Paper Menu >>
Journal Menu >>
 Journal of Minerals & Materials Characterization & Engineering, Vol. 10, No.12, pp.1131-1139, 2011 jmmce.org Printed in the USA. All rights reserved 1131 Growth, Optical, Mechanical and Dielectric Properties of Glycine Zinc Chloride NLO Single Crystals S. Suresh and D. Arivuoli* Crystal Growth Centre, Anna University, Chennai-600 025, India. *Corresponding author: arivuoli@gmail.com ABSTRACT Single crystals of Glycine Zinc Chloride (GZC) were grown from aqueous solution by slow evaporation technique. Single crystal X-ray diffraction analysis reveals that the crystal belongs to orthorhombic system with the space group Pna2 1. The optical transmission study reveals the transparency of the crystal in the entire visible region and the cut off wavelength has been found to be 230 nm. The optical band gap is found to be 3.40eV. The transmittance of GZC crystal has been used to calculate the refractive index (n), the extinction coefficient (K) and the real ε r and imaginary ε i components of the dielectric constant. Mechanical studies were carried out on the as grown crystal. Dielectric constant and dielectric loss measurements were carried out at different temperatures and frequencies. Photoconductivity measurements carried out on the grown crystal reveal the negative photoconducting nature. Key words: Solution growth, Single crystal XRD, Optical transmission, Dielectric constant and loss, Photoconductivity. 1. INTRODUCTION In recent years, there has been extensive investigation on the synthesis of nonlinear optical (NLO) materials because nonlinear optical processes are technologically the basic tools of optoelectronic and photonic applications [1]. The development of highly efficient nonlinear optical materials for optoelectronic applications such as high speed information processing, optical communications and optical data storage have been the subject of intense research activity throughout the world over the past two decades [2–3]. Organic materials show a good efficiency of second harmonic generation. But most of the organic NLO crystals have poor mechanical and thermal stability. In order to increase the mechanical strength and thermal stability, organic compounds are added