldeviano-Vidalon, N. Quispe-Torres and C. Bonilla-Asald, “Multiple Infection with Resistant and Sensitive M. tuberculosis Isolates during Treatment of Pulmonary Tuberculosis Patients,” The International Jour- nal of Tuberculosis and Lung Disease, Vol. 9, No. 10, 2005, pp. 1155-1160. [19] S. A. Watterson, S. M. Wilson, M. D. Yates and F. A. Drobniewski, “Comparison of Three 18 Molecular Assays for Rapid Detection of Rifampin Resistance in M. tuber- culosis,” Journal of Clinical Microbiology, Vol. 36, No. 7, 1998, pp. 1969-1973. [20] C. Dye, S. Scheele, P. Dolin, V. Pathania and M. C. Raviglione, “Global Burden of Tuberculosis: Estimated Incidence, Prevalence and Mortality by Country,” Journal of the American Medical Association, Vol. 282, No. 7, 1999, pp. 677-686. [21] WHO, “Anti-Tuberculosis Drug Resistance WHO/IUALD Global Project on Anti-Tuberculosis Drug Resistance Sur- veillance,” Geneva, 1997, WHO/TB/97.229. [22] C. Dye, Z. Fengzeng, S. Scheele and B. Williams, “Ev al u- ating the Impact of Tuberculosis Control: Number of Deaths Prevented by Short-Course Chemotherapy in China,” International Journal of Epidemiology, Vol. 29, No. 3, 2000, pp. 558-564. doi:10.1093/ije/29.3.558 [23] WHO, “Global Tuberculosis Control Surveillance: Plan- ning, Financing,” Geneva, 2009, pp. 1-303. [24] WHO, “Global Tuberculosis Control,” Geneva, Switzer- land, 2003. [25] WHO, “Anti Tuberculosis Drug Resistance in the World, Prevalence and Trends,” Geneva, Switzerland, 2000. [26] WHO, “Drug-Resistant Isolates of TB Increasing World- wide,” WHO 19, Geneva, 2000. [27] L. Herrera-León, T. Molina, P. Saíz, J. A. Sáez-Nieto and M. S. Jimenez, “New Multiplex PCR for Rapid Detection of Isoniazid-Resistant M. tuberculosis Clinical Isolates,” Antimicrobial Agents and Chemotherapy, Vol. 49, No. 1, 2005, pp. 144-147. doi:10.1128/AAC.49.1.144-147.2005 [28] C. Abe, I. Kobayashi and S. Mitarai, “Biological and Molecular Characteristics of M. tuberculosis Clinical Iso- lates with Low-Level Resistance to Isoniazid in Japan,” Journal of Clinical Microbiology, Vol. 46, No. 7, 2008, pp. 2263-2268. doi:10.1128/JCM.00561-08 [29] S. V. Ramaswamy and J. M. Musser, “Molec ular Genetic Basis of Antimicrobial Agent Resistance in M. tuberculo- sis: 1998 Update,” Tubercle and Lung Disease, Vol. 79, No. 1, 1998, pp. 3-29. doi:10.1054/tuld.1998.0002 [30] C. Plinke, H. S. Cox, S. Kalon, D. Doshetov. Rüsch- Gerdes and S. Niemann, “Tuberculosis Ethambutol Re- sistance: Concordance between Phenotypic and Geno- typic Test Results,” Tuberculosis, Vol. 89, No. 6, 2009, pp. 448-452. doi:10.1016/j.tube.2009.09.001 [31] J. I. Sekig uchi, T. Mi yoshi-Aki yama and E. Augustynowicz- Kopec, “Detection of Multidrug Resistance in Mycobac- terium tuberculosis,” Journal of Clinical Microbiology, Vol. 45, No. 1, 2007, pp. 179-192. doi:10.1128/JCM.00750-06 [32] N. Gonzalez, M. J. Torres, J. Aznar and J. C. Palomares, “Molecular Analysis of Rifampin and Ison iazid Res istance of M. tuberculosis Clinical Isolates in Seville, Spain,” Tubercle and Lung Disease, Vol. 79, No. 3, 1999, pp. 187-190. doi:10.1054/tuld.1998.0195 [33] J. Wang, R. M. Burger and K. Drlica, “Role of Superoxide in Catalase-Peroxidase-Mediated Isoniazid Action against Mycobacteria,” Antimicrobial Agents and Chemotherapy, Vol, 42, No. 3, 1998, pp. 709-711. [34] S. V. Ramaswamy, A. G. Amin, S. Goksel, C. E. Stager, S. J. Dou and H. L. Sahli, “Molecular Genetic Analysis of Nucleotide Polymorphisms