 Vol.3, No.5, 702-713 (2012) Agricultural Sciences http://dx.doi.org/10.4236/as.2012.35085 Long term effects of treated wastewater irrigation on calcisol fertility: A case study of Sfax-Tunisia Nebil Belaid1,2*, Catherine Neel2,3, Monem Kallel4, Tarek Ayoub5, Abdelmoneim A yadi1, Michel Baudu2 1National School of Engineers of Sfax, Radio Analyzes and Environment laboratory (LRAE), Sfax, Tunisia; *Corresponding Author: belaidnebil@yahoo.fr 2University of Limoges, Research Group, Water, Soil and Environment (GRESE), Limoges, France 3CETE Lyon-DLCF, Clermont-Ferrand, France 4National School of Engineers of Sfax, Water, Energy and Environment Laboratory (L.3E), Sfax, Tunisia 5CRDA-Sfax, Sfax, Tunisia Received 25 April 2012; revised 28 May 2012; accepted 16 June 2012 ABSTRACT The us e of trea ted wastewater (TW) for irrigatio n is increasingly being considered as a technical solution to minimize soil degradation and to re- store nutrient content of soils. Indeed, TW usu- ally contain large amounts of nutrient elements. The objective of this study is to evaluate the impact of long-term irrigation by TW on soil fer- tility under real field conditions. In the vicinity of the city of Sfax, a semi-arid region, a calcisol field has been irrigated for more 15 years with organic sodic TW; soil was modeled at three different depths (0 - 30, 30 - 60 and 60 - 90 cm) and along soil pits in the TW irrigated zone and in a nearby non-irrigated zone (control). Several p arameters hav e been measur ed: Soil s pH, CE C, exchangeable cations, nitrate and ammonia, to- tal contents of nitrogen, phosphorus and other essential macro and micro nutrients, electrical conductivity, soil organic carbon and dissolved organic carbon. C/N ratio and SUVA were calcu- lated for each soil layer. The calculation of the isovolumic mass balance on soil profile scale was used to measure macro and micro nutrients supply. The TW irrigation has led to important supply in organic carbon (+100%), phosphorus (+80%) and in most essential nutrients (N, Mn, Zn). Due to the high rate of irrigation and low CEC of the studied soil, the added nutrient cations and nitrate are removed with leaching waters compared to the non-irrigated control soil. Moreover, Sfax’s TW bring about important amounts of salts and Na. Therefore the benefi- cial addition of nutrients could quickly be inhibi- ted by the excessive supply of salts and avail- able nitrogen. Apart from future crops produc- tion risk, groundwater degradation quality and soil fertility will be endangered over the long term. Keywords: Arid Region; Wastewater; Irrigation; Fertility; El Hajeb-Sfax 1. INTRODUCTION In arid and semi-arid regions of countries such as Tunisia which are facing rising serious water shortage problems, the reuse of urban wastewater for non potable purposes, such as agriculture [1-3] has became an usual practice. Indeed, wastewater reuse for irrigation has been the largest field of application because it usually offers some attractive environmental and socio-economic bene- fits, mainly due to the reduction of effluent disposal in receiving water bodies, to the supply of nutrients as fer- tilizers, and to the improvements in crops production during the dry season [4,5]. Benefits apart, planners are aware of the potential disadvantages of wastewater reuse for irrigation which are, aside from pathogenic contami- nation of irrigated crops, mainly related to the specific chemical composition of wastewater being somewhat different from most natural waters used in irrigation [6]. Over time, wastewater irrigation affects some soil para- meters. Increase in soil pH is observed in acidic soils [7,8], whereas slight decrease of pH is mentioned for alkaline soils [5,8-11]. In alkaline calcarous soils, the sligh acidification is due to the leaching of limestone by the leaching water [10]. Wastewater can also supply ammonia anion to soil which is another source of soil acidification [11]. In general, the decrease of pH is also explained by the low C/N ratio of effluents and the subsequent enhancement of the organic mineralization substances [7,10]. Wastewater irrigation thus usually Copyright © 2012 SciRes. OPEN A CCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 703 leads to macro and micro nutrients supply [5,11] which stimulates the microbial activity [12,13] and promotes the mineralization of the soil organic matter. This can lead to the decrease of the soil cation exchange capacity [7,10] and mitigate the soil buffer capacity. By the same way, wastewater usually contains elevated concentrations in metal elements such as Mn, Cu and Zn which cons- titute essential micro nutrients for plants. Over time, the same elements can accumulate in the organic topsoil layer in such manner that they reach their critical level for plant growth. Hence, the use of wastewater for irrigation of crops requires assessment of the balance in supply of macro and micro nutrient over the long-term. Most of previous mentioned studies recorded impacts on contents in macro and micro nutrients without any regard for changes in the total store of these elements in soil. Moreover, specific studies dedicated to highlight long-term impacts (along several decades) have involved soils that have been irrigated by untreated domestic effluents or municipal wastewaters (Rattan et al. [8]: 10 and 20 yr; Yadav et al. [5]: 30 yr; Solis et al. [10]: 50 and 100 yr). Thus, the treatment of wastewater has been generalized after the heighties on the spur of the F.A.O. guidelines for ap- plication in agriculture [4]. The treatment of urban effluents modifies their nutritional value, it is thus of great concern to assess whether the irrigation with treated wastewater (TW) still improves the soil quality or it could cause degradation to its fertility over the long term. The objectives of this study are to evaluate the changes in soil fertility and to balance its essential ele- ments in response to 15-year-long treated wastewater irrigation (TW) within the city of Sfax (the second largest city in Tunisia). In this arid region, there are many signs of extremely low groundwater levels which were registered over the last three decades due to the increas- ing number of wells especially used for irrigation of crops [14]. Treated wastewater