American Journal of Plant Sciences
Vol.5 No.15(2014), Article
ID:47859,5
pages
DOI:10.4236/ajps.2014.515254
Spore Types and Spore Production of Ramularia areola for Screening Cotton Germplasm for Resistance
Jéssica Volponi1, Janaina N. Matos1, Larissa Girotto1, Mariana S. Marangoni1, Rafael Galbieri2, Yeshwant R. Mehta1*
1Instituto Agronômico do Paraná (IAPAR), Londrina, Brazil
2Instituto Mato-Grossense do Algodão (IMA), Primavera do Leste, Brazil
Email: *yeshwantrmehta@gmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 13 May 2014; revised 17 June 2014; accepted 8 July 2014
ABSTRACT
Spore production of Ramularia areola has always been a difficult task. Brazilian isolates of R. areola produce spores of variable size and shape. The typical spores are 3 septate, rarely 4 and 5 septate, together with abundant single celled oblong to round bodies—a phenomenon not reported earlier for R. areola. Budding of spores is a continuous process as observed in our isolates. By repeated culturing the pathogen either ceases to produce typical spores or loses its pathogenic character. To solve this problem, a technique was developed to produce large quantity of typical spores under laboratory conditions. Sporulating cultures produced on Petri plates containing V8 juice-agar were kept on the laboratory bench till they became dry and then stored at 5˚C for reisolation and/or for production of fresh inoculum. In such dried cultures spores remain viable for a period of over 12 months, and hence isolates of R. areola originated from different geographic regions can be maintained in sporulating form. Results of the present investigation would aid cotton breeders and pathologists in screening germplasm resistant to Ramularia and in other genetical studies under glasshouse conditions.
Keywords:Gossypium hirsutum, Ramularia Leaf Blight, Techniques, Fruiting Bodies

1. Introduction
Leaf blight of cotton (Gossypium hirsutum, G. arborium, G. herbaceum and G. barbadense) caused by Ramularia areola is economically important in several countries. The disease is also known as Ramularia, grey mildew, areolate mildew, false mildew and white mold. In the Cerrado region of Brazil, the yield losses caused by this disease are estimated to be around 30%, but in severe cases they can be up to 75% [1] . In countries such as India and Madagascar yield losses of over 60% are reported [2] -[4] . In the USA, the disease is not economically important. A few years ago Ramularia leaf blight was of secondary importance in Brazil, since it used to occur almost at the end of the crop cycle. However in recent years the disease has gained top-most importance in major cotton growing States of Brazil, such as São Paulo, Mato Grosso, Mato Grosso do Sul, Minas Gerais and Bahia. In these States, at present the disease is being partially controlled through 6 - 8 aerial applications of systemic fungicides during the crop cycle. Considering the high cost of fungicidal applications allied with the problem of creation of mutants resistant to some fungicides, efforts are being made to control the disease through varietal resistance.
Screening germplasm for resistance to Ramularia under field conditions is not always reliable mostly because of the unfavorable weather conditions and the necessity for repeated inoculations. Besides, according to Pezenti et al. [5] and Girotto et al. [6] phenotypic and genotypic variations among the isolates of the pathogen exist. These authors did not find any correlation between such variations. It is possible that further investigation may encounter such correlation and may throw some light on the existence of pathotypes of this pathogen.
Resistance to Ramularia in Brazil is governed by at least two different genes [1] . Such a finding opens an avenue for different genetical studies. Most of the genetical studies related to identification of pathotypes, inheritance of resistance, pyramidation of resistance genes and screening germplasm for resistance need to be done under the controlled glasshouse conditions. For this purpose, it is necessary to always maintain sporulating isolates representing different geographical regions of the country—a task which has always been difficult for R. areola. Loss of viability of isolates due to repeated sub-culturing is also a problem with this pathogen. Other than this, the fungus ceases to produce typical spores even on V8 juice-agar medium and within few weeks the colonies change their color from pinkish-white to off-white. In the pinkish-white young colonies the spores at first are 0 - 3 septate, rarely 4 - 5 septate—here onwards referred as “typical spores”. Later, the off-white colonies produce only single celled, oblong to round spores, perhaps as a result of budding. This kind of change in cultural and morphological characteristics of the pathogen has been causing concern among the cotton pathologists in Brazil.
