 World Journal of AIDS, 2012, 2, 245-251 doi:10.4236/wja.2012.23032 Published Online September 2012 (http://www.SciRP.org/journal/wja) 245 Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study Joseph C. Gathe1*, Dean T. Martin2, M. Keith Rawlings3, Benjamin Daquioag1, John E. Fuchs4, Vanessa C. Williams4, Katrina L. Oie4, Gary E. Pakes4 1Therapeutic Concepts P.A., Houston, USA; 2Cigna Healthcare Corp., Phoenix, USA; 3Peabody Clinic, Dallas, USA; 4Glaxo- SmithKline, Research Triangle Park, Durham, USA. Email: *DrGathe@JosephGathe.com Received December 17th, 2011; revised February 21st, 2012; accepted May 18th, 2012 ABSTRACT Objective: The purpose of this study was to investigate whether switching HIV-infected patients stabilized on Trizivir (abacavir 300 mg/lamivudine 150 mg/zidovudine 300 mg) plus lopinavir/ritonavir 400 mg/100mg twice daily to Trizivir alone affects clinical efficacy and tolerability. Methods: This phase 4, open-label, pilot study was conducted over 96 weeks in 23 antiretroviral-naïve, HIV-infected patients. Initially, these patients received induction therapy with Trizivir plus lopinavir/ritonavir 400 mg/100mg twice daily. Patients who achieved a viral load < 50 copies/mL at week 36 or 44 started maintenance therapy with Trizivir alone at week 48 and continued this regimen through week 96. Re- sults: The study population was a median of 37 years of age, predominantly male (78%) and African American (61%, with 39% Caucasians), and had a baseline mean viral load of 5.45 log10 copies/mL and CD4+ count of 232 cells/mm3. Nineteen patients completed induction; of the four who did not, three were lost to follow-up and one withdrew due to gastrointestinal adverse events. In 14 induction completers who had viral load measurements taken at week 48, in- tent-to-treat: observed analysis showed a week 48 viral load < 400 copies/mL attained in 100% (14/14) and <50 cop- ies/mL attained in 71% (10/14). Median week 48 CD4+ cell count was 192 cells/mm3 higher than the baseline count. Twelve patients completed the subsequent 48-week Trizivir-alone maintenance phase, of whom 11 (92%) achieved vi- ral loads of both <400 and <50 copies/mL at week 96. Median week 96 CD4+ cell count was 208 cells/mm3 above baseline. Trizivir-only maintenance was associated with fewer adverse events than the Trizivir-lopinavir/ritonavir in- duction phase and with improvement in total cholesterol, LDL-cholesterol, and triglycerides. Conclusions: Trizivir-alone maintenance after Trizivir-lopinavir/ritonavir induction maintained virologic and CD4+ cell response, and was associ- ated with an improved adverse event and lipid profile. Keywords: Abacavir/Lamivudine/Zidovudine; HIV-1 Infection; Induction-Maintenance Strategy; Kaletra; Lopinavir/Ritonavir; Trizivir 1. Introduction The induction-maintenance antiretroviral treatment strategy involves giving HIV-infected patients induction therapy with multiple antiretroviral agents to achieve virologic suppression, then follow this by deletion of an agent of one class to simplify the regimen. In HIV care, the induction-maintenance strategy was first explored in the late 1990s using different combinations of nucleoside reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), and protease inhibitors (PI) [1]. Antiretroviral induction-maintenance regimen sequences investigated in clinical trials to date have included switching a double NRTI/double PI regi- men to a double PI-alone or a single NRTI/single PI combination [2]; a double NRTI/single PI regimen to ei- ther a double NRTI-alone or double PI-alone regimen [3]; a ritonavir-boosted PI/double NRTI regimen to a boosted PI-alone regimen [4-9]; and a triple NRTI/single NNRTI combination to a triple NRTI-alone regimen [10,11]. The triple NRTI fixed-dose combination Trizivir (GlaxoSmithKline, Research