in Sfax has thus been used for irrigation since 1989. From that period, the irrigation perimeter has regularly been expanded as to reach the area of 600 ha. New extension of the area irrigated by wastewater is planned for twice over its present surface. This study is a part of a research pro- gram which aims at evaluating the impact of waste- water application on both soil and crops properties in the arid region of Sfax. The overall goals are to aid mana- gement of crop irrigation by wastewater, to reduce overexploitation of the local groundwater resources and to improve the water recharge of groundwater. Belaid et al. [15] have evidenced negative impact on the soil salinity and sodicity, especially in the northern part of the irrigation perimeter covered by a deep permeable fluvisol. Therefore, soil salinization and sodification are mitigated by the amount of exchangeable calcium in irrigated cal- cisol fields [15]. The present study thus focuses on this type of calcareous soils, which is mostly found in the Southern part of the irrigation perimeter. 2. MATERIALS AND METHODS 2.1. Study Area The study area is settled at ten kilometres in the West next to the sewage treatment plant (Figure 1) near to the town of Sfax (approximately with one million of habi- tants) in crop fields, which are currently irrigated with treated wastewater whose plant receives domestic as well as industrial effluents from mainly canning factories and textile production. The region has an arid climate with monthly air temperature ranging from 11.3˚C to 26.7˚C, dry summer and annual rainfalls of 200 mm mostly oc- curring from October to December. The average annual potential evaporation of 1200 mm, combined with the low rainfall and high temperatures makes irrigation es- sential for crop production. The present survey has been carried out in the area of the irrigation perimeter that is covered by a calcisol (ac- cording to the FAO World Reference Base for soil re- sources [16]). This soil presents an isohumic character and shows a homogeneous sandy to sandy loam texture. As shown in Table 1, the selected area produces alter- nate cycles of crops, in association with permanent har- vesting of olives, with successive winter and summer harvest of annual crops (oat, sorghum) sectioned every 10 years by a 3-year-long cropping of alfalfa. This kind of cropping system requires irrigation by open surface furrows distributed every 24 m in-between each row of olive trees. The soil has been submitted to wastewater irrigation for 15 years. In order to assess the effects of the wastewater, a nearby field is taken as a control area which produces only olives and has been preserved from any source of irrigation (Figure 1). 2.2. Samples of Preparation and Chemical Analyses Treated wastewater were sampled at the outlet of the Sfax wastewater treatment plant at different times and conserved at 4˚C before characterization. Effluent sam- ples were analyzed for pH and electrical conductivity (ECw) using a pH meter (AFNOR standard method N˚ NF T 90-008 [17]) and a conductimeter (AFNOR N˚ NF EN 27888 [17]) respectively. Chemical oxygen demand (COD), suspended solids (SS), biochemical oxygen de- mand (BOD) and total phosphorus were measured ac- cording to standard methods (AFNOR N˚ NF T 90-018, NF EN 872, NF T 90-103, NF EN 1189 [17]). Cations and anions were measured using chromatography while Copyright © 2012 SciRes. OPEN A CCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 Copyright © 2012 SciRes. 704 Figure 1. Map of studied area with location of the Sfax water treatment plant and of the calcisol site (TW irrigation perimeter in grey and point for the control site). Table 1. Main characteristics of the studied calcisol site. Soil taxonomy Light texture isohumic calcimagnesic soil according to the Tunisian pedological map Soil depth Moderately deep soil laid over a limestone crust approximately 60 cm deep (the crust was dismantled in large part of the irrigated area) Soil texture Sandy to sandy-loam with calcareous nodules in subsurface Soil bulk density (surface layer) 1.4 g/cm3 Total CaCO3 5% to 35% Irrigated area Control area (not irrigated) Cultural system Associated cultivars (olives trees and forages crops) Only olives trees Crop rotation Winter (oat. ray grass) summer (sorghum) annual (alfalfa) - Field area 270 hectares including 90 hectares used for summer crops. 1.5 hectares TW irrigation rate 1000 mm/yr - Irrigation system Surface irrigation by furrows - Irrigation duration 15 years - Number of cores sampled 7 1 carbonates and bicarbonates were estimated by titration with HCl of an aliquot of the effluent samples (AFNOR N˚ NF EN ISO 9963-2 [17]). Soil sampling was performed in October 2006 after the harvest of summer crops and before the seeding of winter crops. Two soil sampling survey have been carried out. The first one concerned each horizon of soil that has been identified along pedological profiles drilled in the control area and in the irrigated field whereas the second sampling was done on plots covered by summer crops only by 30 cm thick soil layers using an Edelman-type auger. In order to account for spatial variations of soil texture and depth, 7 replications were performed in the irrigated field (IWC1 to IWC7). The control area was too small to allow such replications (NIC). Only 2 soil layers down to the depth of 60 cm were sampled in the control field as well as in the replication site IWC5 because of the occurrence of a concrete calcareous crust at depth of 60 cm. This crust of sedimentary origin is irregular and has been generally dismantled in the irrigated field in OPEN ACCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 705 order to help infiltration of irrigation waters, except in the control area, which has never been irrigated. After air-drying, the soil samples were sieved at 2 mm. Soil pHw and pHKCl were measured in a 2.5 soil to wa- ter/KCl 1 M slurry using a glass electrode. Saturation paste extracts of soils were prepared to determine the electrical conductivity of the soil samples (ECs). Soil samples CEC and contents in exchangeable cations were determined at actual soil pH by the cobaltihexamine me- thod [18]. In calcareous soils, Belaid et al., [15] evi- denced that the cobaltihexamine