 Journal of Water Resource and Protection, 2012, 4, 616-621 http://dx.doi.org/10.4236/jwarp.2012.48071 Published Online August 2012 (http://www.SciRP.org/journal/jwarp) Study about Top-Down and Bottom-Up Contr o ls in Regulating the Phytoplankton Biomass in a Eutrophic Reservoir in Northeastern Brazil Juliana dos Santos Severiano1, Ariadne do Nascimento Moura1*, Enaide Marinho de Melo Magalhães2, Viviane Lúcia dos Santos Almeida3 1Graduating Program on Botany, Rural and Federal University of Pernambuco, Recife, Brasil 2Federal University of Alagoas, Institute of Biological Sciences and Health—IBSH, Maceió, Brasil 3Department of the Environment, Prefecture of Recife, Olinda, Brasil Email: *ariadne@db.ufrpe.br, *ariadne_moura@hotmail.com Received May 8, 2012; revised June 17, 2012; accepted June 26, 2012 ABSTRACT This study aims to analyze the effects of nutrients and predation by zooplankton on phytoplankton biomass (chlorophyll a) in a eutrophic reservoir in Brazil (Apipucos Reservoir, State of Pernambuco), through experiments in microcosms. For this, samples of water were placed in 1 L Erlenmeyer flasks and kept for seven days. Treatments included the addition of nutrients (nitrogen combined with phosphorus and isolated additions of nitrogen and phosphorus), with presence and absence of zooplankton and a control which contained the reservoir water without any manipulation. The addition of nutrients did not stimulate phytoplankton growth. However, zooplankton significantly decreased phy- toplankton biomass in the treatments it was added to (p < 0.05). The results of this study showed that for the reservoir studied, predation by zooplankton is the most significant factor in the regulation of phytoplankton, contradicting several studies which show that phytoplankton biomass is more strongly controlled by nutrients (bottom-up control) than by predation (top-down control). Keywords: Chlorophyll a; Nutrients; Zooplankton; Tropical Regions 1. Introduction Top-down (predation) and bottom-up (nutrients) controls have received considerable attention in recent years, mainly due to disputes about which one is more effective in regulating the phytoplankton community. The avail- ability of nutrients has been considered the main regula- tory force of phytoplankton structure and biomass [1,2], since the top-down effects, according to [3], are stronger at the top of the food web and weaken gradually toward the base. Thus, it is believed that the phytoplankton bio- mass is more strongly controlled by nutrients (bottom-up) than predation (top-down). However, studies have also shown that phytoplankton can be strongly regulated by zooplankton [4-8]. Many studies have demonstrated that the addition of nutrients, especially nitrogen (N) and phosphorus (P), can significantly increase the growth of phytoplankton, and influence the taxonomic composition and size class distribution of these organisms [2,9-12]. Zooplankton can have both direct and indirect effects on the phyto- plankton community, through predation and by the re- cycling of nutrients, respectively [5]. Predation gener- ally causes a decrease in phytoplankton biomass thus influencing primary productivity. However, it can also cause positive effects on phytoplankton, since it can stimulate the growth of inedible algae that have a greater competitive advantage for nutrients when the biomass of edible algae decreases [13]. Studies dealing with top-down and bottom-up controls