International Journal of Clinical Medicine
Vol.2 No.5(2011), Article ID:8784,3 pages DOI:10.4236/ijcm.2011.25107
Kohler’s Disease Secondary to Tarsal Pyogenic Arthritis
![]()
1Pediatrics Department, Infanta Sofia University Hospital, Madrid, Spain; 2Rheumathology Department, Infanta Sofia University Hospital, Madrid, Spain; 3Traumatology and Orthopedics Department, Infanta Sofia University Hospital, Madrid, Spain; 4Radiology Department, Infanta Sofia University Hospital, Madrid, Spain; 5Microbiology Laboratory, Infanta Sofia University Hospital, Madrid, Spain.
Email: alfredo.tagarro@salud.madrid.org
Received August 24th, 2011; revised September 30th, 2011; accepted October 20th, 2011.
Keywords: Köhler Disease, Osteochondroses, Septic Arthritis, Children
ABSTRACT
Kohler’s disease is an osteochondrosis of the tarsal navicular bone. The exact causes of osteochondrosis remain unknown. We report a 4-year-old girl with a haematogenous tarsal arthritis who developed a Köhler’s disease immediately after the infection. It is hypothesised that avascular necrosis of the navicular bone was developed secondary to pyogenic arthritis.
1. Introduction
Osteochondroses are a heterogeneous group of injuries to the epiphyses and apophyses of children [1]. The injuries occur because of a disturbance of the endochondral ossification of epiphyseal growth during periods of rapid growth. The aetiology is not known. This disease occurs mainly in the ossification centres of the lower extremity and is associated with characteristic radiographic findings. These findings are decrease in the size, irregularities, and increased density of the involved bone.
Kohler’s disease is an osteochondrosis of the tarsal navicular bone. It tends to affect children between 4 and 9 years of age. They present with onset of insidious foot pain and a limp. There is no evidence that treatment affects outcome, which is almost always complete recovery [2,3], with reossification occurring gradually over the course of 1 - 2 years.
2. Case Report
A 4 year old girl with no previous clinical history was attended at the emergency room for a 4-days pain localized in her right foot, without a history of trauma. Initially diagnosed as a sprain, the pain persisted in spite of immobilization and oral non steroid anti-inflammatory (ibuprofen). Twenty-four hours later, she developed a fever that lasted for 3 days (up to 38.8˚C), and a limp. She consulted again. The physical examination showed swelling, redness, increased local heat and pain in her foot.
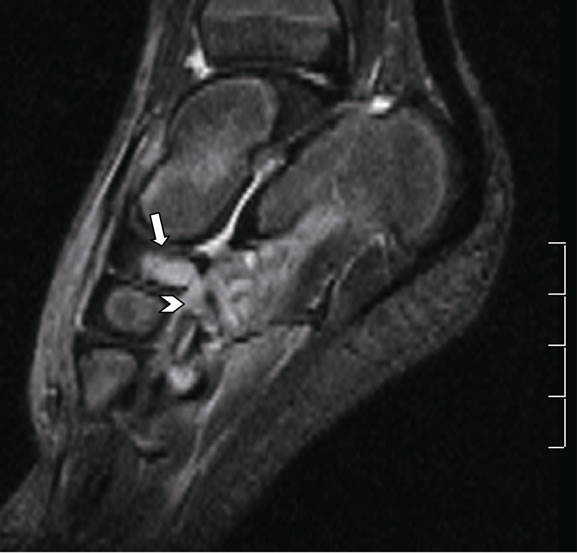
The investigations yielded the following results: white blood count, 19,310/mm3; neutrophil, 14,700/mm3 (76.1%); lymphocytes, 2700/mm3 (14.1%); monocytes, 1400/mm3 (7.2%); eosinophils, 0. Haemoglobin: 12.4 g/dL, platelets: 330,000/mm3. Blood C-reactive protein: 34.9 mg/L. ESR 83 mm in the first hour. Foot X-ray showed no bone changes, but diffusely increased soft tissue. Ultrasonography showed diffusely increased soft tissue. She was then admitted with a presumptive diagnosis of arthritis and cellulitis. She was started with IV cloxacillin at 100 mg/kg/day and oral ibuprofen. Twenty-four hours later, blood