method provided more accurate values than the more usual method using 1 M NH4 acetate solution [19] because of lesser dissolution of carbonates. Concentrations in Ca and Mg were performed by Atomic Absorption Spectrometry (AAS) while Na and K concentrations were determined by Flame Atom Emis- sion Spectrometry (FAES). NO3 and NH4 concentrations were measured in water soluble extracts using ionic chromatography (DIONEX DX-120) after water extraction using 1:5 soil to water ratio for 2 h. Total nitrogen was determined by steam distillation after acid digestion using the Kjeldal proce- dure. Soil organic carbon (SOC) was determined by the Walkley and Black dichromate oxidation method. Water soluble organic C, which is further referred to as dis- solved organic carbon in this paper (DOC), was extracted with deionized water using a 2:1 (water to soil) ratio by shaking at fixed temperature during 3 hours [20]. The extracts were filtered using 0.22 mm filters and DOC concentration was measured using UV-persulfate oxida- tion on a TOC analyzer (TEKMAR DOHRMANN Phoe- nix 8000). UV absorption at 254 nm was measured using a Varian CARY 50 Probe UV-visible spectrophotometer. DOC quality; in terms of aromaticity of organic com- pounds, was determined as specific ultra violet absorb- ance (SUVA) which was calculated by dividing the ab- sorbance at 254 nm by the DOC. 2.3. Statistical Analysis One-sample T-test was used for comparing mean val- ues obtained from replicates of measurements at an irri- gated site to the values measured at the corresponding non irrigated control one. All measured values corre- spond to the average composite samples. However, due to the limited area of the control sites, and since the sam- pling was limited to the central part of them in order to avoid any influence of the neighbouring irrigation then in the absence of sampling replicate in the control area, no variance can be associated with the control values. Hence, we have assumed that the values of the control site represent exact means to be compared with the vari- ance of the mean values (N = 7) measured at the corre- sponding irrigated site. Variance is expected to be larger in the irrigated zone than in the corresponding control one, so that the following sample T-tests can be consid- ered as conservative: mean ofTWE irrigated replicatedvalues-control value standard deviation ofTWEirrigated replicated values T A unilateral T-test was used to calculate the parameters, which are clearly increased or decreased after the irriga- tion by the treated wastewater (ECs, Exchangeable ca- tions, NO3 and NH4). Whereas the bilateral T-test was chosen to identify parameters presenting no obvious re- sponse to the irrigation (pH, CEC, SOC). The global risk increases with the number of simultaneous tests per- formed. Therefore, we have also adopted a more severe individual rule than the usual one to minimize the in- crease of the global risk; the differences are considered significant when p < 1% instead of p < 5%. In case of p ranging between 1% < p < 5%, we conclude that the dif- ferences have to be confirmed. The T-tests were per- formed using SYSTAT Software version 12 http://www.systat.com/Store.aspx. 3. RESULTS 3.1. Treated Wastewater Characteristics During the wastewater survey, the applied treated wastewater (TW) was always remained alkaline with an average basic pH value of 7.7 (Table 2). It also always presented a high level of total dissolved solids (TDS) of 3.7 g·L−1 and of suspended matter (SS). The level of bio- chemical oxygen demand (BOD) and chemical oxygen demand (COD) are ranged respectively between 37 and 220 mg·L−1 and 123 and 700 mg·L−1. The mean electrical conductivity (EC) of effluents reaches 5.7 mS/cm. The sodium absorption ratio (SARw) of the treated wastewa- ter ranges between 9.7 and 15.6. Theses parameters are higher than the usual ranges reported for other Tunisian TW [20] of similar mixed origins (industrial and domes- tic) than those of the Sfax TW. The Sfax’ TW also con- tains great amounts of nitrate, phosphate and potassium which are crucial nutrients for plant growth and soil fer- tility. 3.2. TW Irrigation Impacts on the Soil Proprieties In all sampling points in the irrigated field (Table 3) and in the two soil profiles, the pH (pHw and pHKCl) of the soil water extracts remains alkaline at all depths (Ta- ble 4). There are no significant differences in soil pHw between the wastewater irrigated calcisol and the control calcisol. By the same way, there are no significant differences in the contents of exchangeable Ca2+ between the irri- Copyright © 2012 SciRes. OPEN A CCESS 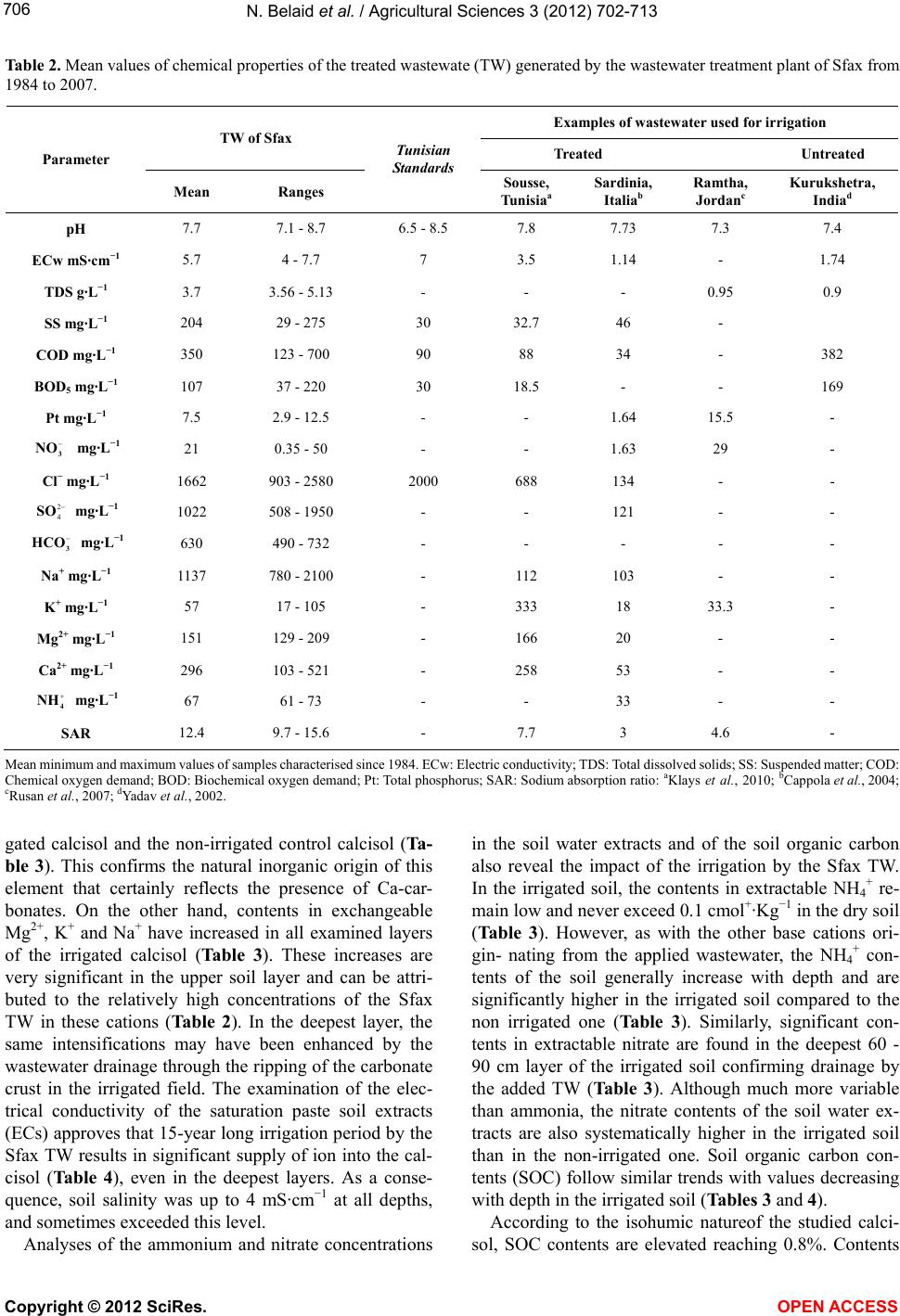 N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 Copyright © 2012 SciRes. 706 Table 2. Mean values of chemical properties of the treated wastewate (TW) generated by the wastewater treatment plant of Sfax from 1984 to 2007. Examples of wastewater used for irrigation TW of Sfax Treated Untreated Parameter Mean Ranges Tunisian Standards Sousse, Tunisiaa Sardinia, Italiab Ramtha, Jordanc Kurukshetra, Indiad pH 7.7 7.1 - 8.7 6.5 - 8.5 7.8 7.73 7.3 7.4 ECw mS·cm−1 5.7 4 - 7.7 7 3.5 1.14 - 1.74 TDS g·L−1 3.7 3.56 - 5.13 - - - 0.95 0.9 SS mg·L−1 204 29 - 275 30 32.7 46 - COD mg·L−1 350 123 - 700 90 88 34 - 382 BOD5 mg·L−1 107 37 - 220 30 18.5 - - 169 Pt mg·L−1 7.5 2.9 - 12.5 - - 1.64 15.5 - 3 NO mg·L−1 21 0.35 - 50 - - 1.63 29 - Cl− mg·L−1 1662 903 - 2580 2000 688 134 - - 2 4 SO mg·L−1 1022 508 - 1950 - - 121 - - 3 HCO mg·L−1 630 490 - 732 - - - - - Na+ mg·L−1 1137 780 - 2100 - 112 103 - - K+ mg·L−1 57 17 - 105 - 333 18 33.3 - Mg2+ mg·L−1 151 129 - 209 - 166 20 - - Ca2+ mg·L−1 296 103 - 521 - 258 53 - - 4 NH mg·L−1 67 61 - 73 - - 33 - - SAR 12.4 9.7 - 15.6 - 7.7 3 4.6 - Mean minimum and maximum values of samples characterised since 1984. ECw: Electric conductivity; TDS: Total dissolved solids; SS: Suspended matter; COD: Chemical oxygen demand; BOD: Biochemical oxygen demand; Pt: Total phosphorus; SAR: Sodium absorption ratio: aKlays et al., 2010; bCappola et al., 2004; cRusan et al., 2007; dYadav et al., 2002. gated calcisol and the non-irrigated control calcisol (Ta- ble 3). This confirms the natural inorganic origin of this element that certainly reflects the presence of Ca-car- bonates. On