with inorganic compounds. Glycine is the simplest form of amino acid which reacts with other inorganic compounds to give a good mechanical and thermal stability. Amino acids are interesting organic materials for NLO applications as  1132 S. Suresh and D. Arivuoli Vol.10, No.12 they contain donor carboxylic acid (COOH) group and the proton acceptor amino (NH 2 ) group in them, known as zwitterions, which create hydrogen bonds, in the form of N–H + —O–C, which are very strong bonds. Hydrogen bonds have also been used in the possible generation of non-centrosymmetric structures, which is a prerequisite for an effective NLO crystal [4]. In the present investigation we report bulk growth, optical, mechanical and dielectric properties of Glycine Zinc Chloride (GZC) single crystals. 2. EXPERIMENTAL PROCEDURE A solution of glycine and zinc chloride was prepared in 3:1 molar ratio and stirred continuously using magnetic stirrer for homogenization and tiny seed crystals were obtained by spontaneous nucleation. The chemical reaction is represented as (ZnCl 2 )+ (C 2 H 5 NO 2 ) 3 → [C 2 H 5 NO 2 .ZnCl 2 ] (1) Recrystallization process was carried out two times and finally the crystals were obtained over a period of 25 days. Fig. 1 shows single crystals of GZC with high degree of transparency. Fig. 1. As-grown single crystals of GZC 3. RESULTS AND DISCUSSIONS 3.1 Single Crystal X-ray Diffraction From the single crystal X-ray diffraction data, it was confirmed that the grown crystal belongs to orthorhombic system with the non-centrosymmetric space group of Pna2 1 . The  Vol.10, No.12 Growth, Optical, Mechanical and Dielectric Properties 1133 cell parameters are: a=15.234 Ǻ, b=11.161 Ǻ and c=15.526 Ǻ; α=β=γ=90 which are in good agreement with the reported values [5]. 3.2 Optical Studies The UV–Vis–NIR spectrum of GZC was recorded with a Lambda 35 spectrophotometer in the range 300–1100 nm with a crystal of thickness 2 mm. From the spectrum, it is evident that GZC crystal has UV cut off below 230 nm, which is sufficient for SHG laser radiation of 1064 nm or other application in the blue region. It further indicates that the crystal has wide transparency window between 300 nm and 1100 nm. The measured transmittance (T) was