on phytoplankton in reservoirs and lakes have been con- ducted in temperate regions, but are rare in tropical en- vironments. Due to the need for broadening and deepen- ing understanding of the structure and dynamics of phytoplankton in these regions, this study aims to ana- lyze the effects of nutrients and predation exerted by zooplankton on the phytoplankton biomass (chlorophyll a) in a eutrophic reservoir in northeastern Brazil (Api- pucos Reservoir, State of Pernambuco), through experi- ments in microcosms. 2. Materials and Methods 2.1. Study Areas and Sampling *Corresponding author. The Apipucos Reservoir (8˚01'14''S e 34˚56'00''W) is a C opyright © 2012 SciRes. JWARP  J. S. SEVERIANO ET AL. 617 eutrophic ecosystem belonging to the Capibaribe river basin, the main river in the state of Pernambuco, Brazil (Figure 1). It has a total area of 2.9 km2, a volume of 556,375 m3 and an average depth of 2.5 m [14]. It has been built for flood containment and recreation, and con- sists of two interconnected subsystems. The occurrence of free-floating macrophytes is expressive, represented mainly by Eichhornia crassipes (Mart.) for its historical, cultural and environmental values; this water body is considered an “Environmental Protection Area”. How- ever, the organic (untreated sewage release) pollution input is high, resulting in elevated eutrophication levels in this system [15]. The annual cycle of the region is characterized by two seasons regulated by precipitation: a rainy season (March to August) and a dry season (Sep- tember to February). Water samples for the laboratory experiment were col- lected on the day that each experiment began: January 10, January 30 and February 27, 2012. Eight liters of water were collected from the surface layer at a single point, with the aid of a van Dorn bottle, of which seven liters were filtered through plankton net with mesh opening of 68 µm to remove most of the zooplanktonic organisms that could interfere with the experimental design. The filtered material was stored in plastic containers and taken to the laboratory where it was used in the experi- ment. Part of this water was destined for nutrient analysis. The following abiotic variables were measured in situ: water temperature (˚C); and dissolved oxygen (mg·L–1), measured through a Schott oximeter, Handylab OX1; water transparency, measured with a Secchi disk; and light intensity (µmol photons m–2, s–1), measured with a photometer. The turbidity and pH values were obtained from measurements in the laboratory with a Hanna In- Figure 1. Geographical location of the Apipucos reservoir, Pernambuco, Brazil. struments turbidimeter HI 93703 and a Digimed poten- tiometer, DMPH-2, respectively. The concentrations of nitrate (µg N-NO3 L –1) and nitrite (µg N-NO2 L –1) were estimated according to the method described by [16]; and ammonia nitrogen (N- NH3 + N-4 NH ) [17], and total phosphorus (µg PT L–1), according to the methodology in [18]. The total nitrogen (TN) was considered as the sum of the concentrations of nitrate, nitrite and ammonia nitrogen. 