culture yielded growing of Staphylococcus aureus resistant to penicillin, sensitive to oxacillin (in two separate blood samples). Magnetic resonance 6 days after admission (Figure 1) showed changes in the intensity of the navicular bone marrow, with hypointensity on T1 weighted images, hiperintensity on T2* and STIR and no enhancement in post-contrast images, findings suggestive of osteonecrosis. There were edematous changes of the surrounding soft tissues and in the area deeper to tarsal navicular, around the flexor tendons of the foot. Small fluid collections with peripheral engaging, coalescing, suggestive of microabscesses were observed. Ultrasound
 (a)
(a)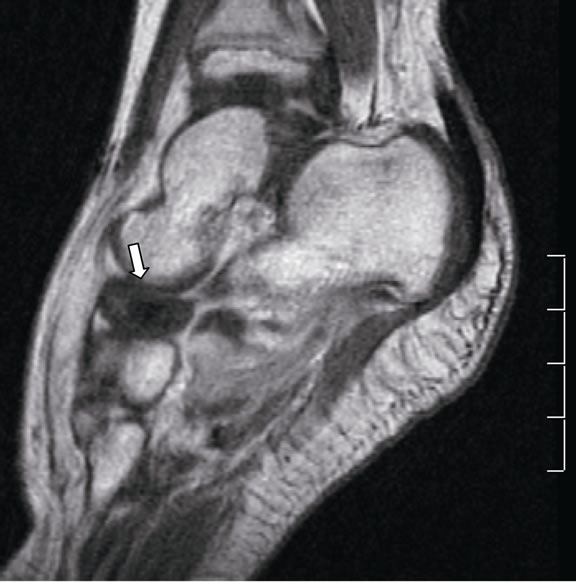 (b)
(b)
Figure 1. Magnetic resonance 6 days after admission. (a) Sagital STIR image shows hyperintensity in navicular ossification nucleus and in surrounding small parts, changes due to edema; (b) Sagital post-contrast T1 image at the same level, depicting non-enhancement of navicular bone, finding suggestive of osteonecrosis.
showed similar findings, with increased Doppler signal and tarsal microabscesses in the synovium.
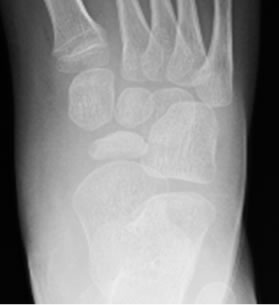
She remained on treatment with cloxacillin IV, oral ibuprofen and ranitidine for 15 days, evolving slowly. Her fever ended 5 days after admission. Roentgenogram a week after the MRI (Figure 2) showed increased density of right navicular bone, consistent with Köhler’s disease.
Two weeks after admission she was switched to oral cefuroxime-axetil (30 mg/kg/day), ibuprofen and ranitidine for 6 weeks. The patient continued to have swelling of the foot with a tarsal diameter difference of 1.5 cm over the following 4 weeks, which gradually decreased to 0.5 cm 3 months later. Subsequent radiographs showed typical 4 phases of osteochondrosis of the navicular bone: sclerosis, fragmentation, flattening and reossification (Figure 2). The patient recovered without sequelae.
3. Discussion
A case of Köhler disease together with hamatogenous tarsal arthritis is reported. We hypothesized that avascular necrosis of the navicular bone was developed after pyogenic arthritis.
The main force causing osteochondrosis is a failure of chondrification of cartilage canals in the growth center, which leads to necrosis of cartilage canal vessels in the subchondral bone and adjacent epiphyseal cartilage [1].
In 1908, Köhler detected avascularity of the tarsal navicular on radiographs [4]. The navicular bone looks sclerotic, fragmented, and collapsed. Most often unilateral
 (a)
(a)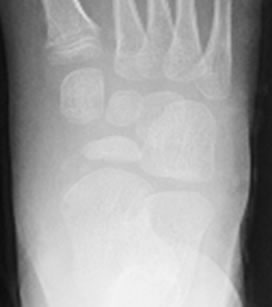 (b)
(b)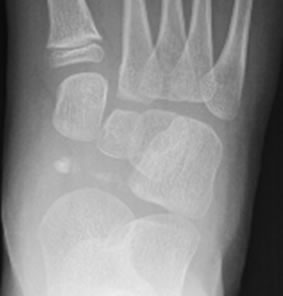 (c)
(c)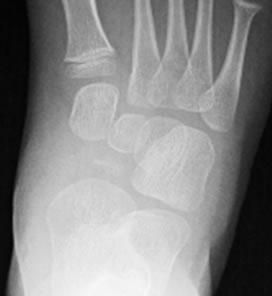 (d)
(d)
Figure 2. AP plain RX views of tarsal bones. Time sequence at (a) time of diagnosis; (b) Two months later; (c) Seven months later and (d) one year later showing evolution with sclerosis, fragmentation, flattening and reossification of tarsal navicular bone.
and affecting boys at age 4 years, Kohler’s disease causes a unique limp in which the patient will avoid bearing weight on the medial edge of the affected foot. The affected foot will be painful, swollen, and tender over the medial arch.