used to calculate the absorption coefficient (α) using the formula t T = 1 log3026.2 α (2) where t is the thickness of the sample. Optical band gap (E g ) was evaluated from the transmission spectra and optical absorption coefficient (α) near the absorption edge is given by [6] )()( 2 ννα hEAh g −= (3) where A is a constant, E g the optical band gap, h the Planck constant and ν the frequency of the incident photons. The band gap of GZC crystal was estimated by plotting 2 )( να h versus ν h as shown in Fig. 3 and extrapolating the linear portion near the onset of absorption edge to the energy axis. From the figure, the value of band gap was found to be 3.40 eV. Extinction coefficient (K) can be obtained from the following equation: π λα 4 =K (4) The transmittance (T) is given by ( ) ( ) ( ) tR tR T α α 2exp1 exp1 2 2 −− −− = (5) Reflectance (R) in terms of absorption coefficient can be obtained from the above equation. Hence, Ttt TtTtTtt R)2exp()exp( )2exp()3exp()exp()exp( 2 αα αααα −+− −+−−−±− = (6) 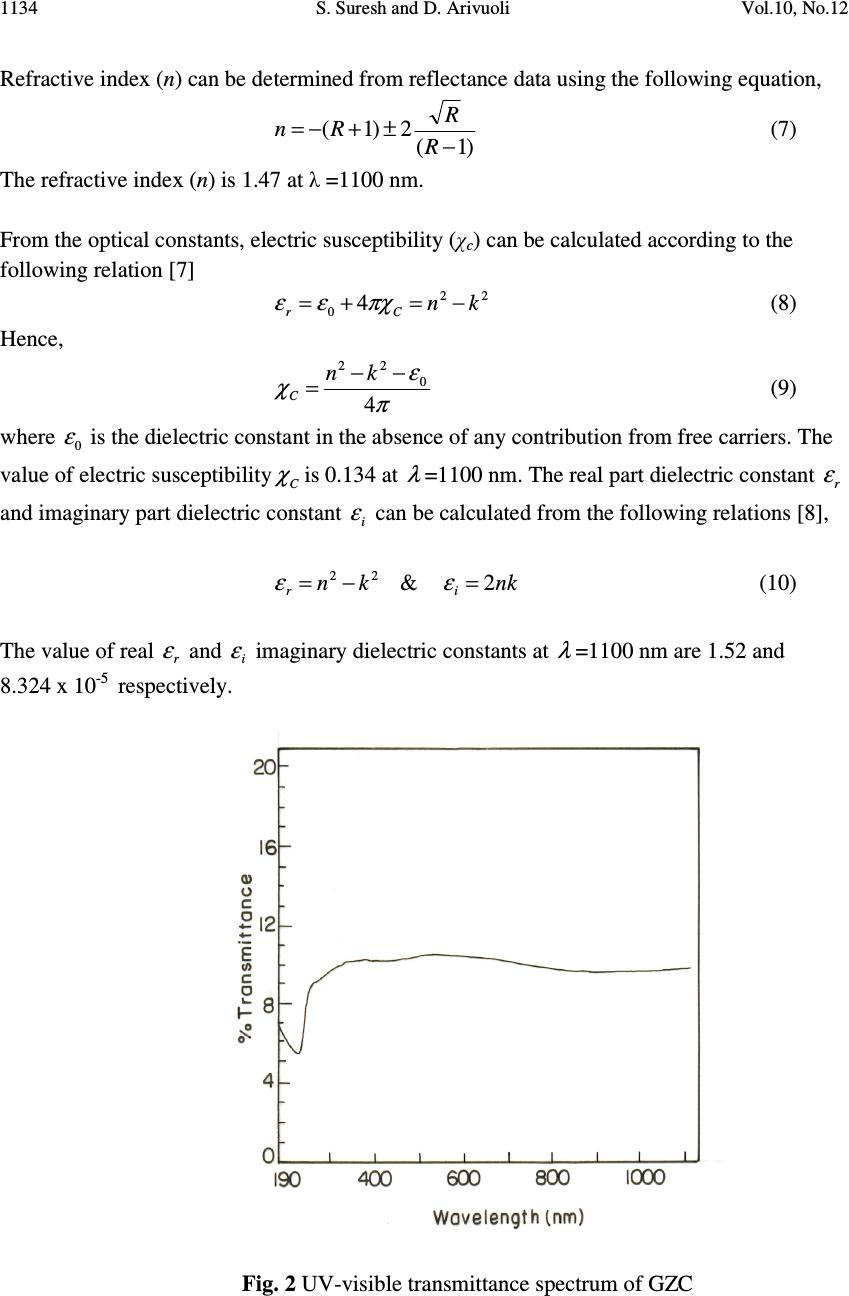 1134 S. Suresh and D. Arivuoli Vol.10, No.12 Refractive index (n) can be determined from reflectance data using the following equation, )1( 2)1( − ±+−= R R Rn (7) The refractive index (n) is 1.47 at λ =1100 nm. From the optical constants, electric susceptibility (χ c ) can be calculated according to the following relation [7] 22 0 4kn Cr −=+= πχεε (8) Hence, π ε χ 4 0 22 −− =kn C (9) where 0 ε is the dielectric constant in the absence of any contribution from free carriers. The value of electric susceptibility C χ is 0.134 at λ =1100 nm. The real part dielectric constant r ε and imaginary part dielectric constant i ε can be calculated from the following relations [8], 22kn r−= ε & nk i 2 = ε (10) The value of real r ε and i ε imaginary dielectric constants at λ =1100 nm are 1.52 and 8.324 x 10 -5 respectively. Fig. 2 UV-visible transmittance spectrum of GZC  Vol.10, No.12 Growth, Optical, Mechanical and Dielectric Properties 1135 Fig. 3 Plot of 2 )( να h versus ν h for GZC single crystal 3.3 Mechanical Property To find surface hardness of the as grown GZC crystal, microhardness was measured from 20 to 100 gram load using HMV Microhardness tester. The Vickers hardness