2.2. Experimental Design Sub-samples of 700 mL were placed in 1 L Erlenmeyer flasks and kept in the laboratory for seven days under temperature conditions of 25˚C ± 1˚C, artificial light with a photoperiod of 12:12 (light and dark), and constant oxygenation. Three experiments were performed, each one with treatments that included the addition of nutrients (nitro- gen combined with phosphorus, nitrogen or phosphorus), with the presence and absence of zooplankton. Therefore, for the environment studied, seven treatments were tested (n = 3): two with the addition of nitrogen and phosphorus, with and without addition of zooplankton, treatments NP and NP + Z, respectively; two with the addition of nitro- gen, with and without the addition of zooplankton, treat- ments N and N + Z, respectively; and two with the addi- tion of phosphorus, with and without the addition of zoo- plankton, treatments P and P + Z, respectively, and the final one, water from the reservoir that had not been ma- nipulated (Control). For treatments with the addition of nutrients, the source of nitrogen was NaNO3 and of phosphorus KH2PO4, and they were added once on the first day of the experi- ments (Table 1). Zooplanktons added to the treatments were collected in the reservoir using plankton net with mesh size of 68 µm. The initial density added to each treatment was twice the average recorded in the natural environment, based on the results of a recent study con- ducted in the studied reservoir [19]. 2.3. Analysis of Phytoplankton The phytoplankton biomass was estimated by the amount of chlorophyll a (µg·L−1), according to the methodology in [20] and, for this, 10 mL subsamples were removed from each treatment on the 1st, 3rd, 5th and 7th days of the experiment. 2.4. Statistical Analysis Statistical differences between treatments were deter- mined using a two-way analysis of variance (ANOVA), with a significance level of p < 0.05, using BioEstat. Ver- sion 5.0. Copyright © 2012 SciRes. JWARP 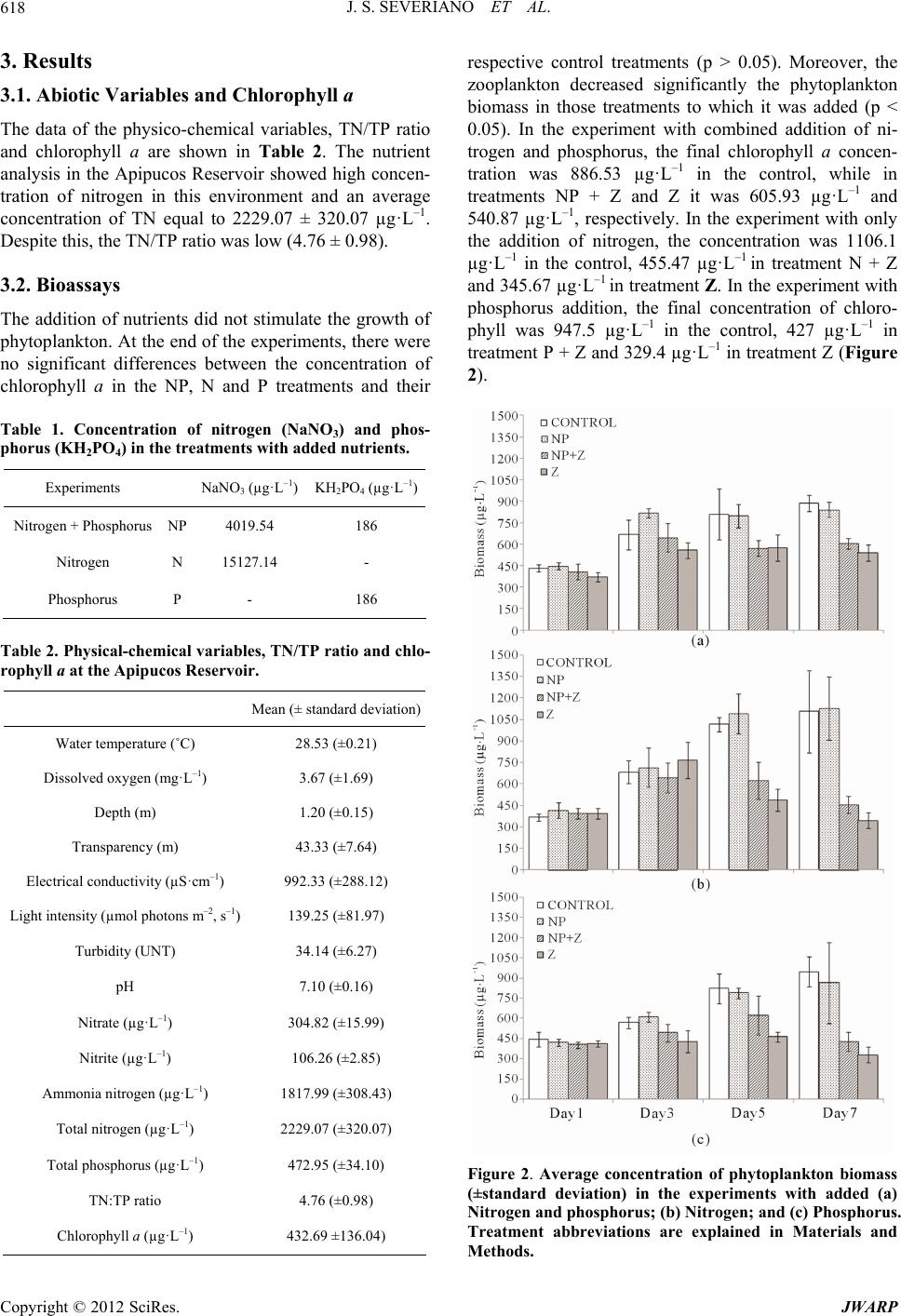 J. S. SEVERIANO ET AL. 618 3. Results 3.1. Abiotic Variables and Chlorophyll a The data of the physico-chemical variables, TN/TP ratio and chlorophyll a are shown in Table 2. The nutrient analysis in the Apipucos Reservoir showed high concen- tration of nitrogen in this environment and an average concentration of TN equal to 2229.07 ± 320.07 µg·L–1. Despite this, the TN/TP ratio was low (4.76 ± 0.98). 3.2. Bioassays The addition of nutrients did not stimulate the growth of phytoplankton. At the end of the experiments, there were no significant differences between the concentration of chlorophyll a in the NP, N and P treatments and their Table 1. Concentration of nitrogen (NaNO3) and phos- phorus (KH2PO4) in the treatments with added nutrients. Experiments NaNO3 (µg·L–1) KH2PO4 (µg·L–1) Nitrogen + Phosphorus NP 4019.54 186 Nitrogen N 15127.14 - Phosphorus P - 186 Table 2. Physical-chemical variables, TN/TP ratio and chlo- rophyll a at the Apipucos Reservoir. Mean (± standard deviation) Water temperature (˚C) 28.53 (±0.21) Dissolved oxygen (mg·L–1) 3.67 (±1.69) Depth (m) 1.20 (±0.15) Transparency (m) 43.33 (±7.64) Electrical conductivity (µS·cm–1) 992.33 (±288.12) Light intensity (µmol photons m–2, s–1) 139.25 (±81.97) Turbidity (UNT) 34.14 (±6.27) pH 7.10 (±0.16) Nitrate (µg·L–1) 304.82 (±15.99) Nitrite (µg·L–1) 106.26 (±2.85) Ammonia nitrogen (µg·L–1) 1817.99 (±308.43) Total nitrogen (µg·L–1) 2229.07 (±320.07) Total phosphorus (µg·L–1) 472.95 (±34.10) TN:TP ratio 4.76 (±0.98) Chlorophyll a (µg·L–1) 432.69 ±136.04) respective control treatments (p > 0.05). Moreover, the zooplankton decreased significantly the phytoplankton biomass in those treatments to which it was added (p < 0.05). In the experiment with combined addition of ni- trogen and phosphorus, the final chlorophyll a