Triangle Park, North Caro- lina, USA), which contains abacavir 300 mg, lamivudine *Corresponding author. Copyright © 2012 SciRes. WJA  Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study 246 150 mg, and zidovudine 300 mg per tablet, has been available for twice-daily treatment of HIV infection since the late 1990s [12]. Use of Trizivir was expected to pre- serve antiretroviral agents of other classes for later use, thereby potentially delaying resistance development. Tri- zivir also offered dose symmetry with other agents simi- larly administered twice daily. Although current antiret- roviral treatment guidelines from the Department of Health and Human Services and IAS-USA do not rec- ommend monotherapy with a triple NRTI combination like Trizivir for initiating antiretroviral therapy [13,14], Trizivir-alone maintenance could be considered by clini- cians desiring to avoid particular adverse events some- times associated with recommended drug regimens. Induction with a triple NRTI plus a boosted PI, fol- lowed by deletion of the PI has undergone little study [15-17]. In view of this data gap, the focus of the present study was to evaluate the efficacy and tolerability of in- duction with Trizivir as the triple NRTI component and lopinavir/ritonavir (LPV/r) as the PI component, fol- lowed by a maintenance period of Trizivir alone. 2. Methods This phase 4 multicenter, open-label, single-arm, pilot study was conducted at three US-based treatment sites: Therapeutic Concepts P.A. (Houston, Texas), Cigna Healthcare Corp (Phoenix, Arizona), and the Peabody Clinic (Dallas Texas). Patients could be enrolled if they were HIV-infected, as documented by HIV-1 antibody enzyme-linked immunosorbent assay (ELISA) and con- firmed by Western blot or HIV plasma viremia, were aged 13 years or older, had screening HIV-1 RNA ≥ 20,000 copies/mL, and were antiretroviral-naïve (defined as having <2 weeks of prior treatment with any NRTI and no exposure to PIs). Women of child-bearing poten- tial could be considered for the study if they had a nega- tive serum pregnancy test (β-human chorionic gonad- otropin) at screening and practiced birth control (used double barrier method of contraception or intrauterine device, or were abstinent) throughout the study. There were no CD4+ cell count restrictions on eligibility. Key exclusion criteria included: active AIDS-defining opportunistic infection or disease (CDC Category C) or pre-existing mental, physical, or substance abuse disorder which, in the opinion of the investigator, would preclude the patient from participation; use of immunomodulators, systemic cytotoxic chemotherapy, or radiation therapy within 4 weeks prior to study entry; exposure to an HIV vaccine within 3 months prior to study entry; clinically relevant hepatitis within 6 months prior to screening; or concurrent use of any agent known to have a phar- macologic interaction with the boosted PI. All patients provided written informed consent to participate in the study, and the protocol was approved by the institutional review boards for the study sites. The study was con- ducted according to Good Clinical Practice and the 2000 Helsinki Declaration. During the induction phase of the study (baseline through week 48), the patients received one Trizivir tab- let plus one LPV/r 400/100 mg fixed-dose combination capsule (Kaletra®, Abbott Laboratories, North Chicago, Illinois) twice daily. Patients achieving a viral load (HIV-1 RNA) < 50 copies/mL at weeks 36 and 44 were eligible to enroll in the maintenance phase, during which LPV/r was discontinued at week 48 and Trizivir alone was continued through week 96. Patients were evaluated during the induction phase at weeks 1, 2, 4, 8, 16, 24, 36, 44, and 48, and during the maintenance phase at weeks 56, 64, 72, 84, and 96. Rou- tine