number (H V ) was calculated using the standard formula, 2 /8544.1 dPH V = (11) where P is the applied load and d is the mean diagonal length of the indentation. The trace is shown in Fig. 4, which shows that the hardness increases with the increase of load. The Meyer's index number was calculated from the Meyer's law, which relates the load and indentation diagonal length as n kdP = (12) dnkP logloglog + = (13) where k is the material constant and ‘n’ is the Meyer's index. In order to find the value of ‘n’, a graph is plotted for log P against log d which gives a straight line. From the slope of the line the Meyer's index number ‘n’ was calculated to be 2.63. According to Onitsch [9] ‘n’ lies between 1 and 1.6 for hard materials and is greater than 1.6 for soft materials [10]. The ‘n’ value observed in the present studies is around 2.63 suggesting that the grown GZC crystal is a relatively soft material. 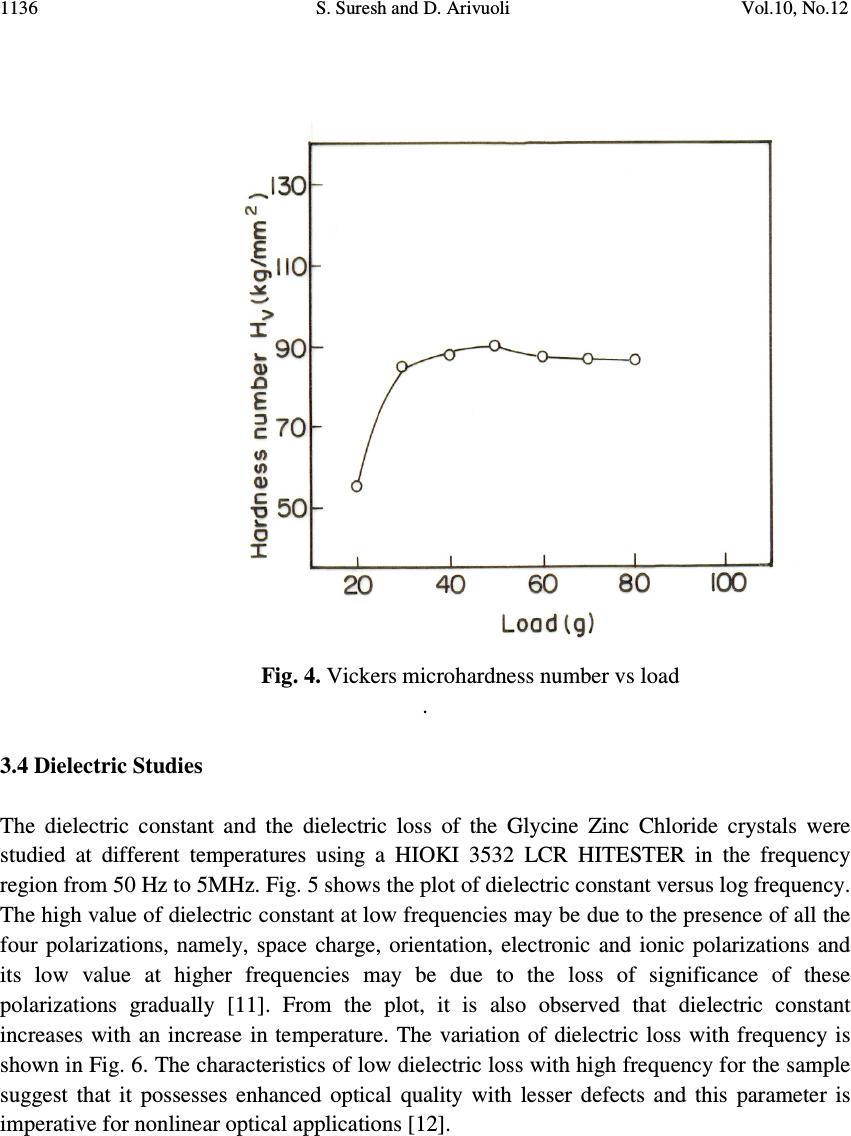 1136 S. Suresh and D. Arivuoli Vol.10, No.12 Fig. 4. Vickers microhardness number vs load . 3.4 Dielectric Studies The dielectric constant and the dielectric loss of the Glycine Zinc Chloride crystals were studied at different temperatures using a HIOKI 3532 LCR HITESTER in the frequency region from 50 Hz to 5MHz. Fig. 5 shows the plot of dielectric constant versus log frequency. The high value of dielectric constant at low frequencies may be due to the presence of all the four polarizations, namely, space charge, orientation, electronic and ionic polarizations and its low value at higher frequencies may be due to the loss of significance of these polarizations gradually [11]. From the plot, it is also observed that dielectric constant increases with an increase in temperature. The variation of dielectric loss with frequency is shown in Fig. 6. The characteristics of low dielectric loss with high frequency for the sample suggest that it possesses enhanced optical quality with lesser defects and this parameter is imperative for nonlinear optical applications [12].  