Associated with Ethambutol Resistance in Human Isolates of M. tuberculosis,” Antim- icrobial Agents and Chemotherapy, Vol. 44, No. 2, 2000, pp. 326-336. doi:10.1128/AAC.44.2.326-336.2000 [35] P. T. Kent and G. P. Kubica, “Public Health Mycobacte- riology: A Guide for the Level III Laboratory. US De- partment of Health and Drug Resistance Pattern of M. tu- berculosis Isolated from Patients Attending a Referral Hospital Wadud et al. Bangladesh,” Journal of Medical Microbiology, Vol. 17, 2000, pp. 1132-1140 [36] J. P. Hosek, P. Svastova, M. Moravkova, I. Pavlik and M. Bartos, “Methods of Mycobacterial DNA Isolation from Different Biological Materia l: A Review,” Veterinary Me- dicine, Vol. 51, No. 5, 2006, pp. 180-192. [37] E. Mazars, S. Lesjean, A. L. Banuls, M. Gilbert, V. Vin- cent, B. Gicquel, M. Tibayrenc, C. Locht and P. H. Supply, “High-Resolution Minisatellite-Based Typing as a Port- able Approach to Global Analysis of M. tuberculosis Molecular Epidemiology,” Proceedings of the National Academy of Sciences, Vol. 98, No. 4, 2001, pp. 1901-1906. doi:10.1073/pnas.98.4.1901 [38] E. B. Hill, L. G. Wayne and M. Gross, “Purification of Mycobacterial Deoxyribonucleic Acid,” Journal of Bac- teriology, Vol. 112, No. 3, 1972, pp. 1033-1039. [39] N. Smittipat and P. Palittapongarnpim, “Identification of Possible Loci of Variable Number of Tandem Repeats in M tuberculosis,” Tubercle and Lung Disease, Vol. 80, No. 2, 2000, pp. 69-74. doi:10.1054/tuld.2000.0236 [40] I. J. Wiid, C. Werely, N. Beyers, P. Donald, P. D. van Helden, “Oligonucleotide (GTG)5 as a Marker for M. tu- berculosis Strain Identification.” Journal of Clinical Mi- crobiology, Vol. 32, No. 5, 1994, pp. 1318-1321. [41] P. Palittapongampim, S. Chomyc, A. Fanning and D. Kunimoto, “DNA Fragment Length Polymorphism Analy- sis of M. tuberculosis Isolates by Arbitrarily Primed Po- lymerase Chain Reaction,” The Journal of Infectious Copyright © 2012 SciRes. AiM  D. K. TRIPATHI ET AL. Copyright © 2012 SciRes. AiM 326 Diseases, Vol. 167, No. 4, 1993, pp. 975-978. doi:10.1093/infdis/167.4.975 [42] R. C. Chan, M. M. M. Hui, E. W. Chan, et al., “Geneti c and Phenotypic Characterization of Drug-Resistant M. tuberculosis Isolates in Hong Kong,” Journal of Antim- icrobial Chemotherapy, Vol. 59, No. 5, 2007, pp. 866- 873. [43] P. M. Gronen, A. E. Bunschoten, D. Van Soolingen and J. D. A. van Embden, “Nature of DNA Polymorphism in the Direct Repeat Cluster of M. tuberculosis: Application for Strain Differentiation by a Novel Typing Method,” Mo- lecular Microbiology, Vol. 10, No. 5, 1993 pp. 1057-1065. doi:10.1111/j.1365-2958.1993.tb00976.x [44] R. M. Warren, N. C. Gey van Pittius, M. Barnard, A. Hesseling, E. Engelke, M. de Kock, M. C. Gutierrez, G. K. Chege, T. C. Victor, E. G. Hoal and P. D. van Helden “Differentiation of M. tuberculosis Complex by PCR Amplification of Genomic Regions of Difference,” The International Journal of Tuberculosis and Lung Disease, Vol. 10, No. 7, 2006, pp. 818-822. [45] WHO, TB Country Profile-Jordan, 2010. www.who.int/tb/data [46] H. Van Deutekom, P. H. Supply, P. E. W. Haas, E. Wil- lery, S. P. Hoijng, C. Locht, R. A. Coutinho and D. van Soolingen, “Molecular Typing of M. tuberculosis by My- cobacterial Interspersed Repetitive Unit Variable-Number Tandem Repeat Analysis, a More Accurate Method for Identifying Epidemiological Links between Patients with Tuberculosis,” Journal of Clinical Microbiology, Vol. 43, No. 9, 2005, pp. 