biochemistry, hematology, and immunology tests were conducted at each assessment. The severity of ad- verse events was graded according to DAIDS criteria [18]. If any patient were to develop a suspected abacavir- related hypersensitivity reaction, Trizivir was to be dis- continued immediately. Median lipid values were as- sessed as to whether they exceeded the National Choles- terol Education Program (NCEP) cutoff levels at which hypolipidemic drug therapy would be indicated [19]. Viral load was measured using the Roche Amplicor MONITOR Ultrasensitive assay (version 1.5; lower limit of quantitation [LLOQ] 50 copies/mL) (Roche Diagnos- tics, Branchburg, New Jersey) and HIV-1 MONITOR Version 1.0 polymerase chain reaction (PCR) assay (LLOQ, 400 copies/mL) (Roche, Nutley, New Jersey). CD4+ lymphocyte cell count was assessed by flow cy- tometry. Adherence was monitored by a self-dministered questionnaire, the PMAQ3 [20]. The primary endpoint of the study was the proportion of patients with plasma HIV-1 RNA < 50 copies/mL at the end of the maintenance phase (week 96). Treatment failure was defined as either virologic failure (repeated [consecutive] detection of plasma HIV-1 RNA > 500 copies/mL after having achieved an HIV-1 RNA < 50 copies/mL) or discontinuation because of toxicity, clini- cal disease progression, or death. For virologic response, both intent-to-treat: missing equals failure (ITT: M = F) and observed analyses were performed. All other pa- rameters were reported using observed analyses only. Descriptive-only statistical analyses were applied to ef- ficacy and safety data and comparisons. 3. Results 3.1. Patient Characteristics A total of 23 patients were enrolled at the three study sites. Most of the patients (18 [78%]) were males and Copyright © 2012 SciRes. WJA  Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study 247 African American (14 [61%], with 9 [39%] Caucasians). The median age was 37 years (range, 20 to 74 years). Median baseline plasma HIV-1 RNA level was 5.45 log10 copies/mL (range, 4.28 to 5.88 log10 copies/mL). Nine patients (39%) had an HIV-1 RNA < 100,000 copies/mL, and 14 (61%) an HIV-1 RNA ≥ 100,000 copies/mL. Median baseline CD4+ cell count was 232 cells/mm3 (range, 19 to 504 cells/mm3), with 12 (52%) ≥ 200 cells/mm3 and 11 (48%) < 200 cells/mm3. The baseline CDC classification was Category A in 16 patients (70%), Category B in six (26%), and Category C in one (4%). 3.2. Virologic and Immunologic Response Of the 23 patients who started induction, 19 completed it, three were lost to follow-up, and one withdrew due to grade 2 adverse events (combination of diarrhea/fatigue/ nausea/vomiting). Of the 19 induction completers, 14 had viral load measurements done at week 48 (five patients missed their week 48 study visit). Week 48 ITT: observed analysis indicated viral load < 400 copies/mL in 100% of the 14 induction completers and < 50 copies/mL in 71% (10) (Figure 1). ITT: M = F analysis showed these levels of viral load suppression in 14/23 (61%) and 10/23 (43%), respectively. As Figure 1 indicates, a drop in percentage of patients with viral load <50 copies/mL occurred between week 32 and week 48 that may possibly have been due to non-adherence in a few patients. Median CD4+ increase above baseline was 192 cells/mm3 at week 48 (Figure 2). Figure 1. Proportion of patients with HIV-1 RNA < 50 cop- ies/mL. Figure 2. Changes in CD4+ cell count. Nineteen patients entered the maintenance phase of the study. Twelve patients completed the entire maintenance phase through week 96, of whom 92% (11/12) achieved viral loads of <400 and <50 copies/mL in the ITT: ob- served analysis and 48% (11/23) in the ITT: M = F analysis (Figure 1). One of the 12 maintenance com- pleters had an insufficient viral load at his last two study visits (470 copies/mL at week 84 and 