the other hand, contents in exchangeable Mg2+, K+ and Na+ have increased in all examined layers of the irrigated calcisol (Table 3). These increases are very significant in the upper soil layer and can be attri- buted to the relatively high concentrations of the Sfax TW in these cations (Table 2). In the deepest layer, the same intensifications may have been enhanced by the wastewater drainage through the ripping of the carbonate crust in the irrigated field. The examination of the elec- trical conductivity of the saturation paste soil extracts (ECs) approves that 15-year long irrigation period by the Sfax TW results in significant supply of ion into the cal- cisol (Table 4), even in the deepest layers. As a conse- quence, soil salinity was up to 4 mS·cm−1 at all depths, and sometimes exceeded this level. Analyses of the ammonium and nitrate concentrations in the soil water extracts and of the soil organic carbon also reveal the impact of the irrigation by the Sfax TW. In the irrigated soil, the contents in extractable NH4 + re- main low and never exceed 0.1 cmol+·Kg−1 in the dry soil (Table 3). However, as with the other base cations ori- gin- nating from the applied wastewater, the NH4 + con- tents of the soil generally increase with depth and are significantly higher in the irrigated soil compared to the non irrigated one (Table 3). Similarly, significant con- tents in extractable nitrate are found in the deepest 60 - 90 cm layer of the irrigated soil confirming drainage by the added TW (Table 3). Although much more variable than ammonia, the nitrate contents of the soil water ex- tracts are also systematically higher in the irrigated soil than in the non-irrigated one. Soil organic carbon con- tents (SOC) follow similar trends with values decreasing with depth in the irrigated soil (Tables 3 and 4). According to the isohumic natureof the studied calci- ol, SOC contents are elevated reaching 0.8%. Contents s OPEN ACCESS 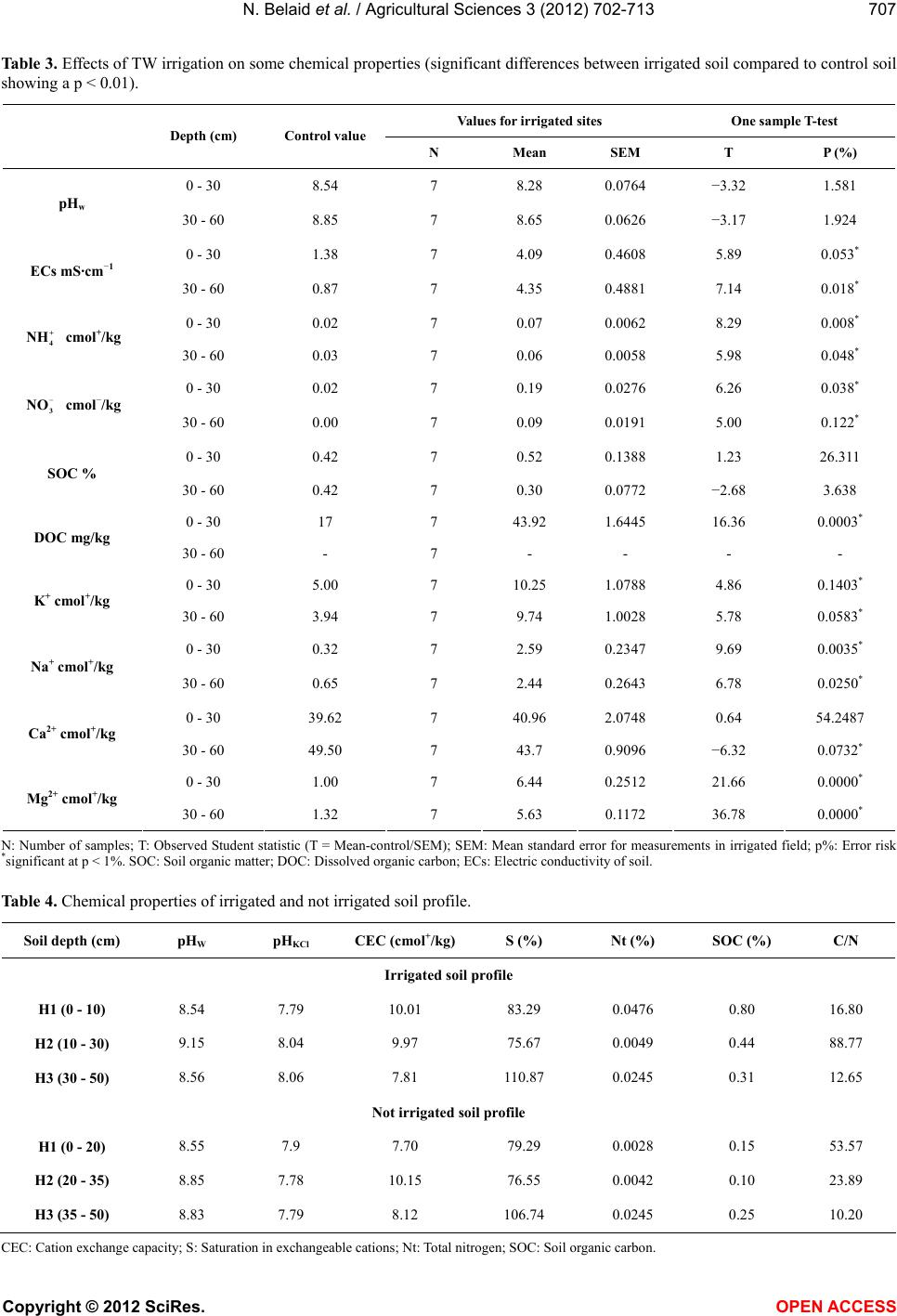 N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 707 Table 3. Effects of TW irrigation on some chemical properties (significant differences between irrigated soil compared to control soil showing a p < 0.01). Values for irrigated sites One sample T-test Depth (cm) Control value N Mean SEM T P (%) 0 - 30 8.54 7 8.28 0.0764 −3.32 1.581 pHw 30 - 60 8.85 7 8.65 0.0626 −3.17 1.924 0 - 30 1.38 7 4.09 0.4608 5.89 0.053* ECs mS·cm−1 30 - 60 0.87 7 4.35 0.4881 7.14 0.018* 0 - 30 0.02 7 0.07 0.0062 8.29 0.008* + 4 NH cmol+/kg 30 - 60 0.03 7 0.06 0.0058 5.98 0.048* 0 - 30 0.02 7 0.19 0.0276 6.26 0.038* 3 NO cmol−/kg 30 - 60 0.00 7 0.09 0.0191 5.00 0.122* 0 - 30 0.42 7 0.52 0.1388 1.23 26.311 SOC % 30 - 60 0.42 7 0.30 0.0772 −2.68 3.638 0 - 30 17 7 43.92 1.6445 16.36 0.0003* DOC mg/kg 30 - 60 - 7 - - - - 0 - 30 5.00 7 10.25 1.0788 4.86 0.1403* K+ cmol+/kg 30 - 60 3.94 7 9.74 1.0028 5.78 0.0583* 0 - 30 0.32 7 2.59 0.2347 9.69 0.0035* Na+ cmol+/kg 30 - 60 0.65 7 2.44 0.2643 6.78 0.0250* 0 - 30 39.62 7 40.96 2.0748 0.64 54.2487 Ca2+ cmol+/kg 30 - 60 49.50 7 43.7 0.9096 −6.32 0.0732* 0 - 30 1.00 7 6.44 0.2512 21.66 0.0000* Mg2+ cmol+/kg 30 - 60 1.32 7 5.63 0.1172 36.78 0.0000* N: Number of samples; T: Observed Student statistic (T = Mean-control/SEM); SEM: Mean standard error for measurements in irrigated field; p%: Error risk *significant at p < 1%. SOC: Soil organic matter; DOC: Dissolved organic carbon; ECs: Electric conductivity of soil. Table 4. Chemical properties of irrigated and not irrigated soil profile. Soil depth (cm) pHW pHKCl CEC (cmol+/kg)S (%) Nt (%) SOC (%) C/N Irrigated soil profile H1 (0 - 10) 8.54 7.79 10.01 83.29 0.0476 0.80 16.80 H2 (10 - 30) 9.15 8.04 9.97 75.67 0.0049 0.44 88.77 H3 (30 - 50) 8.56 8.06 7.81 110.87 0.0245 0.31 12.65 Not irrigated soil profile H1 (0 - 20) 8.55 7.9 7.70 79.29 0.0028 0.15 53.57 H2 (20 - 35) 8.85 7.78 10.15 76.55 0.0042 0.10 23.89 H3 (35 - 50) 8.83 7.79 8.12 106.74 0.0245 0.25 10.20 C EC: Cation exchange capacity; S: Saturation in exchangeable cations; Nt: Total nitrogen; SOC: Soil organic carbon. Copyright © 2012 SciRes. OPEN A CCESS 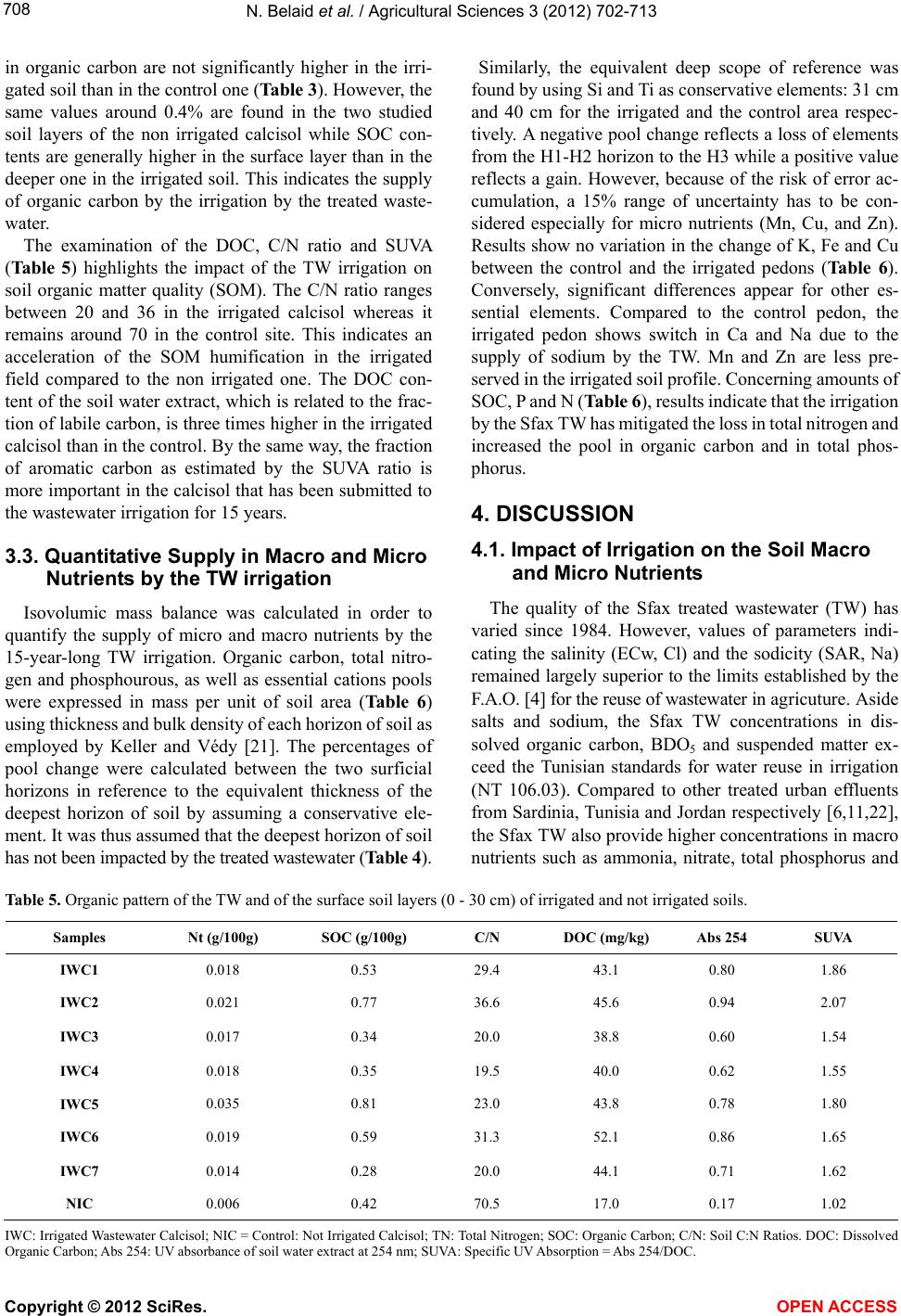 N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 708 in organic carbon are not significantly higher in the irri- gated soil than in the control one (Table 3). However, the same values around 0.4% are found in the two studied soil layers of the non irrigated calcisol while SOC con- tents are generally higher in the surface layer than in the deeper one in the irrigated soil. This indicates the supply of organic carbon by the irrigation by the treated waste- water. The examination of the DOC, C/N ratio and SUVA (Table 5) highlights the impact of the TW irrigation on soil organic matter quality (SOM). The C/N ratio ranges between 20 and 36 in the irrigated calcisol whereas it remains around 70 in the control site. This indicates an acceleration of the SOM humification in the irrigated field compared to the non irrigated one. The DOC con- tent of the soil water extract, which is related to the frac- tion of labile carbon, is three times higher in the irrigated calcisol than in the control. By the same way, the fraction of aromatic carbon as estimated by the SUVA ratio is more important in the calcisol that has been submitted to the wastewater irrigation for 15 years. 3.3. Quantitative Supply in Macro and Micro Nutrients by the TW irrigation Isovolumic mass balance was calculated in order to quantify the supply of micro and macro nutrients by the 15-year-long TW irrigation. Organic carbon, total nitro- gen and phosphourous, as well as essential cations pools were expressed in mass per unit of soil area (Table 6) using thickness and bulk density of each horizon of soil as employed by Keller and Védy [21]. The percentages of pool change were calculated between the two surficial horizons in reference to the equivalent thickness of the deepest horizon of soil by assuming a conservative ele- ment. It was thus assumed that the deepest horizon of soil has not been impacted by the treated wastewater (Table 4). Similarly, the equivalent deep scope of reference was found by using Si and Ti as conservative elements: 31 cm and 40 cm for the irrigated and the control area respec- tively. A negative pool change reflects a loss of elements from the H1-H2 horizon to the H3 while a positive value reflects a gain. However, because of the risk of error ac- cumulation, a 15% range of uncertainty has to be con- sidered especially for micro nutrients (Mn, Cu, and Zn). Results show no variation in the change of K, Fe and Cu between the control and the irrigated pedons (Table 6). Conversely, significant differences appear for other es- sential elements. Compared to the control pedon, the irrigated pedon shows switch in Ca and Na due to the supply of sodium by the TW. Mn and Zn are less pre- served in the irrigated soil profile. Concerning amounts of SOC, P and N (Table 6), results indicate that the irrigation by the Sfax TW has mitigated the loss in total nitrogen and increased the pool in organic carbon and in total phos- phorus. 