concen- tration was 886.53 µg·L–1 in the control, while in treatments NP + Z and Z it was 605.93 µg·L–1 and 540.87 µg·L–1, respectively. In the experiment with only the addition of nitrogen, the concentration was 1106.1 µg·L–1 in the control, 455.47 µg·L–1 in treatment N + Z and 345.67 µg·L–1 in treatment Z. In the experiment with phosphorus addition, the final concentration of chloro- phyll was 947.5 µg·L–1 in the control, 427 µg·L–1 in treatment P + Z and 329.4 µg·L–1 in treatment Z (Figure 2). Figure 2. Average concentration of phytoplankton biomass (±standard deviation) in the experiments with added (a) Nitrogen and phosphorus; (b) Nitrogen; and (c) Phosphorus. Treatment abbreviations are explained in Materials and Methods. Copyright © 2012 SciRes. JWARP  J. S. SEVERIANO ET AL. 619 4. Discussion Studies show that phytoplankton biomass tends to in- crease with the addition of nutrients, especially nitrogen (N) and phosphorus (P), and decrease due to predation by zooplankton [10,21,22]. In this study, the addition of nutrients had no effect on the phytoplankton biomass, measured by the concentration of chlorophyll a. Due to the low N:P ratio recorded in the Apipucos Reservoir, the addition of nitrogen, or nitrogen combined with phosphorus, was expected to stimulate the growth of phytoplankton. According to [23], lakes and reservoirs which have an N:P ratio lower than 20 are limited by nitrogen, those greater than 50 are limited by phosphorus, and those between 20 and 50 are co-limited by N and P. A similar pattern was also proposed by [24,25]. Traditionally, phosphorus has been identified as the main limiting factor for phytoplankton growth, at least in temperate regions, while nitrogen is frequently cited as limiting at low latitudes [26]. However, it has been diffi- cult to establish the main limiting nutrient for phyto- plankton, because this can vary between different envi- ronments and among algal species. Dzialowski et al. (2005) [24], analyzing 19 reservoirs in Kansas, USA, observed that the addition of P stimu- lated growth of phytoplankton in only 8% of all experi- ments in bioassays. On the other hand, limitation by N (16%) and co-limitation by N and P (63%) was often observed. Moreover, just as in the present study, the au- thors observed that in 13% of the reservoirs, the addition of nutrients did not stimulate phytoplankton growth. A similar result was observed by [27], who analyzed five dams along the Manyame River (Harava, Seke, Chivero Lake, Manyame Lake and Bhiri), Mozambique. Nitrogen was the main limiting factor in the Harava, Seke and Manyame Lake dams, and phosphorus was a limiting factor in Bhiri, while no nutrient limited phytoplankton growth in Lake Chivero. In both studies cited above, light was identified as the main limiting factor in the reservoirs where the addition of nutrients did not stimulate algal growth. The possibil- ity of limitation by light is rarely addressed in bioassays for limiting nutrients in freshwater systems [28]; how- ever there is frequent evidence of limitation by light for phytoplankton in lakes and reservoirs [28-30]. According to [31], when the phytoplankton is limited by light it may show little or no limitation by nutrients. The availability of these two resources may vary spa- tially and temporally in most ecosystems [29-32]. Ac- cording to [32], in periods of intense rainfall there is an excessive discharge of nutrients which can directly lessen the