2000 copies/mL at week 96), and non-adherence may have been a causal factor for this. Of the seven patients who dropped out of the maintenance phase prematurely, one had met the cri- teria of virologic failure with two consecutive viral load measurements of >500 copies/mL (at weeks 64 and 72), three had missed their week 96 study visit (all had had a viral load < 50 copies/mL at every maintenance visit through week 84), and three had had a single insufficient viral load at one study visit and subsequently withdrew from the study. Median change from baseline in CD4+ cell count was +208 cells/mm3 at week 96 (Figure 2). 3.3. Safety and Tolerability During the study, all 23 patients experienced at least one adverse event that was grade 1 to 2 (mild to moderate) in severity. Adverse events that investigators considered to be treatment-related were reported by 13 patients (57%); these included diarrhea (two), nausea (four), fatigue (four), rash (two), abdominal pain (two), vomiting (two), and alopecia, abdominal distention, upper abdominal pain, hyperlipidemia, dizziness, erectile dysfunction, and upper respiratory tract infection (one case each). Most of these adverse events were reported within the first 30 days of the induction period, with the exceptions being one report each of diarrhea (at 62 days), fatigue (at 43 days), rash (at 96 days), and upper respiratory tract infec- tion (at 385 days—the only adverse event reported dur- ing the maintenance phase). No suspected abacavir-re- lated hypersensitivity reactions occurred. Median values of all lipids increased above baseline values during the Trizivir-LPV/r induction phase and gen- erally decreased during the Trizivir-only maintenance phase (Figure 3). Median lipid concentrations at base-line, end of induction (week 48), and end of maintenance (week 96) were 165.5, 215.0, and 194.0 mg/dL, respectively, for total cholesterol; 102.5, 129.5, and 119.5 mg/dL, respectively, for LDL-cholesterol; 36.5, 45.0, and 39.0 mg/dL, respectively, for HDL-cholesterol; and 137.0, 208.5, and 141.0 mg/dL, respectively, for triglycerides. Although median total cho- lesterol and triglyceride concentrations exceeded the NCEP cutoff levels at the end of induction, the medians for these lipids fell below the cutoffs during maintenance. Of the 12 patients who completed both phases of the study, the number whose total cholesterol exceeded the NCEP cutoff (200 mg/dL) at baseline, end of induction, and end of Copyright © 2012 SciRes. WJA 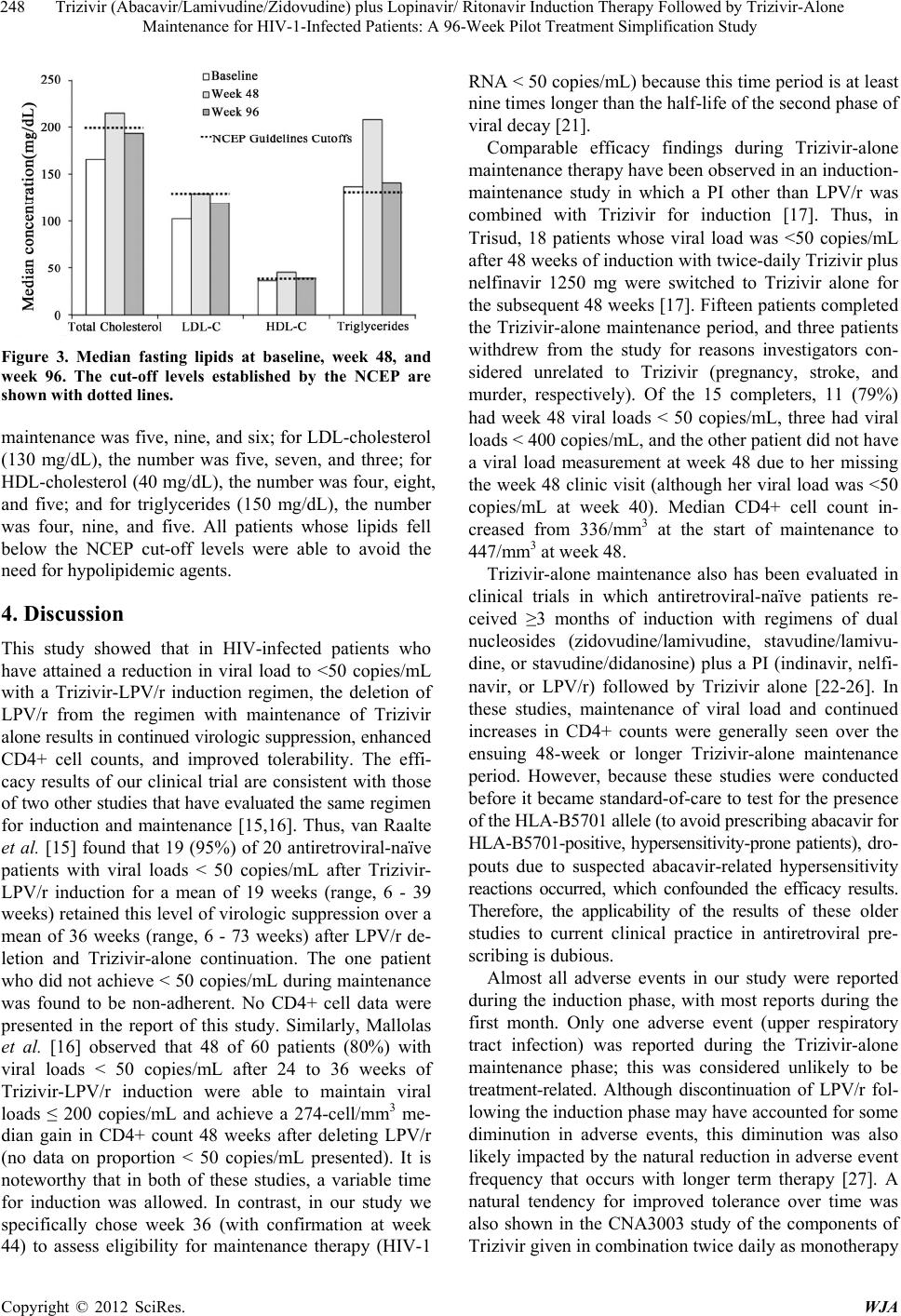 Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study 248 Figure 3. Median fasting lipids at baseline, week 48, and week 96. The cut-off levels established by the NCEP are shown with dotted lines. maintenance was five, nine, and six; for LDL-cholesterol (130 mg/dL), the number was five, seven, and three; for HDL-cholesterol (40 mg/dL), the number was four, eight, and five; and for triglycerides (150 mg/dL), the number was four, nine, and five. All patients whose lipids fell below the NCEP cut-off levels were able to avoid the need for hypolipidemic agents. 4. Discussion This study showed that in HIV-infected patients who have attained a reduction in viral load to <50 copies/mL with a Trizivir-LPV/r induction regimen, the deletion of LPV/r from the regimen with maintenance of Trizivir alone results in continued virologic suppression, enhanced CD4+ cell counts, and improved tolerability. The effi- cacy results of our clinical trial are consistent with those of two other studies that have evaluated the same regimen for induction and maintenance [15,16]. Thus, van Raalte et al. [15] found that 19 (95%) of 20 antiretroviral-naïve patients with viral loads < 50 copies/mL after Trizivir- LPV/r induction for a mean of 19 weeks (range, 6 - 39 weeks) retained this level of virologic