4. DISCUSSION 4.1. Impact of Irrigation on the Soil Macro and Micro Nutrients The quality of the Sfax treated wastewater (TW) has varied since 1984. However, values of parameters indi- cating the salinity (ECw, Cl) and the sodicity (SAR, Na) remained largely superior to the limits established by the F.A.O. [4] for the reuse of wastewater in agricuture. Aside salts and sodium, the Sfax TW concentrations in dis- solved organic carbon, BDO5 and suspended matter ex- ceed the Tunisian standards for water reuse in irrigation (NT 106.03). Compared to other treated urban effluents from Sardinia, Tunisia and Jordan respectively [6,11,22], the Sfax TW also provide higher concentrations in macro nutrients such as ammonia, nitrate, total phosphorus and Table 5. Organic pattern of the TW and of the surface soil layers (0 - 30 cm) of irrigated and not irrigated soils. Samples Nt (g/100g) SOC (g/100g) C/N DOC (mg/kg) Abs 254 SUVA IWC1 0.018 0.53 29.4 43.1 0.80 1.86 IWC2 0.021 0.77 36.6 45.6 0.94 2.07 IWC3 0.017 0.34 20.0 38.8 0.60 1.54 IWC4 0.018 0.35 19.5 40.0 0.62 1.55 IWC5 0.035 0.81 23.0 43.8 0.78 1.80 IWC6 0.019 0.59 31.3 52.1 0.86 1.65 IWC7 0.014 0.28 20.0 44.1 0.71 1.62 NIC 0.006 0.42 70.5 17.0 0.17 1.02 IWC: Irrigated Wastewater Calcisol; NIC = Control: Not Irrigated Calcisol; TN: Total Nitrogen; SOC: Organic Carbon; C/N: Soil C:N Ratios. DOC: Dissolved Organic Carbon; Abs 254: UV absorbance of soil water extract at 254 nm; SUVA: Specific UV Absorption = Abs 254/DOC. Copyright © 2012 SciRes. OPEN A CCESS 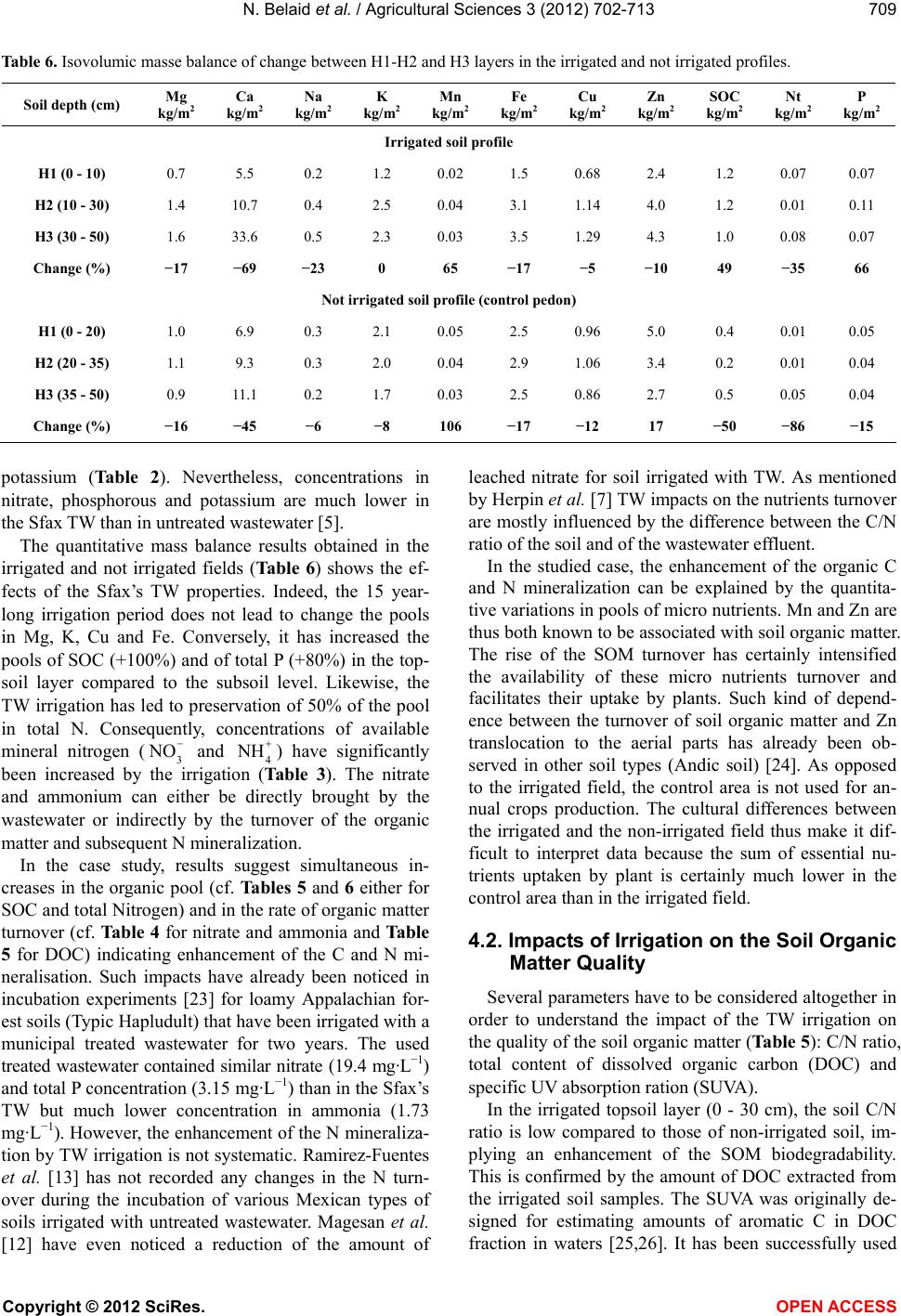 N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 709 Table 6. Isovolumic masse balance of change between H1-H2 and H3 layers in the irrigated and not irrigated profiles. Soil depth (cm) Mg kg/m2 Ca kg/m2 Na kg/m2 K kg/m2 Mn kg/m2 Fe kg/m2 Cu kg/m2 Zn kg/m2 SOC kg/m2 Nt kg/m2 P kg/m2 Irrigated soil profile H1 (0 - 10) 0.7 5.5 0.2 1.2 0.02 1.5 0.68 2.4 1.2 0.07 0.07 H2 (10 - 30) 1.4 10.7 0.4 2.5 0.04 3.1 1.14 4.0 1.2 0.01 0.11 H3 (30 - 50) 1.6 33.6 0.5 2.3 0.03 3.5 1.29 4.3 1.0 0.08 0.07 Change (%) −17 −69 −23 0 65 −17 −5 −10 49 −35 66 Not irrigated soil profile (control pedon) H1 (0 - 20) 1.0 6.9 0.3 2.1 0.05 2.5 0.96 5.0 0.4 0.01 0.05 H2 (20 - 35) 1.1 9.3 0.3 2.0 0.04 2.9 1.06 3.4 0.2 0.01 0.04 H3 (35 - 50) 0.9 11.1 0.2 1.7 0.03 2.5 0.86 2.7 0.5 0.05 0.04 Change (%) −16 −45 −6 −8 106 −17 −12 17 −50 −86 −15 potassium (Table 2). Nevertheless, concentrations in nitrate, phosphorous and potassium are much lower in the Sfax TW than in untreated wastewater [5]. The quantitative mass balance results obtained in the irrigated and not irrigated fields (Table 6) shows the ef- fects of the Sfax’s TW properties. Indeed, the 15 year- long irrigation period does not lead to change the pools in Mg, K, Cu and Fe. Conversely, it has increased the pools of SOC (+100%) and of total P (+80%) in the top- soil layer compared to the subsoil level. Likewise, the TW irrigation has led to preservation of 50% of the pool in total N. Consequently, concentrations of available mineral nitrogen (3 O and 4 H) have significantly been increased by the irrigation (Table 3). The nitrate and ammonium can either be directly brought by the wastewater or indirectly by the turnover of the organic matter and subsequent N mineralization. In the case study, results suggest simultaneous in- creases in the organic pool (cf. Tables 5 and 6 either for SOC and total Nitrogen) and in the rate of organic matter turnover (cf. Table 4 for nitrate and ammonia and Table 5 for DOC) indicating enhancement of the C and N mi- neralisation. Such impacts have already been noticed in incubation experiments [23] for loamy Appalachian for- est soils (Typic Hapludult) that have been irrigated with a municipal treated wastewater for two years. The used treated wastewater contained similar nitrate (19.4 mg·L−1) and total P concentration (3.15 mg·L−1) than in the Sfax’s TW but much lower concentration in ammonia (1.73 mg· L −1). However, the enhancement of the N mineraliza- tion by TW irrigation is not systematic. Ramirez-Fuentes et al. [13] has not recorded any changes in the N turn- over during the incubation of various Mexican types of soils irrigated with untreated wastewater. Magesan et al. [12] have even noticed a reduction of the amount of leached nitrate for soil irrigated with TW. As mentioned by Herpin et al. [7] TW impacts on the nutrients turnover are mostly influenced by the difference between the C/N ratio of the soil and of the wastewater effluent. In the studied case, the enhancement of the organic C and N mineralization can be explained by the quantita- tive variations in pools of micro nutrients. Mn and Zn are thus both known to be associated with soil organic matter. The rise of the SOM turnover has certainly intensified the availability of these micro nutrients turnover and facilitates their uptake by plants. Such kind of depend- ence between the turnover of soil organic matter and Zn translocation to the aerial parts has already been ob- served in other soil types (Andic soil) [24]. As opposed to the irrigated field, the control area is not used for an- nual crops production. The cultural differences between the irrigated and the non-irrigated field thus make it dif- ficult to interpret data because the sum of essential nu- trients uptaken by plant is certainly much lower in the control area than in the irrigated field. 