Vol.10, No.12 Growth, Optical, Mechanical and Dielectric Properties 1137 Fig. 5. Variation of dielectric constant with frequency Fig. 6. Variation of dielectric loss with frequency 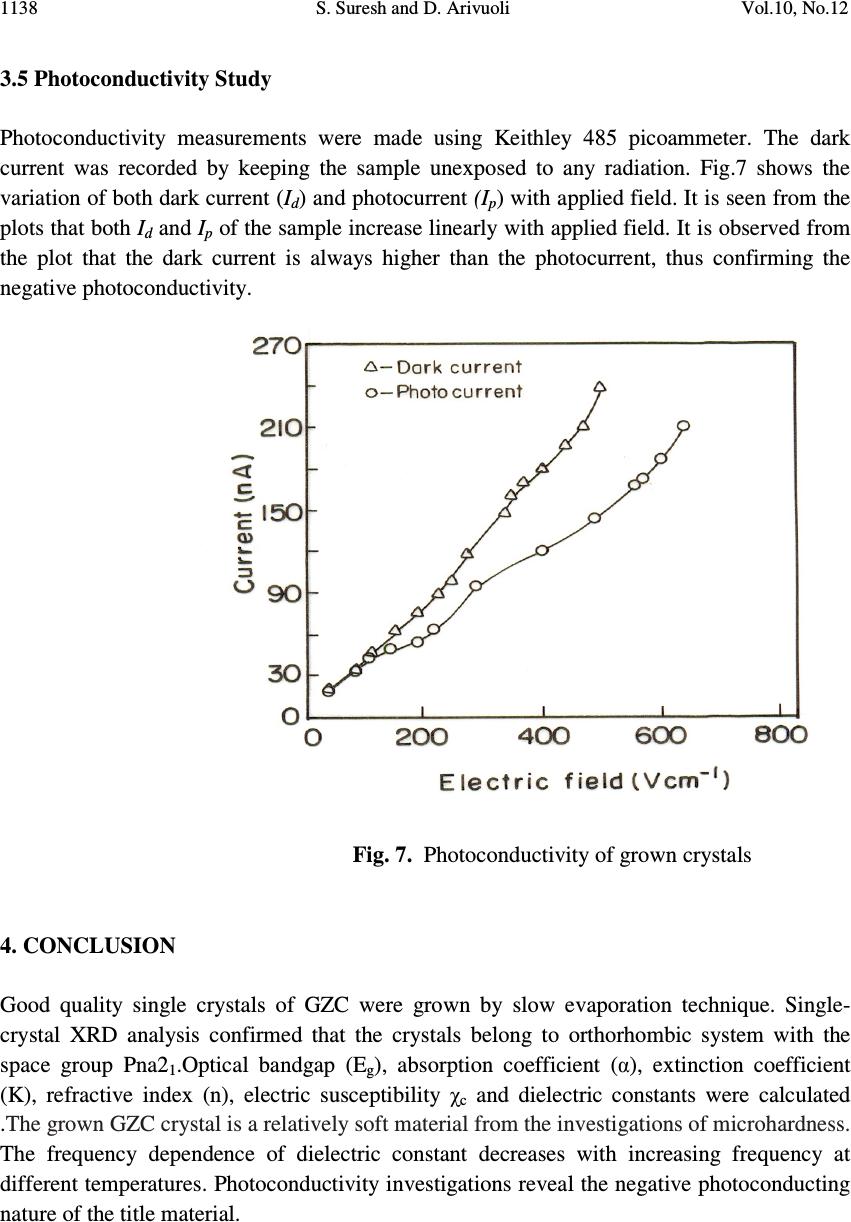 1138 S. Suresh and D. Arivuoli Vol.10, No.12 3.5 Photoconductivity Study Photoconductivity measurements were made using Keithley 485 picoammeter. The dark current was recorded by keeping the sample unexposed to any radiation. Fig.7 shows the variation of both dark current (I d ) and photocurrent (I p ) with applied field. It is seen from the plots that both I d and I p of the sample increase linearly with applied field. It is observed from the plot that the dark current is always higher than the photocurrent, thus confirming the negative photoconductivity. Fig. 7. Photoconductivity of grown crystals 4. CONCLUSION Good quality single crystals of GZC were grown by slow evaporation technique. Single- crystal XRD analysis confirmed that the crystals belong to orthorhombic system with the space group Pna2 1 .Optical bandgap (E g ), absorption coefficient (α), extinction coefficient (K), refractive index (n), electric susceptibility χ c and dielectric constants were calculated .The grown GZC crystal is a relatively soft material from the investigations of microhardness. The frequency dependence of dielectric constant decreases with increasing frequency at different temperatures. Photoconductivity investigations reveal the negative photoconducting nature of the title material.  Vol.10, No.12 Growth, Optical, Mechanical and Dielectric Properties 1139 REFERENCES [1] Prasad PN, Williams DJ, Wiley, New York, 1990. [2] Wong MS, Bosshard C, Pan F, Gunter P, Adv. Mater. 8 (1996) 677. [3] Dmitriev VG, Gurzadyan GG, Nicogosyan DN, Handbook of Nonlinear Optical Crystals, Springer-Verlag, New York, 1999. [4] Frankenbach GM, Etter MC, Chem. Mater. 4 (1992) 272. [5] Tepavitcharov S, Havlícek D, Nemec I, Vojtisek P, Rabadjieva D, Plocek J, J. Mol. Str.918 (2009)55. [6] Ashour A, El-Kadry N, Mahmoud SA.Thin Solid Films. 269 (1995) 117. [7] Gupta V, Mansingh A. J. Appl. Phys. 80 (1996) 1063. [8] Gaffar MA, Abu El-Fadl A, Bin Anooz S. Physica B.327 (2003) 43. [9] Onitsch EM. Microscope. 95(1950) 12. [10] Karan S, Gupta SPS. Mater Sci Eng A. 398(2005) 198–203 [11] Smyth CP, in: Dielectric Behavior and Structure McGraw-Hill, New York, (1965) 132 [12] Balarew C, Duhlew R, J. Solid State Chem. 55 (1984) 1. |