suppression over a mean of 36 weeks (range, 6 - 73 weeks) after LPV/r de- letion and Trizivir-alone continuation. The one patient who did not achieve < 50 copies/mL during maintenance was found to be non-adherent. No CD4+ cell data were presented in the report of this study. Similarly, Mallolas et al. [16] observed that 48 of 60 patients (80%) with viral loads < 50 copies/mL after 24 to 36 weeks of Trizivir-LPV/r induction were able to maintain viral loads ≤ 200 copies/mL and achieve a 274-cell/mm3 me- dian gain in CD4+ count 48 weeks after deleting LPV/r (no data on proportion < 50 copies/mL presented). It is noteworthy that in both of these studies, a variable time for induction was allowed. In contrast, in our study we specifically chose week 36 (with confirmation at week 44) to assess eligibility for maintenance therapy (HIV-1 RNA < 50 copies/mL) because this time period is at least nine times longer than the half-life of the second phase of viral decay [21]. Comparable efficacy findings during Trizivir-alone maintenance therapy have been observed in an induction- maintenance study in which a PI other than LPV/r was combined with Trizivir for induction [17]. Thus, in Trisud, 18 patients whose viral load was <50 copies/mL after 48 weeks of induction with twice-daily Trizivir plus nelfinavir 1250 mg were switched to Trizivir alone for the subsequent 48 weeks [17]. Fifteen patients completed the Trizivir-alone maintenance period, and three patients withdrew from the study for reasons investigators con- sidered unrelated to Trizivir (pregnancy, stroke, and murder, respectively). Of the 15 completers, 11 (79%) had week 48 viral loads < 50 copies/mL, three had viral loads < 400 copies/mL, and the other patient did not have a viral load measurement at week 48 due to her missing the week 48 clinic visit (although her viral load was <50 copies/mL at week 40). Median CD4+ cell count in- creased from 336/mm3 at the start of maintenance to 447/mm3 at week 48. Trizivir-alone maintenance also has been evaluated in clinical trials in which antiretroviral-naïve patients re- ceived ≥3 months of induction with regimens of dual nucleosides (zidovudine/lamivudine, stavudine/lamivu- dine, or stavudine/didanosine) plus a PI (indinavir, nelfi- navir, or LPV/r) followed by Trizivir alone [22-26]. In these studies, maintenance of viral load and continued increases in CD4+ counts were generally seen over the ensuing 48-week or longer Trizivir-alone maintenance period. However, because these studies were conducted before it became standard-of-care to test for the presence of the HLA-B5701 allele (to avoid prescribing abacavir for HLA-B5701-positive, hypersensitivity-prone patients), dro- pouts due to suspected abacavir-related hypersensitivity reactions occurred, which confounded the efficacy results. Therefore, the applicability of the results of these older studies to current clinical practice in antiretroviral pre- scribing is dubious. Almost all adverse events in our study were reported during the induction phase, with most reports during the first month. Only one adverse event (upper respiratory tract infection) was reported during the Trizivir-alone maintenance phase; this was considered unlikely to be treatment-related. Although discontinuation of LPV/r fol- lowing the induction phase may have accounted for some diminution in adverse events, this diminution was also likely impacted by the natural reduction in adverse event frequency that occurs with longer term therapy [27]. A natural tendency for improved tolerance over time was also shown in the CNA3003 study of the components of Trizivir given in combination twice daily as monotherapy Copyright © 2012 SciRes. WJA  Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study 249 [28]. Thus, in the CNA3003 comparison of total inci- dence of