4.2. Impacts of Irrigation on the Soil Organic Matter Quality Several parameters have to be considered altogether in order to understand the impact of the TW irrigation on the quality of the soil organic matter (Table 5): C/N ratio, total content of dissolved organic carbon (DOC) and specific UV absorption ration (SUVA). In the irrigated topsoil layer (0 - 30 cm), the soil C/N ratio is low compared to those of non-irrigated soil, im- plying an enhancement of the SOM biodegradability. This is confirmed by the amount of DOC extracted from the irrigated soil samples. The SUVA was originally de- signed for estimating amounts of aromatic C in DOC fraction in waters [25,26]. It has been successfully used Copyright © 2012 SciRes. OPEN A CCESS 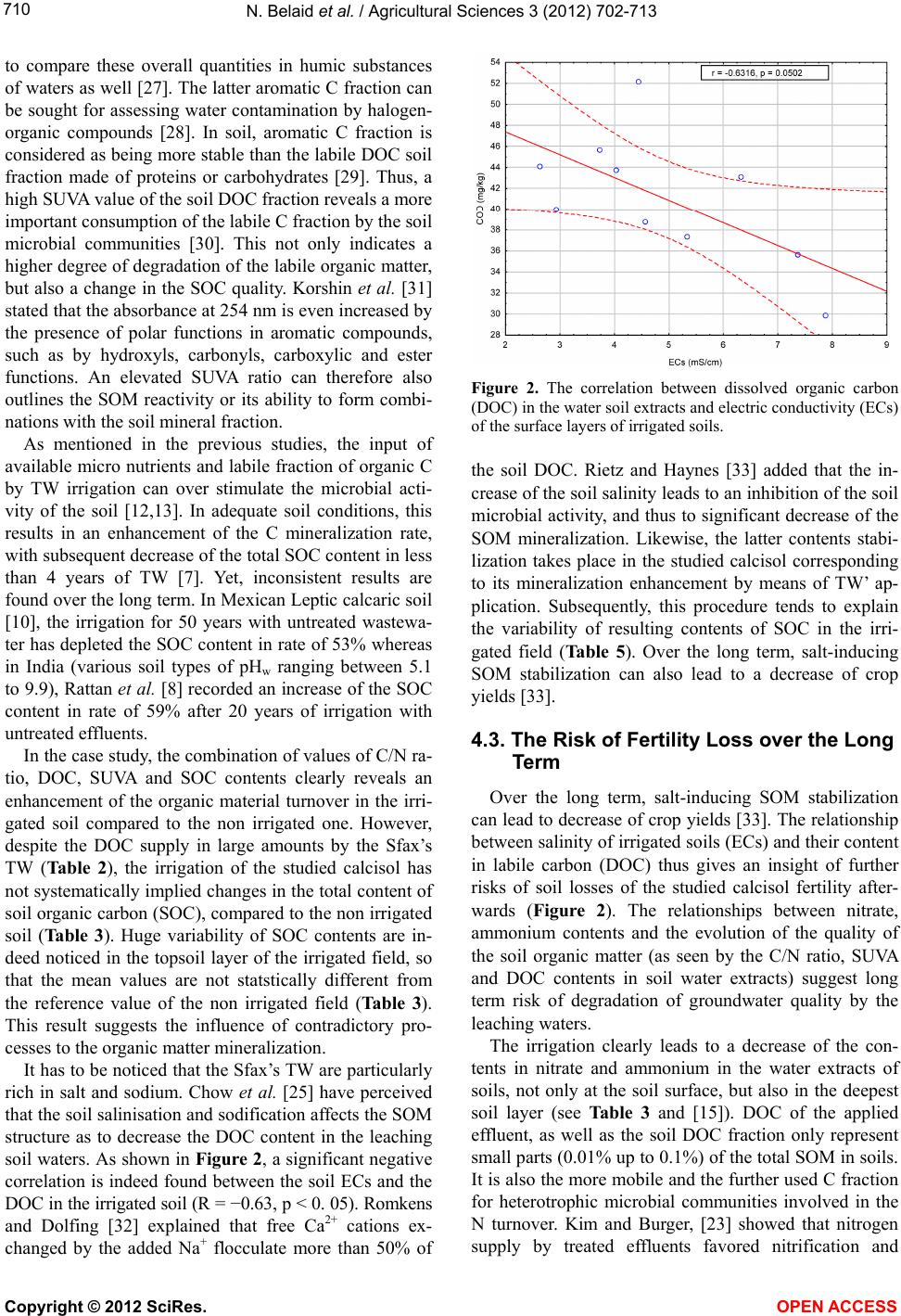 N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 710 to compare these overall quantities in humic substances of waters as well [27]. The latter aromatic C fraction can be sought for assessing water contamination by halogen- organic compounds [28]. In soil, aromatic C fraction is considered as being more stable than the labile DOC soil fraction made of proteins or carbohydrates [29]. Thus, a high SUVA value of the soil DOC fraction reveals a more important consumption of the labile C fraction by the soil microbial communities [30]. This not only indicates a higher degree of degradation of the labile organic matter, but also a change in the SOC quality. Korshin et al. [31] stated that the absorbance at 254 nm is even increased by the presence of polar functions in aromatic compounds, such as by hydroxyls, carbonyls, carboxylic and ester functions. An elevated SUVA ratio can therefore also outlines the SOM reactivity or its ability to form combi- nations with the soil mineral fraction. As mentioned in the previous studies, the input of available micro nutrients and labile fraction of organic C by TW irrigation can over stimulate the microbial acti- vity of the soil [12,13]. In adequate soil conditions, this results in an enhancement of the C mineralization rate, with subsequent decrease of the total SOC content in less than 4 years of TW [7]. Yet, inconsistent results are found over the long term. In Mexican Leptic calcaric soil [10], the irrigation for 50 years with untreated wastewa- ter has depleted the SOC content in rate of 53% whereas in India (various soil types of pHw ranging between 5.1 to 9.9), Rattan et al. [8] recorded an increase of the SOC content in rate of 59% after 20 years of irrigation with untreated effluents. In the case study, the combination of values of C/N ra- tio, DOC, SUVA and SOC contents clearly reveals an enhancement of the organic material turnover in the irri- gated soil compared to the non irrigated one. However, despite the DOC supply in large amounts by the Sfax’s TW (Table 2), the irrigation of the studied calcisol has not systematically implied changes in the total content of soil organic carbon (SOC), compared to the non irrigated soil (Table 3). Huge variability of SOC contents are in- deed noticed in the topsoil layer of the irrigated field, so that the mean values are not statstically different from the reference value of the non irrigated field (Table 3). This result suggests the influence of contradictory pro- cesses to the organic matter mineralization. It has to be noticed that the Sfax’s TW are particularly rich in salt and sodium. Chow et al. [25] have perceived that the soil salinisation and sodification affects the SOM structure as to decrease the DOC content in the leaching soil waters. As shown in Figure 2, a significant negative correlation is indeed found between the soil ECs and the DOC in the irrigated soil (R = −0.63, p < 0. 05). Romkens and Dolfing [32] explained that free Ca2+ cations ex- changed by the added Na+ flocculate more than 50% of Figure 2. The correlation between dissolved organic carbon (DOC) in the water soil extracts and electric conductivity (ECs) of the surface layers of irrigated soils. the soil DOC. Rietz and Haynes [33] added that the in- crease of the soil salinity leads to an inhibition of the soil microbial activity, and thus to significant decrease of the SOM mineralization. Likewise, the latter contents stabi- lization takes place in the studied calcisol corresponding to its mineralization enhancement by means of TW’ ap- plication. Subsequently, this procedure tends to explain the variability of resulting contents of SOC in the irri- gated field (Table 5). Over the long term, salt-inducing SOM stabilization can also lead to a decrease of crop yields [33]. 4.3. The Risk of Fertility Loss over the Long Term Over the long term, salt-inducing SOM stabilization can lead to decrease of crop yields [33]. The relationship between salinity of irrigated soils (ECs) and their content in labile carbon (DOC) thus gives an insight of further risks of soil losses of the studied calcisol fertility after- wards (Figure 2). The relationships between nitrate, ammonium contents and the evolution of the quality of the soil organic matter (as seen by the C/N ratio, SUVA and DOC contents in soil water extracts) suggest long term risk of degradation of groundwater quality by the leaching waters. The irrigation clearly leads to a decrease of the con- tents in nitrate and ammonium in the water extracts of soils, not only at the soil surface, but also in the deepest soil layer (see Table 3 and [15]). DOC of the applied effluent, as well as the soil DOC fraction only represent small parts (0.01% up to 0.1%) of the total SOM in soils. It is also the more mobile and the further used C fraction for heterotrophic microbial communities involved in the N turnover. Kim and Burger, [23] showed that nitrogen supply by treated effluents favored nitrification and Copyright © 2012 SciRes. OPEN A CCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 711 leaching of nitrate because the amounts of available ni- trogen, supplied by the TW, largely exceed the plant de- mand. As seen in Tables 4 and 6, the control calcisol is naturally poor of nitrogen. The presence of active car- bonates in this type of soil also usually causes natural inhibition of microbial activity, which is indicated in this present case by a C/N ratio above 70. Moreover, the CEC of the studied calcisol remains low compared to other equivalent to calcareous soils in which CEC reach values up to 20 cmol+·Kg−1 [15,34]. Due to the low CEC and to the elevated irrigation rate, ammonia cations supplied by the Sfax TW have limited chances of being absorbed by the soil. In such context, nitrate and ammonium contents occurring in the deepest layer are certainly deriving straight from the applied wastewater. The two mineral nitrogen ions are leached down to the root zone and risk to reduce the upper soil layer fertility. As a matter of fact, this can also cause degradation of the soil leaching water and consequently of the free groundwater below roots and transient zones. Apart from risks of degradation of groundwater quality, there are also concerns for the con- dition of the crop harvest. Indeed the excess of available mineral nitrogen in soil usually lead to the breaking of forage crops during the maturation stage (oat and so- ghum). The previous studies have also identified losses in es- sential nutrients (Ca, Mg, K, Na, Cu, Zn, Mn) and soil acidification as others risks of degradation of the fertility of alkaline calcareous soils being irrigated by domestic effluents for several decades [5,8,10,11]. In the studied case, the 15-year long wastewater irrigation has not af- fected the pH of the soil. This result can be explained by an important buffer capacity of the examined calcisol. Calcium represents the most abundant exchangeable base cation (Table 3). As seen along the two pedological pits, horizons of soil are nearly saturated in essential base cations (Table 4). Belaïd et al. [15] already mentioned significant linear correlations (p < 0.05) between the CEC and the contents of exchangeable cations added by the TW: Na+ (R = 0.94), K+ (R = 0.79) and Mg2+ (R = 0.99). The quantitative analysis of pool changes for Ca and Na in the irrigated pedon validates the exchanges of natural Ca2+ by the supplied Na+ (Table 5). Exchange- able contents in Mg2+, K+ and Na+, have generally in- creased only in the irrigated soil (Table 3). These cations developments are corresponding with its relatively high concentrations in the treated wastewater used for irriga- tion. Although total amounts in these essential cations have not significantly changed at the scale of the soil profile (Table 5), there is a risk over the long term of loss of these elements due to the high rate of irrigation and low soil CEC (Table 4). On the other hand, due to the salinity of the Sfax’s TW, important amounts of the sup- plied Na are leached down to the deepest horizon of soil in the irrigated field initiating sodification of groundwa- ter. 5. CONCLUSION The Sfax treated wastewater (TW) is particularly rich in available organic carbon and mineral nitrogen. Irriga- tion for 15 years with the Sfax TW has not significantly changed the pH of the studied calcisol and the SOC con- tent of the topsoil layer. The results of the present study also confirm, under field conditions, processes previ- ously identified in laboratory experiments about the im- pact of wastewater effluents on the quality of the soil organic matter. From a quantitative respect, the 15-year- long irrigation by the Sfax TW has limited the loss of micro nutrients such as Mn and Zn. It has also supplied important amounts in most essential macro nutrients (P and N). The irrigation by the TW has also clearly en- hanced the C and N turnover in the studied calcisol which fertility was naturally limited by the elevated con- tent in active carbonates. If these beneficial effects could reduce the cost of mineral fertilization and aid the pro- duction of crops, the continuous use of the TW arises some questions concerning soil fertility and groundwater protection over the long term. In the case study, because of the elevated concentrations in sodium and salts of the applied TW, the beneficial activation of the microbial activity and the resulting availability of essential ele- ments could be quickly inhibited. Moreover, due to the elevated irrigation rate and the low CEC of the soil, the TW irrigation has clearly increased the leaching of mi- neral nutrients such as nitrate and exchangeable K, Mg, and Na. Further irrigation, even with natural water, could stand for an imminent threat for the quality of the free watertable in the Sfax region by increasing concentra- tions in nitrate, sodium and salt. Apart from this problem, the excessive supply of salts and available nitrogen al- ready constitutes a risk for the future crops production. However, since these speculations only concerned one calcisol filed, the validity of our conclusions need to be verified across a wider study area and for the other types of soils that were irrigated with the Sfax TW. 6. ACKNOWLEDGEMENTS The authors gratefully acknowledge the staff of the CRDA-Sfax for their cooperation during site selection and soils sampling. REFERENCES [1] Bahri, A. (1987) Utilization of treated wastewater and sewage sludge in agriculture in Tunisia. Desalination, 67, 233-244. [2] Bahri, A. (2002) Water reuse in Tunisia: Stakes and pros- pects. Actes de l’atelier du PCSI, Montpellier, 1-11. Copyright © 2012 SciRes. OPEN A CCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 712 [3] Haruvy, N. (1997) Agricultural reuse of wastewater: Na- tion-wide cost-benefit analysis. Agriculture Ecosystem En- vironment, 66, 113-119. doi: 10.1016/S0167-8809(97)00046-7 [4] Pescod, M.B. (1992) Wastewater treatment and use in agriculture. Bulletin FAO 47, Rome, 125. [5] Yadav, B., Goyal, R.K., Sharma, S.K., Dubey, P.S. and Minhas, R.K. (2002) Post-irrigation impact of domestic sewage effluent on composition of soils, crops and ground- water—A case study. Environment International, 28, 481- 486. doi:10.1016/S0160-4120(02)00070-3 [6] Coppola, A., Santini, A., Botti, P., Vacca, S., Comegna, V. and Severino, G. (2004) Methodological approach for evalu- ating the response of soil hydrological behavior to irriga- tion with treated municipal wastewater. Journal of Hy- drology, 292, 114-134. doi:10.1016/j.jhydrol.2003.12.028 [7] Herpin, U., Gloaguen, T.V., da Fonseca, A.F., Montes, C.R., Mendonça, F.C., Piveli, R.P., Breulmann, G., Forti, M.C. and Et Melfi, A.J. (2007) Chemical effects on the soil-plant system in a secondary treated wastewater irri- gated coffee plantationcA pilot field study in Brazil. Ag- ricultur e Water Management, 89, 