The objective of the present investigation was to develop a technique to produce large quantities of typical spores in culture and establish a method for their long term storage.
2. Materials and Methods
Spore types and inoculation procedure: Two isolates of R. areola Nos. 44, and 58.4 from the culture collection of IAPAR, stored in silica gel [7] [8] , were used for the present studies. These isolates were selected considering their differential reaction on three cotton genotypes [6] . Isolates were multiplied on Petri plates containing V8-juice-agar medium for 12 - 15 days at room temperature. Later the Petri plates were flooded with distilled water, fungal colonies were scraped with a soft paint brush and the spore suspension was sprayed on 25 day old three susceptible cotton cultivars (FMT 701, FMT 705 and IAC 25) raised in the glasshouse. These cultivars are susceptible to some isolates and resistant to others [5] [6] . Suspension of the spores of the aggressive isolate No. 44 was adjusted to 104 spores ml−1, considering only the typical spores, whereas for the other isolate No. 58.4 producing only oblong to small single celled bodies, the concentration was not adjusted. Five plates of each isolate were used to produce inoculum using 20 mL of distilled water per plate. The inoculated plants were incubated for four days at 20˚C under alternating cycle of 12 h light/12 h dark and at near saturated humidity, however for the first 18 hours the humidity was kept low (<60%) to avoid run-off of the inoculum. After four days the inoculated plants were transferred to the glasshouse bench without humidity control. Disease reaction was evaluated 25 - 30 days after inoculation, using a visual scale of 0% - 99% of the leaf area infected considering the most severely infected leaf of each plant.
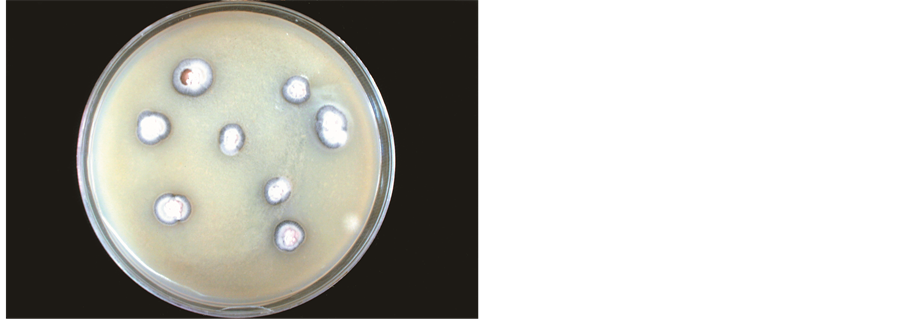
Spore production: The following spore production techniques were used. For production of large quantities of typical spores, at first, monopustular isolation was made directly from the sporulating areas of the leaf. For this purpose, the isolations were made by gently touching the sporulating area by a needle under aseptic conditions and then touching the needle to the V8 juice-agar in the Petri plates. After two weeks small colonies developed with typical spores of R. areola along with some oblong to round single celled bodies. Such colonies were gently scraped by a sterile needle and the spores were streaked on V8 juice-agar plates (Figure 1). The
 (a)
(a)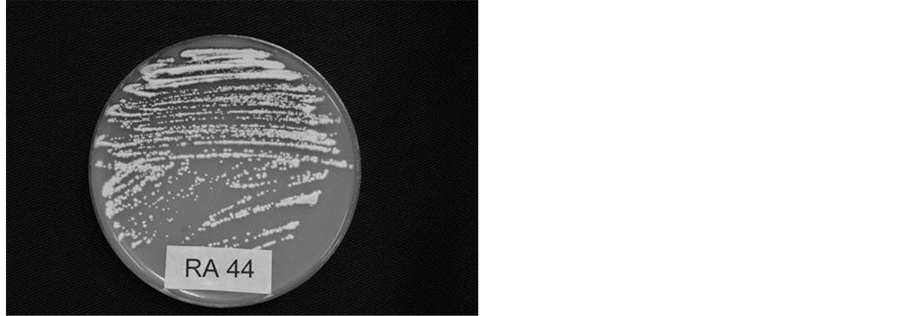 (b)
(b)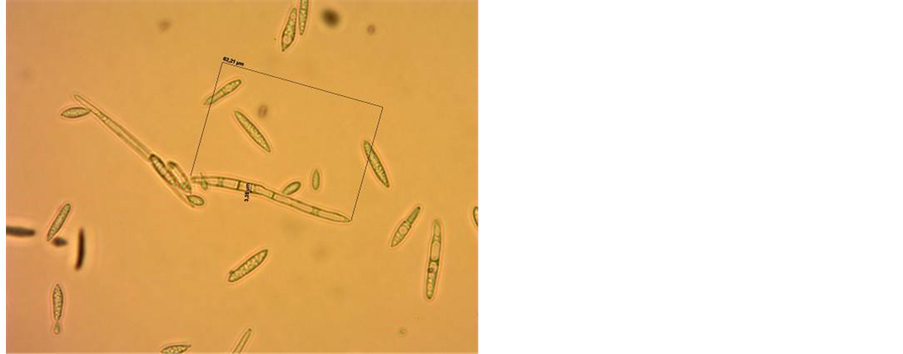 (c)
(c)
Figure 1. (a) Colonies of R. areola developed on V8 juice-agar, 28 days after isolation made directly from the sporulating lesion on infected cotton leaf; (b) Multiplication of R. areola on V8 Juice-agar by streaking; (c) Spores of R. areola showing variable size and shape.