Vascularization of the navicular occurs in 2 ways and is identical in adults and children. A branch from the dorsalis pedis artery crosses the dorsal surface of the navicular and gives off 3 - 5 branches. Some small branches come from the medial plantar artery to supply the plantar surface. These blood vessels create a dense network around the bone and come from the perichondrium toward the center of the cartilage [5].
Kohler suggested that the changes in this disease might be the result of an abnormal strain that acts on a weak navicular, but a definitive answer has not been found. Among the theories to explain the nature of this lesion, a more satisfactory one is a mechanical basis that is associated with a delayed ossification [5]. The navicular is the last tarsal bone to ossify in children. This bone might be compressed between the already ossified talus and the cuneiforms when the child becomes heavier. Compression involves the vessels in central spongy bone, leading to ischemia, which then causes clinical symptoms. It is hypothesised that mechanical stress on the developing center of ossification leads to changes that resemble avascular necrosis [6]. This theory is supported by the absence of the condition in bones in which centres of ossification are present at birth (e.g., calcaneus, talus, or cuboid) [7]. We have also found evidence in the literature that shows a genetic predisposition for certain osteochondroses [8,9].
In this case, osteonecrosis occurred on a previously healthy bone, as seen in first X-ray, and immediately after a tarsal arthritis. Arthritis was clinically diagnosed, and confirmed by ultrasound and MRI. Ultrasound showed increased synovial Doppler signal, and both sonography and MRI showed synovial microabscesses. This arthritis was probably hematogenous in origin, as evidenced by the growth of S. aureus in blood culture. Following anti-inflammatory and antibiotic treatment, blood culture became negative, microabscesses and increased Doppler signal slowly waned. However, Köhler’s disease followed the course, apparently triggered by infection. The eventual clinical and radiographic progression was typical of a tarsal navicular osteochondrosis.
It could be hypothesized that bacteremia caused tarsal arthritis and triggered local inflammation with thrombosis of the network of tiny blood vessels that supply the scaphoid bone, resulting in avascular necrosis.
Generally self-limiting, Kohler’s disease resolves with rest, arch supports, activity modification, and sometimes immobilization for a few weeks in a cast. After the acute phase with avascular necrosis, the perichondral ring of vessels sends the blood supply, allowing rapid revascularization and formation of new bone. The radial arrangement of the vessels of this bone is of great importance in explaining why the prognosis of this lesion is always excellent [5], as it was for this girl.
To our knowledge, this patient represents the first case report in which an association between avascular necrosis of the navicular bone and a bacterial infection is suggested. This association met some criteria of causation (temporal sequence and biological plausibility), but being a single case, it is impossible to determine with certainty whether osteochondrosis was caused by bacterial infection or is just an associated process without unrelated cause and effect.
REFERENCES
- S. M. Doyle and A. Monahan, “Osteochondroses: A Clinical Review for the Pediatrician,” Current Opinion in Pediatrics, Vol. 22, No. 1, 2010, pp. 41-46. doi:10.1097/MOP.0b013e328334579e
- J. L. Borges, J. T. Guille and J. R. Bowen, “Köhler’s Bone Disease of the Tarsal Navicular,” Journal of Pediatrics Orthopedics, Vol. 15, No. 5, 1995, pp. 596-598. doi:10.1097/01241398-199509000-00009
- P. N. Malleson and D. D. Sherry, “Noninflammatory Musculoskeletan Pain Conditions,” Textbook of Pediatric Rheumatology, Cassidy, Philadelphia, 2006, pp. 670-672.
- W. Waugh, “The Ossification and Vascularisation of the Tarsal Navicular and Their Relation to Kohler’s Disease,” Journal of Bone Joint Surgery of Britain, Vol. 40-B, 1958, pp. 765-777.
- B. Vargas, “Kohler Disease,” 2010. http://emedicine.medscape.com/article/1234753-overview
- M. C. Clark, “Overview of the Causes of Limp in Children,” 2011. http://www.uptodate.com.
- R. C. Medlar and E. D. Lyne, “Sinding-Larsen-Johansson Disease. Its Etiology and Natural History,” Journal of Bone and Joint Surgery of America, Vol. 60, No. 8, 1978, pp. 1113-1116.
- G. Kenet, E. Ezra, S. Wientroub, et al., “Perthes’ Disease and the Search for Genetic Associations: Collagen Mutations, Gaucher’s Disease and Thrombophilia,” Journal of Bone Joint Surgery of Britain, Vol. 90, No. 11, 2008, pp. 1507-1511. doi:10.1302/0301-620X.90B11.20318
- A. Al Kaissi, K. Klaushofer and F. Grill, “Osteochondritis Dissecans and Osgood Schlatter Disease in a Family with Stickler Syndrome,” Pediatrics Rheumatology Online Journal, Vol. 7, No. 4, 2009, p. 4. doi:10.1186/1546-0096-7-4