105-115. doi:10.1016/j.agwat.2007.01.001 [8] Rattan, R.K., Datta, S.P., Chhonkar, P.K., Suribabu, K. and Singh, A.K. (2005) Long-term impact of irrigation with sewage effluents on heavy metal content in soils, crops and groundwater—A case study. Agriculture Eco- system and Environment, 109, 310-322. doi:10.1016/j.agee.2005.02.025 [9] Abbas, S.T., Sarfraz, M., Mehdi, S.M., Hassan, G. and Rehman, O.U. (2007) Trace elements accumulation in soil and rice plants irrigated with the contaminated water. Soil & Tillage Research, 94, 503-509. doi:10.1016/j.still.2006.10.004 [10] Solis, C., Andrade, E., Mireles, A., Reyes-Solis, I.E., Gar- cia-Calderon, N., Lagunas-Solar, M.C., Pina, R.G. and Flocchini, C.U. (2005) Distribution of heavy metals in plants cultivated with wastewater irrigated soils during different periods of time. Nuclear Instruments and Meth- ods in Physics Research Section B, 241, 351-355. doi:10.1016/j.nimb.2005.07.040 [11] Rusan, M.J.M., Hinnawi, S. and Rousan, L. (2007) Long term effect of wastewater irrigation of forage crops on soil and plant quality parameters. Desalination, 215, 143- 152. doi:10.1016/j.desal.2006.10.032 [12] Magesan, G.N., Williamson, J.C., Yeates, G.W. and Loyd- Jones, A.R. (2000) Wastewater C:N ratio effects on soil hydraulic conductivity and potential mechanisms for re- covery. Bior esource Technology , 71, 21-27. doi:10.1016/S0960-8524(99)00054-1 [13] Ramirez-Fuentes, E., Lucho-Constantino, C., Escamilla- Silva, E. and Dendooven, L. (2002) Characteristics, and carbon and nitrogen dynamics in soil irrigated with waste- water for different lengths of time. Bioresource Techology, 85, 179-187. doi:10.1016/S0960-8524(02)00035-4 [14] Bouri, S., Abida, H. and Khanfir, H. (2008) Impacts of wastewater irrigation in arid and semi arid regions: Case of Sidi Abid region, Tunisia. Environmental Geology, 53, 1421-1432. doi:10.1007/s00254-007-0751-5 [15] Belaid, N., Neel, C., Kallel, M., Ayoub, T., Ayadi, A. and Baudu, M. (2010) Effects of treated wastewater irrigation on salinity and sodicity of soils: A case study in Sfax (Tu- nisia). Journal of Water Science, 23, 133-145. [16] FAO (1998) World Reference Base for Soil Resources, by ISSS-ISRIC-FAO. World Soil Resources Report No. 84. Rome, 88 p. [17] AFNOR, et al. (1997) Water quality, analysis methodes. 3, 296. [18] Orsini, L. and Remy, J.C. (1976) Utilisation du chlorure de cobaltihexamine pour la détermination simultanée de la capacité d’échange et des bases échangeables des sols. Sciences du sol, bulletin de l’AFES 4, 269-279. [19] Metson, A.J. (1956) Methods of chemical analysis for soil survey samples. New Zealand Soil Bureau, 12. [20] Baker, M.A., Valett, H.M. and Dahm, C.N. (2000) Or- ganic carbon supply and metabolism in a shallow ground- water ecosystem. Ecology, 81, 3111-3148. doi:10.1890/0012-9658(2000)081[3133:OCSAMI]2.0.C O;2 [21] Keller, C. and Védy, J.-C. (1994) Distribution of copper and cadmium fractions in two forest soils. Journal of En- vironmental Quality, 23, 987-999. doi:10.2134/jeq1994.00472425002300050020x [22] Klay, S., Charef, A., Ayed, L., Houman, B. and Rezgui, F. (2010) Effect of irrigation with treated wastewater on geo- chemical properties (saltiness, C, N and heavy metals) of isohumic soils (Zaouit Sousse perimeter, Oriental Tuni- sia). Desalination, 253, 180-187. doi:10.1016/j.desal.2009.10.019 [23] Kim, D.Y. and Burger, J.A. (1997) Nitrogen transforma- tions and soil processes in a wastewater-irrigated, mature Appalachian hardwood forest. Forest Ecology and Man- agement, 90, 1-11. doi:10.1016/S0378-1127(96)03889-3 [24] Néel, C., Soubrand-Colin, M., Piquet-Pissaloux, A. and Bril, H. (2007) Mobility and availability of Cr, Ni, Cu, Zn and Pb in a basaltic grassland: Comparison of selective extractions with quantitative approaches at different scales. Applied Geochemistry, 22, 724-735. [25] Chow, A.T., Tanji, K.K. and Gao, S. (2003) Production of dissolved organic carbon (DOC) and trihalomethane (THM) precursor from peat soils. Water Research, 37, 4475-4485. doi:10.1016/S0043-1354(03)00437-8 [26] Xue, S., Zhao, Q.L., Wei, L.L., Wang, L.N. and Liu, Z.G. (2007) Fate of secondary effluent dissolved organic mat- ter during soil-aquifer treatment. Chinese Science Bulletin, 52, 2496-2505. doi:10.1007/s11434-007-0339-1 [27] Traina, S.J., Novak, J. and Smeck, N.E. (1990) An ultra- violet absorbance method of estimating the percent aro- matic carbon content of humic acids. Journal of Envi- ronmental Quality, 19, 151-153. doi:10.2134/jeq1990.00472425001900010023x [28] Hassouna, M., Theraulaz, F., Lafolie, F. and Massiani, C. (2005) Characterisation and quantitative estimation of the hydrophobic, transphilic and hydrophilic fractions of DOC in soil using direct UV spectroscopy. Geophysical Research Abstracts, 7, 3 p. [29] Kalbitz, K., Schmerwitz, J., Schwesig, D. and Matzner, E. Copyright © 2012 SciRes. OPEN A CCESS  N. Belaid et al. / Agricultural Sciences 3 (2012) 702- 713 Copyright © 2012 SciRes. OPEN A CCESS 713 (2003) Biodegradation of soil-derived dissolved organic matter as related to its properties. Geoderma, 113 , 273-291. doi:10.1016/S0016-7061(02)00365-8 [30] Van Miegroet, H., Boettinger, J.L., Baker, M.A., Nielsen, J., Evans, D. and Stum, A. (2005) Soil carbon distribution and quality in a montane rangeland-forest mosaic in north- ern Utah. Forest Ecology Management, 220, 284-299. doi:10.1016/j.foreco.2005.08.017 [31] Korshin, G.V., Li, C.W. and Benjamin, M.M. (1997) Mo- nitoring the properties of natural organic matter through UV spectroscopy: A consistent theory. Water Research, 31, 1787-1795. doi:10.1016/S0043-1354(97)00006-7 [32] Romkens Paul, F.A.M. and Dolfing, J. (1998) Effect of Ca on the solubility and molecular size distribution of DOC and Cu binding in soil solution samples. Environ- mental Science and Technology, 32, 363-369. doi:10.1021/es970437f [33] Rietz, D.N. and Haynes, R.J. (2003) Effects of irriga- tion-induced salinity and sodicity on soil microbial activ- ity. Soil Biology and Biochemistry, 35, 845-854. doi:10.1016/S0038-0717(03)00125-1 [34] Jalali, M., Merikhpour, H., Kaledhonkar, M.J. and Van Der Zee, S.E.A.T.M. (2008) Effects of wastewater irriga- tion on soil sodicity and nutrient leaching in calcareous soils. Agricultural Water Management, 95, 143-153. doi:10.1016/j.agwat.2007.09.010
|