adverse events at weeks 0 (baseline) to 16 ver- sus at weeks 16 to 48, the following frequency rates were observed: nausea, 47% vs 8%; malaise/fatigue, 37% vs 8%; headache, 31% vs 13%, and nausea with vomiting, 16% vs 5% [28]. Our study demonstrated that lipids increased during the Trizivir-LPV/r induction period, but improved after LPV/r was stopped and Trizivir alone was continued for maintenance. During induction, median concentrations for the treatment group rose above NCEP cutoffs for total cholesterol and triglycerides. Several patients in whom lipid concentrations exceeded NCEP cutoffs during in- duction, and hence were candidates for hypolipidemic agents at that study point, were able to obviate the need for these agents during Trizivir-alone maintenance be- cause their lipids fell below NCEP cutoffs in the latter phase. Mallolas et al. [16] observed a similar pattern of change in total cholesterol, HDL-cholesterol, and triglyc- erides during Trizivir-LPV/r induction (median change from baseline of +30.5, +8, and +65 mg/dL, respectively) and Trizivir-alone maintenance (median change from end of induction to end of study of −25, −2.7, and −62 mg/dL, respectively). Comparable improvement in lipids has also been reported in studies of patients who have switched from Trizivir-efavirenz induction to Trizivir-alone main- tenance [10,11,29]. Our study was limited by its non-randomized design, the small size of the study population, and the unavail- ability of resistance information in patients who had in- sufficient virologic response or failure. However, resis- tance data from the Mallolas et al. study of Trizivir- LPV/r induction with Trizivir-alone maintenance re- vealed that primary treatment-emergent mutations ob- served in virologic failures were either M184V alone or M184V plus a thymidine analogue-associated mutation [16]. Use of a quality-of-life measuring tool also would have been valuable to detect any changes in patient att- itudes toward and satisfaction with their antiretroviral therapy following the change from the induction to the maintenance regimen. In conclusion, in this pilot study, Trizivir-alone main- tenance after Trizivir-LPV/r induction resulted in sus- tained virologic and CD4+ cell response and an im- proved adverse event and lipid profile. 5. Acknowledgements This investigator-initiated study was funded by Glaxo- SmithKline. We gratefully acknowledge the patients who participated in this study. We thank Terry Paul, PhD, of Paul Medical Writing, Inc. for his writing and editorial assistance in the preparation of the manuscript. Results of this study were presented in part in Abstract No. CDB0480 at the XVI International AIDS Conference, Toronto, Canada, August 13-18, 2006. REFERENCES [1] M. E. Curlin, T. Wilkin and J. Mittler, “Induction-Main- tenance Therapy for HIV-1 Infection,” Future of HIV Therapy, Vol. 2, No. 2, 2008, pp. 175-185. doi:10.2217/17469600.2.2.175 [2] M. H. E. Reijers, G. J. Weverling, S. Jurriaans, et al., “Maintenance Therapy after Quadruple Induction Therapy in HIV-1-Infected Individuals: Amsterdam Duration of Antiretroviral Medication (ADAM) Study,” Lancet, Vol. 352, No. 9123, 1998, pp. 185-190. doi:10.1016/S0140-6736(98)06193-5 [3] G. Pialoux, F. Raffi, F. Brun-Vezinet, et al., “A Rando- mized Trial of Three Maintenance Regimens Given after Three Months of Induction Therapy with Zidovudine, Lamivudine, and Indinavir in Previously Untreated HIV- 1-Infected Patients,” New England Journal of Medicine, Vol. 339, No. 18, 1998, pp. 1269-1276. doi:10.1056/NEJM199810293391802 [4] J. R. Arribas, F. Puliod, R. Delgado, et al., “Lopinavir/ Ritonavir as Single-Drug Therapy for Maintenance of HIV-1 Viral Suppression: 48-Week Results of a Rando- mized, Controlled, Open-Label, Proof-of-Concept Pilot Clinical Trial (OK Study),” Journal of