streaking procedure was the same as normally practiced for bacterial cultures. The Petri plates were then kept on the laboratory bench at about 24˚C for 12 - 15 days after which large quantities of typical spores were obtained. Such sporulating cultures were used for screening germplasm for resistance to R. areola, or else dried at room temperature with lid on, and then stored at 5˚C for further use. For inoculation, the dried culture discs are flooded with distilled water, spores are scraped with a soft paint brush and the inoculum is prepared as stated before. Alternatively, severely infected leaves collected from the glasshouse inoculations are blotter paper dried for 48 h and stored at 5˚C for reisolation and production of fresh inoculum.
3. Results and Discussion
The asexual stage of the pathogen is Ramularia areola Atk. The sexual stage belonging to Mycosphaerella areola Ehrlich and Wolf [9] [10] , has not been reported from Brazil. The pinkish-white colonies of both isolates produced typical, oblong to cylindrical, sometimes spindle shaped, 0 - 3 septate, rarely 4 - 5 septate spores, pointed at both ends, measuring between 3.55 ´ 7.28 and 34.71 ´ 62.21 µ. After repeated subculturing the colonies of isolate No. 58.4 gradually transformed into off-white colonies having single celled, oval to round bodies probably due to budding of the typical spores. Budding is a continuous process as observed in both the isolates on V8 juice-agar. According to Rathaiah, [11] [12] and Curvelo et al. [13] , conidia germinate from epical and basal cells. In our isolates other than germination from epical and basal cells, budding occurs from several cells of the spores. Budding of spores results in the formation of single celled, rarely one septate, oblong to round, and sometimes spindle shaped bodies, and could be designated as “microconidia”. Presence of up to five septa and the continuous budding of spores make our isolates different from those reported earlier for R. areola of cotton [9] . We did not study the effect of different culture media on sporulation. Depending upon the repeated subcluturing and the culture media it is possible that the fungus undergoes changes in its cultural and morphological characteristics.
Techniques of spore production, incubation period and incubation conditions used under present investigation were found ideal for germplasm screening as well as for other genetical studies since using such techniques severe symptoms of the disease were obtained consistently within 25 - 30 days after inoculation.
Under glasshouse inoculations both types of colonies produced typical symptoms of Ramularia leaf blight on three cultivars (FMT 701, IAC 25 and FMT 701 (Figure 2)). Isolations from the glasshouse leaf symptoms

Figure 2. Symptoms of Ramularia areola isolate No. 44 on CV. FMT 701, 30 days after inoculation.
made on V8 juice-agar medium yielded pinkish-white colonies within 12 - 15 days producing typical spores as mentioned earlier.
In the present studies we could maintain the sporulating colonies with viable spores on the dried V8 juice-agar medium discs for a period of over 12 months. This is in accordance with Rathaiah and Pavgi [14] , who reported the tolerance of R. carthami to severe dessication. Alternatively, infected leaves can also be filter-paper-dried and stored at 5˚C for production of fresh sporulating cultures.