limitation of this feature. Moreover, there is an in- crease in the discharge and re-suspension of sediments, increasing the intensity of limitation by light, since less penetration of light into the water column can ultimately limit algal growth. Regarding spatial variation, it is believed that the phytoplankton growing in shallow water sites near heavy water flow are exposed to increased sediment discharge and pulses of nutrients when compared to phytoplankton growing in deeper, thermally stratified areas closer to the dam. Hence, phytoplankton from shallow lotic areas may be more limited by light and less limited by nutrients than phytoplankton from deeper areas [32]. Due to the environmental conditions observed in the Apipucos Reservoir, the results of the experiments sug- gest a potential limitation by light, since this reservoir is made up of neighborhoods heavily populated by low- income populations, where sewage is discharged con- tinually, either directly or indirectly [33]. Moreover, its waters are dark and turbid, indicating a high concentra- tion of suspended sediment. One should also consider the possibility of limitation by another nutrient or a seasonal variation in the limiting resource of the phytoplankton in the Apipucos Reservoir. While the addition of nutrients had no effect on the phytoplankton biomass, predation exerted by zooplank- ton caused a significant reduction in the biomass. This result does not corroborate the hypothesis that top-down control exerted by zooplankton in tropical systems is weak when compared to that observed in temperate re- gions [34]. It is believed that the absence of large her- bivorous zooplankton in the tropics, as well as the domi- nance of small-sized species, are the main reasons re- sponsible for this difference [35,36]. Von Rückertand and Giani (2008) [34], while evaluat- ing the effects of predation of three microcrustaceans Daphnia laevis, Moina micrura and Thermocyclops de- cipiens on the phytoplankton community in the Pam- pulha Reservoir (Minas Gerais, Brazil) found that zoo- plankton was inefficient in controlling phytoplankton. Low et al. (2010) [37], when analyzing phytoplankton and zooplankton populations in 12 tropical reservoirs, observed that the zooplankton exerted a top-down control on certain algal communities, such as green algae (An- kistrodesmus, Scenedesmus and Cosmarium) and phy- toflagellates (Peridinium). Cyanobacteria, with the ex- ception of Planktothrix, were not adversely affected by zooplankton. For a better understanding of the interaction between phytoplankton and zooplankton communities in the Apipucos Reservoir, more information about the various factors that may affect the ability of zooplankton to prey on phytoplankton is necessary, namely, the taxonomic composition and biomass of the zooplankton, body size of the zooplankton and phytoplankton, zooplankton se- lectivity and efficiency, resilience of the phytoplankton, the relationship between the nutritional requirements of Copyright © 2012 SciRes. JWARP  J. S. SEVERIANO ET AL. 620 the zooplankton and the nutritional quality of the phyto- planktonic organism [38-44]. 5. Conclusion The results of this study showed that, for the reservoir studied, predation by zooplankton is the most significant factor in the regulation of phytoplankton, contradicting several studies which show that phytoplankton biomass is more strongly controlled by nutrients (bottom-up con- trol) than by predation (top-down control). 