Acquired Immune Deficiency Syndrome, Vol. 40, No. 3, 2005, pp. 280-287. doi:10.1097/01.qai.0000180077.59159.f4 [5] R. E. Campo, R. Lalanne, T. J. Tanner, et al., “Lopinavir/ Ritonavir Maintenance Monotherapy after Successful Viral Suppression with Standard Highly Active Antiretroviral Therapy in HIV-1-Infected Patients,” AIDS, Vol. 19, No. 4, 2005, pp. 447-449. doi:10.1097/01.aids.0000161777.38438.ed [6] P. Vernazza, S. Daneel, V. Schiffer, et al., “The Role of Compartment Penetration in PI-Monotherapy: The Ataza- navir-Ritonavir Monomaintenance (ATARITMO) Trial,” AIDS, Vol. 21, No. 10, 2007, pp. 1309-1315. doi:10.1097/QAD.0b013e32814e6b1c [7] J.-F. Delfraissy, P. Flandre, C. Delaugerre, et al., “Lopi- navir/Ritonavir Monotherapy or plus Zidovudine and Lamivudine in Antiretroviral-Naive HIV-Infected Patients,” AIDS, Vol. 22, No. 3, 2008, pp. 385-393. doi:10.1097/QAD.0b013e3282f3f16d [8] S. Swindells, A. G. DiRienzo, T. Wilkin, et al., “Regimen Simplification to Atazanavir-Ritonavir Alone as Main- tenance Antiretroviral Therapy after Sustained Virologic Suppression,” Journal of the American Medical Asso- ciation, Vol. 296, No. 7, 2006, pp. 806-814. doi:10.1001/jama.296.7.806 [9] F. Pulido, J. R. Arribas, R. Delgado, et al., “Lopinavir- Ritonavir Monotherapy versus Lopinavir-Ritonavir and Two Nucleosides for Maintenance Therapy of HIV,” AIDS, Vol. 22, No. 2, 2008, pp. F1-F9. doi:10.1097/QAD.0b013e3282f4243b [10] M. Markowitz, C. Hill-Zabala, J. Lang, et al., “Induction with Abacavir/Lamivudine/Zidovudine Plus Efavirenz for Copyright © 2012 SciRes. WJA  Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study Copyright © 2012 SciRes. WJA 250 48 Weeks Followed by 48-Week Maintenance with Abacavir/Lamivudine/Zidovudine Alone in Antiretrovi- ral-Naïve HIV-1-Infected Patients,” Journal of Acquired Immune Deficiency Syndrome, Vol. 39, No. 3, 2005, pp. 257-264. doi:10.1097/01.qai.0000169664.15536.20 [11] S. De Wit, M. Johnson, B. Gazzard, et al., “Randomised Comparison of Maintenance Therapy with Trizivir [TZV] + Efavirenz [EFV] vs TZV in Naive HIV-1-Infected Subjects: TIME Study,” 7th International Congress on Drug Therapy in HIV Infection, Glasgow, 14-18 Novem- ber 2004, Abstract PL2.4. [12] P. Keiser and N. Nassar, “Abacavir Sulfate/Lamivudine/ Zidovudine Fixed Combination in the Treatment of HIV Infection,” Expert Opinion in Pharmacotherapy, Vol. 8, No. 4, 2007, pp. 477-483. doi:10.1517/14656566.8.4.477 [13] Panel on Antiretroviral Guidelines for Adults and Adole- scents, “Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents,” Department of Health and Human Services, 2012, pp. 1-239. http://www.aidsinfo.nih.gov/ContentFiles/AdultandAdole scentGL.pdf [14] M. A. Thompson, J. A. Aberg, P. Cahn, et al., “Antire- troviral Treatment of Adult HIV Infection 2010 Recom- mendations of the International AIDS Society, USA Panel,” Journal of the American Medical Association, Vol. 304, No. 3, 2010, pp. 321-333. doi:10.1001/jama.2010.1004 [15] R. van Raalte, B. Heere, R. Regez, et al., “Induction- Maintenance Strategy Re-Evaluated: Initial Boosted-PI in Combination with Triple NRTI, Followed by Triple NRTI Maintenance,” 15th International AIDS Conference, 11-16 July 2004, Bangkok, Abstract TuPeB4597. [16] J. Mallolas, J. Pich, M. Peñaranda, et al., “Induction Therapy with Trizivir Plus Efavirenz or Lopinavir/ Ritonavir Followed by Trizivir Alone in Naïve HIV- 1-Infected Adults,” AIDS, Vol. 22, No. 3, 2008, pp 377- 384. doi:10.1097/QAD.0b013e3282f3db2c [17] J. Reynes, J. M. Ragnaud, B. Delmas, et al., “An Open- Label Study to Evaluate Long-Term (96 Weeks) Safety and Efficacy of Switch to Trizivir After First Line Quadruple Induction Therapy: AZLF3004, Trisud,” 9th European AIDS Conference (EACS), Warsaw, 25-29 October 2003, Poster 7.5/3. [18] National Institute of Allergy and Infectious