4473-4479. doi:10.1128/JCM.43.9.4473-4479.2005 [47] N. Thorne, A. Underwood, S. Gharbia and C. Arnold, “Evolutionary Clues from Comparative Analysis of M. tuberculosis Variable-Number Tandem Repeat Sequences within Genetic Families,” Infection, Genetics and Evolu- tion, Vol. 7, No. 2, 2005, pp. 239-246. doi:10.1016/j.meegid.2006.09.006 [48] ICDDR’B, Health and Science Bulletin, Vol. 1, 2002, pp. 6-10. [49] D. Lina and P. M. Sweta, “Drug Resista nce in Tuberculo- sis,” Bombey Hospital Journal, 1999. www.bhj.org/journal/1999.4103.july99/original.253.htm [50] L. Sandman, N. W. Schluger, A. L. Davidow and S. Bonk, “Risk Factors for Rifampin-Monoresistant Tuberculosis: A Case-Control Study,” American Journal of Respiratory and Critical Care Medicine, Vol. 159, No. 2, 1999, pp. 468-472. [51] A. Hristea, D. Otelea, S. Paraschiv, A. Macri, C. Baicus, O. Moldovan, M. Tinischi, V. Arama and A. Streinu-Cercel, “Detection of M. tuberculosis Resistance Mutations to Rifampin and Isoniazid by Real-Time PCR,” Indian As- sociation of Medical Microbiologists, Vol. 28, No. 3, 2010, pp. 211-216. doi:10.4103/0255-0857.66474 [52] F. A. Post, P. A. Willcox, B. Mathema, L. M. Steyn, K. Shean, S. V. Ram as w am y, E. A. Gravis s , E. Shashkina, B. N. Kreiswirth and G. Kaplan, “Genetic Polymorphism in M. tuberculosis Isolates from Patients with Chronic Mul- tidrug-Resistant Tuberculosis,” The Journal of Infectious Diseases, Vol. 190, No. 1, 2004, pp. 99-106. doi:10.1086/421501 [53] D. Van Soolingen, P. W. M. Hermans, P. E. W. de Haas, D. R. Soll and J. D. van Embden, “Occurrence and Sta- bility of Insertion Sequences in Mycobacterium Tubercu- losis Complex Strains: Evaluation of an Insertion Se- quence-Dependent Polymorphism as a Tool in the Epi- demiology of Tuberculosis,” Journal of Clinical Micro- biology, Vol. 29, No. 11, 1991, pp. 2578-2586. [54] S. M. M. Roring, D. Brittain, A. E. Bunschoten, M. S. Hughes, R. A. Skuce, J. D. A. Van Embden and S. D. Neil, “Spacer Oligotyping of Mycobacterium bovis Iso- lates Compared to Ty ping by Restriction Fragment Length Polymorphism Analysis Using PGRS, DR and IS6110,” Veterinary Microbiology, Vol. 61, No. 1-2, 1998, pp. 111- 120. doi:10.1016/S0378-1135(98)00178-3 [55] K. L. Horan, R. Freeman., K. Weigel, M. Semret, S. Pfaller, T. C. Covert, D. Van Soolingen, S. C. Leao, M. A. Behr and G. A. Cangelosi, “Novel Genetic Polymor- phisms That Further Delineate the Phylogeny of the My- cobacterium tuberculosis Complex,” Journal of Bacteri- ology, Vol. 188, No. 12, 2006, pp. 4271-4287. [56] D. Thierry, M. D. Cave, K . D. Eis enach, J . T. Craw ford, J. H. Bates and B. Gicquel, “IS6110, and IS-like Element of M. tuberculosis Complex,” Nucleic Acids Research, Vol. 18, No. 1, 1990, pp.188-192. doi:10.1093/nar/18.1.188 [57] P. B. Bifani, M. Mathema, S. Campo, B. Moghazeh, E. Nivin, J. Shashkina, S. S. Driscoll, R. Munsiff, R. Froth- ingham and B. N. Kreiswirth, “Molecular Identification of Streptomycin Monoresistant M. tuberculosis Related to Multidrug-Resistant W Strain,” Emerging Infectious Dis- eases, Vol. 7, No. 5, 2001, pp. 842-848. doi:10.3201/eid0705.010512 [58] P. Miotto, F. Piana, V. Penati, F. Canducci, G. B. Migliri and D. M. Cirillo, “Use of Genotype MTBDR Assay for Molecular Detection of Rifampicin and Isoniazid Resis- tance in M. tuberculosis Clinical Isolates Isolated in It- aly,” Journal of Clinical Microbiology, Vol. 44, No. 7, 2006, pp. 2485-2491. doi:10.1128/JCM.00083-06
|