6. Acknowledgements This study was supported by grants from CNPq (Brazil- ian Council for Research and Development (302068/ 2011-2). REFERENCES [1] N. G. Hairston, F. E. Smith and L. B. Slobodkin, “Com- munity Structure, Population Control, and Competition,” The American Naturalist, Vol. 94, No. 879, 1960, pp. 421-425. doi:10.1086/282146 [2] D. Tilman, S. S. Kilham and P. Kilham, “Phytoplankton Community Ecology: The Role of Limiting Nutrients,” Annual Review of Ecology, Evolution, and Systematics, No. 13, 1982, pp. 349-372. doi:10.1146/annurev.es.13.110182.002025 [3] D. J. Mcqueen, M. R. S. Johannes and J. R. Post, “Bot- tom-Up and Top-Down Impacts on Freshwater Pelagic Community Structure,” Ecological Monographs, Vol. 59, No. 3, 1989, pp. 289-309. doi:10.2307/1942603 [4] S. R. Carpenter, J. F. Kitchell and J. R. Hodgson, “Cas- cading Trophic Interactions and Lake Productivity,” Bio- Science, Vol. 35, No. 10, 1985, pp. 634-639. doi:10.2307/1309989 [5] A. M. Bergquist and S. R. Carpenter, “Limnetic Herbi- vory: Effects on Phytoplankton Populations and Primary Production,” Ecology, Vol. 67, No. 5, 1986, pp. 1351- 1360. doi:10.2307/1938691 [6] S. R. Carpenter, J. F. Kitchell, J. R. Hodgson, P. A. Cochran, J. J. Elser, M. M. Elser, D. M. Lodge, D. Kretchmer and X. He, “Regulation of Lake Primary Pro- ductivity by Food Web Structure,” Ecology, Vol. 68, No. 6, 1987, pp. 1863-1876. doi:10.2307/1939878 [7] J. J. Elser and C. R. Goldman, “Zooplankton Effects on Phytoplankton in Lakes of Contrasting Trophic Status,” Limnology and Oceanography, Vol. 36, No. 1, 1991, pp. 64-90. doi:10.4319/lo.1991.36.1.0064 [8] K. Muylaert, S. Declerck, J. V. Wichelen, L. De Meester and W. Vyverman, “An Evaluation of the Role of Daph- nids in Controlling Phytoplankton Biomass in Clear Wa- ter Versus Turbid Shallow Lakes,” Limnologica, Vol. 36, No. 2, 2006, pp. 69-78. doi:10.1016/j.limno.2005.12.003 [9] G. S. Steinhart, G. E. Likens and D. Soto, “Nutrient Limitation in Lago Chaiquenes (Parque Nacional Alerce Andino, Chile): Evidence from Nutrient Enrichment Ex- periments and Physiological Assays,” Revista Chilena de Historia Natural, Vol. 72, 1999, pp. 559-568. [10] E. J. González, “Nutrient Enrichment and Zooplankton Effects on the Phytoplankton Community in Microcosms from El Andino Reservoir (Venezuela),” Hydrobiologia, Vol. 434, No. 1-3, 2000, pp. 81-96. doi:10.1023/A:1004060027616 [11] F. M. Hassan, H. A. Al-Saadi and F. M. Alkam, “Labo- ratory Study on the Effects of Nutrient Enrichment on a Phytoplankton Population in Sawa Lake, Iraq,” Um- Salama Science Journal, Vol. 5, No. 2, 2008, pp. 230- 236. [12] F. R. A. Câmara, A. K. A. Lima, O. Rocha and N. T. Chellappa, “The Role of Nutrient Dynamics on the Phyto- plankton Biomass (Chlorophyll a) of a Reservoir-Channel Continuum in a Semi-Arid Tropical Region,” Acta Lim- nologica Brasiliensia, Vol. 21, No. 4, 2009, pp. 431-439. [13] C. P. Queimaliños, B. E. Modenutti and E. G. Balseiro, “Phytoplankton Responses to Experimental Enhancement of Grazing Pressure and Nutrient Recycling in a Small Andean Lake,” Freshwater Biology, Vol. 40, No. 1, 1998, pp. 41-49. doi:10.1046/j.1365-2427.1998.00326.x [14] S. Neumann-Leitão, J. D. Nogueira-Paranhos and F. B. V. A. Souza, “Zooplâncton do Açude de Apipucos, Recife, PE (Brasil),” Brazilian Archives of Biology and Technol- ogy, Vol. 32, No. 4, 1989, pp. 803-821. [15] V. L. S. Almeida, M. M. G. Melão and A. N. Moura, “Plankton