Diseases (NIAID), “Table for Grading the Severity of Adult and Pediatric Adverse Events, Version 1.0,” Division of Acquired Immunodeficiency Syndrome (DAIDS), Wash- ington DC, 2004. [19] National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III), “Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Final Report,” Circulation, Vol. 106, No. 25, 2002, pp. 3143-3421. [20] M. Duong, L. Piroth, M. Grappin, et al., “Evaluation of the Patient Medication Adherence Questionnaire as a Tool for Self-Reported Adherence Assessment in HIV- Infected Patients on Antiretroviral Regimens,” HIV Clinical Trials, Vol. 2, No. 2, 2001, pp. 128-135. doi:10.1310/M3JR-G390-LXCM-F62G [21] A. S. Perelson, P. Essunger, Y. Cao, et al., “Decay Characteristics of HIV-1-Infected Compartments during Combination Therapy,” Nature, Vol. 387, No. 6629, 1997, pp. 188-191. doi:10.1038/387188a0 [22] E. Martínez, J. A. Arnaiz, D. Podzamczer, et al., “Sub- stitution of Nevirapine, Efavirenz, or Abacavir for Pro- tease Inhibitors in Patients with Human Immuno-Defi- ciency Virus Infection,” New England Journal of Medi- cine, Vol. 349, No. 11, 2003, pp. 1036-1046. doi:10.1056/NEJMoa021589 [23] C. Katlama, S. Fenske, B. Gazzard, et al., “TRIZAL Study: Switching from Successful HAART to Trizivir™ (Abacavir-Lamivudine-Zidovudine Combination Tablet): 48 Weeks Efficacy, Safety and Adherence Results,” HIV Medicine, Vol. 4, No. 2, 2002, pp. 79-86. doi:10.1046/j.1468-1293.2003.00139.x [24] M. Opravil, B. Hirschel, A. Lazzarin, et al., “A Rando- mized Trial of Simplified Maintenance Therapy with Abacavir, Lamivudine, and Zidovudine in Human Immu- nodeficiency Virus Infection,” Journal of Infectious Dis- eases, Vol. 185, No. 9, 2005, pp. 1251-1260. doi:10.1086/340312 [25] N. Clumeck, F. Goebel, W. Rozenbaum, et al., “Simp- lification with Abacavir-Based Triple Nucleoside Therapy versus Continued Protease Inhibitor-Based Highly Active Antiretroviral Therapy in HIV-1-Infected Patients with Undetectable Plasma HIV-1 RNA,” AIDS, Vol. 15, No. 12, 2001, pp. 1517-1526. doi:10.1097/00002030-200108170-00009 [26] H. G. Sprenger, N. Langebeek, P. G. H. Mulder, et al., “Abacavir/Lamivudine/Zidovudine Maintenance after Stan- dard Induction in Antiretroviral Therapy-Naïve Patients: FREE Randomized Trial Interim Results,” AIDS Patient Care and STDs, Vol. 24, No. 6, 2010, pp. 361-366. doi:10.1089/apc.2009.0236 [27] T. Hawkins, “Understanding and Managing the Adverse Effects of Antiretroviral Therapy,” Antiviral Research, Vol. 85, No. 1, 2010, pp. 201-209. doi:10.1016/j.antiviral.2009.10.016 [28] C. Hicks, M. Fischl, S. Greenberg, et al., “Ziagen™ (Abacavir, ABC, 1592) Combined with 3TC and ZDV Provides a Potent and Durable Response Through 48 Weeks in HIV-1-Infected Antiretroviral Therapy (ART)- Naïve Adults (CNA3003),” 37th Annual Meeting of the Infectious Disease Society of America, Philadelphia, 18- 20 November 1999, Abstract/Poster 341. [29] P. de Truchis, D. Mathez, G. Force, et al., “Long-Term Control of Viral Residual Replication under Maintenance Therapy with Trizivir after a Quadruple Induction Regi- men in HIV-1-Infected Adults (Suburbs Trial),” HIV Clinical Trials, Vol. 8, No. 2, 2007, pp. 102-104. doi:10.1310/hct0802-102  Trizivir (Abacavir/Lamivudine/Zidovudine) plus Lopinavir/ Ritonavir Induction Therapy Followed by Trizivir-Alone Maintenance for HIV-1-Infected Patients: A 96-Week Pilot Treatment Simplification Study 251 Appendix CDC: Centers for Disease Control and Prevention; DAIDS: Division of Acquired Immunodeficiency Syn- drome (of the United States National Institutes of Health); LLOQ: lower limit of quantitation; NCEP: National Cholesterol Education Program; NNRTI: non-nucleoside reverse transcriptase inhibitor; NRTI: nucleoside reverse transcriptase inhibitor; PI: protease inhibitor. Copyright © 2012 SciRes. WJA
|