4. Conclusion
1) Our isolates of R. areola produced spores of variable size and shape. In young cultures the spores showed budding and were typically 0 - 3 septate, rarely 4 - 5 septate along with some oblong to round cells—a phenomenon not reported earlier for R. areola of cotton; 2) The young cultures producing “microconidia” are pathogenic to cotton and can be reverted back to their original type of pinkish-white colonies with typical spores; 3) For germplasm screening and for other genetical studies, viable sporulating cultures on V8 juice-agar, which dried at room temperature can be stored for over 12 months.
Acknowledgements
The present research was conducted under the financial support of IMA, MT, Brazil.
References
- Zandoná, C., Novaes, T., Nunes, M.P., Almeida, W.P., Aguiar, P.H., Morello, C.L., Schuster, I. and Mehta, Y.R. (2012) Mechanism of Resistance and Presence of Different Resistance Genes to Ramularia areola in Two Cotton Genotypes. Tropical Plant Pathology, 37, 175-178. http://dx.doi.org/10.1590/S1982-56762012000300002
- Cauquil, J.S. and Sement, G.L. (1973) Faux Mildiou du Cotonnier (Ramularia areola Atk) Dans le Sud-Ouest de Madagascar. Cotton et Fibers Tropicales, 28, 279-286.
- Shivakar, S.K. and Wangikar, P.D. (1992) Estimation of Crop Losses Due to Grey Mildew Disease of Cotton Caused by Ramularia areola. Indian Phytopathology, 45, 74-76.
- Novaes, T.G., Almeida, W.P., Schuster, I. and Mehta, Y.R. (2011) Herança de Resistência do Algodoeiro a Ramularia areola. Summa Phytopathologica, 37, 150-152. http://dx.doi.org/10.1590/S0100-54052011000200012
- Pezenti, L.F., Barbosa, J., Mariane, A.V., Mariana, S.M., Jessica, V., Almeida, W.P., Galbieri, R. and Mehta, Y.R. (2013) Phenotypic Variability among Isolates of Ramularia areola from Brazilian Cotton. Tropical Plant Pathology, 38, 329-331. http://dx.doi.org/10.1590/S1982-56762013005000023
- Girotto, L., Marangoni, M.S., Matos, J.N., Galbieri, R., Almeida, W.P. and Mehta, Y.R. (2013) Identification of Phenotypic and Genotypic Variability among the Isolates of Ramularia areola of Brazilian Cotton. American Journal Plant Sciences, 4, 1893-1898. http://dx.doi.org/10.4236/ajps.2013.49232
- Perkins, D.D. (1962) Preservation of Neurospora Stock Cultures with Anhydrous Silica Gel. Canadian Journal of Microbiology, 8, 591-594. http://dx.doi.org/10.1139/m62-078
- Windels, C.E., Burnes, P.M. and Kommendahl, T. (1988) Five-Year Preservation of Fusarium Species on Silica Gel and Soil. Phytopathology, 78, 107-109. http://dx.doi.org/10.1094/Phyto-78-107
- C.M.I. (1976) Descriptions of Pathogenic Fungi and Bacteria—Ramularia gossypii. C.M.I., Set No. 520, Kew, Surrey, England.
- Gouws, M.A., Prinsloo, G.C. and Van der Linde, E.J. (2001) First Report of Mycosphaerella areola, Teleomorph of Ramulariopsis gossypii, on Cotton in South Africa. African Plant Protection, 7, 115-116.
- Rathaiah, Y. (1976) Reaction of Cotton Species and Cultivars to Four Isolates of Ramularia areola. Phytopathology, 66, 1007-1009. http://dx.doi.org/10.1094/Phyto-66-1007
- Rathaiah, Y. (1977) Spore Germination and Mode of Cotton Infection by Ramularia areola. Phytopathology, 67, 351-357. http://dx.doi.org/10.1094/Phyto-67-351
- Curvelo, C.R.S., Rodrigues, F.A., Berger, P.G. and Rezende, D.C. (2010) Microscopia eletrônica de varredura do processo infeccioso de Ramularia areola em folhas de algodoeiro. Tropical Plant Pathology, 35, 108-113.
- Rathaiah, Y. and Pavgi, M.S. (1971) Ability of Germinated Conidia of Cercospora carthami and Ramularia carthami to Servive Dessication. The Plant Disease Reporter, 55, 846-847.
NOTES

*Corresponding author.