Diversity and Limnological Characterization in Two Shallow Tropical Urban Reservoirs of Pernambuco State, Brazil,” Anais da Academia Brasileira de Ciências, Vol. 84, No. 2, 2012. [16] F. J. H. Mackereth, J. Heron and J. F. Talling, “Water Analysis: Some Revised Methods for Limnologists (Fresh- water Biological Association Scientific Publication, 36),” Titus Wilson & Sons Ltda, Kendall, 1978. [17] F. Koroleff, “Determination of Nutrients,” In: K. Grass- hoff, Ed., Methods of Seawater Analysis, Verlag Chemie, Weinhein, 1976, pp. 117-187. [18] J. D. H. Strickland and T. R. Parsons, “A Manual of Sea- water Analysis (With Special Reference to the More Com- mon Micronutrients and to Particulate Organic Mate- rial),” Fisheries Research Board of Canada, No. 125, 1960, pp. 1-185. [19] V. L. S. Almeida, “Comunidades Planctônicas e Quali- dade da Água em Reservatórios Tropicais Urbanos Com Diferentes Graus de Trofia,” Ph.D. Dissertation, Centro de Ciências Biológicas e da Saúde, São Carlos, 2011. [20] L. Lawton, B. Marsalek, J. Padisák and I. Chorus, “De- termination of Cyanobacteria in the Laboratory,” In: I. Chorus and J. Bartram, Eds., Toxic Cyanobacteria in Water, E & FN Spon, London, 1999, pp. 347-367. [21] K. L. Cottingham, S. E. Knight, S. R. Carpenter, J. J. Cole, M. L. Pace and A. E. Wagner, “Response of Phyto- plankton and Bacteria to Nutrients and Zooplankton: A Mesocosm Experiment,” Journal of Plankton Research, Vol. 19, No. 8, 1997, pp. 995-1010. doi:10.1093/plankt/19.8.995 [22] S. R. Carpenter, J. J. Cole, J. F. Kitchell and M. L. Pace, “Impact of Dissolved Organic Carbon, Phosphorus, and Copyright © 2012 SciRes. JWARP  J. S. SEVERIANO ET AL. Copyright © 2012 SciRes. JWARP 621 Grazing on Phytoplankton Biomass and Production in Experimental Lakes,” Limnology and Oceanography, Vol. 43, No. 1, 1998, pp. 73-80. doi:10.4319/lo.1998.43.1.0073 [23] S. J. Guildford and R. E. Hecky, “Total Nitrogen, Total Phosphorus and Nutrient Limitation in Lakes and Oceans: Is There a Common Relationship?” Limnology and Ocean- ography, Vol. 45, No. 6, 2000, pp. 1213-1223. doi:10.4319/lo.2000.45.6.1213 [24] A. R. Dzialowski, S. Wang, N. Lim, W. W. Spotts and D. G. Huggins, “Nutrient Limitation of Phytoplankton Growth in Central Plains Reservoirs, USA,” Journal of Plankton Research, Vol. 27, No. 6, 2005, pp. 587-595. doi:10.1093/plankt/fbi034 [25] S. Kosten, V. L. M. Huszar, N. Mazzeo, M. Scheffer, L. S. L. Sternberg and E. Jeppesen, “Lake and Watershed Characteristics rather than Climate Influence Nutrient Limitation in Shallow Lakes,” Ecological Applications, Vol. 19, No. 7, 2009, pp. 1791-1804. doi:10.1890/08-0906.1 [26] W. M. J. Lewis, “Tropical Lakes: How Latitude Makes a Difference,” In: F. Schiemer and K. T. Boland, Eds., Perspectives in Tropical Limnology, SPB Academic Pub- lishing, Amsterdam, 1996, pp. 43-64. [27] P. Tendaupenyu, “Nutrient Limitation of Phytoplankton in Five Impoundments on the Manyame River, Zim- babwe,” Water SA, Vol. 38, No. 1, 2012, pp. 97-104. doi:10.4314/wsa.v38i1.12 [28] R. Carignan and D. Planas, “Recognition of Nutrient and Light Limitation in Turbid Mixed Layers: Three Ap- proaches Compared in the Parank Floodplain (Argen- tina),” Limnology and Oceanography, Vol. 39, No. 3, 1994, pp. 580-596. doi:10.4319/lo.1994.39.3.0580 [29] M. F. Knowlton and J. R. Jones, “Temporal and Spatial Dynamics of Suspended Sediment, Nutrients, and Algal Biomass in Mark Twain Lake, Missouri,” Archiv für Hy- drobiologie, Vol. 135, No. 2, 1995, pp. 145-178. [30] E. J. Phlips, M. Cichra, K. Havens, C. Hanlon, S. Badylak, B. Rueter, M. Randall and P. Hansen, “Relationships be- tween Phytoplankton Dynamics and the Availability of Light and Nutrients in a Shallow Sub-Tropical Lake,” Journal of Plankton Research, Vol. 19, No. 3, 1997, pp. 319-342. doi:10.1093/plankt/19.3.319 [31] M. J. Vanni, J. S. Andrews, W. H. Renwick, M. J. Gon- zález and S. J. Noble, “Nutrient and Light Limitation of Reservoir Phytoplankton in Relation to Storm-Mediated Pulses in Stream Discharge,” Archiv für Hydrobiologie, Vol. 167, No. 1-4, 2006, pp. 421-445. [32] B. L. Kimmel, O. T. Lind and L. J. Paulson, “Reservoir Primary Production,” In: K. W. Thornton, B. L. Kimmel and F. E. Payne, Eds., Reservoir Limnology: Ecological Perspectives, John Wiley & Sons, New York, 1990, pp. 133-193. [33] C. B. C. B. Chamixaes, “Ficoflórula do Açude de Apipu- cos (Recife, PE),” Revista Brasileira de Biologia, Vol. 50, No. 1, 1990, pp. 45-60. [34] G. Von Rückert and A. Giani, “Biological Interactions in the Plankton Community of a Tropical Eutrophic Reser- voir: Is the Phytoplankton Controlled by Zooplankton?” Journal of Plankton Research, Vol. 30, No. 10, 2008, pp. 1157-1168. doi:10.1093/plankt/fbn065 [35] C. H. Fernando, “Zooplankton, Fish and Fisheries in Tropical Freshwaters,” Hydrobiologia, Vol. 272, No. 1-3, 1994, pp. 105-123. doi:10.1007/BF00006516 [36] S. S. S. Sarma, S. Nandini and R. D. Gulati, “Life History Strategies of Cladocerans: Comparisons of Tropical and Temperate Taxa,” Hydrobiologia, Vol. 542, No. 1, 2005, pp. 315-333. doi:10.1007/s10750-004-3247-2 [37] E. W. Low, E. Clews, P. A. Todd, Y. C. Tai and P. K. L. Ng, “Top-Down Control of Phytoplankton by Zooplank- ton in Tropical Reservoirs in Singapore?” The Raffles Bulletin of Zoology, Vol. 58, No. 2, 2010, pp. 311-322. [38] C. W. Burns, “Insights into Zooplankton-Cyanobacteria Interactions Derived from Enclosure Studies,” New Zea- land Journal of Marine and Freshwater Research, Vol. 21, No. 3, 1987, pp. 477-482. doi:10.1080/00288330.1987.9516243 [39] A. A. Agrawal, “Algal Defense, Grazers, and Their Inter- actions in Aquatic Trophic Cascades,” Acta Oecologica, Vol. 19, No. 4, 1998, pp. 331-337. doi:10.1016/S1146-609X(98)80037-4 [40] P. Barros, M. L. Fidalgol and A. M. V. M. Soares, “Re- sistance of Cladoceran Species to Toxic Microcystis,” Limnetica, Vol. 20, No. 1, 2001, pp. 173-177. [41] F. Chan, M. L. Pace, R. W. Howarth and R. M. Marino, “Bloom Formation in Heterocystic Nitrogen-Fixing Cyanobacteria: The Dependence on Colony Size and Zoo- plankton Grazing,” Limnology and Oceanography, Vol. 49, No. 6, 2004, pp. 2171-2178. doi:10.4319/lo.2004.49.6.2171 [42] L. Allende and H. Pizarro, “Top-Down Control on Plankton Components in an Antarctic Pond: Experimental Approach to the Study of Low-Complexity Food Webs,” Polar Biology, Vol. 29, No. 10, 2006, pp. 893-901. doi:10.1007/s00300-006-0129-2 [43] M. G. Danielsdottir, M. T. Brett and G. B. Arhonditsis, “Phytoplankton Food Quality Control of Planktonic Food Web Processes,” Hydrobiologia, Vol. 589, No. 1, 2007, pp. 29-41. doi:10.1007/s10750-007-0714-6 [44] R. Sinistro, M. L. Sánchez, M. C. Marinone and I. Izaguirre, “Experimental Study of the Zooplankton Im- pacto on the Trophic Structure of Phytoplankton and the Microbial Assemblages in a Temperate Wetland (Argen- tina),” Limnologica, Vol. 37, No. 1, 2007, pp. 88-99. doi:10.1016/j.limno.2006.09.001
|